预约演示
更新于:2026-05-23
GNKG-168
更新于:2026-05-23
概要
基本信息
药物类型 CpG ODN |
别名 CpG ODN、CPG-ODN、CPG 685 + [3] |
靶点 |
作用方式 激动剂 |
作用机制 TLR9激动剂(Toll样受体9激动剂) |
在研适应症- |
原研机构 |
在研机构- |
权益机构- |
最高研发阶段终止临床1期 |
首次获批日期- |
最高研发阶段(中国)- |
特殊审评- |
登录后查看时间轴
结构/序列
使用我们的RNA技术数据为新药研发加速。
登录
或
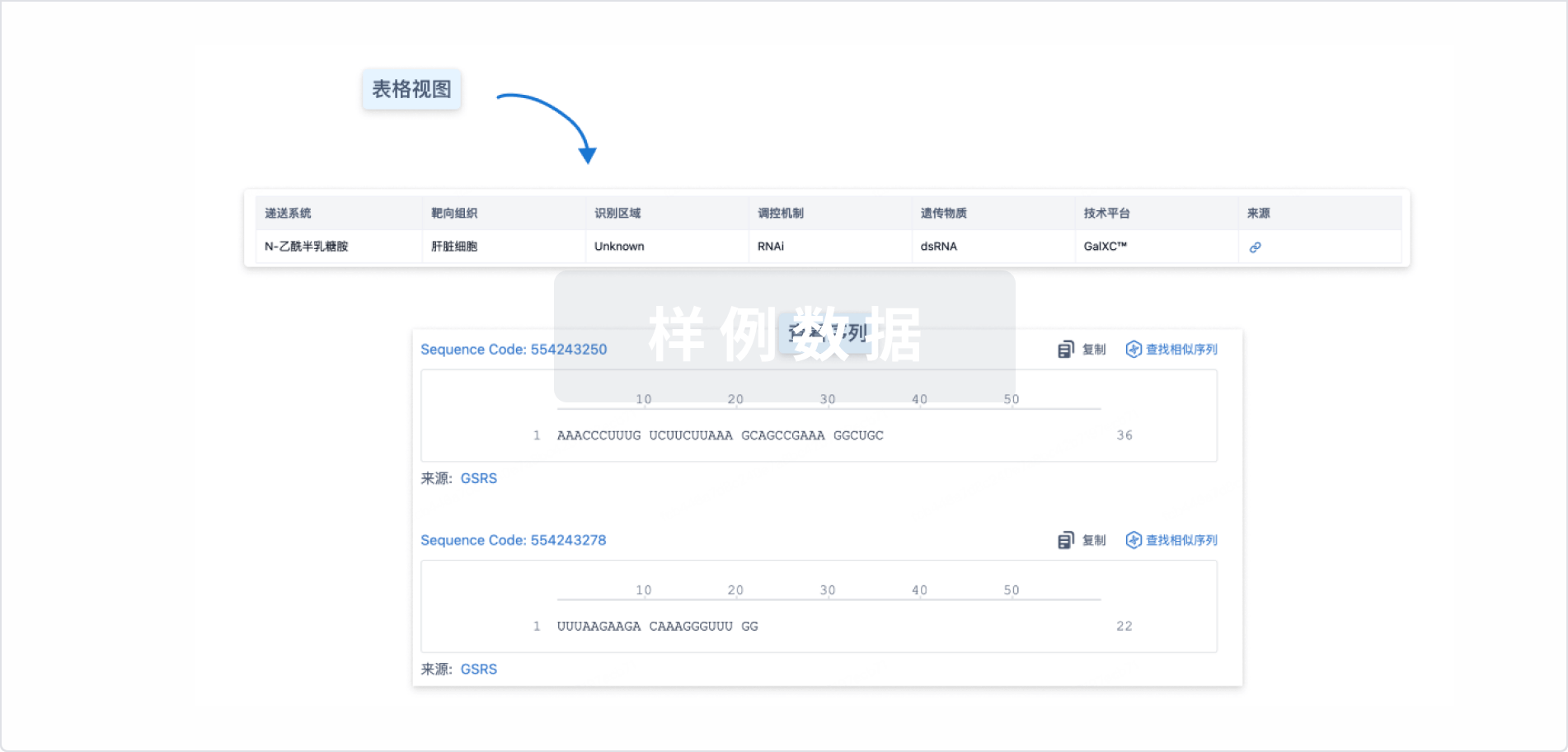
Sequence Code 29304404

来源: *****
关联
4
项与 GNKG-168 相关的临床试验ISRCTN17285423
A phase Ib, open-label, randomised, single-centre, multiple-dose-escalation study of the safety, tolerability, pharmacokinetics and pharmacodynamics of CpG ODN D35 after subcutaneous administration in participants with cutaneous leishmaniasis
开始日期2023-03-01 |
ISRCTN15458851
A Phase I, double-blind, randomised, single-centre, parallel-group, single ascending dose, placebo-controlled study of the safety, tolerability, pharmacokinetics, and pharmacodynamics of CpG ODN D35 after subcutaneous administration in healthy male subjects
开始日期2021-06-01 |
NCT01743807
A Phase I Study of GNKG168 in Pediatric Patients With Acute Lymphoblastic Leukemia or Acute Myeloid Leukemia (IND#113600)
This is a phase I trial of an investigational drug called GNKG168 in patients with relapsed and refractory acute lymphoblastic leukemia (ALL) and acute myelogenous leukemia (AML) who are in morphologic remission but are positive for Minimum Residual Disease (MRD).
开始日期2014-03-26 |
申办/合作机构 |
100 项与 GNKG-168 相关的临床结果
登录后查看更多信息
100 项与 GNKG-168 相关的转化医学
登录后查看更多信息
100 项与 GNKG-168 相关的专利(医药)
登录后查看更多信息
813
项与 GNKG-168 相关的文献(医药)2026-06-01·VETERINARY IMMUNOLOGY AND IMMUNOPATHOLOGY
Immune efficacy evaluation of a PEDV spike protein-derived multi-epitope subunit vaccine candidate in mice
Article
作者: Zhou, Bijun ; Shan, Chunlan ; Cheng, Anchun ; Xu, Fei ; Ji, Taotao ; Cen, Zehong ; Ban, Junjun ; Yin, Huaping ; Luo, Yihong ; Wei, Miaozhan ; Cheng, Zhentao ; Wang, Bin ; Guo, Xiaojiang ; Zhu, Erpeng
Porcine epidemic diarrhea virus (PEDV)-induced piglet diarrhea remains one of the most threatening infectious diseases in the swine industry across many countries. Although commercial vaccines are available, their efficacy in preventing the incidence and prevalence of porcine epidemic diarrhea (PED) remains unsatisfactory. Here we developed three subunit vaccine candidates based on four antigenic domains of the PEDV spike (S) protein expressed in E. coli: core neutralizing epitope (COE), S1 subunit domain A (S1A), and more conservative antigenic epitopes heptad repeat 1 (HR1) and heptad repeat (HR2) in S2 protein. Vaccination of the three vaccine candidates (COE, COE-S1A, and COE-HR1-HR2) induced substantial humoral and cellular expression, as evidenced by increase in specific antibody production and mRNA expression of both Th1 and Th2 cytokines. Among them, the COE-HR1-HR2 construct demonstrated a superior outcome. Furthermore, comparative screening of immune adjuvants was conducted to optimize vaccine efficacy. CpG ODN, in combination with the COE-HR1-HR2 fusion protein, induced enhanced humoral and cellular immune responses compared with Freund's adjuvant and GEL 02, and was comparable to ISA660VG. Taken together, in view of the incomplete protection provided by current vaccines against PEDV, there is an urgent need for next-generation formulations that can effectively reduce disease burden and promote robust herd immunity. Our preliminary findings indicate that the COE-HR1-HR2 candidate, by integrating the core S1 and conserved S2 antigenic epitopes, offers the potential for developing novel multi-epitope vaccine against PEDV that could induce protective immunity thereby reducing disease development.
2026-06-01·INTERNATIONAL JOURNAL OF BIOLOGICAL MACROMOLECULES
Construction, characterization, and evaluation of nanosized curdlan sulfate/quaternary ammonium chitosan/CpG ODN polyelectrolyte complex as an adjuvant for tumor vaccine
Article
作者: Li, Zuyi ; Zhang, Yuhe ; Wang, Fengshan ; Chen, Yipan ; Liu, Minghui ; Jiang, Wenjie ; Li, Pingli
CpG oligodeoxynucleotides (CpG ODNs), as nucleic acid adjuvants, effectively stimulate humoral and cellular immune responses. However, their clinical application is limited by drawbacks such as susceptibility to nuclease degradation and high negative charge, which impede cellular uptake. Previously, we have confirmed that polyelectrolyte complex nanoparticles composed of curdlan sulfate (CS) and O-(2-hydroxyl) propyl-3-trimethyl ammonium chitosan chloride (CS/O-HTCC) could encapsulate negatively charged proteins and enhance their cellular internalization. Hence, in this study, CS/O-HTCC/CpG nanoparticles (C/O/CpG NPs) were prepared by electrostatic adsorption of positively charged CS/O-HTCC NPs with negatively charged CpG ODN and the formulation was optimized. Besides, the immunological activities of C/O/CpG NPs were evaluated both in vitro and in vivo. Moreover, their adjuvant properties and antitumor effects were assessed using ovalbumin (OVA) as a model antigen. The results showed that C/O/CpG NPs were uniformly spherical with a particle size and polydispersity index (PDI) of 119.67 ± 9.07 nm and 0.18 ± 0.01, respectively. C/O/CpG NPs stimulated the activation of antigen-presenting cells, induce antigen-specific cellular and humoral immune responses effectively and showed significant anti-tumor activity. In conclusion, C/O/CpG NPs could be considered as a promising class of adjuvants for tumor vaccines.
2026-05-01·MOLECULES AND CELLS
CpG oligodeoxynucleotide reduces PrPSc accumulation and prolongs survival in prion-infected mice
Article
作者: Kim, Mo-Jong ; Kim, Yong-Sun ; Mostafa, Mohd Najib ; Kwon, Hyung-Joo ; Lee, Younghee ; Choi, Eun-Kyoung ; Choi, Myung-Ju ; Jang, Byungki
Prion diseases, also known as transmissible spongiform encephalopathies, are characterized by the accumulation of misfolded prion proteins (PrPSc), which resists clearance due to impaired removal mechanisms and immune tolerance. Although strategies aimed at enhancing intracellular clearance pathways, including autophagy, have shown promise, effective in vivo interventions remain limited. Innate immune stimulation with CpG oligodeoxynucleotides (CpG ODN), a Toll-like receptor 9 agonist that mimics bacterial DNA containing unmethylated CpG motifs, has been reported to extend survival in prion-infected mice; however, the underlying mechanisms involved remain incompletely defined. In this study, we examined the effects of CpG ODN in both in vitro and in vivo models of prion disease using the 22L scrapie strain. Mice treated with CpG ODN prior to infection exhibited prolonged mean survival compared with vehicle-treated controls and showed reduced PrPSc accumulation in spleen and brain tissues at 60 and 170 days post-infection, respectively. These changes were correlated with increased AMPK (Tyr172) phosphorylation and alterations in autophagy-associated signaling markers in the brain tissue. In 22L scrapie-infected neuronal cells, CpG ODN treatment significantly reduced PrPSc levels, and this effect was attenuated by pharmacological inhibition of autophagy-associated and lysosomal degradative processes. Collectively, these findings indicate that CpG ODN reduces PrPSc accumulation and prolongs survival in prion-infected mice, supporting the involvement of autophagy-associated degradative processes without establishing enhanced autophagic flux.
12
项与 GNKG-168 相关的新闻(医药)2026-03-13
·生物探索
引言
在过去的一个多世纪里,我们试图通过疫苗来治疗癌症的尝试屡屡受挫。然而,近年来基因组测序技术的飞跃、人类白细胞抗原(HLA)I类表位预测算法的大幅优化,以及强大的抗原递送平台的崛起,将这一曾经饱受质疑的治疗理念推向了肿瘤学研究的最前沿。
当我们审视肿瘤细胞的演化过程时,其基因组的异常转录和翻译会产生带有突变信息的异常蛋白质。这些存在于肿瘤细胞而不存在于健康组织中的靶点,正是肿瘤新生抗原(Neoantigen)。它们为激发宿主适应性免疫系统提供了高度特异性的“靶心”。尤其是经历了SARS-CoV-2大流行之后,mRNA疫苗技术在有效性和规模化生产方面展现出了巨大潜力,也为癌症疫苗的临床转化按下了加速键。
3月10日,《Nature Biotechnology》的研究报道“The promises and challenges of neoantigen cancer vaccines”,该综述全面剖析了新生抗原疫苗在临床试验中的最新进展,指出早期的临床数据已经证实,针对新生抗原的疫苗接种能够诱导强大且持久的T细胞免疫反应,这种免疫记忆甚至可能持续数十年。深入探讨了肿瘤新生抗原疫苗如何跨越技术瓶颈,以及在抗原选择、递送策略和临床试验设计上,我们应当如何重新思考未来的癌症治疗法则。
破译肿瘤的专属“指纹”:共享与个性化的靶点博弈
在探讨疫苗的设计之前,研究人员必须回答一个核心问题:我们究竟应该瞄准哪些抗原?目前,绝大多数进入临床开发的新生抗原疫苗,都将焦点聚集在单核苷酸变异(SNVs)和插入/缺失突变(indels)上。基于抗原在不同患者中的分布特征,疫苗策略主要分化为两大阵营:共享新生抗原疫苗(Shared neoantigen vaccines)和个性化癌症疫苗(Personalized cancer vaccines, PCVs)。共享疫苗的“现货”优势与免疫显性困境
共享新生抗原疫苗的逻辑在于,跨越不同肿瘤个体,寻找那些普遍存在的致癌驱动突变。这类“现货型”疫苗绕过了繁琐的定制化生产流程,成本更低,且能在患者确诊后迅速给药。这在晚期肿瘤治疗中尤为关键,因为患者往往没有数周的时间等待疫苗的定制。
然而,共享疫苗的受众范围面临着双重限制。首先,特定的驱动突变仅存在于部分患者中;其次,即使存在突变,患者还必须携带能够呈递该突变表位的特定HLA等位基因。尽管如此,临床数据依然释放了积极的信号。在一项针对胰腺癌和结直肠癌的试验中,研究人员使用了一种通过化学修饰结合白蛋白亲脂基团的合成长肽(SLP)疫苗,以改善淋巴结递送效率。该疫苗靶向KRAS-G12D和KRAS-G12R突变。结果显示,在25名患者中,有21名(84%)在体外检测中表现出了针对突变KRAS抗原的特异性T细胞反应。尤其在接受最高剂量治疗的组别中,这一比例达到了100%,并且这种强烈的免疫原性与患者无进展生存期和总生存期的延长表现出相关性。
为了扩大共享疫苗的适用人群,研究人员尝试将针对多种常见驱动突变(如KRAS和TP53)的抗原池化,整合到单一疫苗中。但这引发了一个深刻的免疫学问题:免疫显性(Immunodominance)。在一项包含20个共享新生抗原的临床试验中,研究人员发现,大多数患者体外T细胞反应主要集中在TP53表位上。值得注意的是,虽然这些患者的HLA等位基因能够呈递这些TP53表位,但他们的肿瘤实际上是KRAS突变型,并不表达这些突变的TP53抗原。
这种现象提示我们,免疫系统在处理多抗原混合物时,可能会产生一种竞争机制。针对“肿瘤不相关”但具有高度免疫显性的抗原引发的T细胞反应,可能会压制针对真正致命的驱动突变(如KRAS)的亚显性T细胞反应。质谱分析数据进一步证实了这一推论:当只包含KRAS新生抗原(G12V、G12D和G12C)时,它们在人类HLA-A*11:01单等位基因细胞系表面的密度较高;但当加入同样预测结合HLA-A*11:01的TP53表位后,KRAS-G12D和KRAS-G12V的表位密度下降了整整两倍。动物模型也验证了这一点,接种全抗原组合的小鼠未能对KRAS-G12D产生免疫反应,但如果将KRAS和TP53新生抗原分装在不同的载体中进行接种,这种免疫显性的压制作用便消失了。这表明,在设计多靶点共享疫苗时,单纯的叠加抗原并非最优解,我们需要更巧妙的抗原排列或多载体策略来平衡免疫反应。个性化疫苗(PCVs)的多靶点饱和攻击
与共享疫苗不同,个性化癌症疫苗(PCVs)通过全外显子组测序和RNA测序,从每个患者特有的肿瘤突变中筛选靶点。由于单个肿瘤中往往存在大量的SNVs和indels,PCVs能够同时靶向数十个不同的新生抗原。鉴于目前的新生抗原预测算法仍难以做到百分之百精准预测哪些表位能引发真正具有杀伤力的T细胞反应,PCVs采用了一种类似“饱和攻击”的策略:通过增加“射门次数”(即包含多个预测的新生抗原),来弥补免疫原性预测不准确的局限性,同时应对肿瘤内部的基因异质性和进化。超越常规边界的隐秘靶点
传统的全外显子测序寻找的是基因组编码区的突变,但这仅仅是冰山一角。目前,研究人员开始将目光投向更广阔、也是以往未曾涉足的领域——非经典抗原(Noncanonical antigens)或隐秘抗原(Cryptic antigens)。
这类抗原来源于肿瘤相关的异常事件,例如非编码DNA区域的异常翻译,或是mRNA的选择性剪接。在对胰腺腺癌、卵巢癌、黑色素瘤和非小细胞肺癌患者的肿瘤组织进行深入分析时发现,这些肿瘤中HLA I类分子结合的肽段有很大一部分是非经典肽。更重要的是,许多此类肽段具有肿瘤排他性,能够在多个患者的肿瘤中共享,并能引发肿瘤特异性的T细胞反应。
内源性逆转录病毒(ERVs)是另一个极具潜力的新生抗原来源。人类基因组中潜伏着大量的古老病毒序列,它们通常处于沉默状态,但在肿瘤相关的基因组事件中可能被重新激活。例如,在肾细胞癌(RCC)中,由于von Hippel-Lindau肿瘤抑制基因的失活,会导致缺氧诱导因子(HIF)转录因子的上调,进而诱导多个ERVs的表达。研究证实,由这些内源性逆转录病毒衍生出的肽段,不仅能在人类RCC细胞系和实际的肾癌组织中被HLA I类分子呈递,还能在对免疫检查点抑制剂(ICB)产生应答的患者体内诱导出特异性的T细胞反应。这意味着,对于那些常规突变负荷(TMB)较低的肿瘤,非经典抗原和ERVs可能会成为开辟新战场的关键。运载技术的巅峰对决:RNA与多肽的深层进化
识别了靶点之后,如何将这些抗原信息安全、高效地递送给免疫系统(尤其是树突状细胞等专业抗原呈递细胞),决定了疫苗的最终成败。目前,核酸(RNA、DNA)和合成长肽(SLP)是临床试验中最为主流的递送平台,它们各自在免疫学特性和工程改造上展现出了截然不同的演进路径。RNA疫苗:自带佐剂效应的精准微调
mRNA疫苗的核心结构包括5'帽、5'非翻译区(UTR)、抗原编码区、3'UTR和poly(A)尾。RNA分子本身具有内在的免疫佐剂特性:单链和双链RNA可以作为病原体相关分子模式(PAMPs),触发1型干扰素的产生。这一过程虽然在一定程度上会干扰RNA在细胞内的蛋白质翻译效率,但对于激发强效的抗肿瘤T细胞反应而言,这种由Toll样受体(TLR)介导的先天免疫激活却是必不可少的。
由于游离RNA在体内极不稳定,必须依靠脂质纳米颗粒(LNP,常用于肌肉注射)或脂质复合物(Lipoplex,常用于静脉注射)进行包裹递送。现阶段,针对RNA序列元素的化学修饰已经成为提升疫苗效能的关键。例如,通过深度学习和人工智能优化5'UTR和3'UTR的序列,不仅能增强RNA抗核酸外切酶降解的能力,还能显著提升翻译效率。
此外,递送策略也在不断突破。在一项临床前研究中,研究人员将编码白细胞介素-12(IL-12)的mRNA与编码卵清蛋白(OVA)或病毒抗原的mRNA-LNP共同包裹并给药。结果显示,这种共递送策略极大地增强了CD8+ T细胞的效应功能和记忆能力。在B16F0黑色素瘤模型中,与单独的疫苗相比,加入IL-12 mRNA-LNP显著提升了治疗效果。不仅是IL-12,未来包括IL-15、IL-7,甚至是树突状细胞生长因子和共刺激分子的mRNA,都可能作为“模块化”的免疫增强剂与抗原mRNA组合,形成更为强大的治疗网络。合成长肽(SLP)疫苗:从微米到纳米的结构重塑
合成长肽疫苗在肿瘤治疗领域有着数十年的研究历史。早期的肽疫苗多采用8至10个氨基酸的短肽,旨在直接嵌入HLA I类分子的结合槽以激活CD8+ T细胞。但这种策略存在致命缺陷:短肽无法被HLA II类分子有效呈递,且无需专业抗原呈递细胞的处理,导致T细胞启动效果不佳,更无法有效动员CD4+ T细胞(CD4+ T细胞在维持抗肿瘤免疫中同样扮演着核心角色)。
因此,现代的SLP疫苗多采用包含多个抗原表位的长肽。肽疫苗本身不具备免疫原性,必须配合免疫佐剂使用。这一“缺点”反而成为了一种优势:它允许我们将抗原递送载体与佐剂(通常是引发毒性的主要原因)解耦,从而在临床开发中获得更好的安全性。然而,针对高突变负荷肿瘤设计包含大量新生抗原的多肽疫苗时,序列特异性的合成难题和溶解度限制,依然是制约其定制化速度的瓶颈。
为了突破多肽在体内的递送效率限制,纳米技术被巧妙地引入。在一项研究中,研究人员将新生抗原多肽与TLR激动剂(CpG ODN)共同配制在可溶性高密度脂蛋白(sHDL)纳米盘中。这种纳米盘能够极其高效地进入淋巴结,实现抗原的持续呈递和T细胞的交叉启动。在接种OVA模型的小鼠中,使用sHDL和CpG ODN配制的多肽,其产生的疫苗特异性CD8+ T细胞频率达到了循环CD8+ T细胞的20%,而使用可溶性肽加CpG佐剂的对照组仅为1%至3%。在更为接近真实肿瘤的MC38模型中,使用sHDL-Adpgk加上CpG配制疫苗并联合PD-1阻断疗法,实现了100%小鼠的肿瘤完全消退;相比之下,可溶性抗原加PD-1抑制剂组的完全消退率仅为25%。
另一项名为SNP-7/8a的纳米颗粒多肽疫苗则展现了更为深层的免疫重塑能力。该技术将抗原多肽的N端和C端连接到经过电荷修饰和疏水处理的寡肽上,使其自组装成纳米胶束。这种设计使得引流淋巴结中携带疫苗抗原的CD11c+树突状细胞比例从约1%激增至约35%。静脉注射SNP-7/8a不仅诱导了幅度提高20倍的CD8+ T细胞反应,产生的T细胞还具有干细胞样特征,并显著减少了具有免疫抑制特征的单核细胞髓源性抑制细胞。这表明,先进的递送平台不仅仅是在“运输”抗原,更是在系统性地重塑肿瘤微环境。唤醒免疫记忆的临床实录:从体外数据到体内杀伤
随着底层技术的成熟,新生抗原疫苗在人体试验中逐渐展现出令人瞩目的免疫学响应。早期的临床试验主要集中在黑色素瘤等高突变负荷的实体瘤中。研究人员不仅观察到了疫苗特异性免疫反应,更发现了一个极其关键的生物学现象:表位扩展(Epitope spreading)。
在针对黑色素瘤、非小细胞肺癌和尿路上皮癌的SLP个性化疫苗联合ICB的试验中,绝大多数患者体内不仅产生了针对疫苗本身包含的新生抗原的T细胞,还衍生出了针对疫苗未包含的、肿瘤其他新生抗原的T细胞反应。这种“表位扩展”意味着疫苗成功诱导T细胞杀死了部分肿瘤细胞,肿瘤细胞破裂后释放出的新抗原被免疫系统捕获,进而引发了更广泛、更全面的抗肿瘤免疫反应带。更令人振奋的是,在这几项研究中,表位扩展的出现与患者无进展生存期(PFS)的延长存在显著关联。在一项包含黑色素瘤患者的试验中,通过对连续核心肿瘤活检的病理评估,发现9名患者在接种疫苗后实现了从部分病理缓解到完全缓解的转化。
在一项纳入13名实体瘤和血液恶性肿瘤患者的I期试验中,研究人员使用了一种包含最多10个靶点的SLP个性化疫苗。通过体外IFN-γ ELISpot检测,在总共126个免疫新生抗原中,有44%成功引发了T细胞反应。通过对未刺激和新生抗原刺激后的T细胞进行大规模TCR Vβ测序,研究发现接种疫苗后患者外周血中出现了数百个新的TCR克隆。当在疫苗接种后从肿瘤浸润淋巴细胞(TILs)中重建这些TCR并进行特异性筛选时发现,疫苗特异性的TCR几乎排他性地存在于接种后的肿瘤组织内。这提供了直接的证据:疫苗成功地驱使特异性杀伤T细胞从外周血侵入了肿瘤内部。
在基于mRNA脂质复合物递送的个性化疫苗(Autogene cevumeran)的最大规模临床试验中,90名有血液样本可供检测的患者中,有64名(71%)表现出明显的体外IFN-γ ELISpot反应。在这些患者体内,每个疫苗(最多包含20个抗原)平均能引发针对2个抗原(范围1-8个)的特异性反应。重要的是,这些引发的T细胞绝大多数是“从头产生(De novo)”的,即在接种前并未在体内检测到,而且表现出明显的CD4+ T细胞主导特征。然而,在针对晚期、经过大量既往治疗的实体瘤患者(剂量递增队列的57名可评估患者)中,仅有3名患者达到了客观缓解。这一数据冷静地提醒我们:对于肿瘤负荷极高、免疫微环境已严重耗竭的晚期癌症,单靠疫苗往往难以力挽狂澜。决战术后防线:高光时刻的降临
癌症疫苗发挥作用的机制决定了它不是一种“起效极快”的疗法。从抗原被摄取、呈递,到T细胞的激活、大规模扩增,再到渗透进肿瘤组织发挥杀伤作用,这是一个需要数周甚至数月才能完成的生物学过程。因此,在肿瘤负荷被外科手术降至最低、体内仅存微小残留病灶(MRD)的“辅助治疗(Adjuvant therapy)”阶段,疫苗有了最充裕的时间去构建坚不可摧的免疫防线。正是基于这一逻辑,新生抗原疫苗在临床试验中迎来了真正的高光时刻。
在迄今唯一一项展示出明确临床疗效的随机、2期个性化疫苗研究(KEYNOTE-942)中,157名IIIB-IV期高危黑色素瘤患者在手术切除后,被随机分配接受Moderna修饰的mRNA个性化疫苗(最多靶向34个新生抗原,mRNA-4157)联合帕博利珠单抗(Pembrolizumab,标准治疗),或仅接受帕博利珠单抗单药治疗。
随访数据的对比令人震撼:在中位随访时间为两年时,联合治疗组在18个月时的无复发生存率(RFS)达到了79%,而单药组仅为62%。这意味着,加入个性化疫苗使患者的复发风险大幅降低了44%(风险比HR=0.56,P=0.052)。更为关键的是,在评估远处转移风险时,疫苗的保护效力更为惊人——远处转移风险急剧下降了65%(HR=0.347,P=0.01)。数据曲线显示,两条生存曲线大约在治疗开始后的30周(对于无复发生存)和50周(对于无远处转移生存)开始明显分离。这种明显的时间延迟,在机制上完美契合了免疫系统逐步建立、扩增并巩固疫苗诱导的抗肿瘤免疫记忆所需的时间窗口。
另一项针对可切除胰腺导管腺癌(PDAC,一种以极度缺乏免疫原性和预后极差著称的冷肿瘤)的研究,则在更为微观的细胞层面揭示了这种免疫保护的本质。在这项试验中,16名患者在术后接受了阿替利珠单抗(Atezolizumab),随后进行了8周的BioNTech脂质复合物mRNA个性化疫苗(Autogene cevumeran)初免,接着接受标准化疗(mFOLFIRINOX)及一次疫苗加强针。
研究人员根据外周血免疫反应将这16名患者分为两组:8名“应答者”在ELISpot检测中对一种或多种疫苗新生抗原产生了明显的T细胞反应,并检测到了疫苗扩增的TCRβ克隆;而另外8名“无应答者”则未能产生可检测的免疫反应。临床结局的差异是极其分明的:经过中位18个月的随访,8名无应答者中有6人出现了肿瘤复发;而8名应答者无一复发。更令人振奋的是,在长达3.2年的后续随访中,这种无复发生存状态在应答者中得到了稳定维持。
通过追踪外周血中TCRβ克隆型的转录本,研究人员得出了一个不可思议的结论:这些由疫苗诱导并扩增的TCR克隆,其平均估计寿命长达7.7年,部分克隆的预计存活时间甚至达到了惊人的100年以上!将单细胞RNA测序与单细胞TCR测序结合后发现,这些疫苗激活的T细胞在经历早期的增殖和收缩后,进入了一种高度稳定的“组织驻留记忆(Tissue-resident memory)”状态,表现为ZNF683基因的显著过表达;相反,那些代表常规中央记忆或效应记忆的标志物(如SELL、TCF1和IL7R),以及与T细胞耗竭相关的标志物,在这些长寿克隆中均未显现。
此外,当研究人员在患者接种疫苗3年多之后,提取其外周血单核细胞并用之前确认具有免疫原性的新生抗原进行体外重新刺激时,6名(在8名应答者中)患者的T细胞依然能够迅速分泌效应细胞因子并展现出强大的细胞溶解能力。更重要的是,相比于野生型对应的TCR,这些长寿克隆来源的TCR在受到同源新生表位刺激时,普遍表现出更高的亲和力,部分TCR的亲和力甚至达到了纳摩尔(Nanomolar)级别的超高水平。
这些细致入微的免疫学数据,为我们勾勒出了一幅癌症疫苗起效的完整图景:疫苗不仅能够在体内从头唤醒对特定肿瘤靶点的杀伤反应,更能够跨越时间的长河,将这些带有极高识别精度的杀伤细胞固化为患者体内的长效“巡逻兵”,实现对微小残留病灶长达数年乃至数十年的持续压制。这也正是癌症疫苗区别于所有其他药物干预的根本魅力所在。重新定义临床坐标:从终末期走向全周期干预
面对前期试验呈现出的数据差异(晚期肿瘤的低反应率与辅助治疗阶段的高效保护),学术界和产业界开始深刻反思并重构癌症疫苗的临床开发策略。TMB的悖论:低突变负荷肿瘤的潜在优势
在早期的试验设计中,研究人员倾向于选择黑色素瘤和非小细胞肺癌等高肿瘤突变负荷(TMB)和对PD-1抑制剂敏感的癌种。逻辑很简单:突变越多,能够被免疫系统识别的新生抗原“靶点”就越丰富。然而,目前的药物合成技术(无论是多肽还是mRNA)受限于药理学和制造工艺,单个个性化疫苗能够容纳的新生抗原数量通常存在上限(如目前多为20到40个)。
这就产生了一个有趣的悖论:在那些具有成百上千个突变的高TMB肿瘤中,疫苗所包含的几十个靶点仅仅覆盖了整个肿瘤抗原图谱中极小的一部分,一旦肿瘤通过下调这些特定抗原的表达来进行免疫逃逸,疫苗的效力就会大打折扣。反观如胰腺癌(PDAC)和胶质母细胞瘤这样低TMB的肿瘤,由于其总突变数量本来就少,一个包含数十个靶点的疫苗实际上可能已经覆盖了该肿瘤绝大部分,甚至全部的新生抗原库。这种对靶点的“全面覆盖”,反而可能使得疫苗在低TMB肿瘤中具备意想不到的治疗优势。一项测试一线Autogene cevumeran联合帕博利珠单抗治疗晚期黑色素瘤的随机IIb期试验数据似乎也印证了这一点:尽管该试验未达到无进展生存期(PFS)的主要终点,但在亚组分析中,研究人员发现总生存期的延长仅出现在低TMB患者群体中,而在高TMB患者中并未观察到获益。围手术期设计的智慧演进
鉴于疫苗需要充裕的时间来构建免疫记忆,除了将疫苗的介入时机提前至辅助治疗阶段外,研究人员正在探索更为巧妙的围手术期(Perioperative)临床试验设计。
近期的多项大型研究表明,在黑色素瘤、非小细胞肺癌和头颈癌中,手术前先进行免疫检查点抑制剂(ICB)的新辅助治疗,其长期生存获益明显优于单纯的术后辅助治疗。将个性化癌症疫苗(PCVs)融入这一极具前景的治疗模式中,是未来的重要方向。但PCVs的定制化生产通常需要数周时间,这与新辅助治疗的时间窗存在冲突。
为了解决这一难题,一种“基于风险分层(Risk-adapted)”的设计应运而生:在所有符合条件的患者确诊并准备接受新辅助ICB治疗的第一时间,立即采集样本启动肿瘤全外显子测序和RNA测序,开始新生抗原的挖掘。在患者接受新辅助治疗并完成手术切除后,临床医生会对切除的肿瘤组织进行细致的病理评估。对于那些达到主要病理缓解(MPR)或完全病理缓解的患者,由于其复发风险极低,仅需按照标准方案进行常规辅助治疗或单纯的长期随访监测,无需接种疫苗;而对于那些未能达到主要病理缓解(非MPR)的高危患者,此时预先启动生产的个性化疫苗刚好制备完成,可以立即作为辅助治疗方案无缝衔接,集中火力清剿体内可能残存的耐药癌细胞。
同时,在此期间,如果存在针对该癌种的“现货型”共享新生抗原疫苗,完全可以在PCVs定制期间先行给药,作为一种免疫系统的早期“预警启动”。重塑评估指标:循环肿瘤DNA(ctDNA)的崛起
对于确实需要在晚期或转移性肿瘤中进行评估的疫苗试验,传统的基于影像学(如RECIST标准)的客观缓解率(ORR)往往是一种滞后且不完全准确的评估工具。疫苗诱导的免疫反应最初可能引发肿瘤病灶的炎症性浸润,在影像学上甚至可能表现为“假性进展”。
因此,医学界迫切需要更灵敏、更早期的替代指标。在近期的一项涉及7名既往接受过治疗的微卫星稳定型结直肠癌(MSS-CRC,对传统ICB治疗极度不敏感)患者的试验中,患者接受了腺病毒载体初免叠加自扩增mRNA(samRNA)加强的新生抗原疫苗,并联合静脉注射纳武利尤单抗和皮下注射伊匹木单抗。尽管部分患者在影像学上表现为疾病稳定或进展,但研究人员发现,外周血中循环肿瘤DNA(ctDNA)水平的下降与患者总生存期的改善呈现出高度的关联性。结合外周血中高强度的体外IFN-γ ELISpot特异性T细胞反应数据,这种以血液学指标和深度免疫分型为核心的疗效评估体系,正逐渐成为衡量晚期肿瘤疫苗活性的新标准。
站在基因组学与免疫学深度融合的历史节点上,肿瘤新生抗原疫苗已经从理论概念蜕变为拥有坚实临床数据支撑的治疗实体。尽管目前不同技术路线孰优孰劣尚未有头对头的临床试验证据定论,且针对靶点筛选精度、复杂脂质载体制剂稳定性的工艺优化仍面临诸多挑战,但随着涉及数千名患者的III期注册临床试验的全面铺开,我们正处于见证历史的边缘。
疫苗或许无法解答癌症治疗的所有难题,但这种赋予人体免疫系统以“精准记忆”的策略,其所展现出的彻底清除残留病灶、提供长达数十年的免疫保护的潜力,不仅是对现有外科手术、放化疗和靶向治疗体系的强有力补充,更是推动人类最终走向“治愈”癌症宏伟目标的核心拼图。在与肿瘤的漫长博弈中,我们终于握住了那把能够真正“教化”免疫系统、直击敌人要害的密钥。
参考文献
Ott PA. The promises and challenges of neoantigen cancer vaccines. Nat Biotechnol. 2026 Mar 10. doi: 10.1038/s41587-026-03018-2. Epub ahead of print. PMID: 41807825.
声明:本文仅用于分享,不代表平台立场,如涉及版权等问题,请尽快联系我们,我们第一时间更正,谢谢!
往期热文:
Nature | 潜伏与反杀:HIV真实储库克隆的动态抗原表达与免疫抵抗密码
Nature Medicine | ChatGPT医疗分诊能力深度解析:当人工智能直面生死抉择
Nature | 破解罕见病诊断的“黑盒”:基于可追溯推理的智能体系统重塑临床决策
Nature Biotechnology | 告别数据“强行整合”!scHiCAR实现单细胞转录、表观与三维基因组的“真·同源”解析
Science | 破解呼吸道机械清除屏障:百日咳杆菌靶向宿主细胞微管的动态感染策略
Cell | 记忆衰退的“解药”藏在肝脏里?揭示运动逆转大脑老化的血管密码
Nature | 攻防的极致博弈:个体化mRNA疫苗激发三阴性乳腺癌持久免疫
Cell | 增殖还是耐药?深度解析胰腺癌细胞如何利用ECM感知在“建设者”与“幸存者”间灵活切换
Nature Biotechnology | 自身免疫病的“一次性治愈”曙光:细胞免疫疗法的范式转移
Nature | 结构生物学的“暗物质”觉醒:利用生成式AI填补AlphaFold在无序蛋白(IDR)预测中的空白
2026-03-11
·同花顺
据石家庄发布消息,位于石家庄高新区的华普生物疫苗产业化生产基地项目一期正加紧建设,预计2027年建成投产后将具备年产数千万剂次疫苗的能力。该项目总投资达52亿元,规划占地177亩,是河北省重点项目,旨在推动疫苗产业升级,让更多创新疫苗从石家庄走向世界。华普生物是一家深耕免疫学领域20余年的国家高新技术企业,自2022年落户石家庄高新区以来,已建立TLR9激动剂CpG技术平台,建成“百公斤级”CpG产业化生产基地,可满足全球CpG佐剂供应需求。CpG佐剂作为该企业的核心技术,被称为下一代疫苗的“发动机”,能推动疫苗产业从传统技术向精准免疫调控升级。该项目分三期打造疫苗生产“超级矩阵”。其中一期投资13亿元,建设疫苗原液车间、制剂中心、质检中心等。随着项目加速建成,使用华普生物CpG佐剂的带状疱疹疫苗、乙肝疫苗和流感疫苗将实现产业化量产,石家庄将形成从佐剂生产到疫苗研发的完整产业链条。华普生物已建立起完善的CpG技术平台,拥有42项授权国际发明专利,并联合中国食品药品检定研究院起草撰写《疫苗用CpG ODN佐剂质量控制研究技术共识》,填补了新型佐剂质量控制标准的行业空白。企业表示将以项目建设为契机,持续加大研发投入,依托石家庄生物医药产业集群优势,深耕CpG佐剂技术迭代和疫苗研发,为“中国好药·石家庄造”品牌添彩,为石家庄高质量发展注入新动能。原文:让更多创新疫苗从石家庄走向世界——华普生物疫苗产业化生产基地项目一期建设稳步推进(来源:石家庄发布)
关注同花顺财经(ths518),获取更多机会
疫苗临床3期
2026-03-11
·搜狐新闻
春日融融,万物竞发。3月10日,走进位于石家庄高新区的华普生物疫苗产业化生产基地项目现场,工程车辆往来穿梭,建设者们正抢抓春日施工黄金期,全力以赴赶工期、抢进度,现场一派热火朝天的施工景象。“眼下正在施工的是华普生物疫苗产业化生产基地项目的一期,预计2027年建成,投产后将具备年产数千万剂次疫苗的能力。”华普生物副总经理刘光辉站在项目展板前,向记者描绘着未来的蓝图,“作为河北省重点项目,这个项目建成后,这里将崛起一座现代化的疫苗生产车间,让更多创新疫苗从石家庄走向世界。”这个总投资达52亿元、规划占地177亩的重点项目,究竟有何特别之处?为何能成为石家庄生物医药产业高质量发展的“标杆项目”?“华普生物疫苗产业化生产基地项目是我们布局疫苗产业化的核心载体,是推动疫苗产业升级的关键一步。”华普生物副总经理刘光辉给出了答案。作为一家深耕免疫学领域20余年的国家高新技术企业,华普生物自2022年落户石家庄高新区以来,建立了TLR9激动剂CpG技术平台,建成了“百公斤级”CpG产业化生产基地,可满足全球CpG佐剂供应需求。“CpG佐剂就是我们的核心技术,它被称为下一代疫苗的‘发动机’,能推动疫苗产业从传统技术向精准免疫调控升级。”刘光辉说,与沿用百年的传统铝佐剂相比,这种人工合成的寡脱氧核苷酸,可激活体内天然免疫和获得性免疫,大幅提高人体免疫应答。看似小小的佐剂,背后是数十年的技术攻坚。经过三次迭代开发,华普生物已建立起完善的CpG技术平台,拥有42项授权国际发明专利,联合中国食品药品检定研究院起草撰写《疫苗用CpG ODN佐剂质量控制研究技术共识》,填补了新型佐剂质量控制标准的行业空白,为全球疫苗安全、有效、可及贡献了“中国智慧”。而这座正在加速建设的产业化基地,正是将华普的创新成果转化为民生福祉的关键载体。据介绍,基地分三期打造疫苗生产“超级矩阵”,其中一期投资13亿元,建设疫苗原液车间、制剂中心、质检中心等,随着项目的加速建成,使用华普生物CpG佐剂的带状疱疹疫苗、乙肝疫苗和流感疫苗将实现产业化量产,石家庄将形成从佐剂生产到疫苗研发的完整产业链条。春光正好,实干正当时。刘光辉表示,华普生物将以项目建设为契机,持续加大研发投入,依托石家庄生物医药产业集群优势,深耕CpG佐剂技术迭代和疫苗研发,力争让更多创新药在石家庄落地生根,为“中国好药·石家庄造”品牌添彩,为石家庄高质量发展注入新的强劲动能。石家庄发布(shijiazhuangfabu)运营|石家庄广播电视台新媒体中心来源|石家庄日报记者 宋子婷编辑|李雪莹 王圆圆责编|胡光返回搜狐,查看更多
疫苗临床3期寡核苷酸
100 项与 GNKG-168 相关的药物交易
登录后查看更多信息
研发状态
10 条进展最快的记录, 后查看更多信息
登录
| 适应症 | 最高研发状态 | 国家/地区 | 公司 | 日期 |
|---|---|---|---|---|
| 复发性急性淋巴细胞白血病 | 临床1期 | 美国 | 2014-03-26 | |
| 治疗相关性急性髓系白血病 | 临床1期 | 美国 | 2014-03-26 | |
| 慢性淋巴细胞白血病 | 临床1期 | 美国 | 2009-09-01 |
登录后查看更多信息
临床结果
临床结果
适应症
分期
评价
查看全部结果
| 研究 | 分期 | 人群特征 | 评价人数 | 分组 | 结果 | 评价 | 发布日期 |
|---|
No Data | |||||||
登录后查看更多信息
转化医学
使用我们的转化医学数据加速您的研究。
登录
或

药物交易
使用我们的药物交易数据加速您的研究。
登录
或

核心专利
使用我们的核心专利数据促进您的研究。
登录
或

临床分析
紧跟全球注册中心的最新临床试验。
登录
或

批准
利用最新的监管批准信息加速您的研究。
登录
或

特殊审评
只需点击几下即可了解关键药物信息。
登录
或

生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用


