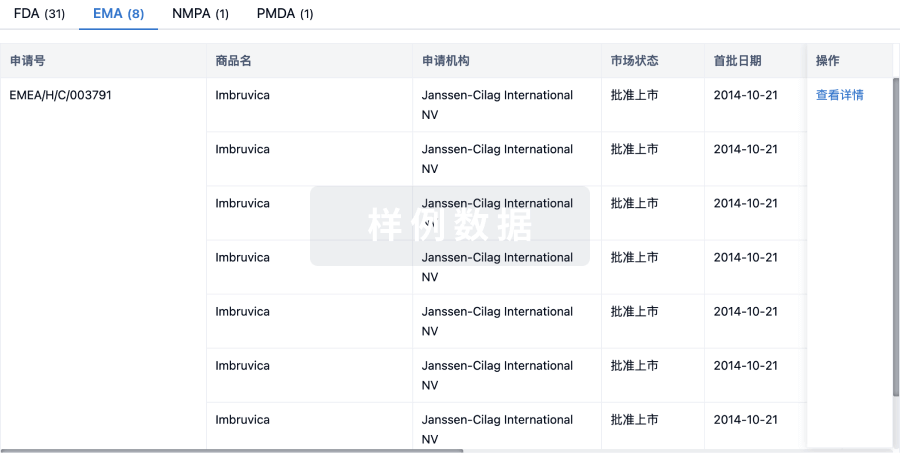
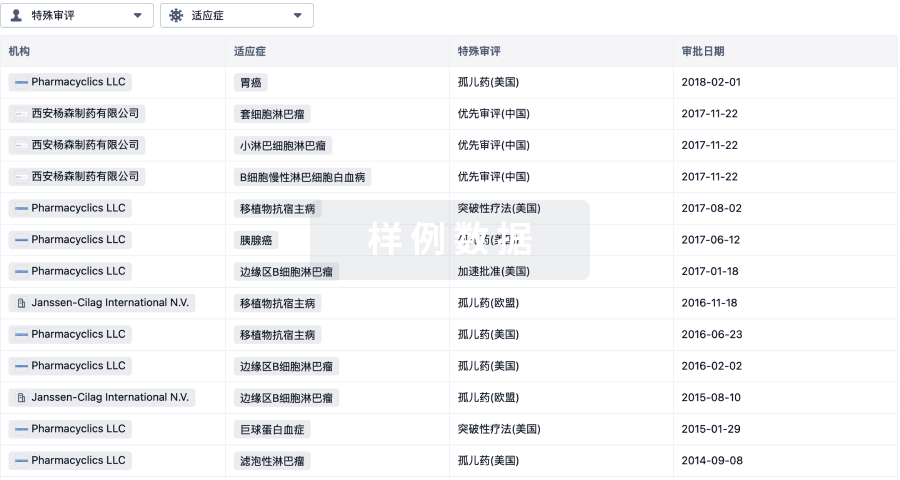
1
项与 Autologous adipose derived MSCs therapy(Arkansas Heart Hospital) 相关的临床试验Phase I/Phase II Safety and Preliminary Efficacy Study Using Low Frequency Ultrasound in Addition to Adipose Derived Stem Cells (ADSCs) in Patients With Moderate to Severe Lower Extremity Peripheral Arterial Disease (PAD)
This is a single center, open label, prospective, randomized, controlled study in patients with non- revascularizable moderate or severe lower extremity Peripheral Arterial Disease (PAD) receiving autologous stem cells therapy (minimally manipulated). This study will enroll a maximum of 10 subjects in this study. The patients will be divided into two groups. One group will receive Adipose Derived Stem Cells (ADSCs) alone and the other group will receive low frequency ultrasound before the addition of ADSCs. The randomization will be 1:1. If the results of this pilot study are not clearly different, there will be a need for a phase II study to support the results.
Baseline, 6 week, 3 month and 6 month assessments of 6 min walking distance (6MWT), Ankle Brachial Indexes (ABIs)/Toe Brachial Indexes (TBIs), Transcutaneous Partial Oxygenation (TcPO2) and Rutherford assessments will be performed for the study group. Prior to the stem cell treatment, the ultrasound group patient will receive noninvasive transcutaneous pulsed focused ultrasound around the involved vessel(s) in the affected extremity. All patients will then receive 200 million autologous stromal vascular fraction cells containing adipose derived stem cell therapy: 1/3 of the cells will be delivered intra-venous proximal to the lesion (not retrograde), 1/3 of the cells will be delivered intra- adventitia proximal to the lesion and 1/3 of the cells will be delivered by intra- muscular injection along the vessel path (20-30 injections each separated by1.5 inch). Standard therapy for PAD patients will not be interrupted for the duration of the study. Standard therapy for PAD is defined as cardiovascular general measures like lifestyle changes, smoke cessation, exercise, cardiovascular rehabilitation, etc. All participants have already received maximal non-surgical therapy. Safety will be monitored on an ongoing basis.
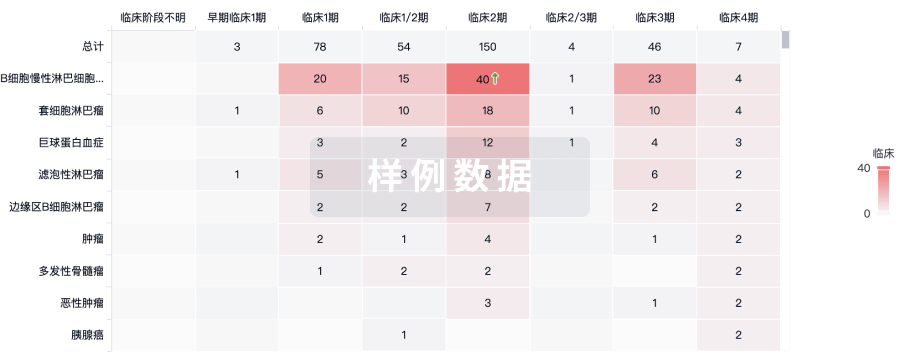
100 项与 Autologous adipose derived MSCs therapy(Arkansas Heart Hospital) 相关的临床结果
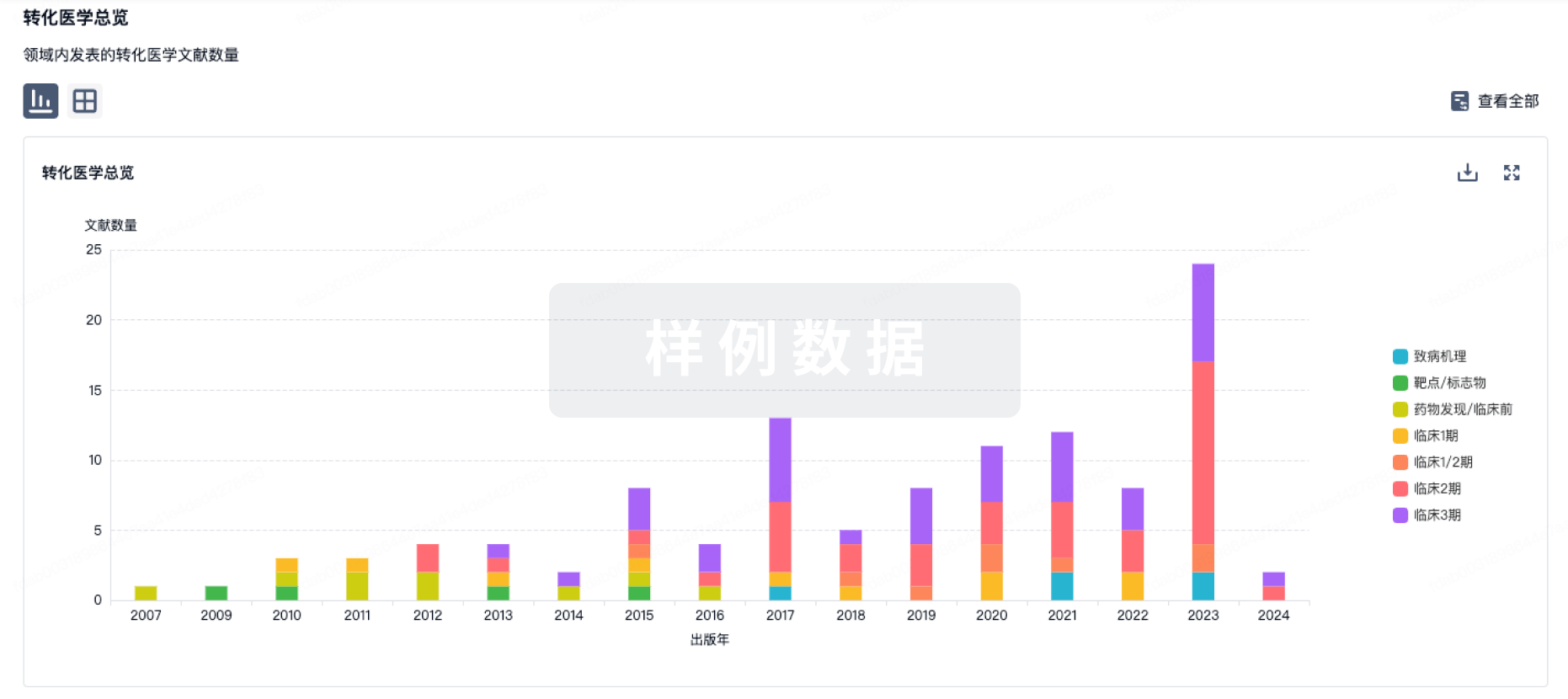
100 项与 Autologous adipose derived MSCs therapy(Arkansas Heart Hospital) 相关的转化医学
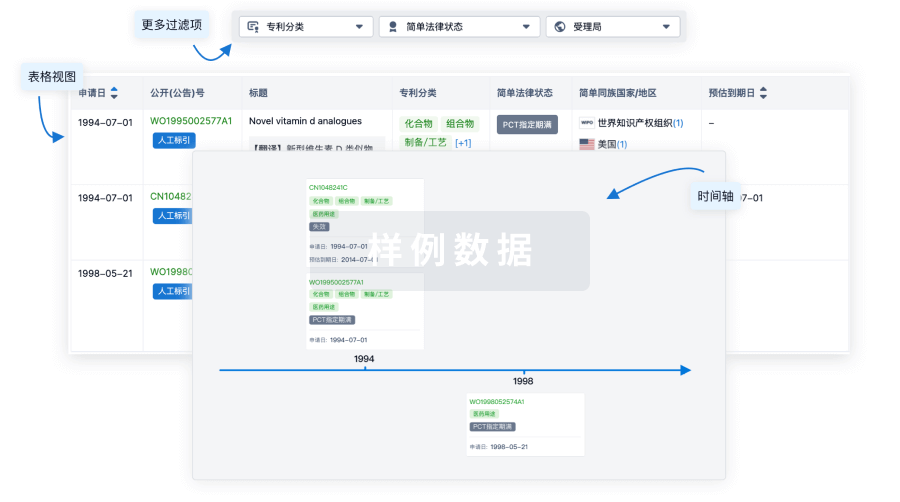
100 项与 Autologous adipose derived MSCs therapy(Arkansas Heart Hospital) 相关的专利(医药)
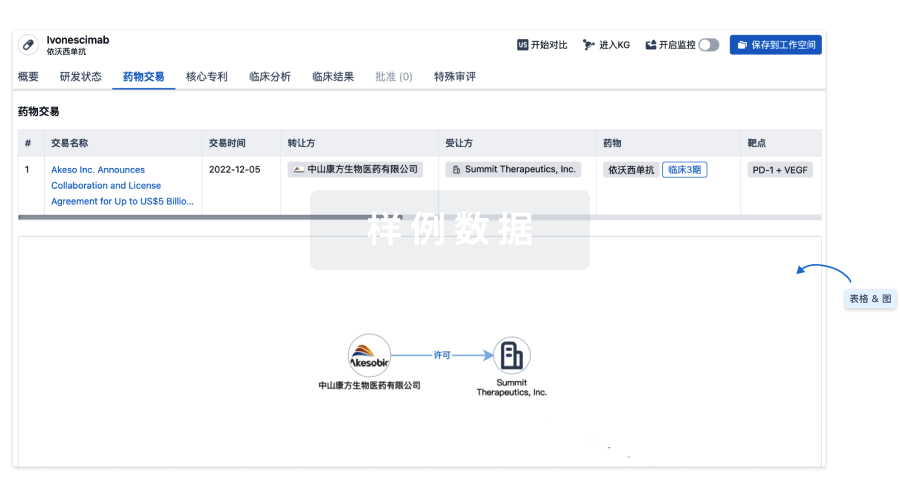
100 项与 Autologous adipose derived MSCs therapy(Arkansas Heart Hospital) 相关的药物交易