更新于:2023-04-29
Heilongjiang Agriculture Co., Ltd.
更新于:2023-04-29
概览
关联
100 项与 黑龙江北大荒农业股份有限公司 相关的临床结果
登录后查看更多信息
0 项与 黑龙江北大荒农业股份有限公司 相关的专利(医药)
登录后查看更多信息
5
项与 黑龙江北大荒农业股份有限公司 相关的文献(医药)2018·Journal of Plant Nutrition
Effects of low-molecular-mass organic acids on P nutrition and some plant properties of Hordeum vulgare
作者: Oral, Adem ; Uygur, Veli
One of the strategies of the plants growing in phosphorus (P)-deficient environments is to exudate low-mol.-weight organic acids (LMWOA). Thus, we aimed to investigate the effect of LMWOA on phosphorus uptake of barley from either fertilizer or inherited soil phosphorus. The experiment was set up in full factorial arrangement in completely randomised design with two phosphorus (0 and 50 mg P kg-1), five organic acids, LMWOA (malic acid, oxalic acid, citric acid, acetic acid, ascorbic acid), and four organic acid rates (0, 10, 20, and 30 mmol kg-1). The effects of LMWOA on yield in descending order were: oxalic acid > ascorbic acid > malic acid > acetic acid > citric acid. The maximum P concentration in grain was obtained at 30 mmol kg-1 LMWOA treatments. As a result, it was found that oxalic acid was the most effective LMWOA in increasing nutrient uptake induced grain yield with and without phosphorous fertilizer application.
2015-09-01·Proceedings of the National Academy of Sciences of the United States of America1区 · 综合性期刊
Hybrid mimics and hybrid vigor in Arabidopsis
1区 · 综合性期刊
Article
作者: Wang, Li ; Greaves, Ian K. ; Groszmann, Michael ; Wu, Li Min ; Dennis, Elizabeth S. ; Peacock, W. James
F1 hybrids can outperform their parents in yield and vegetative biomass, features of hybrid vigor that form the basis of the hybrid seed industry. The yield advantage of the F1 is lost in the F2 and subsequent generations. In Arabidopsis, from F2 plants that have a F1-like phenotype, we have by recurrent selection produced pure breeding F5/F6 lines, hybrid mimics, in which the characteristics of the F1 hybrid are stabilized. These hybrid mimic lines, like the F1 hybrid, have larger leaves than the parent plant, and the leaves have increased photosynthetic cell numbers, and in some lines, increased size of cells, suggesting an increased supply of photosynthate. A comparison of the differentially expressed genes in the F1 hybrid with those of eight hybrid mimic lines identified metabolic pathways altered in both; these pathways include down-regulation of defense response pathways and altered abiotic response pathways. F6 hybrid mimic lines are mostly homozygous at each locus in the genome and yet retain the large F1-like phenotype. Many alleles in the F6 plants, when they are homozygous, have expression levels different to the level in the parent. We consider this altered expression to be a consequence of transregulation of genes from one parent by genes from the other parent. Transregulation could also arise from epigenetic modifications in the F1. The pure breeding hybrid mimics have been valuable in probing the mechanisms of hybrid vigor and may also prove to be useful hybrid vigor equivalents in agriculture.
2007-10-20·International Journal of Food Microbiology1区 · 农林科学
Mycotoxins in rice
1区 · 农林科学
Review
作者: Tanaka, Kenji ; Sago, Yuki ; Zheng, Yazhi ; Nakagawa, Hiroyuki ; Kushiro, Masayo
Mycotoxin contamination in rice is usually lower as in wheat or corn. However, there are some reports that rice has been contaminated with mycotoxins such as aflatoxin B1, B2, G1, G2 (AFS), citrinin, deoxynivalenol (DON), fumonisin B1, B2, B3 (FMS), fusarenon-X (Fus.-X), nivalenol (NIV), ochratoxin A (OTA), sterigmatocystin (STE), and zearalenone. Rice in Japan is preserved in warehouses where moisture content and temperature are regulated. Therefore, mycotoxin contamination from post harvest fungal growth occurs very seldom. Trichothecenes, aflatoxins, and STE in rice were recently analyzed in our laboratory. In 1998, a typhoon struck before rice harvesting in Japan, and the unpolished rice was found to be stained brown. Samples were collected and analyzed for the presence of trichothecenes. Mycotoxins DON, Fus.-X, and NIV were detected and confirmed with GC-MS. The quantity of trichothecenes was determined using GC-ECD. STE is a carcinogenic mycotoxin produced by Aspergillus versicolor and some other fungi. STE contamination of rice was studied in our laboratory since 1973. GC-MS, LC-MS, LC-MS/MS, and LC-UV methods for STE determination were examined, giving good results for the LC-UV method using a photo diode array detector. Different techniques for the extraction of STE from rice were also studied. Finally, brown rice was ground, and the ground rice was extracted with acetonitrile-water. An Autoprep MF-A 1000 column was used to clean up AFS and STE. The cleaned-up extract was analyzed with HPLC-UV. Forty-eight brown rice samples were analyzed, and none of them were contaminated with STE. These rice samples were also analyzed for AFS and FMS, and none of the samples were contaminated. The Ministry of Agriculture, Forestry and Fisheries in Japan is making the appropriate Institutes develop analytical methods for mycotoxins and survey mycotoxin contamination on rice as well as wheat, corn, and some other cereals.
7
项与 黑龙江北大荒农业股份有限公司 相关的新闻(医药)2022-11-29
TORONTO, Nov. 29, 2022 (GLOBE NEWSWIRE) -- Mountain Valley MD Holdings Inc. (the “Company” or “MVMD”) (CSE: MVMD) (OTCQB: MVMDF) (FRA: 20MP) is pleased to provide a business update and details of its current commercialization strategy. MVMD is an early-stage biotech business which, since inception, has focused its resources on investing in multiple research and development (“R&D”) initiatives. The Company is pleased to confirm that it has shifted focus to the commercialization strategy for those of its projects and technologies that it believes will best provide viable revenue streams for long-term company health and shareholder value. There are three primary lines of business MVMD is currently working towards commercial advancement in: (1) novel innovations that improve the administration and efficacy of nutraceutical health and wellness products; (2) agricultural plant signaling technology that organically drives increases in crop yields and supports the reduction of fertilizer usage; and (3) the application of solubilized drugs to positively impact husbandry animal health. “This is a significant milestone for MVMD as we transition from an R&D-focused biotech start-up business into a commercially focused biotech entity with revenue-generating activities,” stated Dennis Hancock, President & CEO of MVMD. “We believe we have identified a great pathway to generate positive cashflow as early as the 2023 calendar year that will support our goal to create shareholder value. I believe this next phase of growth for MVMD will be an exciting validation of our progress.” In support of its commercialization efforts, MVMD holds a license from Agrarius Corp. for the rights to distribute its Agrarius plant signaling technology, and currently has two wholly owned technologies; a) patented Quicksome™ molecule formulation and delivery technology, and b) patented Quicksol™ solubility formulation and delivery technology. LINE OF BUSINESS UPDATES: Nutraceuticals As previously disclosed, following evaluations of North American GMP manufacturing options for MVMD’s nutraceutical product strategy, the Company entered into a license agreement with its selected third-party lead production partner in the United States. The Company strategically secured its lead manufacturing partner as a licensee, who will in turn produce nutraceutical products based on, or embodying, MVMD’s proprietary technologies for third parties, who will then be approved by MVMD and who will have an agreement with Company. The Company believes this strategy will help to ensure product quality, support the ability to scale production, streamline the audit process for royalty agreements, and provide the necessary protection of its technology and trade secrets, versus having numerous licensed partners, each replicating the manufacturing process for their own products. Securing the lead manufacturer and finalizing the scaled GMP production environment aligns with MVMD’s anticipated increased business development efforts and the Company’s plans to secure additional nutraceutical licensing partnerships for production in the 2023 calendar year. This timeline has been updated from the anticipated late 2022 date due to the licensed third-party lead production partner’s current process to establish a scaled GMP production line capable of larger volume outputs, including the installation of equipment to support the required manufacturing process that embeds MVMD’s Quicksome™ formulation approach. MVMD’s production partner has been working to support the Company’s license agreement with Circadian Wellness Corp. (“Circadian”) by way of finalizing the proprietary formulations for mushroom-infused products Circadian intends to market under its Eons brand. Circadian’s license from MVMD supports the desire to achieve an increase in overall molecule efficacy with the Company’s Quicksome™ technology applied across a variety of rapid dissolve sublingual and dermal products in the functional mushroom space. The initial product formulation work and dissolution testing has been positive in MVMD’s view. In early November, Circadian launched its initial products in the nutraceutical space for the United States market: a mushroom infused “smart coffee” and a chewable gummy product designed to aid in sleep. The focus in the next product phase for the Eons brand is mushroom-infused sublingual sleep and energy products and a pain management cream that embodies MVMD’s technology. Although outside of MVMD’s control, it is anticipated that Circadian will be introducing consumer products that embody MVMD’s technology under its Eons brand for sale in the United States in the first half of the 2023 calendar year. Agriculture In September 2022, the Company announced the initiation of commercial sales activity and related business development investments to support in-field trialing of Agrarius, a certified organic product owned by Agrarius Corp. (“AC”). AC isa private US corporation in which the Company holds an equity interest and from which it has licensed certain rights regarding the distribution of AC’s product, Agrarius . The Agrarius product is delivered in a liquid concentrate form that gets mixed with water at the point of application and then applied via sprayer to agricultural crop plants ideally twice during a plant’s lifecycle. Agrarius has been tested across numerous major agricultural crops and has demonstrated its ability to naturally increase crop yields from approximately ten to fifty percent or more depending on crop type, reduce fertilizer usage by more than thirty percent, and increase general resilience to pests and climate change forces such as drought. “We believe Agrarius presents a significant opportunity to support MVMD’s growth objectives given the rapidly changing global agricultural landscape,” continued Mr. Hancock. “With the significant global agricultural pressures we are seeing with mass fertilizer shortages, unprecedented climate change impact, and the urban sprawl limiting available farmlands, we believe we are in the perfect place to positively help address some of the key global food supply issues.” The Agrarius product works by activating the plants’ “defense mechanisms” at the cellular level, without the actual stress factor. The intended effect of Agrarius is that treated plants grow deeper roots and open up their foliage to optimize the effect of photosynthesis, thus increasing growth hormones, plant efficiency for water use and nutrients, decreasing the requirement of fertilizer where used, and increasing overall resistance to diseases and stressed climate conditions. Agrarius technology enables direct plant communication by mimicking naturally occurring organic molecules that correspond to various plant survival instincts, which improves the natural defense mechanisms of the plant to overcome environmental stresses. Agrarius works by sending signals that a plant understands and propagates through its roots and mycelial networks, allowing nearby plants to benefit from the signals naturally while reducing the actual amount of product that is used on a field. These signals literally send information that stimulates plant growth, subsequently increasing yields, volatility resistance, and growth speeds. By virtue of its arrangement, MVMD is an authorized reseller of Agrarius products and has commenced business development activity with large scale farming operations to induce trial activity on crops that would demonstrate the benefit to improving crop yields and the corresponding reduction in fertilizer usage where used in the specific farming operation. Additionally, the Company is working through a strategy of targeting large food supply businesses where significant purchasing power of agricultural products exists. The Company anticipates revenue from Agrarius sales activity in the 2023 calendar year related to the crop trialing cycle, whereby farming organizations would personally validate the anticipated positive impact on their crop yields and/or fertilizer reduction after it has been trialed on their farm. Revenue and the related ramp-up of sales growth in many cases is anticipated to take one full crop cycle. The Company currently facilitates Agrarius product information and product trialing and order flow through its wholly owned and developed website IncreaseCropYields.com, including translation for Spanish and Portuguese languages. The website presents product benefits, enables clients to calculate crop-specific ROI and facilitates product trialing and purchase processes. Husbandry Animals / Aquatic Species As previously disclosed, the Company has applied its Quicksol™ technology to the drug ivermectin and believes a more solubilized format versus current in-market products would have novel applications across the broad husbandry animal and aquatic species marketplace. The Company’s overall husbandry and aquatic species strategy was to develop its scientific assets with a view to licensing to third parties. Subject to final legal review and the Company’s execution of the final licensing agreement, MVMD has finalized the framework negotiations with a third party for licensing of its Soluvec™ 1% product to pursue sales in the husbandry and aquatic species marketplace inside Bangladesh. Related to the framework agreement and necessary government approvals, the pharmacokinetic trials conducted inside Bangladesh across husbandry and aquatic species categories have been completed by a third-party Contract Research Organization (CRO) and MVMD believes the results of these trials will positively support the value proposition necessary to secure necessary government approvals to commercialize inside Bangladesh in the 2023 calendar year. Management believes that commercialization of Soluvec™ 1% inside Bangladesh by the licensee will commence in the first half of the 2023 calendar year, provided the Company’s final legal review and execution of the final licensing agreement is complete. Currently, the Company is also in negotiations with a third party for licensing of its Soluvec™ 1% product worldwide (excluding Bangladesh) to further develop, produce, distribute and sell Soluvec™ 1%, or products embodying the Soluvec™ 1% technology, in the husbandry and aquatic species marketplace. The licensee would also service the production and supply of product to support the Bangladesh license agreement described above. Management believes that this is in line with its manufacturing approach to nutraceuticals, to engage with a limited number of licensees that will be able to produce/commercialize products embodying the Company’s technology, with the aim of allowing for greater protection of MVMD’s intellectual property as well as greater ease in managing licensees and resulting revenue streams. Red White and Bloom Brands In November 2021, the Company entered into an agreement with Red White and Bloom Brands Inc. (“RWB”), a publicly traded company that operates in the cannabis space in the United States. The agreement has been amended to be non-exclusive as RWB is evaluating its skew and product approach going forward and is uncertain at this point in time if RWB will expand their skew line to include MVMD’s technology. The original agreement established the terms upon which the Company’s Quicksome™ technology and novel solubilization techniques could be applied by RWB to various product applications in the space that they operate in. The Company has no immediate control of the decisions made by RWB that would bring any products with MVMD’s technology to market and is not actively pursuing any product applications in the cannabis space at this time. The decision and agreement to change the arrangement to a non-exclusive would allow MVMD to benefit from the arrangement in the future but not limit MVMD's ability to benefit from a similar arrangement with third parties. Research and Development In line with the commercialization phase described above, MVMD is shifting to a more traditional use of capital aligned to revenue growth activities. The Company will continue to assess the ability, viability, and desire to continue to develop its other projects and pre-clinical R&D work, however R&D expenditures are anticipated to be more narrowly focused on business expansion within the above three commercialization focus areas of the business, namely (1) Nutraceuticals, (2) Agricultural, and (3) Husbandry Animals. ABOUT MOUNTAIN VALLEY MD HOLDINGS INC. Mountain Valley MD is building a world-class organization centered around the implementation, licensing and reselling of key technologies and formulations: patented Quicksome™ oral formulation and delivery technologies,patented Quicksol™ solubility formulation technologylicensed product reseller of Agrarius™, a novel agricultural plant signalling technology Consistent with its vision towards “More Life”, MVMD applies its owned and licensed technologies to its work for advanced delivery of molecules for human and husbandry animal applications, including the development of products for pain management, weight loss, energy, focus, sleep, anxiety, and more. Additionally, MVMD’s work with Agrarius is focused on generating a positive impact on crop yields and reducing fertilizer usage. MVMD’s patented Quicksome™ technology utilizes proprietary formulations and stabilizing molecules to encapsulate and formulate active ingredients into highly efficient product formats. The result is a new generation of product formulations that could be capable of delivering nutraceutical and drug molecules into the body faster, with greater impact, efficiency and accuracy. MVMD’s patented Quicksol™ technology covers all highly solubilized macrocyclic lactones that could be effectively applied in multiple viral applications that could positively impact human and animal health globally. MVMD’s licensed Agrairus™ agricultural plant signalling technology that could be capable of application to agricultural crops to naturally increase yields, reduce fertilizer usage, and increase general resilience to pests and climate change. For more Company information and contact details, visit www.MVMD.com.SOURCE: Mountain Valley MD Holdings Inc. www.MVMD.com CAUTIONARY STATEMENT REGARDING FORWARD-LOOKING INFORMATION Certain statements contained in this news release may constitute forward-looking information. Forward-looking information is often, but not always, identified by the use of words such as "anticipate", "plan", "estimate", "expect", "may", "will", "intend", "should", and similar expressions. Forward-looking information involves known and unknown risks, uncertainties and other factors that may cause actual results or events to differ materially from those anticipated in such forward-looking information. The Company's actual results could differ materially from those anticipated in this forward-looking information as a result of regulatory decisions, competitive factors in the industries in which the Company operates, prevailing economic conditions, and other factors, many of which are beyond the control of the Company. The Company is making forward-looking statements, including but not limited to: the transition from an R&D phase to a commercialization phase of the Company’s business; the three (3) business areas of focus selected for the commercialization phase currently; the impact of the foregoing, including that these areas will best provide viable revenue streams for long-term company health and shareholder value; the ability and timing with respect to the generation of revenues; the commercialization phase being a validation of the Company’s progress; the use of a third party—lead production partner for its nutraceuticals business, the involvement of third parties and the direct agreements of such third parties with the Company, and the result of the approach being the protection of MVMD’s intellectual property, the ability to scale production, the positive impact on product quality, and the streamlining of the audit process for royalty agreements; the Company’s business development plans and goals to secure additional nutraceutical partnerships in the 2023 fiscal year; the production of consumer goods by Circadian embodying MVMD’s technology under its Eons brand, and the location and timing of such production and sales; the licensing and distribution by MVMD of Agrarius product and the relationship between such parties, and all matters related thereto, including but not limited to timing and business development efforts and details; the anticipated timing of revenues arising from Agrarius and the connection to the crop trialing cycle; the license arrangement between the Company and a third party with respect to its Quicksol™ technology in Bangladesh and all matters related thereto, including but not limited to the anticipating timing of commercialization; the anticipated arrangement with a third party regarding the development, production distribution and sale of its Soluvec™ product worldwide (excluding Bangladesh), and related matters, including but not limited to the ability of such third party to support the agreement with the third party in Bangladesh, and the impact of such an agreement, including it resulting in greater protection of MVMD”s intellectual property and greater easy in managing licensees and resulting revenue streams; MVMD”s agreement and future arrangement with RWB and the impact on MVMD of the amendment to a non-exclusive arrangement; the continued assessment of the Company’s ability, viability and desire to continue to develop its other projects and pre-clinical R&D work; and the focus of expenditures on the three (3) selected areas of business. The Company believes that the expectations reflected in the forward-looking information are reasonable, but no assurance can be given that these expectations will prove to be correct and such forward-looking information should not be unduly relied upon. Any forward-looking information contained in this news release represents the Company's expectations as of the date hereof and is subject to change after such date. The Company disclaims any intention or obligation to update or revise any forward-looking information whether as a result of new information, future events or otherwise, except as required by applicable securities legislation. Neither the CSE nor OTC has reviewed or approved the contents of this press release.
引进/卖出
2022-11-22
The coalition aims to spotlight the many ways the equine industry benefits and enriches communities across California.
LOS ANGELES, Nov. 22, 2022 /PRNewswire/ -- Today, equine industry leaders and stakeholders across California announced the launch of the California Horse Power Coalition (CHPC). The new coalition's mission is to preserve and protect California's vibrant equestrian culture by bringing together equine industry leaders, workers, families and fans of the sport to illustrate its significant benefits.
Founded by Del Mar Thoroughbred Club, Santa Anita Park, and Thoroughbred Owners of California, the coalition is creating a space for the public to learn about the significant impacts that the industry has on California's economy, tourism, agricultural sustainability, and culture.
"As one of the oldest and most celebrated sports in the world, horse racing contributes billions to California's economy annually and employs tens of thousands of workers," said Bill Nader, Thoroughbred Owners of California, President & CEO. "I am proud to be a part of a community that not only brings together family and friends but also continuously challenges itself to be the model of care and best practices for equine communities around the world."
This unprecedented partnership between Thoroughbred associations, backstretch workers and jockeys, aftercare providers, and veterinarians is a testament of the industry's commitment to working together and pushing the industry to new heights across core impact areas.
Economic Impact
The equine industry is proud to be a sustainable, stable, and ever-growing industry in California.
$8.3 Billion in contributions to California's economy through horse racing annually.
$1.7 Billion in tourism spending generated by participants and spectators.
Jobs and Labor
The industry supports a diverse range of skills, employing hard-working individuals whose work in the industry is often a multi-generational tradition.
77,000+ workers employed by the horse racing industry.
Workforce includes veterinarians, carpenters, jockeys and backstretch workers such as grooms, exercise riders, hot walkers, and more.
Agriculture and Sustainability
The industry contributes to California's cyclical and sustainable agricultural ecosystem.
15-20 pounds of hay locally sourced and fed to every horse daily, supporting local farms' overall profits and sustainability.
8,000 tons of straw waste diverted from landfills to sustainable farms each year.
"Our community strives to create a culture of inclusivity and accessibility to local communities of all ages, genders and economic backgrounds, which has resulted in a long tradition of deep cultural ties to our industry. In fact, over 30% of households in California report having a horse enthusiast in their home," said Amy Zimmerman, Santa Anita Park, Senior Vice President and Executive Producer. "I look forward to seeing our industry continue to prosper, garner more fans, and foster a sense of belonging."
To learn more or to become a member, visit: californiahorsecoalition.com
About the California Horse Power Coalition
The California Horse Power Coalition is a growing partnership among thoroughbred associations, organizations representing backstretch workers and jockeys, animal wellness advocates and more. Founded by Santa Anita Park, Del Mar Thoroughbred Club, and Thoroughbred Owners of California, the coalition's members include California Retirement Management Account, California Thoroughbred Breeders Association, California Thoroughbred Horsemen's Foundation, California Thoroughbred Trainers, Edwin J. Gregson Foundation, and Winner's Foundation.
Media Contact:
Stefan Friedman [email protected]
SOURCE California Horse Power Coalition
2022-08-08
DULUTH, Ga., Aug. 8, 2022 /PRNewswire/ -- This weekend, nearly 600 veterinary students representing three dozen veterinary schools from across the US, Canada, France, Germany and the Netherlands marked the conclusion of a special summer research experience with a presentation of their findings at the National Veterinary Scholars Symposium.
Mentored this summer by leading researchers from across academia, the US Department of Agriculture (USDA) and National Institutes of Health, the students – all participants in the Veterinary Scholars Program – conducted original research, relevant to both animal and human health, in such fields of study as emerging infectious diseases, toxicology, oncology and chronic diseases as well as advances in conservation and sustainable agriculture. This longstanding collaboration between public and private organizations, academia and government agencies provides veterinary students hands-on research experience in the laboratory and field, and exposes them to potential careers in biomedical research.
The Veterinary Scholars Program participants summarize their research findings and work over the course of the summer on a scientific poster, which they present to public health officials and industry experts as well as leading biomedical researchers and other veterinary students at the National Veterinary Scholars Symposium. This year's Symposium was hosted by the University of Minnesota's College of Veterinary Medicine, and also sponsored by Boehringer Ingelheim, the American Association of Veterinary Medical Colleges, American Veterinary Medical Association and American Veterinary Medical Foundation, National Institutes of Health, USDA and the Burroughs Wellcome Fund.
This summer marked the first time that Veterinary Scholars had the opportunity to work directly with scientists at the Agricultural Research Service (ARS) of the U.S. Department of Agriculture, researching diseases that could affect livestock and public health and advancing sustainable approaches for agriculture and food production. Under this new collaboration between the USDA and Boehringer Ingelheim, 12 students spent the summer at one of nine USDA sites working with an ARS scientist on a research project, with Boehringer Ingelheim and USDA covering all costs for the students, including a monthly stipend and costs associated with traveling to and from their schools to the USDA centers and the opportunity to present their research at the National Veterinary Scholar Symposium.
"It's clear from the students' presentations that they have learned a great deal from their mentors and their research work this summer. We hope that this summer's experience has introduced them to the many ways that veterinarians are contributing to science, innovation and the well-being of animals and humans, and that it will inspire them throughout their careers.," said Caroline Belmont, head of U.S. Animal Health Innovation for Boehringer Ingelheim, in her welcome address to participants at the National Veterinary Scholars Symposium.
Boehringer Ingelheim Research Awards
Several veterinary students and one graduate veterinary researcher are recognized annually at the National Veterinary Scholars Symposium with Boehringer Ingelheim awards in recognition of their early contributions to research advancing animal and human health.
Dr. Ashley M. Rasys, from the University of Georgia, received the
2022 Boehringer Ingelheim Veterinary Research Award for Graduate Veterinarians, which recognizes graduate veterinarians who have completed or will soon complete a Ph.D. program or are in their final years of residency training in veterinary pathology, medicine, surgery, radiology/ imaging, or laboratory animal medicine. Dr. Rasys' work has focused on genome editing in reptiles, with the intention to develop a cheaper and more efficient method to edit the chicken genome in order to help mitigate pathogen spread and disease among birds. Dr. Rosemary Bayless, from North Carolina State University's College of Veterinary Medicine, also was recognized as Veterinary Graduate honorable mention.
Sydney Womack, from Cornell University's College of Veterinary Medicine, received the
2022 Boehringer Ingelheim Veterinary Research Award for Veterinary Students. Womack's research has focused on the exploration of proteomics across species to identify potential biomarkers for osteoarthritis in animal and humans.
Lauren Ellison, from Mississippi State University's College of Veterinary Medicine, and Myranda Gorman, from University of Tennessee's College of Veterinary Medicine, also received recognition as honorable mention Boehringer Ingelheim Veterinary Research Scholars.
The
Boehringer Ingelheim Veterinary Scholars Program was established more than 30 years ago to introduce veterinary medical students to biomedical research. At each participating school, Boehringer Ingelheim Veterinary Scholars are assigned a mentor and laboratory. Each scholar conducts a hypothesis-driven research project. The research project is typically conducted over a 10-12-week period during the summer, with students presenting their work at the conclusion. More than 5,000 students have received stipends from Boehringer Ingelheim to conduct research since the program started. More information is available at .
About Boehringer Ingelheim Animal Health USA
Boehringer Ingelheim Animal Health is working on first-in-class innovation for the prediction, prevention, and treatment of diseases in animals. For veterinarians, pet owners, producers, and governments in more than 150 countries, we offer a large and innovative portfolio of products and services to improve the health and well-being of companion animals and livestock.
As a global leader in the animal health industry and as part of the family-owned Boehringer Ingelheim, we take a long-term perspective. The lives of animals and humans are interconnected in deep and complex ways. We know that when animals are healthy, humans are healthier too. By using the synergies between our Animal Health and Human Pharma businesses and by delivering value through innovation, we enhance the health and well-being of both.
Learn more about Boehringer Ingelheim Animal Health USA at
SOURCE Boehringer Ingelheim
基因疗法First in Class
100 项与 黑龙江北大荒农业股份有限公司 相关的药物交易
登录后查看更多信息
100 项与 黑龙江北大荒农业股份有限公司 相关的转化医学
登录后查看更多信息
管线布局
2024年05月07日管线快照
无数据报导
登录后保持更新
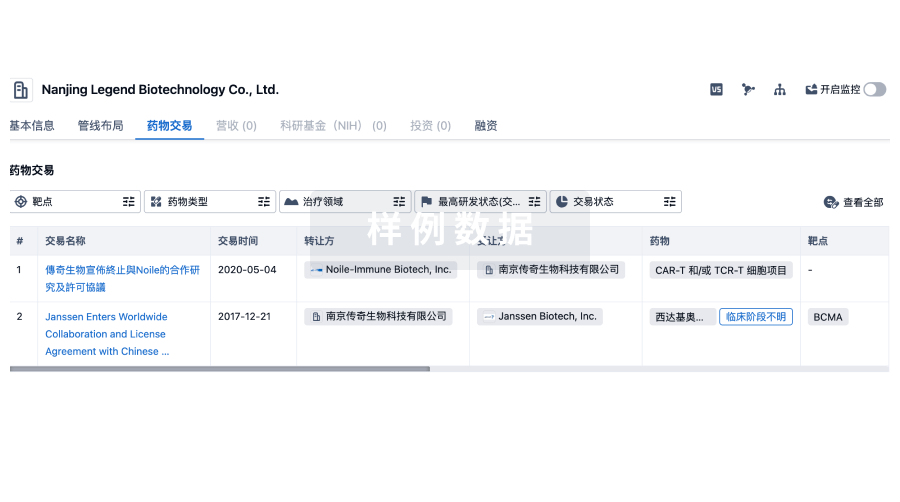
药物交易
使用我们的药物交易数据加速您的研究。
登录
或
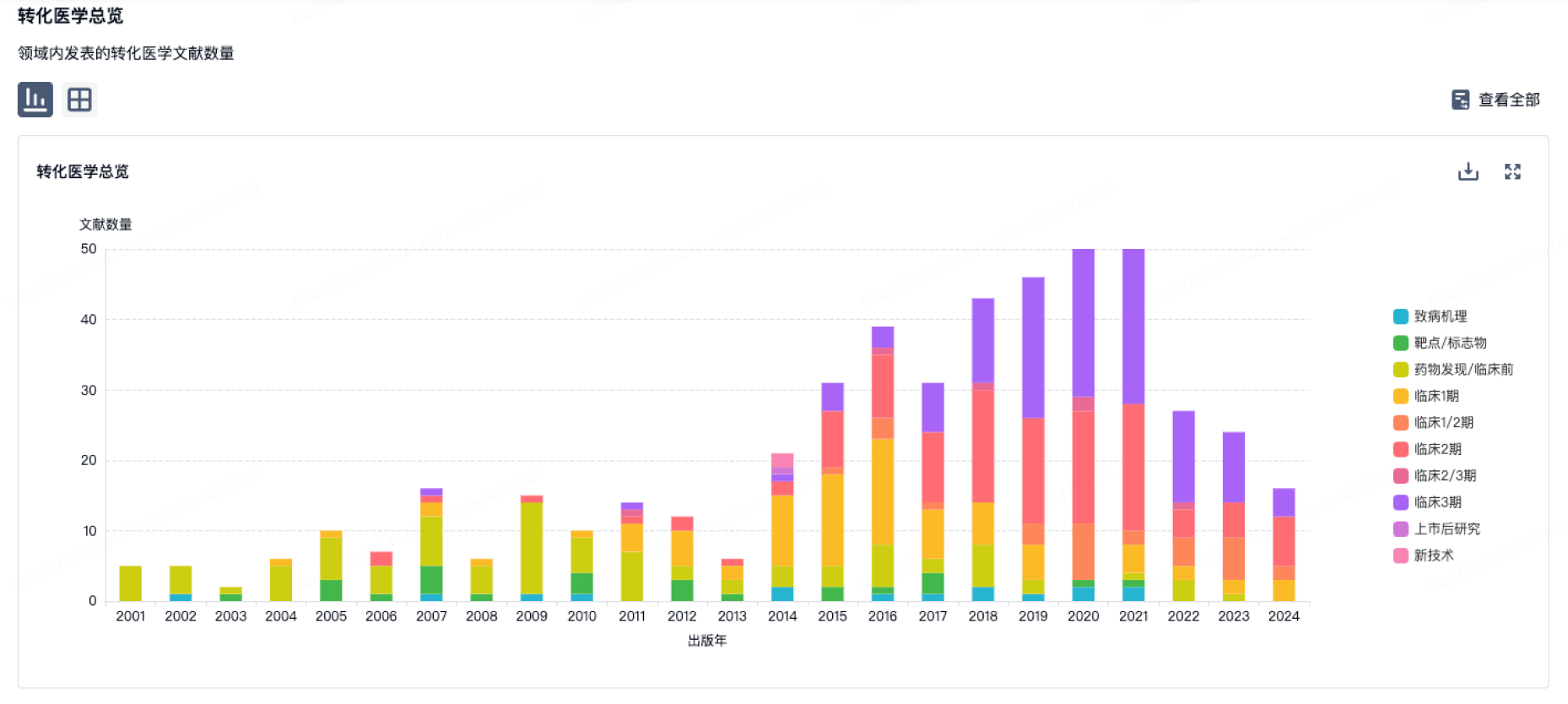
转化医学
使用我们的转化医学数据加速您的研究。
登录
或
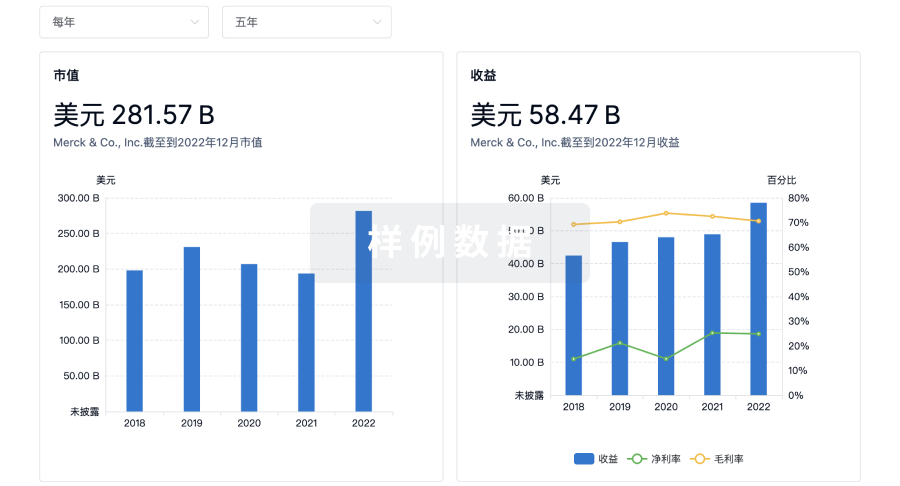
营收
使用 Synapse 探索超过 36 万个组织的财务状况。
登录
或
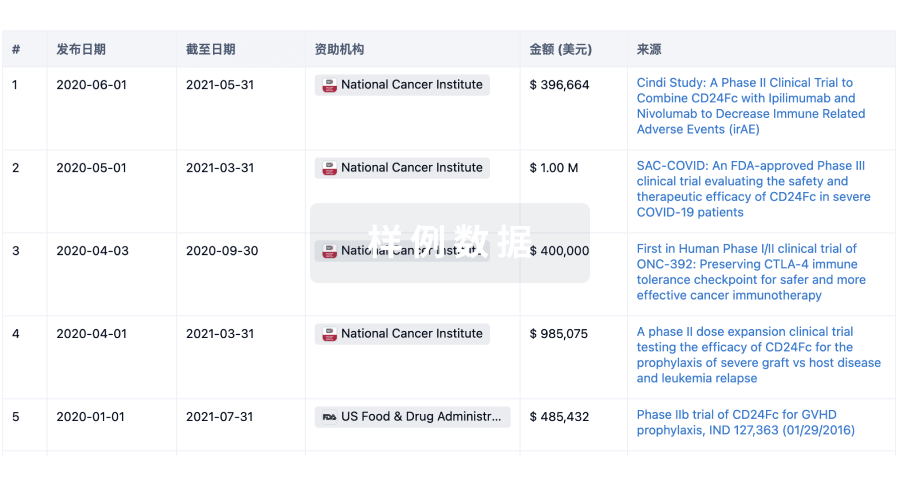
科研基金(NIH)
访问超过 200 万项资助和基金信息,以提升您的研究之旅。
登录
或
投资
深入了解从初创企业到成熟企业的最新公司投资动态。
登录
或
融资
发掘融资趋势以验证和推进您的投资机会。
登录
或
标准版
¥16800
元/账号/年
新药情报库 | 省钱又好用!
立即使用
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用