2
项与 E6 TCR cells(NCI) 相关的临床试验A Phase I Study of Immunotherapy With E6 T Cell Receptor T Cells for Vulvar High-Grade Squamous Intraepithelial Lesions
Background:
Vulvar high-grade squamous intraepithelial lesion (HSIL) is caused by infection of the vulva with human papillomavirus (HPV). In a small percentage of cases, vulvar HSIL can turn into cancer. The risk of cancer can be reduced by treating HSIL. A personalized immune treatment might rid the body of HPV infection and thereby cure vulvar HSIL. The immune treatment in this study is called T cell therapy. The cells are E6 T Cell Receptor (TCR) T cells. Participants will also get aldesleukin (IL-2) to help the cells last longer.
Objective:
To find a safe dose of E6 TCR T cells combined with aldesleukin to use in people with vulvar HSIL.
Eligibility:
Design:
Participants will be screened with:
Physical exam
Medical history
Blood, lab, and pregnancy tests
Heart tests
Chest x-ray
Sample of tissue taken from the vulva (biopsy).
Participants will have leukapheresis. Blood will be removed by a needle in one arm. A machine removes white blood cells from the blood. The rest of the blood is returned by needle in the other arm. The white blood cells will be changed into E6 TCR T cells and grown in a lab. About 3 weeks later, participants will be admitted to the hospital for about 5 days. They will get the cells through a tube placed in a vein. They will get IL-2 the same way. Participants will recover 1-3 days in the hospital. They will be monitored closely. They will have blood and lab tests. Participants will have follow-up visits with lab tests and a physical exam every few months for 5 years. At some visits they will also have leukapheresis, blood tests, or vulvar biopsy.
A Phase I/II Study of T Cell Receptor Gene Therapy Targeting HPV-16 E6 for HPV-Associated Cancers
Background:
The National Cancer Institute (NCI) Surgery Branch has developed an experimental therapy for treating patients with cancer that involves taking white blood cells from the patient, growing them in the laboratory in large numbers, genetically modifying these specific cells with a type of virus (retrovirus) to attack only the tumor cells, and then giving the cells back to the patient. This type of therapy is called gene transfer. Researchers want to test this on human papilloma virus (HPV)-associated cancers.
Objective:
- The purpose of this study is to determine a safe number of these cells to infuse and to see if these particular tumor-fighting cells (Anti-HPV E6) can shrink tumors associated with HPV and test the toxicity of this treatment.
Eligibility:
- Adults age 18-66 with an HPV-16-associated cancer.
Design:
Work up stage: Patients will be seen as an outpatient at the National Institutes of Health (NIH) clinical Center and undergo a history and physical examination, scans, x-rays, lab tests, and other tests as needed
Leukapheresis: If the patients meet all of the requirements for the study they will undergo leukapheresis to obtain white blood cells to make the anti HPV E6 cells. {Leukapheresis is a common procedure, which removes only the white blood cells from the patient.}
Treatment: Once their cells have grown, the patients will be admitted to the hospital for the conditioning chemotherapy, the anti HPV E6 cells and aldesleukin. They will stay in the hospital for about 4 weeks for the treatment.
Follow up: Patients will return to the clinic for a physical exam, review of side effects, lab tests, and scans about every 1-3 months for the first year, and then every 6 months to 1 year as long as their tumors are shrinking. Follow up visits take up to 2 days.
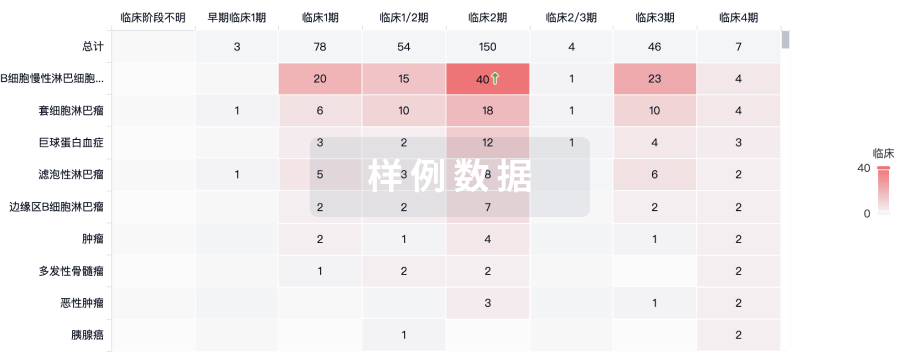
100 项与 E6 TCR cells(NCI) 相关的临床结果
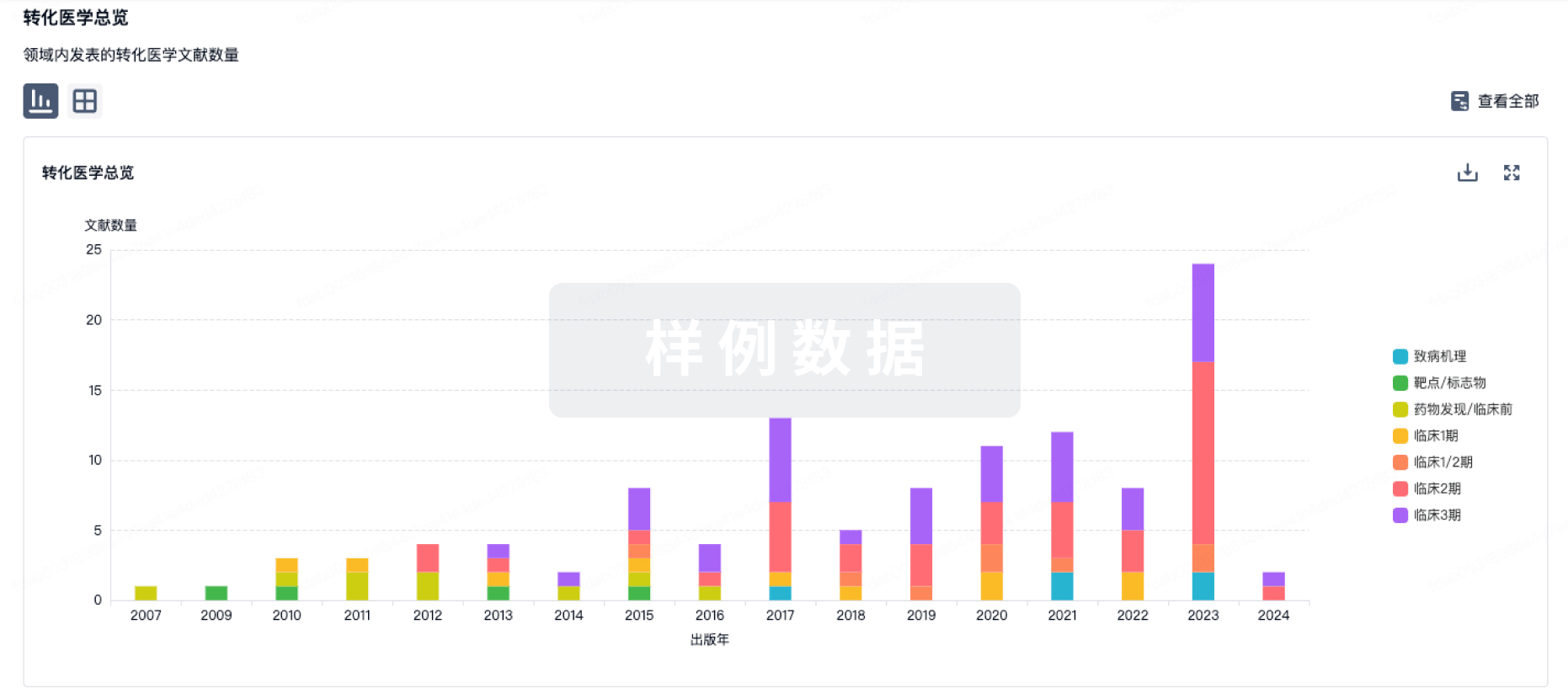
100 项与 E6 TCR cells(NCI) 相关的转化医学
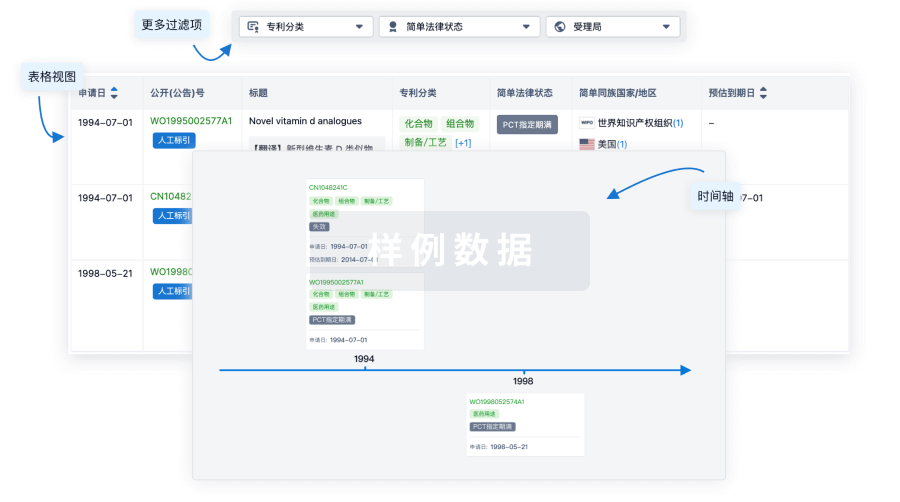
100 项与 E6 TCR cells(NCI) 相关的专利(医药)
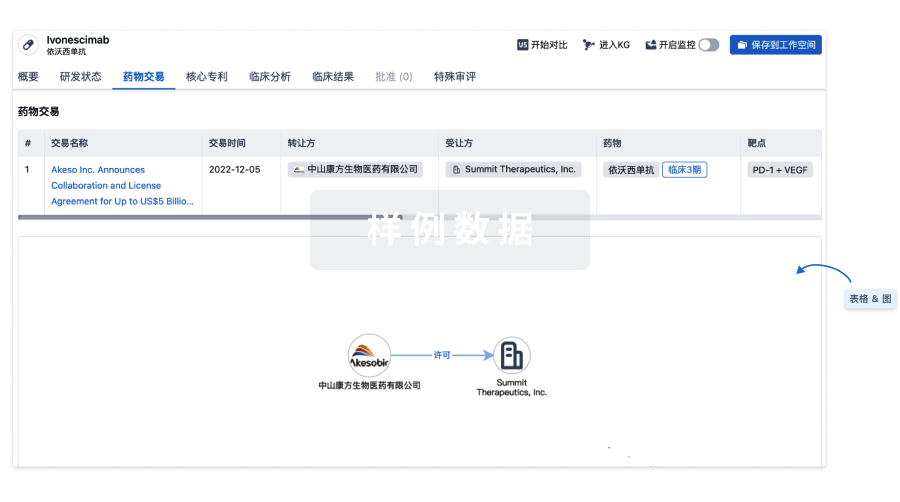
100 项与 E6 TCR cells(NCI) 相关的药物交易