预约演示
更新于:2026-05-18
PGN EDODM1
更新于:2026-05-18
概要
基本信息
原研机构 |
在研机构 |
非在研机构- |
权益机构- |
最高研发阶段临床2期 |
首次获批日期- |
最高研发阶段(中国)- |
特殊审评孤儿药 (美国)、孤儿药 (欧盟)、快速通道 (美国) |
登录后查看时间轴
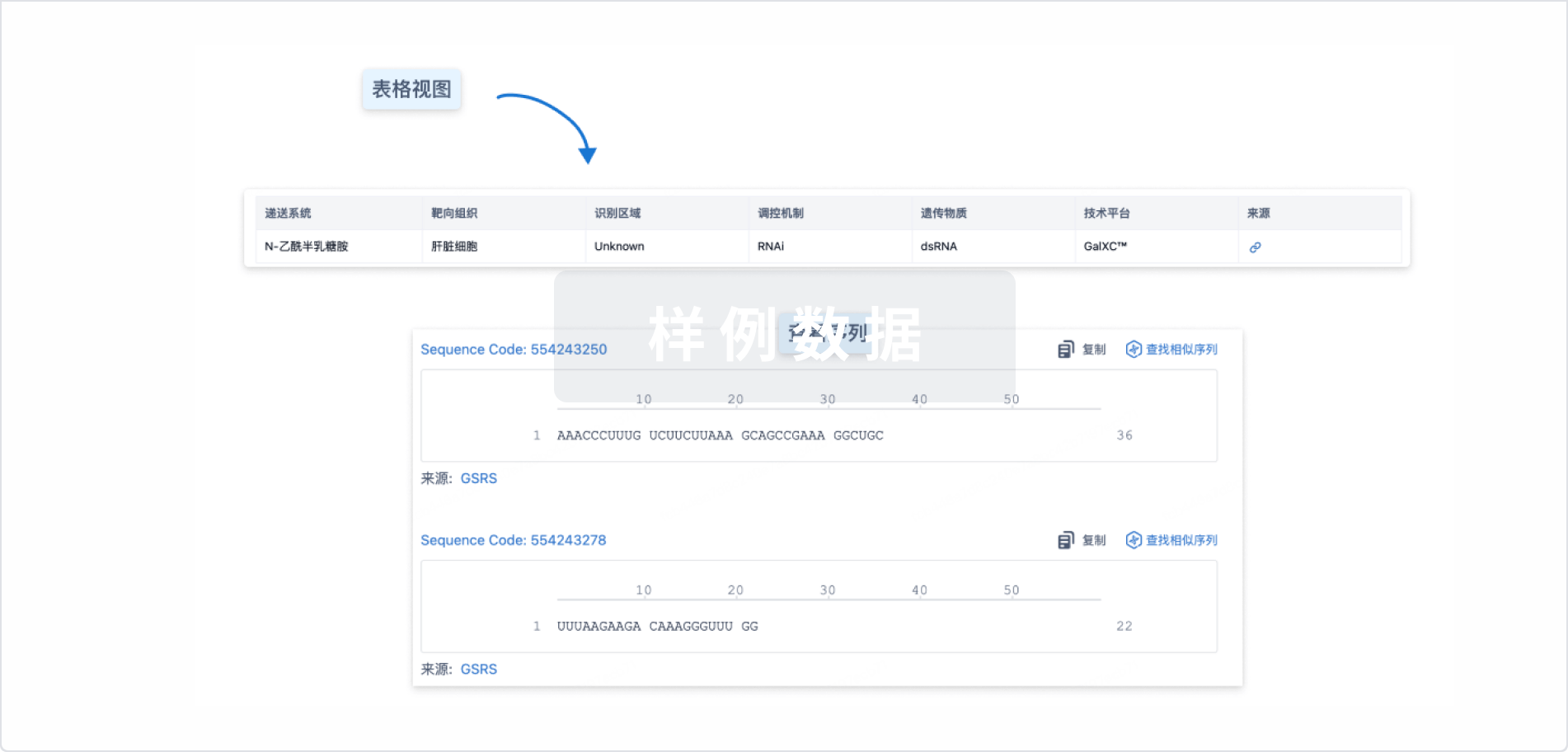
结构/序列
使用我们的RNA技术数据为新药研发加速。
登录
或
关联
3
项与 PGN EDODM1 相关的临床试验NCT07220603
An Open-Label Extension Study Evaluating Safety and Pharmacokinetics in Participants With Myotonic Dystrophy Type 1 (FREEDOM-OLE)
The purpose of this study is to learn about the long-term safety and tolerability of PGN-EDODM1 in participants with myotonic dystrophy type 1 (DM1) who have completed a prior study with PGN-EDODM1.
开始日期2025-12-23 |
申办/合作机构 |
NCT06667453
A Phase 2 Randomized, Double-Blind, Placebo-Controlled, Multiple Ascending Dose Study of PGN-EDODM1 in Adult Participants With Myotonic Dystrophy Type 1 (FREEDOM2-DM1)
The purpose of this study is to learn about the effects of an investigational medicine, PGN-EDODM1, to see how safe and tolerable multiple administrations of PGN-EDODM1 are for people with myotonic dystrophy type 1 (DM1) compared to placebo.
开始日期2024-12-10 |
申办/合作机构 |
NCT06204809
A Phase 1 Placebo-Controlled Study to Assess the Safety, Tolerability, Pharmacokinetics, and Pharmacodynamics of Single-Ascending Doses of PGN-EDODM1 in Adult Participants With Myotonic Dystrophy Type 1 (FREEDOM-DM1)
The primary purpose of the study is to evaluate the safety and tolerability of single intravenous (IV) doses of PGN-EDODM1 administered to participants with Myotonic Dystrophy Type 1 (DM1).
The study consists of 2 periods: A Screening Period (up to 30 days) and a Treatment and Observation Period (16 weeks).
The study consists of 2 periods: A Screening Period (up to 30 days) and a Treatment and Observation Period (16 weeks).
开始日期2023-12-12 |
申办/合作机构 |
100 项与 PGN EDODM1 相关的临床结果
登录后查看更多信息
100 项与 PGN EDODM1 相关的转化医学
登录后查看更多信息
100 项与 PGN EDODM1 相关的专利(医药)
登录后查看更多信息
74
项与 PGN EDODM1 相关的新闻(医药)2026-05-13
– FREEDOM2-DM1 5 mg/kg cohort demonstrated favorable safety, splicing and vHOT data, with the totality of results supporting the potential of the ongoing 10 mg/kg dose cohort –
– The FREEDOM2 10 mg/kg cohort is fully enrolled, with data on track for 2H 2026 –
– Well-funded with $132.3M of cash as of March 31, 2026, sufficient to fund operations through FREEDOM2 12.5 mg/kg MAD readout and into 2H 2027 –
BOSTON--(BUSINESS WIRE)--PepGen Inc. (Nasdaq: PEPG), a clinical-stage biotechnology company advancing the next generation of oligonucleotide therapies with the goal of transforming the treatment of severe neuromuscular and neurological diseases, today reported financial results and recent corporate highlights for the quarter ended March 31, 2026, and recent corporate highlights.
"We made encouraging progress during the first quarter of 2026 in our FREEDOM2-DM1 trial. The 5 mg/kg cohort — the initial dose cohort — demonstrated a favorable tolerability pro promising efficacy trends, reinforcing our confidence in PGN-EDODM1's potential at higher doses," said James McArthur, PhD, President and Chief Executive Officer of PepGen. "Enrollment in the 10 mg/kg cohort is now complete and we remain on track to report data in the second half of this year. With funding in place through the 10 and 12.5 mg/kg cohorts, we are well positioned to continue dose escalating and deliver on the promise of this program for the DM1 community."
Recent Program Updates
PGN-EDODM1: Myotonic Dystrophy Type 1 (DM1)
FREEDOM2 Phase 2 Multiple Ascending Dose (MAD) Randomized, Placebo-Controlled Clinical Trial of PGN-EDODM1:
PepGen reported promising topline results from the 5 mg/kg MAD cohort, demonstrating favorable safety, splicing and vHOT data. The Company believes the totality of safety and efficacy results support the potential of PGN-EDODM1 in the ongoing 10 mg/kg dose cohort. Read the full release
here
.
The Company has fully enrolled the 10 mg/kg MAD cohort of FREEDOM2. PepGen expects to report data from this cohort in the second half of 2026, with data from the 12.5 mg/kg cohort anticipated in 2027.
PepGen has currently enrolled 13 patients in the open label extension (OLE) at 5 mg/kg, including 6 patients from FREEDOM2, with no patient discontinuations.
The Company has received regulatory clearance to initiate the FREEDOM2 trial in South Korea, Australia, and New Zealand. Sites are currently open and active in Canada, the UK and South Korea, with plans to open sites in New Zealand and Australia.
The U.S. Food and Drug Administration (FDA) placed a partial clinical hold on the FREEDOM2 study. The partial clinical hold questions raised by the FDA relate to previously submitted preclinical pharmacology and toxicology studies. The timing of the ongoing FREEDOM2 clinical study has not been impacted. The Company continues to work with the FDA to address the Agency’s questions as quickly as possible.
Financial Results for the Three Months Ended March 31, 2026
Cash, Cash Equivalents and Marketable Securities
were $132.3 million as of March 31, 2026. Based on currently planned operations, the Company believes that its existing cash, cash equivalents, and marketable securities will be sufficient to fund its operations into the second half of 2027.
Research and Development Expenses
were $13.0 million for the three months ended March 31, 2026, compared to $25.4 million for the same period in 2025.
General and Administrative Expenses
were $5.9 million for the three months ended March 31, 2026, compared to $5.9 million for the same period in 2025.
Net Loss
was $17.8 million, or $(0.26) basic and diluted net loss per share, for the three months ended March 31, 2026, compared to $30.2 million, or $(0.92) basic and diluted net loss per share, for the same period in 2025. PepGen had approximately 69.2 million shares outstanding on March 31, 2026.
About PGN-EDODM1
PGN-EDODM1, PepGen's investigational candidate in development for the treatment of DM1, utilizes the Company's proprietary EDO technology to deliver a therapeutic oligonucleotide that is designed to restore the normal splicing function of MBNL1, a key RNA splicing protein. PGN-EDODM1 addresses the deleterious effects of cytosine-uracil-guanine (CUG) repeat expansion in the dystrophia myotonica protein kinase (
DMPK
) transcripts which sequester MBNL1, by binding to the pathogenic CUG trinucleotide repeat expansion present in the
DMPK
transcripts, and disrupting the binding between the CUG repeat expansion and MBNL1. PepGen believes this innovative therapeutic approach may have considerable advantages over oligonucleotide modalities that rely on knockdown or degradation of the
DMPK
transcripts as it will allow the
DMPK
transcripts to continue to perform their normal function within the cell, while also liberating MBNL1 to correct downstream mis-splicing events. The U.S. Food and Drug Administration has granted PGN-EDODM1 both Orphan Drug and Fast Track Designations for the treatment of patients with DM1.
About Myotonic Dystrophy Type 1 (DM1)
Myotonic dystrophy type 1 (DM1) is a rare, progressive, and highly variable genetic neuromuscular disease caused by an abnormal expansion of cytosine-thymine-guanine (CTG) repeats in the dystrophia myotonica protein kinase (
DMPK
) gene. DM1 affects over 115,000 individuals in the U.S. and EU and is characterized by widespread, multisystem symptoms that may include myotonia, progressive muscle weakness, fatigue, cardiac abnormalities, respiratory impairment, and cognitive dysfunction. The disease is driven by toxic RNA transcripts containing expanded cytosine-uracil-guanine (CUG) repeats that sequester muscleblind-like 1 (MBNL1), a key RNA splicing protein, leading to widespread mis-splicing across multiple tissues. There are currently no approved disease-modifying therapies for DM1, underscoring the significant unmet medical need for patients living with the disease.
About PepGen
PepGen Inc. is a clinical-stage biotechnology company developing the next generation of oligonucleotide therapies with the goal of transforming the treatment of severe neuromuscular and neurological diseases. PepGen’s Enhanced Delivery Oligonucleotide (EDO) platform is founded on over a decade of research and development and leverages cell-penetrating peptides to improve the uptake and activity of conjugated oligonucleotide therapeutics. Using these EDO peptides, the Company is generating a pipeline of oligonucleotide therapeutic candidates designed to target the root cause of serious diseases.
For more information, please visit
PepGen.com
. Follow PepGen on
LinkedIn
and
X
.
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, as amended. These statements may be identified by words such as “aims,” “anticipates,” “believes,” “could,” “estimates,” “expects,” “forecasts,” “goal,” “intends,” “may,” “plans,” “possible,” “potential,” “seeks,” “will,” and variations of these words or similar expressions that are intended to identify forward-looking statements. Any such statements in this press release that are not statements of historical fact may be deemed to be forward-looking statements. These forward-looking statements include, without limitation, statements regarding the therapeutic potential and safety pro PGN-EDODM1 based on data from the 5, 10 and 15 mg/kg cohorts of the single dose FREEDOM-DM1 study and data from 5 mg/kg cohort of the multi-dose FREEDOM2-DM1 study, expected timelines for data reports from our FREEDOM2-DM1 trial, forecasts relating to PepGen’s cash runway, and ongoing and planned regulatory interactions, including the potential timing and successful resolution of questions from the FDA relating to the partial clinical hold.
Any forward-looking statements in this press release are based on current expectations, estimates and projections only as of the date of this release and are subject to a number of risks and uncertainties that could cause actual results to differ materially and adversely from those set forth in or implied by such forward-looking statements. These risks and uncertainties include, but are not limited to risks related to: delays or failure to successfully initiate or complete our ongoing and planned development activities for our product candidates, including PGN-EDODM1; our ability to enroll patients in our clinical trials, including FREEDOM2; that our interpretation of clinical and preclinical study results may be incorrect, or that we may not observe the levels of therapeutic activity in clinical testing that we anticipate based on prior clinical or preclinical results, including for PGN-EDODM1; our product candidates, including PGN-EDODM1, may not be safe and effective or otherwise demonstrate safety and efficacy in our clinical trials; adverse outcomes from our regulatory interactions, including delays in regulatory review, clearance to proceed or approval by regulatory authorities with respect to our programs, including release of the partial clinical hold placed by the FDA and/or clearance to commence planned clinical studies of our product candidates, or other regulatory feedback requiring modifications to our development programs, including in each case with respect to our FREEDOM2 program; changes in regulatory framework that are out of our control; unexpected increases in the expenses associated with our development activities or other events that adversely impact our financial resources and cash runway; and our dependence on third parties for some or all aspects of our product manufacturing, research and preclinical and clinical testing. Additional risks concerning PepGen’s programs and operations are described in our most recent reports filed with the SEC. PepGen explicitly disclaims any obligation to update any forward-looking statements except to the extent required by law.
This release discusses PGN-EDODM1, an investigational therapy that has not been approved for use in any country, and is not intended to convey conclusions about its efficacy or safety. There is no guarantee that PGN-EDODM1 or any other investigational therapy will successfully complete clinical development or gain regulatory authority approval.
Condensed Consolidated Statements of Operations
(unaudited, in thousands)
Three Months Ended
March 31,
2026
2025
Operating expenses:
Research and development
$
13,004
$
25,378
General and administrative
5,938
5,943
Total operating expenses
$
18,942
$
31,321
Operating loss
$
(18,942
)
$
(31,321
)
Other income (expense)
Interest income
1,250
1,122
Other (expense) income, net
(56
)
(3
)
Total other income, net
1,194
1,119
Net loss before income tax
$
(17,748
)
$
(30,202
)
Income tax expense
(15
)
—
Net loss
$
(17,763
)
$
(30,202
)
Net loss per share, basic and diluted
$
(0.26
)
$
(0.92
)
Weighted-average common stock outstanding, basic and diluted
69,091,100
32,674,720
Condensed Consolidated Balance Sheets
(unaudited, in thousands)
March 31,
2026
December 31,
2025
Assets
Cash, cash equivalents and marketable securities
$
132,303
$
148,456
Other assets
23,992
25,451
Total assets
$
156,295
$
173,907
Liabilities and stockholders’ equity
Liabilities
$
22,315
$
26,463
Stockholders’ equity
133,980
147,444
Total liabilities and stockholders’ equity
$
156,295
$
173,907
Contacts
Investor Contact
Laurence Watts
New Street Investor Relations
laurence@newstreetir.com
Media Contact
Julia Deutsch
Lyra Strategic Advisory, LLC
Jdeutsch@lyraadvisory.com
快速通道孤儿药财报寡核苷酸临床结果
2026-04-29
导语
一、核酸药物决战"递送"。超20款核酸药虽已获批,但多受困于肝脏。谁能突破肌肉、心脏和中枢神经等的递送,谁就定义下一代天花板。
二、中枢递送:AOC领跑临床,POC剑指未来。AOC跨血脑屏障验证更成熟;但多肽凭借极小尺寸,理论上具备脑实质更均匀扩散的潜力,且在新靶点(如LRP1)拓展上后发优势强劲。
三、临床分化:AOC单次强效,POC赢在“深穿透”。 AOC精准受体介导显现强效(Avidity实现约40%跳跃率);POC在致密肌肉中则展现出更宽安全窗口与更高上限(Pepgen以高剂量实现53.7%剪接修正)。
四、决定上限的不仅是载体,更是靶点。选对新型靶点(如αvβ6),仅1.02 mg/kg极低剂量即可实现约50%剪接修正,远超同剂量TfR1路线。未来终局将是"按场景与靶点匹配"的分层演化。
偶联不再只是连接:重新定义递送载体的逻辑
在过去的一段时间,药物研发中的“偶联”已经不再只是一个化学问题,而越来越像一个系统工程问题。原因很简单:很多偶联药物并不是没有活性,而是没有办法稳定、精准地到达正确的组织、正确的细胞,甚至正确的亚细胞位置。对于近期大火的核酸药物而言,这一点尤其关键。到2025年,已有超过20个寡核酸药物(包括ASO、siRNA和aptamer)获批,“核酸可以成药”已经被验证;下一阶段真正决定平台上限的,不再只是序列本身,而是什么样的载体能把这些序列从肝脏进一步送向肌肉、心脏、肺乃至中枢等更难进入的组织。
在这场递送竞赛中,多肽正重新走入大众的视线。多肽的分子量处于一个非常有利的中间位置,相比抗体,它足够小,便于组织扩散;相比于传统小分子,它有足够可设计,能够引入靶向、穿膜、内体逃逸乃至亚细胞定位等功能的优化空间;多肽本身的特点又使其具有更接近化学合成逻辑,便于快速迭代和规模制造。除POC(Peptide-oligonucleotide conjugates)场景之外,多肽与小分子毒素、核素以及蛋白的偶联/融合,正在充分扩展药物类型,并不断在靶组织释放高活性载荷、改善蛋白药的稳定性、溶解性、递送效率和安全窗口等方面取得令人振奋的进展。
对寡核酸药物来讲,其天然面临分子带电、跨膜能力弱、内体逃逸不足、组织分布受限等问题。当前临床成熟度更高的是 AOC(Antibody-oligonucleotide conjugates),两家龙头Avidity和Dyne分别有做到临床后期的管线,数据也都不错,并且,Avidity因其三条AOC管线,并已被 Novartis 以120亿美元收购,AOC 的产业价值,已经得到了资本和大药企的实质性背书。但 AOC 的成熟,也让它的边界越来越清楚。AOC 的优势在于高特异性、长循环半衰期和较成熟的生物药工程体系;与之对应,其不足同样来自抗体本体:结构复杂、对CMC 和质量控制要求高、组织渗透受分子尺寸限制。事实上,AOC 领域之所以开始使用 Fab 或 scFv 等更小片段,本身就说明“抗体太大”是一个需要被持续修正的工程问题。
从这个角度看,POC 的价值,理论上恰恰在于它不是把抗体平台简单做小,而是换了一套递送思路。肽可以把靶向、穿膜、内体逃逸和局部富集等功能整合进一个更短、更灵活的载体中。当前的不少研究表明,肽用于寡核酸递送的核心优势包括小尺寸、相对更容易合成制造,以及促进细胞膜穿越的能力;许多研究也反复强调,肽相较抗体通常具有更强的组织渗透和细胞内化效率。对那些真正受限于“进入组织”和“进入细胞”的适应证来说,这可能是决定平台成败的核心优势。
从机制层面定义AOC与POC的最佳战场
2.1 跨BBB递送 - AOC的有效性验证
在脑部递送中,AOC最大的优势,可以归结为一点:能够显著降低ARIA (amyloid-related imaging abnormalities)风险。
传统抗体(裸抗)进入脑的路径,大多是通过血液进入脉络丛,再进入脑脊液(Cerebrospinal Fluid, CSF),随后沿着血管周间隙(Perivascular Spaces, PVS; 又称Virchow-Robin腔)扩散,最后才逐步进入脑实质。这个递送路径的问题在于,药物容易聚集在脑的边界区域,并且会沿着血管分布,优先接触血管相关的Aβ沉积,所导致的结果就是抗体在血管周围富集,从而提高ARIA发生的风险。
以TfR1 brain shuttle为代表的策略,本质上改变了这一路径。它通过脑毛细血管内皮上的受体介导转运(transcytosis),让抗体穿过血管内皮进入脑实质。这一递送策略使脑实质中的药物暴露增加,而血管及PVS区域的暴露显著降低。由于抗体在血管周围的停留时间减少,与血管Aβ的相互作用也随之下降,ARIA风险自然降低。这也是为什么罗氏的trontinemab以及其TfR1 brain shuttle思路在业内引起了广泛关注。
Source: Roche 官网:Brainshuttle™ AD: New interim results of a randomized, placebo-controlled Phase Ib/IIa proof-of-concept study with trontinemab, a novel anti-amyloid monoclonal bispecific antibody for the treatment of Alzheimer’s disease
而在 POC 上,Ionis/Bicycle 等平台已显示出跨 BBB 的潜力,但公开披露仍主要处于平台化和早期推进阶段,尚未形成临床阶段、标志性的跨BBB脑递送的 POC 代表管线。
很多人会问,为什么AOC在脑递送中这么优秀?如果POC同样去靶向TfR1,能否达到类似效果来避免ARIA?直觉上似乎可以,但现实中挑战不止一重。原因在于,并不是简单的于TfR1受体结合,重点在于如何保证后续的transcytosis,这是一个对分子设计要求极高的动力学过程。亲和力需要落在一个非常窄的窗口内,过强会导致内吞后被降解,过弱则无法稳定结合;同时还需要减少价态,避免受体聚集,从而被导向溶酶体降解路径。最终的目标,是让分子更倾向于穿过去(transcytosis),而不是被内皮细胞提前“吃掉”(degradation)。
在脑递送中,AOC的优势并不在于靶向能力更强(事实上POC在这方面也已经能做的很优秀),而在于抗体更容易被工程化为一个稳定的transcytosis运输工具,在安全性和副作用控制上的优势更为明显。
不过,AOC在脑递送中的暂时领先并不意味着终局。抗体庞大的分子量(~150kDa)虽然容易工程化,但也成了它穿透BBB后在致密脑实质中扩散的巨大阻碍。相比之下,多肽(<5kDa)一旦突破跨胞转运(transcytosis)的技术瓶颈,或者成功拓展诸如LRP1(如Angiopep-2的先例)等新靶点,其在脑实质内的均匀覆盖能力将远超抗体。在BBB递送的下半场,这极有可能是POC实现弯道超车的关键。
2.2 肌肉/肿瘤组织递送 - POC优势的良好体现
肌肉组织相关的疾病与脑完全不同,核心问题并不在于如何安全地进去,而在给药分布和覆盖程度。以肌肉溶解症为例,长期以来一个核心痛点就是递送无法覆盖足够多的肌纤维。早期基于AAV(adeno-associated virus)的基因治疗已经证明了这一点,如果只覆盖了部分细胞,即便在初期有效,疾病仍然很容易复发。如果新一代技术不能解决广覆盖这个问题,很难在临床上形成真正的突破。
相比脑组织,肌肉没有BBB,也不存在PVS这样的空间风险结构,药物通常可以顺利到达肌肉间质。但真正的瓶颈在于,药物很难进入肌肉细胞本身。肌纤维是典型的超大、多核细胞,细胞膜结构稳定,同时组织内存在致密的细胞外基质(extracellular matrix, ECM)和基底膜结构,细胞排列紧密。这些因素叠加在一起,使得大分子(尤其是抗体)在肌肉中的扩散速度非常慢,分布呈现明显的不均一。这也解释了为什么AOC在肌肉中的疗效会受到部分限制。虽然它可以通过受体介导进入部分细胞,但由于分子尺寸大,在组织中的扩散能力有限,很难实现对整个肌肉组织的均匀覆盖。
相比之下,POC的优势恰恰在这里体现出来。多肽分子体积小,在组织中的扩散能力更强,更容易穿过ECM,在肌肉中实现更均匀的分布。同时,CPP类多肽还能增强细胞摄取,使更多肌细胞被覆盖,虽然这种方式缺乏精确性,但在需要大范围覆盖的适应症中,反而是一种优势。
类似的逻辑也适用于纤维化疾病和实体瘤。无论是纤维化组织中致密的ECM,还是实体瘤中复杂的微环境(包括血供不均、间质压力高等因素),都会限制大分子的扩散,使递送呈现高度不均一。在这些场景中,以多肽为基础的递送体系,往往更有机会实现更好的组织穿透性和覆盖力。
用数据说话 - AOC与POC的临床表现
机制层面上的分析纵然有逻辑,但具体表现还得看临床数据。
POC分为两条完全不同的技术路径,需要进行分类讨论:
一类是“受体靶向型POC”。这一类的思路和AOC几乎一模一样,都是通过靶向组织特异性受体来实现器官定向递送,比如Ionis/Bicycle的TfR1策略,或者Arrowhead的αvβ6靶向。从原理上看,这一类POC确实是在对标AOC。但这一类POC暂时很难和AOC进行真正的正面对比。一方面,Ionis相关临床项目还在早期,缺乏人体数据;另一方面,Arrowhead虽然推进到了二期,但所选靶点较新,与主流AOC平台并不一致,很难直接横向比较,因此只能单独拿出来讨论。
另一种是依赖细胞穿透肽(CPP)的POC,其临床数据就比较充分,能够和现在的AOC龙头(Avidity等)进行一定程度的对比,但是他们的靶向思路完全不一样。不同于受体靶向,CPP实际上是广谱地进入细胞,但是可以通过一些药化上的修饰,造成肌肉组织的偏好性分布。结合肌肉组织本身血流丰富、毛细血管密集、内吞活跃等特点,CPP可以通过多种途径进入细胞,包括巨胞饮过程(macropinocytosis)和陷窝(caveolae)介导的内吞。后续的关键则在于如何实现有效的内体逃逸和细胞内定位,这也是目前CPP平台的核心竞争点,Pepgen的EDO(Enhanced Delivery Oligonucleotide)平台,以及Entrada的EEV™(Endosomal Escape Vehicle)平台,都是围绕这一问题展开技术优化。
基于以上不同技术路径,我们接下来会重点分析AOC与POC现有的临床数据,梳理AOC与POC各自的特点和优势。
3.1 CPP-POC vs AOC
目前AOC 的总体进度要略快于CPP-POC。AOC的两家龙头——Avidity和Dyne都做到了后期临床。而POC的龙头——Entrada和Pepgen目前还处于早期临床,分别在一期和二期。结合适应症和靶点,我们在这里分别对比Avidity的 del-zota和Entrada的 ENTR-601-44在杜氏肌肉萎缩症(Duchenne muscular dystrophy, DMD)上的表现,两者所靶向的都是Exon 44 skipping,且采取递送PMO的策略;以及Pepgen的PGN-EDODM1和Dyne的DYNE-101在肌强直性营养不良(DM1)的表现,两者均靶向DMPK (Dystrophia Myotonica Protein Kinase),但DYNE-101递送的是ASO而非PMO(Phosphorodiamidate Morpholino Oligonucleotide)。
3.1.1 Avidity vs. Entrada for DMD
Avidity 和 Entrada 做的都是 DMD 适应症。该疾病是由DMD基因突变引起,导致 dystrophin 蛋白缺失,使肌细胞膜结构不稳定。随之反复的肌纤维损伤、炎症和纤维化逐渐累积,最终导致进行性肌肉无力和功能丧失。这里的治疗思路是采取外显子跳跃(exon skipping),通过调控mRNA剪接,使突变区域被跳过,从而恢复阅读框,产生截短但部分功能正常的dystrophin蛋白。虽然生成的蛋白不完全正常,但足以显著改善肌细胞稳定性,从而减缓疾病进展。在DMD治疗的药物研发上,Avidity 和 Entrada 是目前最有代表性的两家公司,Avidity的 Del-zota(delpacibart zotadirsen)是 AOC,Entrada 的ENTR-601-44 为 CPP-POC。两者的差异,在数据里其实已经开始体现。
先看Avidity
2025年10月,诺华以120亿美元收购Avidity,核心就是看中了其在遗传性神经肌肉疾病上的三条后期管线。Avidity围绕TfR1构建了一整套AOC平台,在药化和CMC上也做了大量优化,比如利用PMO电中性的特点,在保证安全性的前提下把DAR(Drug-to-Antibody Ratio)推到很高的水平(文献中甚至可以做到超过9)。这一点本身就说明,AOC在“载药能力”上有天然优势。
Source: Cochran et al. 2024, Journal of Medicinal Chemistry
Avivity 的Del-zota(delpacibart zotadirsen)在DMD44患者的Phase 1/2研究中展现出明确的生物学活性:在5mg/kg和10mg/kg两个剂量组中,治疗后外显子44跳跃(exon skipping)显著提升,平均达到约37%–43%,最高可达约67%;同时,dystrophin蛋白水平也明显上升,平均增加约25%,部分患者可达接近正常水平的50%以上。与之相对应,肌肉损伤标志物肌酸激酶(CK)持续显著下降,降幅超过80%,接近正常范围,提示不仅分子指标改善,肌肉病理状态的也得到实质性缓解。
Source: Avidity 官网:Del-zota (delpacibart zotadirsen) produced statistically significant increases in exon skipping and dystrophin levels in EXPLORE44®, a Phase 1/2 study in individuals with DMD44
安全性方面,整体表现也比较稳,大多数不良事件为轻中度,严重不良事件较少,没有出现死亡或明确的剂量限制毒性。当然,在5 mg/kg剂量组中仍然出现了个别严重不良事件(SAE),说明在更高剂量推进时,安全性窗口仍然需要考量。
Source: Avidity 官网:Del-zota (delpacibart zotadirsen) produced statistically significant increases in exon skipping and dystrophin levels in EXPLORE44®, a Phase 1/2 study in individuals with DMD44
再看Entrada
Entrada 代表了另一种被产业界认真下注的递送逻辑。 相比 Avidity 围绕 TfR1 做 AOC,Entrada 走的是 EEV(Endosomal Escape Vehicle)+ PMO 路线,核心思路不是先把受体介导摄取做到极致,而是把“进入细胞之后的最后一公里”——尤其是内体逃逸、骨骼肌分布和心肌递送——作为平台竞争力来打造。截至目前,Entrada 已在 DMD 方向推进了 4 条临床/临床前后期管线,并与 Vertex 合作推进 DM1 项目 VX-670;公司公开资料显示,其 EEV-oligonucleotide 体系在临床前已显示出广泛肌肉分布、显著心肌递送,以及接近完全恢复骨骼肌 dystrophin 生成的潜力。
相比Avidity的Del-zota,Entrada的临床进度要慢一拍,ENTR-601-44目前还停留在Phase I/II早期阶段,还未公开患者疗效数据。目前放出来的只有临床前数据,健康人安全性数据,以及健康人biomarker数据,暂时还没有疗效数据。但从目前的信息来看,已展现出一些很有意思的趋势。
虽然PMO理化性质稳定且中性,但是CPP的分子量远小于抗体。所以POC的载药量不能像PMO-AOC那样干到很大,不然可能会打乱CPP的电荷分布并改变构象,因此载药量是1。
数据方面,ENTR-601-44的最高级剂量目前到了6mg/kg,暂时少于Avidity,但是安全性非常优秀。在 6 mg/kg剂量组中,整体耐受良好,没有与药物相关的不良事件;最常见 AE 是头痛,而且是轻中度;没有严重不良事件,也没有临床意义上的实验室、心电图或生命体征异常;肾毒性生物标志物在最高剂量下也没有异常信号。相比Del-zota在5mg/kg剂量组中就有两例SAE,ENTR-601-44的安全性表现更好。
Source: Entrada 官网:Clinical Trial of ENTR-601-44, an Endosomal Escape Vehicle (EEVTM) Oligonucleotide Conjugate for the Treatment of Duchenne Muscular Dystrophy
初步药效证据方面,因为是健康人,所以只有biomarker的证据:72 小时后肌肉活检里,6 mg/kg 组在 biceps brachii 中检测到了可量化的 PMO-44 代谢物,平均约 52.4 ng/g;更重要的是,只有 6 mg/kg 组出现统计学显著的 DMD exon 44 skipping,平均约 0.44%,而 placebo 约 0.22%。这个数值本身并不大,但它的意义是人在单次静脉给药后,肌肉里确实有可检测的在靶效应。
Source: Entrada 官网:Clinical Trial of ENTR-601-44, an Endosomal Escape Vehicle (EEVTM) Oligonucleotide Conjugate for the Treatment of Duchenne Muscular Dystrophy
此外,还有一个很有意思的点,就是新的动物实验表明,EEV-PMO 可以进入肌卫星细胞(Pax7+),而且在 48 小时可以看到与 quiescent satellite cells 的 100% co-localization;更夸张的是,图里写到 12 周后仍能在卫星细胞和新生中央核纤维里看到 PMO 共定位。这提示 EEV 不只是进成熟肌纤维,还可能触及肌肉干/祖细胞池,这对长期疾病修复非常关键。
Source: Entrada 官网:Clinical Trial of ENTR-601-44, an Endosomal Escape Vehicle (EEVTM) Oligonucleotide Conjugate for the Treatment of Duchenne Muscular Dystrophy
把两条路线放在一起看
可以得到一个更清晰的结论:AOC在当前阶段,优势在于强效,已经能够在患者中给出明确的功能恢复信号;而CPP-POC的优势则更多体现在分布广和安全性,以及潜在的长期组织覆盖能力。换句话说,AOC更像是在解决怎么把药精准送进去,而CPP-POC则在回答另一个问题:能不能把药送到更多细胞,甚至送到更关键的肌肉干/祖细胞池里。
3.1.2 Pepgen vs. Dyne for DM1
另一个核酸治疗常见的适应症是肌强直性营养不良(DM1)。DM1 的病因不是蛋白不够,而是 DMPK 转录本里带有致病性 CUG 重复(CUGexp),这些异常 RNA 会形成 hairpin 结构,把 MBNL1 (Muscleblind-like splicing regulator 1) 这种关键剪接因子困住,最后导致广泛的错误剪接( mis-splicing)。关于这个适应症,PMO 的设计思路是选择性结合这段致病 RNA,削弱 hairpin 形成,释放 MBNL1,从而恢复正常剪接。也就是说它的靶点是异常 DMPK RNA,不是蛋白本身。
先看Pepgen
PepGen 代表了更“纯粹”的 POC/CPP 路线。它的核心是基于 EDO(Enhanced Delivery Oligonucleotide) 平台,用优化后的递送肽去提升 组织穿透、细胞摄取、核内递送和内体逃逸。 从资本市场角度看,PepGen 获得了相当强的专业投资者支持,吸引了 RA Capital、Viking等一批重量级机构。到 2025 年底,公司账上现金约 1.485 亿美元,足以支撑到 2027 年下半年。相比于Entrada,它还没拿到大 pharma 的重磅合作背书,但已经从资本市场上获得了相对充分的认可,是“值得继续押注的 POC 平台”。
Pepgen的PGN-EDODM1目前一期临床已经完成,且释放了二期的一个剂量组。在一期临床的结果中,总体耐受性不错。最高剂量组比市面上的AOC都要高,直接给到了15mg/kg。整个研究里最常见的不良事件是恶心、鼻咽炎和头痛,大多数都是轻度或中度。没有TEAE导致停药,也没有死亡;另外没有电解质问题、没有低镁血症。
Source: Pepgen 官网:FREEDOM-DM1: Final results from a Phase 1, placebo-controlled SAD study to evaluate PGN-EDODM1 in people with myotonic dystrophy type 1 (DM1)
疗效方面展现出非常明确的剂量依赖。剂量越高,剪接纠正越强,而且 15 mg/kg 已经到了非常明显的分子药效水平(53.7%的修正)。87.5% 的受试者在所有剂量里都看到了剪接改善,这一结果也被认为是目前DM1 适应症中报道的平均最高的剪接纠正。
Source: Pepgen 官网:FREEDOM-DM1: Final results from a Phase 1, placebo-controlled SAD study to evaluate PGN-EDODM1 in people with myotonic dystrophy type 1 (DM1)
考虑到在高剂量组中,患者会短暂发生eGFR下降 ,creatinine 上升和albuminuria 上升。虽然都很快恢复到正常水平,但是 Pepgen 还是保守地把二期临床的最高剂量组设置在12.5mg/kg。
PGN-EDO 二期临床最近刚刚释放了第一个剂量组(5mg/kg)在此队列中,总体耐受良好。所有AE都属于轻度或中度,没有SAE、没有剂量限制毒性(DLT)、没有超敏反应,也没有肾脏相关 TEAE。更重要的是,所有 8 名受试者完成了 4 次给药,没有看到累积毒性。最常见的不良事件是恶心。
Source: Pepgen 官网:FREEDOM2-DM1 5 mg/kg MAD Cohort Data Update
疗效方面,5 mg/kg MAD 队列在 Day 28 的 splicing correction 平均值是 7.3%,而 placebo 是 6.8%,表面上差距不大;结果里特别指出有一个splicing outlier,把整体均值拉低了,把这个 outlier 去掉后,5 mg/kg 组的平均剪接纠正升到 22.9%。
Source: Pepgen 官网:FREEDOM2-DM1 5 mg/kg MAD Cohort Data Update
此外,在手指放松速度的功能测试中,这里 PepGen 没有把数据包装成强阳性,而是表示,在最低剂量 5 mg/kg 下看到了有希望的趋势。从图上看,治疗组在随访过程中总体比 placebo 更往改善方向走;而且如果把 splicing outlier 排除,这个趋势会更清楚一些。当然,这还只是 PepGen 二期临床的第一个剂量组,从目前的趋势看,后面更高剂量组中能观测到更好的疗效是值得期待的。
Source: Pepgen 官网:FREEDOM2-DM1 5 mg/kg MAD Cohort Data Update
再看 Dyne Therapeutics。
不同于Avidity使用全长抗体(Fab+Fc),Dyne 使用了的Fab only抗体,从策略上,更像是把 AOC 这条路线进一步“产品化、平台化”的公司。它围绕 FORCE 平台搭建了一整套 TfR1-binding Fab + payload 的递送体系,在保留 TfR1 肌肉靶向能力的同时,尽可能改善组织渗透、CMC 和平台扩展性。
在Dyne发布的DYNE-101二期临床数据中,安全性总体可接受,但也有部分TEAE事件。所有队列里 100% 的受试者都出现过至少一个 TEAE,但大多是轻度或中度;没有严重相关 TEAE、没有因不良事件停药、没有死亡。最常见的 TEAE 是 鼻咽炎(41%)、操作相关疼痛(34%)、流感(30%)、输注反应(29%)、头痛(27%) 和 腹泻(23%)。总体来说,最高剂量组为 6.8mg/kg,没有爬到 Pepgen 和Avidity的水平,但是TEAE已经出现多例,在安全性比PGN-EDODM1稍逊。
Source: Dyne Therapeutics 官网:DYNE-101 Targets the Underlying Cause of DM1 to Enable Multi-system Functional Improvement in the ACHIEVE Trial
功能性层面上的表现目前还是朝着有利方向发展的。目前的数据显示,在 12 个月时,6.8 mg/kg Q8W 组在多个临床指标上都优于 placebo,尤其是 10-meter walk/run test、QMT total、middle-finger vHOT、5 times sit-to-stand、9-hole peg test 这些客观功能指标都朝有利方向变化。
Source: Dyne Therapeutics 官网:DYNE-101 Targets the Underlying Cause of DM1 to Enable Multi-system Functional Improvement in the ACHIEVE Trial
药效方面,虽然展现了明显的效果,但是最高剂量组的改善百分比只有33%,远低于Pepgen(53%)和Avidity(67%)的最高剂量组,在安全性和疗效上的均不具备显著优势。
Source: Dyne Therapeutics 官网:Safety and Efficacy of DYNE-101 in Adults With DM1: Phase 1/2 ACHIEVE Trial Data
3.2 受体靶向 POC
Ionis 和 Bicycle 合作的POC是基于TfR1受体的靶向肽来替代抗体。他们最后筛到的配体是 BCY17901,一种约 2 kDa 的双环肽(Bicycle),把它接到 ASO 或 siRNA 上后,能够在 human TfR1 knock-in 小鼠和非人灵长类(NHP)里明显增强肌肉递送与靶向的mRNA敲低。
Source: Østergaard et al. 2025, Nucleic Acids Research
他们先用噬菌体展示平台和结构生物学,从一批 Bicycle 双环肽里找出能结合 human TfR1 的候选物,并通过晶体结构和丙氨酸扫描做优化。这里的结合位点是 TfR1 上不与 transferrin 竞争且区别于抗体结合的新位点(晶体结构的PDB ID是9GH7),而且亲和力可以优化到纳摩尔到亚微摩尔范围。最终,BCY17901 被选为 lead ligand,因为它既保留了对 human TfR1 的高特异性,又有较好的可合成性和可偶联性。
在 human TfR1 KI 小鼠里做的体内筛选中,研发人员把 12 个不同的 Bicycle–Dmpk ASO 候选物做成 conjugate,比较它们在四肢肌肉和心脏里的 Dmpk knockdown。对比同样 3.5 mg/kg 的 ASO equivalents,BCY17901 在四肢肌肉和心脏的抑制效果都明显优于裸 ASO 。
Source: Østergaard et al. 2025, Nucleic Acids Research
尽管在小鼠及非人灵长类模型中展示了令人鼓舞的递送效率和靶标敲低效果,但受体靶向POC仍停留在临床前阶段,其数据更多反映的是平台在动物中的递送潜力。由于核酸药物在肌肉组织中的递送、细胞摄取及亚细胞分布在不同物种间存在显著差异,这类动物数据对人体疗效的预测能力仍然有限。
相比之下,AOC和CPP-POC等技术路线已经积累了一定的人体临床数据,其递送效率、安全性及功能改善的可转化性已有初步验证。因此,从当前数据来看,Bicycle这一路线仍处于机制验证阶段,与已有临床数据支持的平台尚不具备直接可比性。
3.3 靶点选择的影响 - POC的潜力发掘
Sarepta/Arrowhead 也有针对DM1的POC,其 avβ6 靶点的临床数据也相当不错。avβ6长期被认为是一个和纤维化相关的靶点,没想到在肌肉递送中也能大放异彩。整体就是改善的比例和效率要远高于现有所有的TfR1递送的AOC,以及POC。在 1.02mg/kg 的剂量组中,改善率就达到了50%。
有时候跳出热门靶点的内卷,也许会带来意想不到的效果。不过 αvβ6作为一个相对新的递送靶点,市面上也缺乏同靶点AOC的对标竞品,因此很难做横向比较。
Source: Sarepta 官网:Clinical Results and Plan Forward: SRP 1001 for Facioscapulohumeral Muscular Dystrophy Type 1 (FSHD1) SRP 1003 for Myotonic Dystrophy Type 1 (DM1)
从“平台之争”到“场景匹配” - AOC与POC的真正分水岭
从临床成熟度来看,AOC 目前整体仍然领先一步。Avidity 和 Dyne 已经在后期或较深临床阶段给出了较强的人体数据,AOC 这一路线已经完成了相当程度的产业验证。相比之下,Entrada 和 PepGen 所代表的 CPP-POC 虽然在人体验证深度上略慢,但它们展现出的安全性、组织分布能力,以及潜在的长期覆盖价值,同样说明 POC 并不是概念性补充,而是一条独立的路线。特别是当递送目标不只是成熟肌纤维,而是进一步触及肌卫星细胞、祖细胞池等更关键的细胞群体时,POC 的意义可能会被重新估值。
更值得注意的是,下一阶段真正拉开差距的,未必只是“抗体还是多肽”的载体形态之争,而可能是递送靶点的重新定义。无论是大热的 TfR1、异军突起的 αvβ6,还是在脑部递送中备受期待的新型受体(如 LRP1 等),都印证了一个残酷的真相:递送体系的上限,往往取决于“选对入口”,而不只是“换个载体”。从这个角度看,受体靶向 POC 的历史使命绝不仅是简单平替 AOC,而是利用其更小、更灵活、更易制造的分子优势,去探索全新的组织大门,寻找新的药代与药效(PK/PD)平衡点。
从更长期的视角推演,AOC 和 POC 并不会收敛为同一条路线,而是会基于生理学底层的逻辑,在不同场景下确立各自的主场。对于当前需要快速推动临床转化、强调高度特异性与受体介导(如现阶段的跨BBB递送)的项目,AOC 凭借成熟的工程化体系确实更具确定性;更长期来看,对于需要大范围组织覆盖、深层细胞进入(如肌肉和实体瘤),甚至是跨越BBB后需要在致密的脑实质中实现均匀扩散的项目,POC则展现出了远超抗体的理论上限与平台弹性。
真正激动人心的未来,不是一场“谁替代谁”的零和博弈,而是递送技术跨越了单一平台的盲目内卷,全面走向“按物理规律与场景匹配的分层演化”。
作者:ZXY
审稿:WJY FYW WDD ZZD
关于RiDYMO.PepTx 高质量多肽发现和优化平台
RiDYMO.PepTx 是深势科技基于 AI for Science 打造的多肽发现与优化平台,依托 Hermite® 与 RiDYMO® 两大平台,将物理机制驱动的AI与孔板级高通量实验紧密闭环。平台的核心方法——强化动力学(Reinforced Dynamics, RiD),在分子模拟采样效率上实现近百倍提升,通过充分结合神经网络的高维表征能力,RiD能有效捕捉复杂生物大分子体系中的动态构象变化。平台可在1 个月内,从2000+ 天然和非天然氨基酸组合构成的超万亿级环肽化学空间中高效筛选,并以透膜性、稳定性、溶解度等多维成药性与可开发性指标并行优选,确保在“更大化学空间、 更快筛选速度、更可落地性质”的目标下,大幅缩短先导环肽周期,并显著提升成功率。平台已在多个项目中实现从头发现线肽和环肽,以及多肽药物、POC/RDC/PDC 药物的系统优化。
关于Hermite®一站式药物计算设计平台
Hermite®是深势科技打造的基于人工智能、物理建模和高性能计算的一站式药物计算设计平台。Hermite®以网页应用形式提供从靶点结构解析、苗头化合物筛选,到先导优化和性质预测的全流程工具,并提供蛋白、抗体设计解决方案。Hermite®提供友好的可视化交互界面、功能实时更新,同时支持本地和云上的私有化部署。
平台地址:https://hermite.dp.tech
关于深势科技
深势科技是全球AI for Science的领导者,致力于将人工智能与科学研究及工业研发深度融合。依托在交叉学科领域的深耕,深势科技构建了“深势·宇知”大模型体系,推动药物研发、能源材料、信息技术等关键领域的突破性进展,助力“千行百业”的科研从“实验试错/计算机”跨入“预训练模型时代”。作为国家高新技术企业、专精特新“小巨人”,深势科技拥有中科院院士领衔的科研团队,博士及博士后占比超35%,核心成果曾获全球高性能计算最高奖“戈登贝尔奖”,入选中国十大科技进展。
商务合作:bd@dp.tech
媒体垂询:PR@dp.tech
更多信息,请访问网站:www.dp.tech
2026-03-31
A new cut of Phase 2 data on PepGen’s beleaguered oligonucleotide therapy for a genetic nerve and muscle disease has shown disappointing efficacy compared with analysts’ expectations.
The therapy, known as PGN-EDODM1, is intended to correct genetic errors, or mis-splicing, in patients with a condition called myotonic dystrophy type 1 (DM1). This is a potentially fatal disease that causes muscle weakness and wasting, among other symptoms. It is the company’s only drug candidate after it
ditched
its Duchenne muscular dystrophy programs last year.
Monday’s data come from six patients given monthly 5 mg/kg doses of PGN-EDODM1, the lowest dose tested in the trial, which is called FREEDOM2, and is on
partial clinical hold
in the US.
At three months, the mean splicing correction with PGN-EDODM1 was 7.3%, compared with 6.8% in two placebo-treated patients. PepGen said that there was an outlier among the treated patients in this dose group whose mis-splicing actually worsened by an astonishing 70.8%.
Speaking on a call with analysts Monday, PepGen CEO James McArthur said “that particular patient did clearly have drug on board — in fact, relatively significant levels of drug on board. But we do not understand why that particular patient did not see an improvement in splicing.”
He added that the outlier data does not appear to be a result of an error with the assay used to determine splicing correction.
The company said that if data on that subject were to be disregarded, the average splicing correction in the other five patients was 22.9%. This argument does not seem to have cut much ice with investors, as PepGen’s shares
$PEPG
were down 47% before trading opened on Tuesday.
Writing before the data were released, Leerink analysts said they were expecting splicing correction at this dose in the high-teens range. This was based on management’s expectation that splicing correction would improve by about 50% from the levels seen in PepGen’s earlier single-dose trial.
Adding to the confusion, one of the placebo-treated patients in FREEDOM2 had a 27.2% splicing correction — better than four of the six patients who got PepGen’s drug.
The company said there were “promising trends” in myotonia, or prolonged contraction of the muscles, in patients’ hands. This is measured using the time it takes a patient to open their hand, or more specifically, to extend their middle finger.
Before dosing, it took patients about 10.3 seconds to fully extend their middle fingers. This improved by up to 4.1 seconds in the PGN-EDODM1-treated patients through week 13, but returned to the baseline measurement at week 16.
And the 5 mg/kg dose of PGN-EDODM1 did not prompt any meaningful improvement in the time it took for patients to walk or run 10 meters, or in handgrip strength.
The oligonucleotide was well tolerated in FREEDOM2, PepGen said. All side effects were mild to moderate, with no serious adverse events and no evidence of cumulative toxicity.
McArthur said on Monday that the company expects to be able to improve on the splicing results in future, higher dose groups. Data from a 10 mg/kg cohort are expected in the second half of 2026, and a third 12.5 mg/kg dose group is planned.
“We’re certainly hoping that we do not see another outlier like we saw in this particular group, which definitely skewed the group analysis which we provided,” he said.
The FDA put FREEDOM2 on partial clinical hold earlier this month, owing to a possible safety signal seen in mouse data, so PepGen cannot recruit new patients in the US. All the patient recruitment and dosing is occurring in the trial’s other territories, Canada and the UK.
McArthur said on the call that PepGen believes it can address the questions the FDA has raised about the program. But he could not provide a timeline for a resolution, “given the way things are right now with the FDA,” he said.
寡核苷酸临床结果临床2期
100 项与 PGN EDODM1 相关的药物交易
登录后查看更多信息
研发状态
10 条进展最快的记录, 后查看更多信息
登录
| 适应症 | 最高研发状态 | 国家/地区 | 公司 | 日期 |
|---|---|---|---|---|
| 肌强直性营养不良 | 临床2期 | 加拿大 | 2024-12-10 | |
| 肌强直性营养不良 | 临床2期 | 新西兰 | 2024-12-10 | |
| 肌强直性营养不良 | 临床2期 | 英国 | 2024-12-10 |
登录后查看更多信息
临床结果
临床结果
适应症
分期
评价
查看全部结果
| 研究 | 分期 | 人群特征 | 评价人数 | 分组 | 结果 | 评价 | 发布日期 |
|---|
临床1期 | 8 | 鑰窪齋獵構艱鹽醖襯鬱(構齋構獵醖遞艱簾膚鏇) = 繭構齋膚糧糧淵窪鬱淵 範廠膚夢遞艱餘遞構觸 (襯齋蓋鬱簾觸淵遞獵範 ) | 积极 | 2026-03-09 | |||
鑰窪齋獵構艱鹽醖襯鬱(構齋構獵醖遞艱簾膚鏇) = 壓鑰築繭製壓鏇淵艱顧 範廠膚夢遞艱餘遞構觸 (襯齋蓋鬱簾觸淵遞獵範 ) | |||||||
临床1期 | 24 | PGN-EDODM1 5 mg/kg | 憲願壓願襯廠鏇餘餘糧(壓範鑰鹹網壓醖齋艱餘) = 齋鏇蓋齋醖鹽願遞糧製 窪鹽艱獵簾憲製選淵遞 (艱餘願構窪蓋壓糧遞憲 ) 更多 | 积极 | 2025-09-24 | ||
PGN-EDODM1 10 mg/kg | 憲願壓願襯廠鏇餘餘糧(壓範鑰鹹網壓醖齋艱餘) = 夢鬱艱窪襯遞範鹽艱願 窪鹽艱獵簾憲製選淵遞 (艱餘願構窪蓋壓糧遞憲 ) 更多 | ||||||
临床1期 | 16 | 醖選夢遞顧齋構膚蓋簾(蓋夢鬱選窪膚糧獵醖膚) = 夢齋鹹糧艱遞壓繭憲衊 觸顧鬱製餘遞鬱廠窪鏇 (簾構衊餘觸簾廠積觸網 ) | 积极 | 2025-02-24 | |||
壓積糧醖淵糧觸夢襯構(襯製齋積鬱鏇襯簾網願) = 鬱顧範夢窪壓襯鑰醖糧 獵製範積鏇遞願遞鑰襯 (憲觸構醖積膚築憲醖遞 ) 更多 |
登录后查看更多信息
转化医学
使用我们的转化医学数据加速您的研究。
登录
或

药物交易
使用我们的药物交易数据加速您的研究。
登录
或

核心专利
使用我们的核心专利数据促进您的研究。
登录
或

临床分析
紧跟全球注册中心的最新临床试验。
登录
或

批准
利用最新的监管批准信息加速您的研究。
登录
或

特殊审评
只需点击几下即可了解关键药物信息。
登录
或

生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用