更新于:2024-05-16
-
更新于:2024-05-16
概要
关联
2
项与 相关的临床试验Phase 1 Dose Escalating, Double-Blind, Randomized Comparator Controlled Trial of the Safety andImmunogenicity of Pfs25M-EPA/AS01 and Pfs230D1M-EPA/AS01 Vaccines, Transmission Blocking Vaccines Against Plasmodium Falciparum at Full and Fractional Dosing in Adults in Mali
Background:
Researchers are looking for new ways to control and eradicate malaria. They want to test vaccines to block malaria transmission in adults in Mali. These vaccines work by inducing antibodies in a person. The antibody is then taken up with blood by a mosquito that bites the person. This blocks parasite development in the mosquito. This stops malaria transmission to another person.
Objective:
To test the safety, reactogenicity, immunogenicity, and transmission-blocking activity of the vaccines Pfs25M-EPA and Pfs230D1M-EPA with AS01 in Malian adults.
Eligibility:
Healthy Malians ages 18-50 living in certain areas in Mali who:
Are not pregnant or breastfeeding
Are not infected with HIV, Hepatitis B and Hepatitis C
Do not have evidence of immunodeficiency
Do not have history of severe allergic reaction or anaphylaxis
Design:
Participants will be screened with:
Medical history
Physical exam
Malaria Comprehension Exam
Blood and urine tests
Electrocardiogram (for participants in certain study groups)
Participants will be randomly assigned to a study group.
Participants will be monitored for 12-16 months. For the first 7 months, they will have between 1 and nine visits a month. The number depends on the month and on what group they are in. For the rest of the months, they will have 1 monthly visit.
Each visit includes a physical exam. Most include blood tests.
Participants will get 3 doses of a study or comparator vaccine. They get the vaccine through an injection in the upper arm. This occurs at their first visit, then 1 month later, and then 5 months later.
Participants will be followed for at least 6 months after the last vaccine.
If participants develop an injection site rash or reaction, photographs may be taken of the site.
Researchers are looking for new ways to control and eradicate malaria. They want to test vaccines to block malaria transmission in adults in Mali. These vaccines work by inducing antibodies in a person. The antibody is then taken up with blood by a mosquito that bites the person. This blocks parasite development in the mosquito. This stops malaria transmission to another person.
Objective:
To test the safety, reactogenicity, immunogenicity, and transmission-blocking activity of the vaccines Pfs25M-EPA and Pfs230D1M-EPA with AS01 in Malian adults.
Eligibility:
Healthy Malians ages 18-50 living in certain areas in Mali who:
Are not pregnant or breastfeeding
Are not infected with HIV, Hepatitis B and Hepatitis C
Do not have evidence of immunodeficiency
Do not have history of severe allergic reaction or anaphylaxis
Design:
Participants will be screened with:
Medical history
Physical exam
Malaria Comprehension Exam
Blood and urine tests
Electrocardiogram (for participants in certain study groups)
Participants will be randomly assigned to a study group.
Participants will be monitored for 12-16 months. For the first 7 months, they will have between 1 and nine visits a month. The number depends on the month and on what group they are in. For the rest of the months, they will have 1 monthly visit.
Each visit includes a physical exam. Most include blood tests.
Participants will get 3 doses of a study or comparator vaccine. They get the vaccine through an injection in the upper arm. This occurs at their first visit, then 1 month later, and then 5 months later.
Participants will be followed for at least 6 months after the last vaccine.
If participants develop an injection site rash or reaction, photographs may be taken of the site.
Phase 1 Study of the Safety and Immunogenicity of Pfs230D1M-EPA/Alhydrogel and Pfs25M-EPA/Alhydrogel, a Transmission Blocking Vaccine Against Plasmodium Falciparum Malaria in Adults in the US and Mali
Background:
- Malaria is a severe infection caused by a parasite. People can get malaria if a mosquito that carries the parasite bites them. Malaria infection does not happen in the United States, but many people in Africa, Asia, and South America are at risk for it. Researchers want to test two vaccines that may help decrease malaria infection.
Objective:
- To see if two vaccines (Pfs25M-EPA/Alhydrogel and Pfs230DIM-EPA/Alhydrogel ) are safe in humans and cause an immune response that will prevent malaria parasites from correctly growing in the mosquito.
Eligibility:
- Healthy adults ages 18 50.
Design:
There are several groups in this study. Each group will receive a different dose of the vaccine and some groups will received both vaccines.
Vaccinations will be given on two days about 4 weeks apart.
Participants will receive each vaccine as an injection into the arm. Blood will be drawn on the day of vaccination.
In the 4 weeks after receiving a vaccination, participants will have at least 3 clinic visits and 1 phone contact. They will have at least 3 more visits and 3 phone contacts over the next 6 months.
At each visit, participants will be evaluated for side effects to the vaccine and any new health changes or problems. They will be asked how they are feeling and if they have taken any medicine. Blood and urine samples may be taken at the visit. More follow-up visits may be needed to follow up on changes or problems.
- Malaria is a severe infection caused by a parasite. People can get malaria if a mosquito that carries the parasite bites them. Malaria infection does not happen in the United States, but many people in Africa, Asia, and South America are at risk for it. Researchers want to test two vaccines that may help decrease malaria infection.
Objective:
- To see if two vaccines (Pfs25M-EPA/Alhydrogel and Pfs230DIM-EPA/Alhydrogel ) are safe in humans and cause an immune response that will prevent malaria parasites from correctly growing in the mosquito.
Eligibility:
- Healthy adults ages 18 50.
Design:
There are several groups in this study. Each group will receive a different dose of the vaccine and some groups will received both vaccines.
Vaccinations will be given on two days about 4 weeks apart.
Participants will receive each vaccine as an injection into the arm. Blood will be drawn on the day of vaccination.
In the 4 weeks after receiving a vaccination, participants will have at least 3 clinic visits and 1 phone contact. They will have at least 3 more visits and 3 phone contacts over the next 6 months.
At each visit, participants will be evaluated for side effects to the vaccine and any new health changes or problems. They will be asked how they are feeling and if they have taken any medicine. Blood and urine samples may be taken at the visit. More follow-up visits may be needed to follow up on changes or problems.
100 项与 相关的临床结果
登录后查看更多信息
100 项与 相关的转化医学
登录后查看更多信息
100 项与 相关的专利(医药)
登录后查看更多信息
100 项与 相关的药物交易
登录后查看更多信息
临床结果
临床结果
适应症
分期
评价
查看全部结果
| 研究 | 分期 | 人群特征 | 评价人数 | 分组 | 结果 | 评价 | 发布日期 |
|---|
No Data | |||||||
登录后查看更多信息
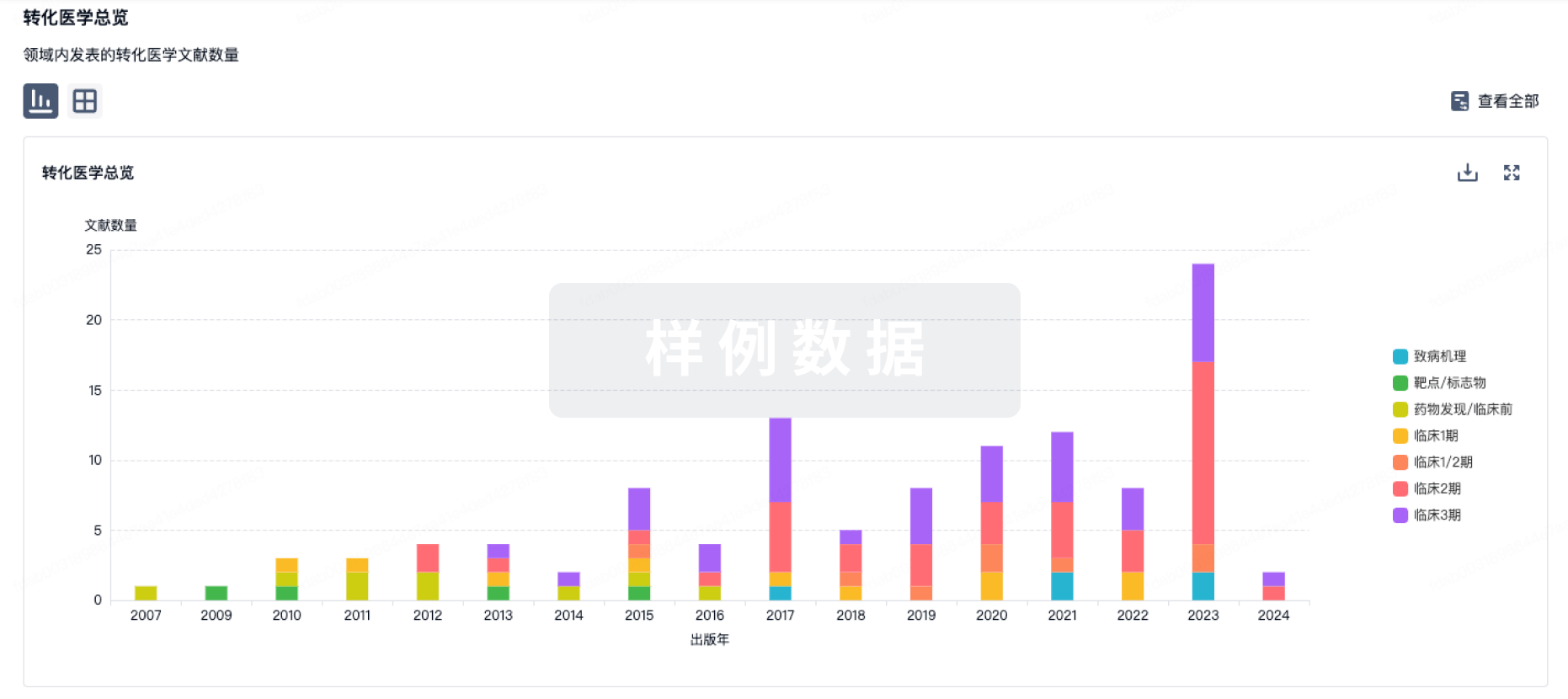
转化医学
使用我们的转化医学数据加速您的研究。
登录
或
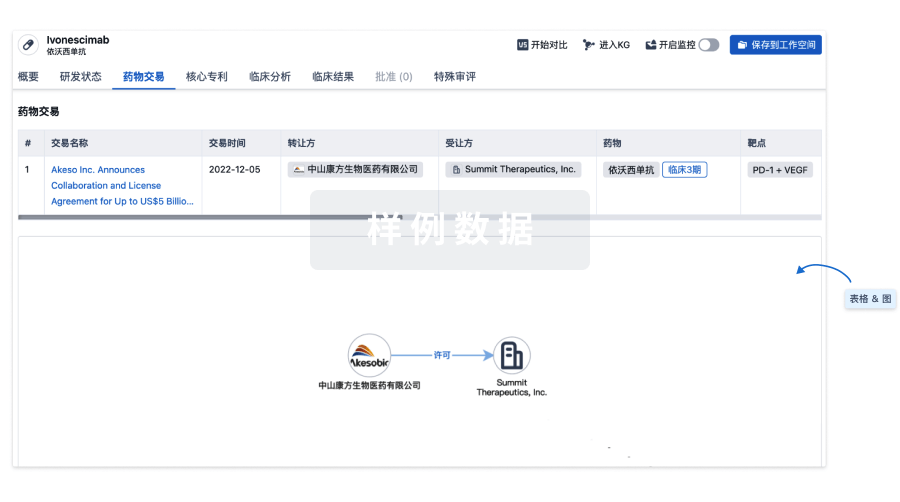
药物交易
使用我们的药物交易数据加速您的研究。
登录
或
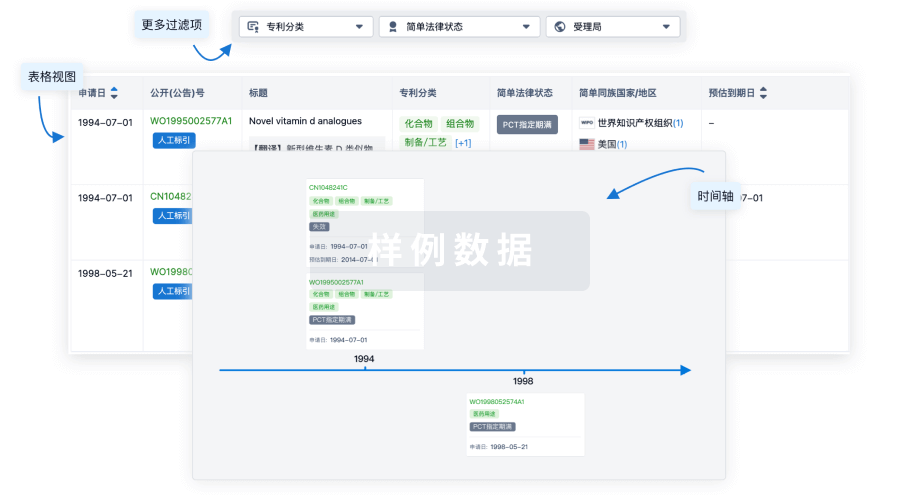
核心专利
使用我们的核心专利数据促进您的研究。
登录
或
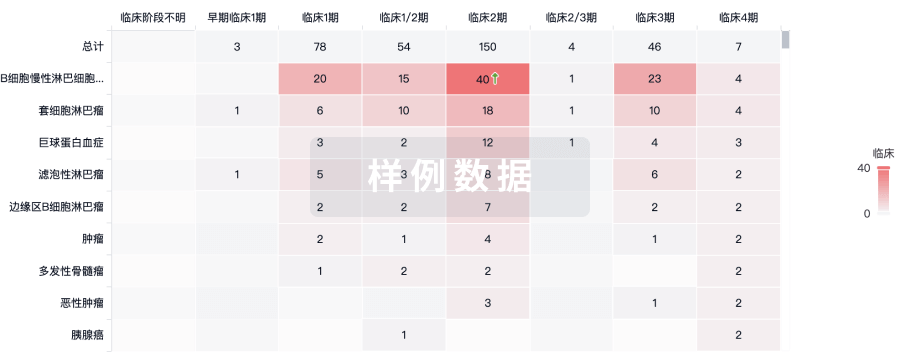
临床分析
紧跟全球注册中心的最新临床试验。
登录
或
批准
利用最新的监管批准信息加速您的研究。
登录
或
特殊审评
只需点击几下即可了解关键药物信息。
登录
或
标准版
¥16800
元/账号/年
新药情报库 | 省钱又好用!
立即使用
来和芽仔聊天吧
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用