预约演示
更新于:2026-04-18
Tonlamarsen
更新于:2026-04-18
概要
基本信息
在研机构 |
权益机构- |
最高研发阶段临床2期 |
首次获批日期- |
最高研发阶段(中国)- |
特殊审评- |
登录后查看时间轴
结构/序列
使用我们的RNA技术数据为新药研发加速。
登录
或
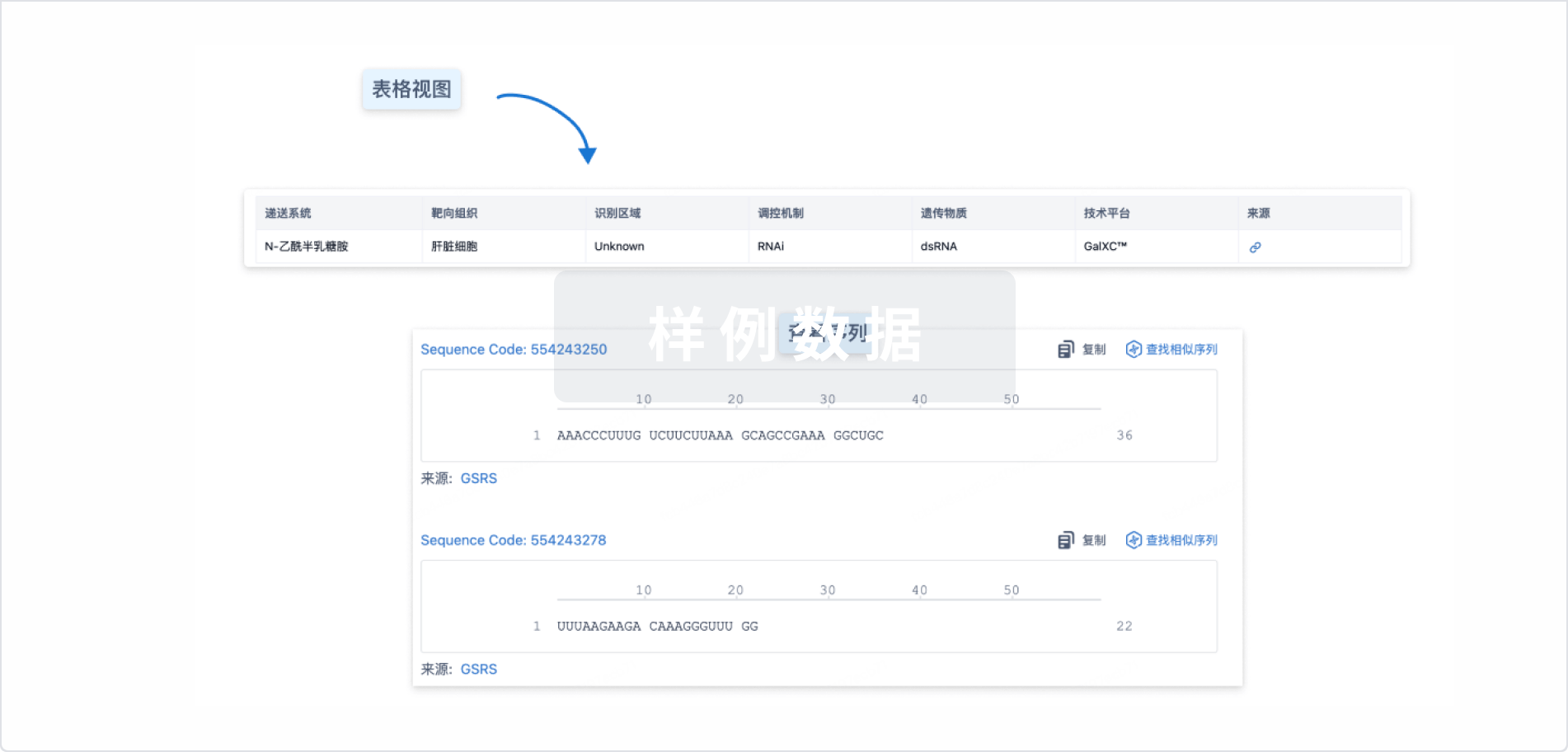
Sequence Code 317493258

来源: *****
关联
4
项与 Tonlamarsen 相关的临床试验NCT07511361
A Phase 2b, Randomized, Double-Blind, Placebo-Controlled, Multicenter Study to Evaluate the Efficacy and Safety of Tonlamarsen in Participants With Acute Severe Hypertension Recently Discharged From the Hospital
The purpose of this study is to evaluate the blood pressure-lowering effect of tonlamarsen in adult participants who were recently discharged from the hospital, after treatment for acute severe hypertension.
开始日期2026-04-20 |
申办/合作机构 |
NCT06864104
A Randomized, Double-Blind, Placebo-Controlled Study to Assess the Efficacy and Safety of Monthly Subcutaneous Administration of Tonlamarsen in Participants With Uncontrolled Hypertension (KARDINAL)
The purpose of this study is to evaluate the blood pressure (BP)-lowering efficacy of tonlamarsen in adult participants who, despite taking 2 or more antihypertensive medications, have not achieved their target blood pressure (BP).
开始日期2025-04-10 |
申办/合作机构 |
NCT05314439
A Double-Blind, Placebo-Controlled, Phase 2 Study to Assess the Safety, Tolerability and Efficacy of Monthly Subcutaneous Administration of ION904 in Patients With Uncontrolled Hypertension
The purpose of this study is to evaluate the effect of ION904 on plasma angiotensinogen (AGT) in participants with uncontrolled hypertension.
开始日期2022-04-29 |
申办/合作机构 |
100 项与 Tonlamarsen 相关的临床结果
登录后查看更多信息
100 项与 Tonlamarsen 相关的转化医学
登录后查看更多信息
100 项与 Tonlamarsen 相关的专利(医药)
登录后查看更多信息
33
项与 Tonlamarsen 相关的新闻(医药)2026-04-13
·知新行馆
老年患者血压轨迹与心血管结局:来自STEP试验的见解
期刊: J Am Coll CardiolPMID: 41949524
简述:该研究基于STEP试验分析7296例接受强化降压治疗的高血压患者,识别出7种收缩压控制轨迹,证实快速且稳定的血压控制模式心血管风险最低,血压控制的速度、稳定性和质量是心血管结局的独立预测因素,高血压管理应优先实现快速、稳定、持续的血压控制。摘要翻译:
背景:强化血压控制是预防心血管疾病的基础。然而,个体反应的异质性——具体而言是控制速度、稳定性和控制质量——仍不明确。
目的:本研究旨在明确血压反应的多样性,描述强化治疗期间不同的收缩压控制轨迹特征,并评估其对心血管结局的预后价值。
方法:研究分析了来自老年高血压患者血压干预策略(STEP)试验的7296例接受强化降压治疗的高血压患者,包括原始强化治疗组以及延长期内转为强化治疗的标准治疗组患者。基于前12个月的血压数据,根据控制速度(达标时间)和稳定性(随访间平均真实变异性)对患者进行分组。同时为每位患者计算四项连续指标:达标时间、平均真实变异性、目标范围内时间和累积收缩压负荷。主要结局为复合心血管事件。在对潜在混杂因素进行序贯校正后,采用Fine-Gray亚分布风险模型估计风险比。
结果:接受强化治疗的患者间存在显著的异质性,研究共识别出7种轨迹。理想的快速-稳定组主要结局发生率最低,且安全性良好。与快速-稳定组相比,其他所有轨迹均与心血管风险显著且进行性升高相关。具体而言,未控制轨迹组风险最高(风险比≥2.0),不稳定组(风险比1.83)、延迟达标组(风险比1.81~1.89)和快速-不稳定组(风险比1.50)风险亦显著升高。此外,连续动态指标是心血管结局的独立决定因素。达标时间每延迟1个月,心血管风险增加3%(风险比1.03;95%置信区间1.01~1.04)。更高的变异性(每增加1个标准差,风险比1.13)、更高的收缩压负荷(每增加1个标准差,风险比1.21)和更低的目标范围内时间(每升高10%,风险比0.95)均与不良结局显著相关。
结论:血压控制轨迹可有效预测心血管风险。快速-稳定模式可获得最佳获益,而任何偏离该模式的情况——以延迟、不稳定或控制失败为特征——均会显著升高风险。血压控制的速度、稳定性和质量是心血管结局的独立预测因素。高血压管理应优先实现快速、稳定且持续的血压控制。
肥胖与射血分数降低的心力衰竭:综述
期刊: J Am Coll CardiolPMID: 41949514
简述:该综述指出肥胖与射血分数降低的心力衰竭常共存,现有研究对二者关联的认知不足,BMI作为肥胖替代指标存在局限性,还存在肥胖悖论;肥胖是射血分数降低心力衰竭的可修饰性疾病放大器,减重干预具有潜在应用价值,但相关循证证据有限,新型减重药物尚未在该人群中专项评估。摘要翻译:
肥胖与射血分数降低的心力衰竭常同时存在。事实上,大多数射血分数降低的心力衰竭患者存在超重或合并肥胖。尽管患病率极高,但射血分数降低的心力衰竭相关肥胖问题受到的关注有限,因为近期大多数综述均聚焦于广义的心力衰竭或射血分数保留的心力衰竭表型。这种关注度不足的部分原因在于,肥胖与射血分数保留的心力衰竭之间存在更强、更一致的病理生理和流行病学关联,而肥胖与射血分数降低的心力衰竭之间的关系仍未得到充分阐明。这种不确定性很大程度上源于各项研究中关于体重指数是否能独立预测新发射血分数降低的心力衰竭的结果不一致,而体重指数作为肥胖替代标志物的局限性已被广泛认可。射血分数降低的心力衰竭患者中存在的肥胖悖论——即更高的体重指数通常与更好的结局相关——进一步加剧了这些挑战。尽管如此,在射血分数降低的心力衰竭中,肥胖可被更好地定义为一种可修饰的疾病放大器,影响疾病进展和临床轨迹,而非主要病因驱动因素。肥胖药物治疗等减重干预措施已被证实对肥胖合并射血分数保留的心力衰竭患者安全有效,肥胖相关的病理生理过程(包括全身炎症、神经激素激活、局部旁分泌信号传导和机械负荷)是该疾病发生的明确诱因。尽管这些过程在射血分数降低的心力衰竭中的相对作用可能不同,但过度肥胖仍可通过这些途径加剧疾病进展,提示减重干预可能适用于该人群。迄今为止,射血分数降低的心力衰竭相关研究证据仍有限,且主要来自方法学受限的非肥胖专项试验。尽管多种新型减重药物已研发问世,但尚无药物在射血分数降低的心力衰竭患者中开展专项评估,这限制了对其在该人群中潜在临床价值的认知。
健康与心血管疾病中的线粒体来源囊泡和线粒体细胞外囊泡
期刊: Circ ResPMID: 41955324
简述:该综述总结线粒体来源囊泡和线粒体细胞外囊泡的生成机制、分离方法,阐述二者在线粒体质量控制、细胞器及细胞间通讯中的作用,重点探讨线粒体细胞外囊泡在心血管疾病中的致病作用与治疗潜力。摘要翻译:
线粒体来源囊泡和线粒体细胞外囊泡是线粒体动力学的两种相关延伸形式,将细胞器维持与细胞内及细胞间通讯联系起来。线粒体来源囊泡是直接从线粒体出芽形成的小型囊泡,可选择性包裹线粒体外膜、内膜或基质成分。该囊泡作为局部质量控制机制,可清除氧化或受损物质,无需启动完整的线粒体自噬机制。出芽后,线粒体来源囊泡通常进入内溶酶体途径,与晚期内体或溶酶体融合以降解内容物。部分线粒体来源囊泡还可靶向其他细胞器(尤其是过氧化物酶体),参与细胞器通讯、脂质代谢和氧化还原平衡。相比之下,释放到细胞外间隙的线粒体细胞外囊泡包含完整的功能性线粒体、线粒体内容物(蛋白质、DNA/RNA、脂质等)和非线粒体货物(如mRNA、非编码RNA等),可转移至受体细胞,进而产生致病或有益效应。因此,线粒体细胞外囊泡参与代谢协同、免疫调节、组织重塑和衰老过程。本综述总结了线粒体来源囊泡和线粒体细胞外囊泡多种生成机制的最新研究进展,以及二者的分离方案。重点阐述了线粒体来源囊泡和线粒体细胞外囊泡在调控线粒体质量/数量控制、健康与疾病状态下细胞内细胞器及不同细胞类型间多层级通讯中的作用。最后,还探讨了线粒体细胞外囊泡介导的心血管疾病致病作用和治疗潜力。
外泌体-线粒体杂合膜靶向递送一氧化碳改善心肌缺血再灌注损伤、抑制线粒体功能障碍和细胞焦亡
期刊: ACS NanoPMID: 41945782
简述:该研究构建外泌体-线粒体杂合膜载体包裹一氧化碳,可靶向递送至心肌细胞线粒体,减少线粒体活性氧生成、维持线粒体功能、抑制细胞焦亡,促进巨噬细胞极化减轻炎症,有效改善心肌缺血再灌注损伤、减小梗死面积并保护心功能。摘要翻译:
心肌缺血再灌注损伤会加重临床血运重建后的心脏功能障碍和心力衰竭。其主要机制包括活性氧异常蓄积,诱导线粒体功能障碍、触发细胞焦亡并放大免疫炎症反应。本研究构建了外泌体-线粒体杂合膜载体包裹一氧化碳,用于靶向递送一氧化碳以减轻心肌缺血再灌注损伤。得益于外泌体的黏附特性和线粒体膜的同源线粒体靶向能力,该载体可实现从梗死心肌到心肌细胞线粒体的序贯靶向。线粒体中释放的一氧化碳可减少异常线粒体活性氧生成,维持线粒体功能,进而减少线粒体DNA释放,在体内外抑制细胞焦亡。此外,单次静脉注射该载体可通过促进巨噬细胞从M1型向M2型极化,减轻心脏组织的炎症放大效应,有效减少促炎细胞因子释放、抑制炎症反应,从而减轻心肌梗死并改善心脏功能。综上,本研究结果表明,通过靶向气体疗法恢复线粒体功能、清除活性氧并抑制细胞焦亡具有改善心肌缺血再灌注损伤的潜力。
细胞间线粒体转移:对心血管健康的意义
期刊: Circ ResPMID: 41955329
简述:该综述总结心血管系统中细胞间线粒体转移的证据与功能结局,明确该领域的知识空白和关键问题,探讨研究面临的挑战与机遇,细胞间线粒体转移可在心血管细胞间及器官间通讯,其效应具有双重性。摘要翻译:
线粒体是调控代谢稳态、细胞命运和存活的重要细胞器。新兴证据表明,线粒体并非局限于细胞内。细胞间线粒体转移在多种细胞间(包括心血管系统主要细胞类型)的发生已得到越来越多的认可。共培养体系、遗传谱系示踪技术和动物模型的研究结果证实,线粒体可在心肌细胞、成纤维细胞、内皮细胞、血管平滑肌细胞、心脏巨噬细胞和间充质基质细胞之间转移。也有研究报道线粒体可在远端器官(如脂肪组织)与心脏之间转移,提示线粒体转运不仅可介导单个细胞间的通讯,还可实现跨器官通讯。目前已报道两种主要的细胞间线粒体转移方式:一种是通过膜接触或纳米管进行直接、接触依赖性的线粒体转运;另一种是线粒体以膜结合囊泡包裹形式或游离形式释放到细胞外间隙,再被受体细胞摄取。细胞间线粒体转移的效应可有益或有害,具体取决于细胞类型和转移发生的条件。然而,线粒体转移的具体机制及其效应尚未完全阐明,因此对细胞间线粒体转移在心血管健康中的作用仍存在一定的推测性。本综述首先总结了心血管系统中细胞间线粒体转移的证据及观察到的功能结局,随后明确该领域的知识空白和亟待解决的关键问题,最后探讨了推动该领域发展的挑战与机遇。
弥合线粒体介导的免疫与线粒体转移之间的关联
期刊: Circ ResPMID: 41955328
简述:该综述阐述线粒体除供能外的免疫调控作用,血小板是循环线粒体的主要来源,细胞外线粒体可激活先天免疫、参与自身免疫及心血管疾病,线粒体移植具有治疗潜力,明确线粒体免疫与转移的关联对心血管疾病研究至关重要。摘要翻译:
线粒体是除提供能量外还参与调控多种细胞功能的细胞器。在病毒感染或线粒体复制缺陷时,机体通过线粒体孔形成启动复杂的自我破坏机制,导致线粒体DNA释放到细胞质中,进而触发先天免疫反应。血小板是循环线粒体的主要来源,越来越多的证据表明血小板可主动释放线粒体,部分线粒体被包裹在细胞外囊泡中。该过程在自身免疫性疾病中增强,发生于血小板储存期间,并与血小板输注后的不良反应相关。细胞外线粒体可作为损伤相关分子模式的载体,在抗磷脂综合征、心肌病等多种疾病中成为抗体的作用靶点。此外,抗线粒体抗体水平升高还与系统性红斑狼疮患者的死亡率增加和心血管风险升高相关。线粒体移植(即用健康线粒体替换组织或器官中受损的线粒体)作为一种治疗手段受到越来越多的关注。因此,明确细胞外线粒体与免疫系统的相互作用愈发重要。本综述总结了当前关于线粒体多维度免疫作用的研究进展,重点关注血小板及血小板来源线粒体作为核心生物学场景的研究。
儿童运动员的心脏评估
期刊: Eur Heart JPMID: 41967042
简述:该临床共识由欧洲心脏病学会相关协会发布,首次针对16岁以下儿童运动员提出心脏筛查建议,推荐结合病史、体格检查和心电图筛查,12岁前启动筛查,必要时行超声心动图,明确心电图判读标准,指出需进一步优化筛查策略并加强专科人才培养。摘要翻译:
儿童运动员并非简单的“小型成人”。现有的大多数儿童运动员心脏筛查建议主要基于成人证据,专为成人运动员设计。由于心脏生理、发育和生长的特殊性,年龄相关的疾病表现、改良的诊断路径、运动训练适应性以及相关伦理问题,需要制定儿童专属的筛查建议。本临床共识文件由欧洲心脏病学会欧洲预防心脏病学协会和欧洲儿科与先天性心脏病协会联合发布,首次基于专家共识及现有儿童运动员人群研究数据,提出儿童运动员专属心脏筛查建议。撰写组专家对核心建议条款进行匿名投票,达成共识需≥80%的同意率。本文件所有建议适用于16岁以下儿童运动员(包括12岁以下儿童)。文件建议,儿童运动员心脏筛查应包含个人及家族病史采集、体格检查和12导联静息心电图检查,且筛查启动时间不晚于12岁。实施筛查项目需确保具备必要的医疗资源。在具备基线诊断评估完善基础设施的前提下,可进行一次经胸超声心动图检查,以识别心电图无法检出的高危结构性心脏病。文件还明确了儿童运动员心电图正常、临界和异常结果的定义标准,并针对初始检查发现可疑结果后的进一步评估提供了详细建议。文件强调,需进一步开展研究以优化筛查策略、准确评估和量化心源性猝死风险,并为合并心脏疾病的儿童运动员提供基于证据的参赛资格建议。同时指出,需增加儿童运动心脏病学专科培训机会,为儿童运动员群体提供充足的医疗保障。
工程化免疫抑制树突状细胞抵御心脏重构
期刊: NaturePMID: 41951742
简述:该研究构建靶向纤维化的工程化免疫抑制树突状细胞,在小鼠和非人灵长类心肌梗死模型中均可减轻心脏炎症纤维化、改善心肌灌注和收缩功能,其通过抑制免疫基质细胞活化、扩增调节性T细胞发挥作用,为心脏重构和心力衰竭提供新治疗策略。摘要翻译:
心力衰竭仍是导致发病和死亡的主要原因,但目前尚无获批的疗法能有效预防或逆转病理性心脏纤维化及伴随的心脏功能下降。慢性炎症是缺血或血流动力学应激后病理性纤维化的核心驱动因素,但缺乏在不引发全身免疫抑制的前提下局部平衡损伤性与修复性免疫反应的治疗策略。树突状细胞是免疫激活和免疫耐受的关键调控因子,为心脏疾病的治疗性免疫重编程提供了契机。本研究证实,工程化免疫抑制、靶向纤维化的树突状细胞可有效抵御病理性心脏重构。在缺血再灌注损伤、心肌梗死和压力负荷小鼠模型中,该细胞治疗可减轻炎症性心脏纤维化、改善心肌灌注并保留心肌收缩力。机制上,工程化树突状细胞可通过直接抑制免疫和基质细胞活化,或间接促进调节性T细胞克隆扩增,发挥持续的心脏保护作用。重要的是,在非人灵长类心肌梗死模型中,该细胞治疗同样可减轻心脏纤维化、改善心肌灌注和收缩功能,且未引发全身毒性。这些研究结果证实,病灶靶向免疫调节是控制心脏纤维化的可行策略,工程化树突状细胞是治疗心脏重构和心力衰竭的极具潜力的治疗平台。
载脂蛋白B、非高密度脂蛋白胆固醇和低密度脂蛋白胆固醇目标用于一级预防降脂治疗的成本效益
期刊: JAMAPMID: 41949879
简述:该经济评估通过计算机模拟模型证实,与低密度脂蛋白胆固醇、非高密度脂蛋白胆固醇相比,以载脂蛋白B为目标指导降脂治疗强化方案具有成本效益,可改善人群健康,是一级预防降脂治疗的优选标志物。摘要翻译:
重要性:载脂蛋白B是降脂治疗患者残余动脉粥样硬化性心血管疾病风险的优质标志物,优于低密度脂蛋白胆固醇和非高密度脂蛋白胆固醇。目前尚未明确低密度脂蛋白胆固醇、非高密度脂蛋白胆固醇和载脂蛋白胆固醇目标的成本效益。
目的:明确基于低密度脂蛋白胆固醇、非高密度脂蛋白胆固醇和载脂蛋白B目标强化降脂治疗用于一级预防的相对成本效益。
设计、场所和研究对象:本经济评估采用计算机模拟模型,评估根据低密度脂蛋白胆固醇、非高密度脂蛋白胆固醇或载脂蛋白B目标,使用高强度他汀类药物或依折麦布强化降脂治疗的成本效益。研究队列纳入25万名符合他汀治疗指征、无动脉粥样硬化性心血管疾病的美国成人,数据来源于2005-2016年美国国家健康与营养检查调查参与者(4149例)。受试者在血脂筛查后进入模拟流程,并根据2018年美国心脏协会/美国心脏病学会指南接受他汀治疗。模型输入数据来源于全国调查数据、汇总纵向队列研究和已发表文献。采用传统和概率敏感性分析探索结果不确定性。
干预措施:若受试者未达到治疗后低密度脂蛋白胆固醇<100mg/dL、非高密度脂蛋白胆固醇<118mg/dL或载脂蛋白B<78.7mg/dL的目标,则强化降脂治疗。
主要结局和指标:终身质量调整生命年和成本(2025年美元计价),每年按3.0%贴现。主要结局为增量成本效益比。若每获得一个质量调整生命年的成本低于120000美元,则认为该策略具有成本效益。
结果:与低密度脂蛋白胆固醇目标相比,非高密度脂蛋白胆固醇目标可增加965个质量调整生命年(95%不确定区间-3551~5341),同时减少210万美元成本(95%不确定区间-9420万~9200万美元)。与非高密度脂蛋白胆固醇目标相比,载脂蛋白B目标可增加1324个质量调整生命年(95%不确定区间-2602~5669),同时增加4020万美元成本(95%不确定区间-4360万~1.34亿美元),增量成本效益比为每质量调整生命年30300美元。在每质量调整生命年120000美元的支付意愿阈值下,65%的概率分析显示载脂蛋白B目标为最优策略,25%显示非高密度脂蛋白胆固醇目标为最优策略。载脂蛋白B检测成本极低,成本升高主要源于预期寿命延长和预防性治疗周期延长。
结论和意义:本计算机模拟研究结果表明,载脂蛋白B可作为指导一级预防降脂治疗的成本效益标志物,改善人群健康。
嗅觉受体激活通过肌动蛋白细胞骨架重塑降低血小板反应性与动脉血栓形成
期刊: CirculationPMID: 41944007
简述: 本研究将血小板中嗅觉受体家族的孤儿G蛋白偶联受体作为新型抗血栓治疗靶点,通过工程化报告细胞系高通量筛选出8000种非气味生物活性化合物,鉴定出6种OR2L13特异性激动剂;先导激动剂CCF0054500可磷酸化血小板HSP27并破坏肌动蛋白细胞骨架,在动物动脉损伤与心肌梗死模型中显著降低血小板聚集和反应性,且不影响纤维蛋白生成与止血功能,为抗血小板治疗提供了全新作用通路。摘要翻译:
背景: 即便接受抗血小板治疗,部分患者仍因药物无反应性或高残留血小板反应性处于缺血高风险状态。本研究旨在靶向血小板中嗅觉受体家族的孤儿G蛋白偶联受体,以此作为新型抗血栓策略。
方法: 采用表达人源OR2L13的工程化报告细胞系(该孤儿G蛋白偶联受体与限制血小板反应性相关),对8000种非气味生物活性化合物进行高通量筛选并开展反向筛选验证。后续研究评估了健康受试者以及冠心病、外周动脉疾病患者的血小板功能;磷酸化蛋白质组学分析揭示了关键信号通路,同时通过体外及体内实验评估先导化合物对血小板信号传导、生物力学特性以及动静脉血栓形成的影响。
结果: 研究鉴定出6种OR2L13特异性激动剂,这类激动剂可通过多种受体抑制血小板聚集与α颗粒胞吐作用,提示存在共同的下游介导因子。先导激动剂CCF0054500可使血小板热休克蛋白27发生磷酸化,破坏肌动蛋白细胞骨架并减少血块收缩(血块面积70.6对比5.2;P<0.0001),该效应可通过抑制HSP27逆转。在小鼠动脉损伤模型中,CCF0054500使血小板聚集量降低88.9%(P<0.0003),且不影响纤维蛋白生成或止血功能。在高残留血小板反应性的心肌梗死模型中,CCF0054500可降低血小板反应性(P<0.0001)并改善左心室功能(P=0.0007)。
结论: 本研究通过全面探究靶向孤儿血小板G蛋白偶联受体的新型血小板抑制剂,首次报道并表征了一种非嗅觉探针,该探针可通过下游HSP27抑制血小板活化与血栓形成。
新发心房颤动患者卒中风险预测的可解释机器学习模型
期刊: NPJ Digit MedPMID: 41946928
简述: 心房颤动是缺血性卒中的主要病因,现有CHA₂DS₂-VASc评分预测效能有限。本研究仅基于年龄、合并症与用药情况,构建并外部验证了逻辑回归、普拉特校准极端梯度提升两种可解释机器学习模型,用于预测新发房颤患者1年卒中风险;模型内部与外部验证AUC显著优于传统评分,校准度与临床实用性良好,长期随访证实其风险分层与治疗反应评估价值,可指导真实世界直接口服抗凝药的启用。摘要翻译:
心房颤动是最常见的持续性心律失常,也是缺血性卒中的主要病因。现有风险评分(如CHA₂DS₂-VASc评分)预测准确性有限,无法捕捉复杂的临床特征模式。为提升模型泛化性与临床应用价值,本研究仅采用年龄、合并症与用药情况,构建并外部验证了具有临床可解释性的机器学习模型,用于预测新发心房颤动患者的1年卒中风险。逻辑回归与普拉特校准极端梯度提升模型在内部验证队列中AUC分别为0.915与0.914,外部验证队列中AUC为0.877-0.886,均显著优于CHA₂DS₂-VASc评分(AUC为0.614-0.621;p<0.001)。校准曲线与决策曲线分析证实模型具备良好的临床应用价值。长期随访结果显示,逻辑回归定义的高危组在风险分层与治疗反应评估方面表现更优。该类模型可提供准确、个体化的卒中风险评估结果,为真实世界医院场景下直接口服抗凝药的启用提供指导。
线粒体向内皮细胞的转移:机制、证据与治疗潜力
期刊: Circ ResPMID: 41955325
简述: 本篇综述系统阐述线粒体作为血管健康核心调控因子的多重作用,其功能远超能量代谢,还参与氧化还原平衡、钙稳态调控与生物合成支持;近年研究证实线粒体可参与血管系统的细胞间通讯,外源性线粒体递送已从临床前概念验证推进至人体首次试验,可实时分离并输注线粒体至危重患者且早期结局良好。本文梳理线粒体功能与内皮细胞生理的关联、血管线粒体转移的新兴领域进展,以及线粒体移植的临床转化路径,为血管保护与疾病治疗开辟新方向。摘要翻译:
线粒体日益被认为是血管健康的核心调控因子,其对内皮细胞功能的调控作用远不止于能量产生。除协调氧化还原平衡、钙动力学与生物合成支持外,近期研究发现线粒体可参与细胞间通讯,且血管系统中存在线粒体转移的相关证据。相关研究同步推动外源性线粒体的主动递送从临床前概念验证阶段发展至人体首次试验,证实可实时分离新鲜细胞器并输注至危重患者,且早期结局良好。上述获益的潜在机制尚未完全明确,高效、可规模化的递送策略仍在探索中。本篇综述重点梳理线粒体功能与内皮细胞生理相关的最新证据,聚焦血管系统中线粒体转移这一新兴且快速发展的领域,并探讨线粒体移植如何从实验概念向临床转化推进。这些研究进展共同为保护血管完整性、治疗相关疾病指明了新的治疗途径。
T-World虚拟人类心肌细胞Ⅰ:构建、验证与细胞心律失常发生
期刊: Circ ResPMID: 41944014
简述: 心血管疾病是全球发病与死亡首要原因,现有虚拟心肌细胞模型通用性不足,限制其转化应用。本研究开发新型T-World虚拟人类心肌细胞模型,基于数据驱动微分方程构建性别特异性兴奋-收缩偶联、机械收缩与β肾上腺素能信号通路,优化钙信号相关通道与转运体参数;模型可重现频率依赖性动作电位、钙处理与收缩特性,首次完整模拟所有关键致心律失常细胞机制,证实女性心肌细胞更易出现早期后除极与动作电位时程陡复极,为心律失常多尺度研究提供开源工具。摘要翻译:
背景: 心血管疾病是全球发病率与死亡率的首要原因,亟需新技术提升机制认知并指导治疗策略。以人类为核心的心脏模拟研究极具应用前景,但现有细胞模型仅能重现少数致心律失常行为,且与实验数据存在显著差异。本研究旨在构建一种新型模型,克服现有模型通用性不足的缺陷,该缺陷显著限制了虚拟心肌细胞的预测能力与转化应用价值。
方法: 本研究开发了新型T-World虚拟人类心肌细胞模型,采用数据驱动微分方程描述性别特异性兴奋-收缩偶联、机械收缩、β肾上腺素能信号传导及其对细胞靶点的调控作用。该模型包含多项关键创新,包括L型钙通道与雷诺定受体偶联的新方法,优化前者的钙依赖性失活特性,构建后者全新的钙诱导不应期特征并完成全面参数重定;同时重新构建钠钾泵模型,大幅优化钠钙交换体的作用模式。
结果: T-World模型在频率依赖性动作电位、钙处理与收缩特性方面与实验数据高度吻合。经独立数据集广泛验证后,该模型展现出优异的预测性能,例如可模拟药物诱导的动作电位变化。模型可重现交感神经刺激的效应,包括动作电位时程缩短、钙瞬变振幅与收缩力增强。重要的是,该模型首次重现了所有驱动危及生命心律失常的关键细胞机制(早期与延迟后除极、交替波动、陡S1-S2复极),包括交感激活、肌浆网钙ATP酶抑制、动作电位延长等干预措施的实验观测反应。结合模型模拟电生理性别特异性差异的能力,研究揭示女性心肌细胞更易出现早期后除极与动作电位时程陡复极。
结论: T-World是一款通用性与预测性俱佳的开源人类心室心肌细胞计算机模型,适用于探究心律失常发生决定因素的多尺度研究。
T-World虚拟人类心肌细胞Ⅱ:器官水平模拟与应用
期刊: Circ ResPMID: 41948815
简述: 机械性心脏模拟广泛应用于科研、药物研发与监管科学,但现有人类心肌细胞模型缺乏跨尺度预测通用性。本研究将T-World模型嵌入临床影像来源的双心室电生理与机电耦合解剖模型,开展器官水平模拟;药物安全性评估准确率达87%、特异性100%,揭示美西律治疗2型长QT综合征的作用机制,阐明2型糖尿病心律失常与肌浆网钙ATP酶降低的关联,证实NaV1.8异位表达可直接触发早期后除极,为多尺度心脏研究、药物研发与数字孪生构建提供统一开源平台。摘要翻译:
背景: 机械性心脏模拟在科研、药物研发与监管科学中的应用日益广泛,但现有多数人类心肌细胞模型缺乏跨尺度预测转化所需的通用性。本团队近期开发的T-World模型克服了这一障碍,可重现所有主要细胞致心律失常机制,并与实验及临床数据高度吻合。本研究旨在验证T-World模型在器官水平与转化研究中的应用价值,涵盖心律失常离子机制至整体心脏生理 emergent 特征。
方法: 将T-World模型嵌入基于临床影像构建的双心室电生理与机电耦合解剖真实模型,开展器官水平模拟。采用更新CredibleMeds注释的60种化合物处理单细胞模型群,评估药物安全性;通过2型长QT综合征模型变体开展美西律机械性疗效研究;疾病应用包括人类2型糖尿病心律失常机制与心脏病中异位表达的神经元钠通道NaV1.8的致心律失常潜能研究。
结果: T-World模型可重现人类样心电图形态与心室力学特征(射血分数61%),无需参数调整即可在生理相关缺血条件下诱发性心室颤动。在扭转型室速风险药物安全性评估中,T-World模型准确率达87%、特异性100%,并揭示基于体外检测的利多卡因与西洛他唑药理学描述存在局限性。美西律模拟研究证实,抑制晚钠电流与L型钙电流共同介导其对2型长QT综合征的抗心律失常获益。2型糖尿病重构的细胞模拟研究解释了患者对早期后除极易感性升高、交替波动风险增加与舒张功能障碍的关联,机制上与肌浆网钙ATP酶表达降低相关。最后,即便NaV1.8表达量极低,也可通过其独特的右移激活与失活特性直接触发早期后除极。
结论: T-World提供了统一的人类特异性开源平台,实现细胞机制与器官水平动力学、转化结局的衔接。其在心律失常、收缩功能、药物安全性/机制与疾病生理方面的预测性能,使其成为多尺度心脏研究、治疗药物研发与下一代心脏数字孪生构建的强大工具。
基于心外膜脂肪放射组学表型的常规心脏CT早期预测心力衰竭
期刊: J Am Coll CardiolPMID: 41949519
简述: 心外膜脂肪是代谢活跃的内脏脂肪库,可感知并调控心肌生物学特性。本多中心队列研究纳入72751例无心力衰竭或心梗史、接受冠脉CT血管造影的成人,自动化提取心外膜脂肪1655项放射组学特征,构建心力衰竭脂肪放射组学特征(FRPHF);模型内部与外部验证C统计量分别为0.869、0.850,FRPHF每升高25百分位,心力衰竭风险升高近4倍,最高十分位患者风险较最低组升高近20倍,可显著优化传统风险模型的预测效能,为心力衰竭精准预防提供影像学工具。摘要翻译:
背景: 心外膜脂肪组织是代谢活跃的内脏脂肪库,既是心肌生物学的感知因子也是调控因子,可响应心肌旁分泌信号发生成分改变。本研究假设,通过常规冠脉CT血管造影对心外膜脂肪进行放射组学表征,可无创捕捉这种不良重塑并实现心力衰竭早期风险分层。
目的: 构建并外部验证与新发心力衰竭相关的可重复性心外膜脂肪放射组学特征。
方法: 开展多中心队列研究,纳入英国9个中心2007-2022年间72751例无已知心力衰竭或心肌梗死病史、接受冠脉CT血管造影检查的成人。采用全自动流程分割心外膜脂肪组织,提取1655项容积、形态与高阶放射组学纹理特征;基于统一的生存自编码器架构,推导心力衰竭脂肪放射组学特征。模型在7个中心的59327例受试者中构建(年龄57±13岁,女性占47.5%),并在2个地理独立中心的13424例参与者中外部验证(年龄58±12岁,女性占49.4%)。生存模型校正年龄、性别与传统危险因素,包括冠心病严重程度与心外膜脂肪体积。
结果: 内部与外部验证队列中位随访时间分别为5.1年与4.0年,分别有1737例(2.9%)与363例(2.7%)受试者发生心力衰竭。心力衰竭脂肪放射组学特征展现出稳健的区分能力(内部验证C统计量:0.869,95%CI:0.850-0.889;外部验证C统计量:0.850,95%CI:0.831-0.870)。该特征每升高25百分位,校正后心力衰竭风险升高近4倍(内部校正风险比:3.90,95%CI:3.13-4.84;外部校正风险比:3.79,95%CI:3.01-4.76;均P<0.001),最高十分位患者风险较最低十分位升高近20倍。在外部队列中,将该特征加入包含心外膜脂肪体积与冠心病严重程度的传统风险模型,可显著提升5年预测区分度(ΔAUC:0.047;95%CI:0.029-0.065)与净重新分类指数(NRI:0.39;95%CI:0.29-0.48),决策曲线分析提示存在净临床获益。该关联在不同人口学亚组与射血分数谱中均保持一致。
结论: 基于常规冠脉CT血管造影的自动化心外膜脂肪放射组学表型分析,可在临床发病前实现可规模化、生物学指导的心力衰竭未来风险分层,使基于影像学的内脏脂肪无创分析成为精准预防的潜在工具。
急性失代偿性心力衰竭儿童的评估与管理:美国心脏协会科学声明
期刊: CirculationPMID: 41937629
简述: 全球范围内合并先天性与获得性心脏病的儿童及青少年急性失代偿性心力衰竭发病率呈上升趋势,该人群住院后发病率、死亡率更高且医疗资源消耗更大,目前缺乏规范化的评估、管理与出院计划指导。该人群诊疗涉及急诊、心脏科、外科、重症医学与心理学等多学科交叉,本科学声明梳理了该领域现有研究证据,指出关键知识缺口,为高危儿童的综合管理提供指导依据。摘要翻译:
全国范围内,合并先天性与获得性心脏病并表现为急性失代偿性心力衰竭的儿童及青少年数量呈上升趋势。与成人患者相比,该类儿童住院后发病率与死亡率更高,且消耗的医疗资源显著更多。目前,针对表现为急性失代偿性心力衰竭的儿童,如何开展评估、管理并制定有效的出院计划,相关指导意见较为匮乏。鉴于该人群涉及急诊医学、心脏科、外科、重症医学与心理学等多学科交叉领域,亟需制定针对这一高危人群综合管理的指导文件。本科学声明反映了该领域现有研究证据现状,并指出了该领域存在的重要知识缺口。
用于心血管疾病即时诊断的深度学习增强型双模式多路复用光学传感器
期刊: Light Sci ApplPMID: 41951574
简述: 心肌梗死与心力衰竭的快速心脏生物标志物检测对及时诊疗至关重要,现有检测系统存在耗时久、动态范围窄、单指标检测等缺陷。本研究开发深度学习增强型双模式多路垂直流分析光学传感器,整合比色与化学发光检测,23分钟内可通过50μL血清同时定量检测心肌肌钙蛋白I、肌酸激酶同工酶MB、N末端B型利钠肽前体,灵敏度覆盖临床范围,92例患者样本中量化相关系数>0.96,为心血管疾病即时快速、精准诊断提供便携高效工具。摘要翻译:
快速、可及的心脏生物标志物检测对于心肌梗死与心力衰竭的及时诊断和风险评估至关重要,这两种相互关联的疾病常合并存在,导致反复住院且死亡率较高。然而,现有实验室与即时检测系统存在检测周转时间长、生物标志物动态范围窄、单分析物检测无法反映心血管疾病复杂性等局限。本研究提出一种深度学习增强型双模式多路复用垂直流分析系统,配备便携式光学读取器与基于神经网络的量化流程。该光学传感器在单一纸质检测卡中整合比色与化学发光检测,可互补覆盖高低丰度生物标志物的宽动态范围(跨度约6个数量级),同时保持定量准确性。该光学传感器仅需50μL血清,即可在23分钟内同时定量检测心肌肌钙蛋白I、肌酸激酶同工酶MB与N末端B型利钠肽前体。该多路垂直流分析系统对心肌肌钙蛋白I的检测灵敏度达皮克级以下,对肌酸激酶同工酶MB与N末端B型利钠肽前体的检测灵敏度达纳克级以下,覆盖上述生物标志物的临床相关范围。基于92例患者血清样本训练并盲测的神经网络模型,展现出优异的量化性能(与对照检测方法的皮尔逊相关系数>0.96)。该双模式多路垂直流分析传感器将高灵敏度、多路检测与自动化特性整合于紧凑经济的光学传感器中,可实现心血管疾病的即时快速定量诊断。
清除受损线粒体的两条途径:降解与分泌
期刊: Circ ResPMID: 41955326
简述: 本文为心脏领域综述,线粒体是心肌细胞产生ATP的核心细胞器,功能异常线粒体蓄积会引发心肌损伤,心肌细胞通过多层质控机制清除缺陷线粒体;研究总结了心脏中线粒体降解(主要经自噬)与分泌(经细胞外囊泡)的核心机制,阐明了两条途径在心脏病进展中的作用,以及作为心血管疾病治疗靶点的潜在价值。摘要翻译:
线粒体是高度动态的双层膜细胞器,在心肌细胞中产生大部分三磷酸腺苷(ATP),同时维持细胞稳态并参与信号转导。功能异常线粒体的蓄积会促进心肌细胞丢失、损害收缩功能,最终导致心肌损伤。为维持线粒体完整性,心肌细胞依赖多层质控机制清除缺陷线粒体。该过程主要有两条途径:主要通过自噬实现的降解途径,以及通过细胞外囊泡实现的分泌途径。本综述总结了心脏中线粒体降解和分泌的机制,重点阐述了其在心脏病进展中的作用及作为治疗靶点的潜力。
线粒体转移:从基础研究到临床转化
期刊: Circ ResPMID: 41955327
简述: 本文为心血管领域综述,细胞间线粒体转移是心血管系统组织修复与适应的核心机制,线粒体可通过隧道纳米管、细胞外囊泡等方式在细胞间转移;研究解析了线粒体转移的分子机制,提出4种临床转化策略,同时分析了该疗法从基础走向临床的关键障碍,为心血管疾病线粒体靶向治疗提供了方向。摘要翻译:
细胞间线粒体转移已成为心血管系统组织适应和修复的核心机制,对心血管、神经、代谢及炎症性疾病具有重要意义。线粒体曾被认为是静态的,如今被证实是可移动的细胞器,可通过隧道纳米管、细胞外囊泡以及游离线粒体的形式在细胞间移动。这些通路支撑线粒体通讯的两条互补轴:补充拯救轴,即健康线粒体或线粒体组分可恢复受体细胞的生物能量代谢和抗应激能力;释放缓解轴,即受损线粒体被排出并降解,以维持细胞稳态并减轻炎症反应。本文总结了调控隧道纳米管形成、线粒体来源囊泡生物发生、细胞外囊泡分选以及游离线粒体释放与摄取的分子机制,并探讨了这些过程对器官功能的影响。基于这些机制研究成果,本文概述了4种转化策略:(1)捐赠健康线粒体或清除受损线粒体的细胞疗法;无细胞疗法包括(2)含线粒体的细胞外囊泡或(3)纯化线粒体;(4)增强内源性线粒体更新和细胞间交换的药物、营养及生活方式干预。最后,本文讨论了临床转化的关键障碍,包括炎症和致癌风险、线粒体核不相容性、对转移线粒体的命运和持久性认知不足,以及缺乏标准化的制备、效力检测和长期储存方法。将机制生物学与生物工程、监管科学持续结合,是推动心血管疾病中线粒体转移疗法安全地从基础研究走向临床应用的关键。
慢性冠状动脉完全闭塞患者经皮冠状动脉介入治疗或药物治疗后的生活质量:EUROCTO与DECISION-CTO荟萃分析
期刊: J Am Coll CardiolPMID: 41949523
简述: 本研究为两项随机对照试验的事后汇总分析,纳入518例单纯慢性冠状动脉完全闭塞无合并冠脉病变的患者,对比经皮冠状动脉介入治疗(PCI)与最优药物治疗(OMT)的效果;结果证实PCI在改善心绞痛、生活质量方面显著优于OMT,两组主要不良心血管事件发生率无差异,为临床治疗决策提供了依据。摘要翻译:
背景:由于现有随机对照试验(RCTs)数量有限,慢性冠状动脉完全闭塞(CTOs)患者行经皮冠状动脉介入治疗(PCI)对比最优药物治疗(OMT)在改善临床症状和生活质量(QoL)方面的获益仍存在争议。
方法:本研究为两项随机对照试验EUROCTO和DECISION-CTO的事后汇总分析,纳入518例单纯慢性冠状动脉完全闭塞且无其他显著冠脉病变的患者,两项试验均对比了PCI与OMT的疗效;DECISION试验中PCI与OMT的随机分组比例为1:1,EUROCTO试验为2:1。采用西雅图心绞痛问卷(SAQ)在基线和12个月后评估临床状态,并监测3年临床事件。
结果:PCI手术成功率为92.2%。意向性治疗分析显示,在基线至随访期的心绞痛频率评分变化(12.2 vs 8.6;P=0.009)、生活质量评分(19.5 vs 11.3;P<0.001)及西雅图心绞痛问卷总分(13.8 vs 8.5;P<0.001)方面,PCI均优于OMT。在身体受限评分方面,差异仅达到多重检验的邦费罗尼校正水平(P=0.01)。西雅图心绞痛问卷评分的变化存在广泛变异性。对于生活质量,显著改善的主要决定因素为基线评分较低和接受PCI治疗,而性别、糖尿病或病变复杂程度无影响。平均随访3.1年期间,两组心源性死亡或非致死性心肌梗死的临床终点发生率相似(OMT vs PCI:2.7% vs 5.1%;P=0.17)。卒中或出血住院发生率相似,仅OMT组靶病变血运重建率更高(18.8% vs 10.6%;P=0.005)。
结论:在两项随机对照试验的事后分析中,针对单纯慢性冠状动脉完全闭塞且无显著合并病变的患者,PCI较OMT能更好地改善生活质量、心绞痛频率和西雅图心绞痛问卷总分,且未出现临床终点不良事件增加的信号。
存在心血管异常表现的大师级运动员:欧洲心脏病学会预防心脏病学协会与美国心脏病学会临床共识声明
期刊: Eur Heart JPMID: 41947397
简述: 本国际临床共识声明指出,规律运动可降低心血管风险,但大师级运动员心血管异常发生率更高,现有诊疗指南不适用于该群体;声明针对房颤、心肌纤维化等7类心血管异常,提出了标准化的诊断、管理方案,明确了未来的临床研究方向,填补了运动员心血管诊疗的空白。摘要翻译:
运动训练可改善心血管健康,降低未来心血管事件发生风险和死亡率。然而,新证据表明,与活动量较少的人群相比,大师级运动员心房心律失常、冠状动脉粥样硬化、主动脉扩张和心肌纤维化等心血管异常的发生率更高。大师级运动员的临床管理存在挑战,因为现有相关疾病诊疗指南主要基于有症状的久坐患者数据,不适用于高水平训练人群。大师级运动员临床评估的其他独特挑战包括:与久坐人群相比症状表现不同、对启动药物治疗的潜在抵触,以及可提供心血管健康相关信息的消费级可穿戴健康设备的普及。本欧洲预防心脏病学协会/欧洲心脏病学会与美国心脏病学会联合临床共识声明旨在全面更新大师级运动员心血管异常表现的现有研究进展。本文针对(1)心房颤动、(2)缓慢性心律失常、(3)室性心律失常、(4)冠状动脉粥样硬化、(5)主动脉扩张、(6)心肌纤维化和(7)运动诱发的心律失常性心肌病,提出了基于专家意见的诊断评估、管理和预后方案。本文讨论了临床挑战、争议领域、不确定性及潜在机制,同时提出了未来展望和研究方向,包括开展临床结局研究、针对运动员的专属随机对照试验,以及建立多样化人群的纵向随访国际登记库,以评估心脏异常的自然病程,并为存在心血管异常的大师级运动员制定循证临床管理策略。
运动中的多器官生理缺陷可识别射血分数保留型心力衰竭的临床和分子易感因素
期刊: CirculationPMID: 41944041
简述: 本研究通过有创心肺运动试验、代谢组学与基因组学联合分析,证实射血分数保留型心力衰竭(HFpEF)患者存在多器官运动生理缺陷,其代谢特征可精准预测疾病发生与预后,优化风险分层;研究揭示了HFpEF与肥胖、糖尿病等合并症共享的代谢遗传机制,为疾病分型和靶向治疗提供了新范式。摘要翻译:
背景:运动可暴露射血分数保留型心力衰竭(HFpEF)特征性的多器官系统储备能力缺陷,但运动缺陷的代谢和遗传基础,及其对HFpEF严重程度和预后的累积影响尚未完全明确。
方法:本研究采用有创心肺运动试验(iCPET)、代谢物谱分析和基因组学技术,同时表征HFpEF患者的7项运动生理缺陷:运动每搏输出量和心率降低、肺毛细血管楔压/心输出量(PCWP/CO)斜率升高、肺血管阻力升高、运动时肺机械受限、外周氧摄取受损,以及肥胖相关的运动启动代谢消耗异常增加。首先绘制这些运动缺陷的分布、功能和预后意义;随后应用LASSO回归分析确定每项运动缺陷的代谢物特征,并在多民族动脉粥样硬化研究(MESA)的6345名受试者中(随访约20年),评估这些特征与临床人口学特征、心脏磁共振成像及新发心力衰竭的关系;最后将与缺陷相关的代谢物映射到约200万心力衰竭人群的组织特异性遗传变异,以及最大规模的HFpEF合并症(肥胖、肾病、糖尿病)全基因组关联研究(GWAS)中,评估HFpEF病理生理的共享代谢机制。
结果:本研究的有创心肺运动试验HFpEF队列(年龄61.7±14.1岁,54%为女性,体重指数30.6±6.7 kg/m²)存在广泛的复合心脏和心外运动缺陷。存在≥5项运动缺陷的受试者,发生心血管事件或死亡的风险升高近4倍(风险比3.90,95%置信区间1.74-8.75,P<0.0001)。运动肺毛细血管楔压/心输出量斜率的代谢物特征,使MESA研究中新发心力衰竭的风险比每标准差增加1.43(95%置信区间1.20-1.71,P<0.001)。将所有有创心肺运动试验缺陷代谢特征纳入单一模型,较传统HFpEF危险因素的连续净重新分类改善率约为20%。运动缺陷代谢组相关基因在心力衰竭全基因组关联研究(约200万人)中富集,并与肥胖、肾功能不全和糖尿病共享,揭示了心力衰竭(包括HFpEF)及其合并症的终身共享易感机制。
结论:器官对运动的特异性反应及其循环代谢物特征与HFpEF的发生和预后密切相关。这些结果为解析HFpEF亚表型和优先研究HFpEF代谢机制提供了新范式。
合成类和生物类改善病情抗风湿药的安全性:为2025年欧洲抗风湿病联盟类风湿关节炎管理指南更新提供依据的系统文献综述
期刊: Ann Rheum DisPMID: 41951459
简述: 本系统文献综述纳入2022-2025年的相关研究,评估了各类改善病情抗风湿药(DMARDs)的安全性;结果显示生物类DMARDs严重感染风险更高,JAK抑制剂带状疱疹、静脉血栓栓塞风险显著升高,研究为2025年欧洲抗风湿病联盟类风湿关节炎诊疗指南的更新提供了核心安全性证据。摘要翻译:
目的:本研究旨在开展一项系统文献综述(SLR),评估合成类和生物类改善病情抗风湿药(DMARDs)的安全性,为2025年欧洲抗风湿病联盟(EULAR)类风湿关节炎(RA)管理指南更新提供依据。
方法:检索Medline、Embase、Cochrane CENTRAL和Web of Science数据库,纳入2022年1月14日至2025年1月22日发表的、以传统合成(cs-)、生物(b-)、靶向合成DMARDs及糖皮质激素安全性为主要终点的观察性研究和随机对照试验;同时单独检索数据库建库至2025年1月22日的DMARDs监测相关研究;所有纳入研究均需设置对照组,且涵盖所有安全性结局。
结果:共检索到3837篇文献,321篇进入全文筛选,最终纳入71篇。所有研究中,感染是最常评估的结局:13项研究评估了严重或住院感染(通常为细菌、机会性感染和带状疱疹的复合终点),1项研究评估了非严重感染。生物类DMARDs的严重感染发生率高于合成类DMARDs。Janus激酶抑制剂(JAKis)的带状疱疹风险高于生物类DMARDs。与生物类DMARDs相比,JAK抑制剂未增加结核风险;但英夫利昔单抗和阿达木单抗的结核风险高于依那西普。15项研究评估了恶性肿瘤,其中一半分析所有恶性肿瘤,另一半排除非黑色素瘤皮肤癌(NMSC);2项研究聚焦黑色素瘤,2项聚焦非黑色素瘤皮肤癌。与普通人群相比,使用DMARDs的类风湿关节炎患者非黑色素瘤皮肤癌发生率升高,且与特定DMARDs无关。20项研究报告了心血管和血栓栓塞事件。无一致证据表明JAK抑制剂较生物类DMARDs增加主要不良心血管事件风险。JAK抑制剂的静脉血栓栓塞风险高于生物类DMARDs,主要由肺栓塞驱动。14项研究报告了药物留存率和不良事件相关停药情况,8项研究评估了其他特定不良事件。胃肠道穿孔和脱髓鞘疾病各有3项研究报道。DMARDs监测相关检索未发现符合标准的文献。
结论:评估安全性结局的研究数量显著增加,其中大部分为观察性研究,主要聚焦恶性肿瘤、血栓栓塞和心血管事件,多数研究关注JAK抑制剂的安全性。本系统文献综述中,大量研究依靠理赔数据库评估安全性结局,该方法存在重要的方法学局限性。令人意外的是,过去3年中探讨糖皮质激素安全性的研究数量较少。本系统文献综述联合DMARDs疗效相关系统文献综述,为2025年EULAR类风湿关节炎合成类和生物类DMARDs管理指南更新提供了依据。
β2肾上腺素能受体调控心肌梗死后中性粒细胞的脱边缘化及向缺血心肌的募集
期刊: Nat CommunPMID: 41965377
简述: 本研究通过雄性小鼠实验证实,心肌梗死早期募集至缺血心肌的中性粒细胞来源于血管边缘池,而非骨髓新生细胞;β2肾上腺素能受体信号是调控中性粒细胞脱边缘化的核心靶点,短暂抑制该信号可改善心梗后心脏重构与功能,为急性心梗的炎症靶向治疗提供了新策略。摘要翻译:
心肌梗死(MI)后中性粒细胞迅速在心脏内聚集,但骨髓粒细胞生成的峰值出现较晚,提示存在其他早期来源。本研究通过雄性小鼠实验证实,募集至缺血心肌的第一波中性粒细胞来源于血管边缘池,而非骨髓或脾脏新生的细胞。通过造血干细胞消融、流式细胞术和蛋白质组学分析发现,心肌梗死早期诱导的中性粒细胞增多症具有应激性脱边缘化的特征,该过程中黏附的中性粒细胞快速释放进入血液循环。在雄性小鼠中,药物或基因抑制去甲肾上腺素合成或β肾上腺素能受体信号,可抑制中性粒细胞脱边缘化并减少心脏浸润。尽管持续性β肾上腺素能受体阻断无法改善长期预后,但短暂抑制该信号可增强心脏重构和功能。这些研究结果证实,应激性脱边缘化是心肌梗死后早期中性粒细胞的主要来源,提示时间靶向性的β2肾上腺素能受体调节可能有助于抑制急性炎症损伤。
遗传性转甲状腺素蛋白淀粉样变性家系的级联基因筛查:诊断和预后影响
期刊: Eur Heart JPMID: 41955077
简述: 本回顾性研究分析了意大利15个中心近20年的家系数据,证实级联基因筛查可实现遗传性转甲状腺素蛋白淀粉样变性(ATTRv)的早期诊断,显著改善患者生存率;不同基因突变携带者的疾病转化率差异显著,部分携带者发病早于预期,基因型特异性随访是优化预后的关键。摘要翻译:
背景与目的:遗传性转甲状腺素蛋白淀粉样变性(ATTRv)是一种外显率可变的常染色体显性遗传病。级联基因筛查可实现早期诊断和干预,但其对预后的影响尚不明确。
方法:本研究回顾性分析了2004年至2024年意大利15个转诊中心431个家系的967例受试者。受试者分为ATTRv先证者、症状携带者(基因型阳性/表型阳性[G+/P+])和无症状携带者(基因型阳性/表型阴性[G+/P-])。评估临床特征、疾病转化率和生存率。
结果:共纳入398例先证者,对1243名亲属进行基因筛查后发现569例携带者(461例G+/P-,108例G+/P+)。在461例G+/P-携带者中,中位随访5.3(1.7-9.8)年,77例(16.7%)患者确诊为ATTRv:Glu89Gln突变(42.2%,95%置信区间28.8-56.9)、Phe64Leu突变(24.7%,95%置信区间16.1-35.8)、Val30Met突变(13.1%,95%置信区间7.4-22.1)、Ile68Leu突变(7.3%,95%置信区间4.1-12.8)、Val122Ile突变(5.1%,95%置信区间1.3-18.3),其他突变占22.9%(95%置信区间14.5-34.1)。值得注意的是,62例携带者中有11例(17.7%)的发病时间较预测年龄提前10年以上。G+/P+患者的生存率优于先证者(风险比0.43,95%置信区间0.24-0.79),混合表型患者的预后差于心脏表型患者。疾病修饰治疗与死亡率降低独立相关(风险比0.11,95%置信区间0.01-0.17)。
结论:级联基因筛查有助于早期诊断,且与生存率改善相关,这可能与疾病早期识别和及时启动治疗有关。基因型特异性随访至关重要,因为部分携带者的发病时间早于预期。系统性、基于基因型的监测是优化ATTRv患者预后的关键。
Zodasiran治疗高脂血症患者的降胆固醇和甘油三酯效果:1期篮子试验最终报告
期刊: Nat MedPMID: 41946917
简述: 本1期临床试验评估了靶向ANGPTL3的小干扰RNA药物Zodasiran的安全性与疗效,药物安全性良好,无严重治疗相关不良事件,可显著且持久降低血清ANGPTL3和甘油三酯水平,为广谱血脂异常的靶向治疗提供了可靠的临床前期依据。摘要翻译:
血管生成素样蛋白3(ANGPTL3)基因的功能缺失变异与甘油三酯和低密度脂蛋白胆固醇水平降低相关,同时与心血管风险降低相关。本文报道了Zodasiran(一种靶向ANGPTL3的小干扰RNA)治疗接受降脂治疗的高脂血症患者的16周1期试验,试验分为三组:高脂血症组(设安慰剂对照组,n=9;男性7例,女性2例)、家族性高胆固醇血症组(n=17;男性9例,女性8例)和中重度高甘油三酯血症组(n=6;男性4例,女性2例)。患者在第1天和第29天皮下注射Zodasiran,随后对家族性高胆固醇血症队列(n=13;男性7例,女性6例)进行48周开放标签扩展,每12周给药一次Zodasiran。试验主要终点为无严重治疗相关不良事件,结果未观察到该类事件;此外,未出现肝氨基转移酶、胆红素或糖化血红蛋白升高,无药物停药情况。所有队列在第16周(末次给药后12周)均出现血清ANGPTL3(≤-85.4%)和甘油三酯(≤-67.1%)水平降低(次要终点)。家族性高胆固醇血症队列中,ANGPTL3的降低效果持续至开放标签扩展结束。这些结果表明Zodasiran具有良好的安全性,有望用于治疗单纯性高胆固醇血症和中重度高甘油三酯血症,支持进一步开展Zodasiran治疗广谱血脂异常的研究。
肾可清除硫化铋纳米花的肿瘤选择性自噬阻断用于精准光热治疗
期刊: ACS NanoPMID: 41943257
简述:传统肿瘤靶向策略依赖复杂的表面修饰,本研究开发了一种生物行为驱动的Bi2S3@3-MA纳米平台,通过MMP2响应性肽段偶联实现肿瘤选择性细胞死亡;该纳米材料可在肿瘤微环境中释放自噬抑制剂,诱导纳米花聚集破坏肿瘤细胞溶酶体,增强光热消融效果,同时可经肾脏快速清除降低正常组织毒性,还能通过CT成像实时监测分布与治疗反应。摘要翻译:
传统策略依赖复杂的表面修饰,而非利用纳米材料的内在生物学行为来实现肿瘤选择性。本研究介绍了一种生物行为驱动的纳米平台Bi2S3@3-MA,该平台通过简单的表面偶联MMP2响应性3-甲基腺嘌呤肽段(3-MA)对硫化铋纳米花进行改造,从而实现选择性肿瘤细胞死亡。中等尺寸的Bi2S3@3-MA(370纳米)优先在肿瘤组织中富集。在肿瘤微环境(TME)中,高表达的MMP2会切割肽段连接臂,触发肿瘤微环境特异性释放自噬抑制剂3-MA。这种肿瘤选择性的自噬阻断会促使硫化铋纳米花在酸性溶酶体环境中聚集形成微米级结构,最终导致肿瘤细胞的溶酶体膜破裂。
此外,肿瘤细胞内的微米级聚集体表现出增强的光热消融作用,克服了保护性自噬诱导的热疗耐药性。相比之下,pH响应性降解颗粒(pH约6.5-7.4)的快速肾脏清除可最大限度减少正常组织的脱靶暴露,而保护性自噬则维持正常细胞的溶酶体完整性。Bi2S3@3-MA通过光热消融与自噬抑制的协同作用,在小鼠乳腺癌模型中实现了肿瘤的完全根除。此外,硫化铋固有的CT造影特性允许实时可视化纳米颗粒的生物分布和治疗反应。总之,这些结果建立了一种范式,即通过合理整合纳米材料的内在生物学行为,实现高选择性的癌症治疗,同时将全身毒性降至最低。
纳武利尤单抗新辅助与辅助治疗联合不可逆电穿孔用于BCLC A期高复发风险肝细胞癌患者(NIVOLEP试验)
期刊: HepatologyPMID: 41950497
简述:本项多中心2期NIVOLEP试验评估纳武利尤单抗联合不可逆电穿孔(IRE)治疗BCLC A期肝细胞癌的疗效,患者接受新辅助纳武利尤单抗、根治性IRE及辅助纳武利尤单抗治疗;主要终点为1年无局部复发生存率,结果显示该联合方案可诱导与免疫激活相关的病理缓解,展现出抗肿瘤效果。摘要翻译:
背景:不可逆电穿孔(IRE)是一种非热消融技术,适用于难治性肝细胞癌(HCC),其1年局部复发(LR)率超过50%。
目的:我们假设围手术期免疫治疗可与IRE发挥协同作用,降低局部复发率。
设计:NIVOLEP是一项多中心2期试验,评估纳武利尤单抗联合IRE治疗BCLC A期肝细胞癌患者的疗效。患者接受两次纳武利尤单抗新辅助输注、根治性IRE治疗,以及12次纳武利尤单抗每月辅助输注。在基线和IRE治疗期间进行肿瘤活检。主要终点为1年无局部复发生存率(LRFS)。
结果:本研究纳入43例患者的62个肝细胞癌结节(平均大小:30.0毫米),患者平均年龄71岁,男性占88%,81%合并肝硬化。所有患者均接受新辅助纳武利尤单抗治疗,35例患者接受根治性IRE治疗(其余8例:4例IRE治疗失败、3例肝细胞癌进展、1例死亡)。新辅助纳武利尤单抗治疗后,分别有24.2%和26.3%的结节出现影像学或病理缓解。意向性治疗分析显示,1年无局部复发生存率为70.6%(95% CI:55.3 - 85.9),2年总生存率为74.2%。2例患者出现纳武利尤单抗相关的3级或4级不良事件,1例患者因纳武利尤单抗死亡。新辅助纳武利尤单抗治疗后肿瘤的RNA测序分析显示,与白细胞迁移、T细胞活化或CD8+T细胞和B细胞浸润相关的通路富集,该现象与病理缓解相关。循环蛋白变化与病理或影像学缓解以及局部复发相关。
结论:对于符合IRE治疗条件的BCLC A期肝细胞癌患者,纳武利尤单抗新辅助与辅助治疗可诱导与免疫激活相关的病理缓解,并展现出抗肿瘤效果。
重新思考线粒体:细胞外维度
期刊: Circ ResPMID: 41955330
简述:线粒体是维持细胞能量代谢与多种功能的关键细胞器,传统认为其仅存在于细胞内,而最新证据表明线粒体及其衍生成分可释放至细胞外形成细胞外线粒体;本文综述了细胞外线粒体的形态、释放机制及病理生理意义,强调其在心血管系统稳态与疾病进展中的作用,以及作为治疗靶点的潜力。摘要翻译:
线粒体是至关重要的细胞器,可将代谢底物中的能量转化为三磷酸腺苷(ATP),同时参与众多细胞过程。传统上线粒体被严格认为是细胞内细胞器,而越来越多的证据表明,线粒体及线粒体衍生成分也可释放到细胞外空间,形成细胞外线粒体。细胞外线粒体具有显著的异质性,形态从完整的细胞器到单个分子成分不等,存在游离状态或囊泡包裹结构,功能状态从严重损伤到具有代谢活性均有覆盖。
其释放由活细胞和死亡细胞中严格调控的机制介导,并受细胞应激、激活状态以及控制线粒体选择、区室化、运输和排出的通路影响。细胞外释放过程在机体内发挥多种功能,包括质量控制、细胞特性调节、炎症信号传导以及对受体细胞的功能支持。在心血管系统中,细胞外线粒体参与机体稳态维持与疾病进展。本综述总结了目前关于细胞外线粒体形态、释放机制和病理生理意义的研究进展,并强调了其在心血管病理生理及其他领域中作为治疗靶点的新兴潜力。
基于个性化人工智能的左心室射血分数与收缩功能障碍评估
期刊: NPJ Digit MedPMID: 41951962
简述:左心室射血分数(LVEF)是评估心功能的核心指标,常规需依赖超声心动图等影像学技术;本研究基于大规模心电图(ECG)数据开发人工智能模型,单独或结合结构化特征评估LVEF,个性化模型显著提升评估精度,可高效筛查左心室收缩功能障碍。摘要翻译:
左心室(LV)射血分数(LVEF)是评估心脏功能的核心指标,通常需通过经胸超声心动图(TTE)等资源密集型影像学技术评估。本研究纳入191941例患者的236623组心电图/超声心动图配对数据,评估心电图作为一种便捷可及的替代数据用于估算LVEF的价值。我们仅使用心电图数据或结合结构化特征,构建了卷积神经网络和概率神经网络模型,用于估算LVEF并量化其不确定性。
仅心电图模型的平均绝对误差(MAE)为7.71%,均方根误差(RMSE)为10.36%;混合模型的平均绝对误差为7.84%,均方根误差为10.52%。个性化模型显著提升了评估性能,仅心电图模型平均绝对误差降至5.98%,混合模型降至6.75%。该模型识别左心室收缩功能障碍(LVEF≤40%)的曲线下面积(AUC)为0.88,灵敏度为0.92,阴性预测值为0.98。本研究构建的模型在估算左心室射血分数和筛查左心室收缩功能障碍方面表现出优异的性能。
心脏手术后新发房颤的管理与预后:VISION心脏手术队列研究
期刊: Eur Heart JPMID: 41964448
简述:新发术后房颤是心脏手术最常见的并发症,本项前瞻性多中心队列研究分析其发生率、管理方式与长期预后的关联;结果显示近三分之一心脏手术患者术后30天内发生房颤,其抗栓与抗心律失常管理存在差异,术后房颤患者1年内临床房颤及全因死亡风险显著升高。摘要翻译:
背景与目的:新发房颤(AF)是心脏手术最常见的并发症。目前对术后房颤(POAF)的认知主要来自注册研究和单中心研究,本研究旨在通过前瞻性多中心队列描述术后房颤的发生率、管理方式及其与长期预后的关联。
方法:VISION心脏手术研究是一项前瞻性队列研究,纳入12个国家接受心脏手术的成年患者。采用多变量Cox模型,校正患者特征、手术特征及抗栓治疗后,评估术后房颤与术后30天至1年预后的相关性。
结果:在12234例患者中(55.3%为单纯冠状动脉旁路移植术),31.8%的患者术后30天内发生术后房颤。出院时,术后房颤患者中仅接受抗凝治疗的比例为15.6%,仅接受抗血小板治疗的比例为54.3%,联合接受抗凝与抗血小板治疗的比例为23.9%,未接受任何治疗的比例为6.3%;48.8%的患者使用胺碘酮。术后1年,术后房颤患者的临床房颤发生率为6.9%,而非术后房颤患者为0.6%(校正风险比aHR:11.30;95% CI:8.17-15.70)。卒中或血管性死亡的主要复合终点发生率,术后房颤患者为2.3%,非术后房颤患者为1.5%(校正风险比aHR:1.32;95% CI:0.99-1.77)。术后房颤患者的全因死亡风险更高(3.0% 对比 1.7%;校正风险比aHR:1.54;95% CI:1.18-2.00)。
结论:三分之一的心脏手术患者会发生新发术后房颤,其抗栓和抗心律失常管理方式存在差异。术后房颤患者在术后1年内发生临床房颤和全因死亡的风险均升高。
MARCH2通过稳定NR1H2并促进凋亡心肌细胞清除预防阿霉素诱导的心肌病
期刊: Nat CommunPMID: 41963318
简述:阿霉素诱导的心肌病与心脏巨噬细胞清除凋亡心肌细胞的胞葬功能受损相关;本研究发现MARCH2-NR1H2信号轴是调控该过程的关键,MARCH2缺失会加重心肌病、抑制胞葬并加剧炎症,药物激活NR1H2可缓解疾病进展,该轴成为阿霉素诱导心肌病的潜在治疗靶点。摘要翻译:
阿霉素诱导的心肌病(DiCM)涉及心脏巨噬细胞对凋亡心肌细胞清除功能(胞葬作用)的受损。本研究揭示了MARCH2-NR1H2轴在该过程中的核心作用。我们发现,在阿霉素诱导心肌病小鼠和人类扩张型心肌病患者的心脏巨噬细胞中,MARCH2表达显著降低。全身敲除MARCH2或特异性敲除心脏驻留巨噬细胞中的MARCH2,均会加重阿霉素诱导的心肌病,损伤胞葬作用并增加炎症反应。机制上,MARCH2通过K27连接的多聚泛素化增强核受体NR1H2的蛋白稳定性,进而上调胞葬受体MERTK的表达。相反,巨噬细胞特异性NR1H2缺陷会抑制胞葬作用并加重心功能障碍。重要的是,药物激活NR1H2可缓解阿霉素诱导心肌病的进展。这些发现证实MARCH2-NR1H2轴是巨噬细胞胞葬作用的关键调控因子,也是阿霉素诱导心肌病的潜在治疗靶点。
子痫前期中滋养细胞分化缺陷、内皮功能障碍与免疫失调共同汇聚于胎盘以VGLL3为中心的基因网络
期刊: CirculationPMID: 41953989
简述:子痫前期是严重的妊娠并发症,本研究通过单细胞、空间分析及体内外模型发现,子痫前期胎盘中Hippo通路转录共调节因子VGLL3表达上调;VGLL3可诱导免疫激活、损伤滋养细胞分化、引发内皮功能障碍,靶向抑制VGLL3可预防并缓解子痫前期病变,成为该病的新型治疗靶点。摘要翻译:
背景:子痫前期约影响十分之一的妊娠,会给母体和子代带来严重并发症及长期健康风险。尽管病因尚不明确,但子痫前期与自身免疫性疾病及月经初潮时间相关。
方法:本研究通过人类单细胞和空间分析,联合体外、体内及离体模型,证实Hippo通路中的转录共调节因子VGLL3在子痫前期胎盘中表达上调。
结果:VGLL3可促进免疫激活、损害滋养细胞分化并诱导内皮功能障碍,这些作用共同导致妊娠相关高血压、胎儿生长受限及子代死亡。本研究数据显示,VGLL3作用于子痫前期相关过程的上游,包括关键生物标志物可溶性fms样酪氨酸激酶1(sFLT1)的产生。值得注意的是,通过小鼠胎盘基因敲除或人类胎盘治疗性抑制靶向VGLL3,可预防子痫前期并减轻疾病病理改变。
结论:这些研究结果将VGLL3定位为子痫前期极具潜力的新型治疗靶点。
构建社会心理表型以解析心力衰竭患者对数字健康技术的使用依从性
期刊: NPJ Digit MedPMID: 41935162
简述: 本研究依托AIM-ACT框架开展混合方法研究,纳入146名接受6个月多设备数字干预的心力衰竭成人患者,通过k-medoids聚类分析结合调查、生态瞬时评估与设备日志数据,鉴定出三种社会心理表型,辅以定性访谈明确社会心理特征与数字健康技术使用质量的关联机制,为设计适应性公平数字干预方案提供依据。摘要翻译:
可穿戴设备、智能手机及互联设备等数字健康技术(DHTs)在慢性病管理的自我照护支持中具备巨大潜力,但其使用依从性差异极大,且往往仅通过使用指标进行衡量。本研究依托AIM-ACT框架开展混合方法研究,探究社会心理与情境因素如何塑造146名心力衰竭成人患者的数字健康技术使用依从性,这些患者完成了为期6个月、涉及多种设备的数字干预(ClinicalTrials.gov 编号 NCT05056129)。研究通过对调查问卷、生态瞬时评估数据及设备日志数据进行k-medoids聚类分析,鉴定出三种截然不同的社会心理表型——困境生存型、主动学习型、积极自我调节型,这些表型反映了患者在动机、社会心理资源及与数字健康技术交互方式上的差异。研究对26名参与者进行的定性访谈为这些表型提供了情境阐释,并揭示了社会心理特征与数字健康技术使用质量相关联的内在机制。研究结果强调了社会心理表型分型在解析数字健康技术使用依从性异质性方面的价值,可为设计可在不同人群中规模化推广且持续有效的适应性、公平性数字干预方案提供参考。
低收入高血压患者的多元化血压控制策略
期刊: N Engl J MedPMID: 41950472
简述: 本研究将美国路易斯安那州与密西西比州的联邦合格健康中心诊所随机分组,对比多元化团队干预方案与强化常规护理对低收入高血压患者的血压控制效果,结果显示干预组18个月收缩压降幅显著更大,血压管理依从性评分更高,且安全性良好。摘要翻译:
背景:未控制的高血压在健康差距显著的人群中更为高发,目前缺乏针对低收入患者采用多元化团队协作策略实现血压控制的有效性与实施相关数据。
方法:本研究将路易斯安那州和密西西比州的联邦合格健康中心诊所随机分配至高血压控制多元化实施策略组(干预组)或强化常规护理组(对照组)。干预措施包括团队协作诊疗、基于方案的强化血压管理、血压审计与反馈、生活方式改变及用药依从性健康指导、家庭血压监测;强化常规护理仅为医师提供高血压临床指南教育。主要有效性结局为基线至18个月收缩压的平均变化值,主要实施结局为依从性总评分(0-4分,分值越高代表血压管理依从性越好)。
结果:共36家诊所完成随机化,纳入1272例40岁及以上未控制高血压患者,其中干预组642例、对照组630例。患者平均年龄58.8岁,女性占56.7%,黑人占63.4%,75.9%处于失业状态,73.4%家庭年收入低于25000美元。18个月时,干预组收缩压较基线平均降低15.5mmHg(95%CI:-17.4~-13.6),对照组降低9.1mmHg(95%CI:-11.0~-7.2),组间差异为-6.4mmHg(95%CI:-9.0~-3.8;P<0.001)。18个月随访期间,干预组平均依从性总评分为2.8(95%CI:2.7~2.9),对照组为2.1(95%CI:2.0~2.2),组间差异0.7分(95%CI:0.6~0.8;P<0.001)。严重不良事件发生率干预组为20.9%,对照组为21.7%。
结论:在低收入高血压患者中,多元化团队协作实施策略较强化常规护理可显著降低收缩压水平。
Mir147通过抑制非泡沫巨噬细胞作用限制动脉粥样硬化进展
期刊: CirculationPMID: 41944070
简述: 本研究探究miR-147-3p在动脉粥样硬化中的作用,发现其主要高表达于斑块核心的非泡沫巨噬细胞,通过靶向半乳糖凝集素-3抑制非泡沫巨噬细胞对内皮细胞的损伤、增强其凋亡DNA清除能力,从而减轻动脉粥样硬化坏死核心扩张与血栓形成风险。摘要翻译:
背景:高胆固醇血症与高脂饮食通过诱导泡沫巨噬细胞脂滴积聚、激活非泡沫巨噬细胞炎症反应,促进动脉粥样硬化相关的两种巨噬细胞亚型分化。微小RNA是巨噬细胞功能的关键调控因子,例如miR-10a-5p可减轻动脉粥样硬化并改善泡沫巨噬细胞线粒体功能,而miR-155-5p则通过损伤胞葬作用加速动脉粥样硬化。miR-147-3p在巨噬细胞炎症刺激及动脉粥样硬化斑块中表达上调,提示其参与非泡沫巨噬细胞的功能调控。
方法:在Apoe-/-Mir147flox/floxLysMCre+小鼠中,探究表达或不表达增强型绿色荧光蛋白的髓系细胞中miR-147-3p对动脉粥样硬化的作用。通过活体斑块4D共聚焦成像评估脂滴、半胱天冬酶-3活化、凋亡DNA、胆固醇结晶形成及线粒体功能,同时成像分析巨噬细胞迁移、凋亡DNA吞噬及管状膜延伸结构形成。采用海马实验检测活体斑块组织线粒体功能,利用Apoe-/-LSL-tAgo2/Mir147flox/floxLysMCre+小鼠及对照小鼠的动脉粥样硬化主动脉组织,开展绿色荧光蛋白标记的Argonaute 2免疫共沉淀联合引物RNA测序。通过4D活体斑块成像研究半乳糖凝集素-3抑制剂GB1107的作用。
结果:与泡沫巨噬细胞不同,非泡沫巨噬细胞主要定位于斑块核心,且在小鼠及人类动脉粥样硬化组织中均呈现更高的miR-147-3p表达水平。敲除髓系细胞Mir147可加重动脉粥样硬化,使坏死核心内胆固醇结晶形成增多、凋亡DNA积聚。敲除Mir147会降低非泡沫巨噬细胞(而非泡沫巨噬细胞)的线粒体活性,升高半胱天冬酶-3活性,并降低斑块巨噬细胞的储备呼吸能力。此外,敲除Mir147会损伤非泡沫巨噬细胞对凋亡DNA的摄取,增加细胞外凋亡DNA含量,促进胆固醇结晶形成。非泡沫巨噬细胞中Mir147缺失还会诱导内皮细胞半胱天冬酶-3活化,促进泡沫巨噬细胞突起跨内皮延伸。Mir147敲除后,标记Argonaute 2免疫共沉淀产物中编码半乳糖凝集素-3的Lgals3转录本水平降低,且Lgals3 3'-UTR区域存在miR-147-3p结合位点并经功能验证。GB1107处理可逆转巨噬细胞中Mir147敲除的效应。
结论:miR-147-3p通过靶向半乳糖凝集素-3,抑制非泡沫巨噬细胞对内皮细胞的损伤并增强其凋亡DNA清除能力,从而减轻动脉粥样硬化。因此上调miR-147-3p表达或可延缓坏死核心扩张,降低非泡沫巨噬细胞介导内皮损伤引发的动脉血栓形成风险。
USP18通过去泛素化酶PTEN-L抑制Parkin介导线粒体自噬加重心肌缺血再灌注损伤
期刊: Mil Med ResPMID: 41953055
简述: 本研究证实USP18在心肌缺血再灌注损伤中表达上调,通过PTEN-L-Parkin轴抑制线粒体自噬,加重心肌损伤与不良心室重构,STEMI患者血清PTEN-L水平升高,靶向该通路可为心肌缺血再灌注损伤提供新治疗策略。摘要翻译:
背景:线粒体质量控制对于限制缺血再灌注(I/R)诱导的心肌损伤至关重要,该损伤是再灌注治疗后不良预后的主要原因。本研究旨在探究去泛素化酶泛素特异性蛋白酶18(USP18)是否调控心肌缺血再灌注损伤过程中的线粒体自噬,进而成为减轻心肌缺血再灌注损伤的潜在治疗靶点。
方法:构建心肌特异性USP18敲除小鼠并建立心肌缺血再灌注损伤模型,通过RNA测序、蛋白质组质谱、透射电镜及线粒体自噬实验阐明USP18在线粒体自噬调控与心肌缺血再灌注损伤中的作用。同时采用9型腺相关病毒(AAV9)介导USP18过表达、Parkin与PTEN-L敲低及抗PTEN-L中和抗体给药等手段解析内在机制。此外,收集ST段抬高型心肌梗死(STEMI)患者血清样本评估临床相关性。
结果:USP18在缺血再灌注损伤小鼠心脏及人类缺血心肌组织中表达上调。心肌特异性USP18缺失可减轻缺血再灌注诱导的急性心肌损伤、线粒体功能障碍及不良心肌重构,而过表达USP18则会加重上述病理改变。机制上,USP18与PTEN-L相互作用,后者结合并抑制Parkin的磷酸化及线粒体转位,进而抑制线粒体自噬。敲低Parkin可消除USP18缺失带来的心肌保护作用,而敲低PTEN-L则可逆转USP18过表达的损伤效应。此外,PTEN-L还通过旁分泌机制发挥致病作用,抗体中和PTEN-L可减轻心肌缺血再灌注损伤。STEMI患者血清PTEN-L水平升高,尤其在介入治疗后更为显著。
结论:USP18通过PTEN-L-Parkin轴损伤线粒体自噬并加重心肌缺血再灌注损伤,该过程涉及细胞内与旁分泌双重机制。靶向USP18-PTEN-L通路或可成为减轻心肌缺血再灌注损伤的新型治疗策略。
细胞外囊泡介导的心脏内交互对话减轻阿霉素诱导的心脏毒性
期刊: Circ ResPMID: 41948821
简述: 本研究发现运动后心肌细胞来源的细胞外囊泡富集保护性piRNA(EPPIR),其在扩张型心肌病及阿霉素心脏毒性模型中表达降低,通过调控KDM6B-H3K27me3-Dtna表观遗传轴及抑制Tp53发挥心脏保护作用,为阿霉素心脏毒性提供新治疗靶点。摘要翻译:
背景:细胞外囊泡(EVs)参与运动诱导的心脏保护作用,但这些囊泡中组织特异性分子载荷(包括PIWI相互作用RNA,piRNA)的作用及内在机制尚不明确,尤其是运动后心脏内细胞外囊泡相关piRNA对阿霉素诱导心脏毒性(DCT)的调控机制尚未阐明。
方法:采用转基因报告小鼠及心肌细胞特异性Rab27a敲除策略,探究心肌细胞来源的细胞外囊泡在运动诱导的抗阿霉素心脏毒性保护作用中的贡献。通过分析扩张型心肌病患者心脏组织及阿霉素心脏毒性实验模型(包括心肌细胞特异性敲除小鼠、人诱导多能干细胞衍生心肌细胞、原代小鼠心肌细胞),筛选具有功能相关性的心脏保护性piRNA载荷。
结果:研究发现运动后心肌细胞来源的细胞外囊泡富集一种心脏特异性保护性piRNA(piR-mmu-57256903),将其命名为运动诱导保护性piRNA(EPPIR)。EPPIR在扩张型心肌病患者及阿霉素心脏毒性模型的心脏组织中表达显著降低。功能上,EPPIR通过调控KDM6B(赖氨酸特异性去甲基化酶6B)-H3K27me3-Dtna表观遗传轴保护心脏免受阿霉素毒性损伤,同时作为心肌细胞特异性Tp53(肿瘤蛋白p53)抑制因子发挥作用。
结论:本研究鉴定出心肌细胞来源细胞外囊泡相关piRNA EPPIR在介导运动诱导心脏保护中的全新作用,EPPIR通过协同调控KDM6B-Dtna轴及心肌细胞特异性Tp53抑制发挥保护效应,为阿霉素心脏毒性的治疗提供机制依据与潜在策略。
急诊科常规头颅CT用于机会性心血管风险评估
期刊: J Am Coll CardiolPMID: 41949516
简述: 本项回顾性队列研究旨在开发并验证深度学习模型,通过急诊科常规头颅CT预测新发心血管疾病、评估冠状动脉钙化评分,并与临床风险因素对比预测性能。研究纳入斯坦福医疗中心急诊科27990例无已知心血管疾病的成人患者与2313例联合检查患者,结果显示头颅CT模型的心血管风险预测效能显著优于美国心脏协会PREVENT风险模型,可联合传统风险因素完成心血管风险分层,无需额外医疗成本与辐射暴露即可识别年轻高危患者的亚临床疾病。摘要翻译:
背景: 常规非心脏计算机断层扫描成像可能包含心血管风险相关信息,头颅CT是临床最常用的影像学检查之一,每年有数百万患者接受该项检查,但其在心血管风险评估中的应用价值尚未被研究。
目的: 本研究旨在开发并验证深度学习模型,通过头颅CT预测新发心血管疾病、评估冠状动脉钙化评分,并对比其与临床风险因素的预测性能。
方法: 本回顾性队列研究纳入2020年8月至2024年8月斯坦福医疗中心急诊科的数据,心血管疾病队列包含27990例接受头颅CT检查且无已知心血管疾病的成人患者,冠状动脉钙化评分队列包含2313例同时接受头颅CT与冠状动脉CT血管造影的患者。研究采用预训练深度学习模型从头颅CT中提取影像学特征,从电子健康记录中提取其他风险因素,结局指标为新发心血管并发症(心肌梗死、卒中、心力衰竭)与冠状动脉钙化评分分层。采用一致性指数和受试者工作特征曲线下面积评估模型性能,并与采用美国心脏协会PREVENT风险模型变量的基线模型进行对比。
结果: 27990例患者中4%(1110例)发生心血管事件,患者中位年龄63.0岁,女性占51.7%。头颅CT模型的一致性指数为0.82(95%CI:0.78-0.85),PREVENT模型为0.75(95%CI:0.70-0.79),两组差值为0.07(95%CI:0.04-0.10)。在冠状动脉钙化评分评估中(2313例患者,中位年龄65.0岁,女性占53.5%),头颅CT联合PREVENT模型对冠状动脉钙化评分>100的预测一致性指数为0.76(95%CI:0.72-0.80),受试者工作特征曲线下面积为0.80(95%CI:0.73-0.85)。15.7%的患者被重新分层,重新归类为高危的患者更年轻,且血管钙化(30.2% vs 24.8%,P=0.001)和脑梗死(20.1% vs 5.8%,P<0.001)发生率更高。
结论: 常规头颅CT检查可补充传统风险因素用于心血管风险分层,识别风险评分良好的年轻患者的亚临床疾病。将该技术整合至临床实践,可在不增加成本和辐射的前提下,提升心血管疾病的筛查与预防水平。
护士指导下的电话干预用于卒中后高血压控制:加纳一项多中心3期随机试验
期刊: CirculationPMID: 41953982
简述: 本项多中心、随机、开放标签、终点盲法3期试验在加纳10家医院开展,针对低收入地区卒中后高血压患者,评估护士主导的移动健康电话干预的疗效与安全性。干预方案包含家庭血压自测、电话用药提醒、每周方言语音健康教育,结果显示干预组12个月血压控制率显著高于常规护理组,该方案可安全有效改善资源有限地区卒中幸存者的血压控制情况。摘要翻译:
背景: 应对低收入国家日益加重的卒中疾病负担,需要针对主要危险因素制定实用、可推广的干预措施。常规医疗条件下,仅有不足10%的高血压成人患者实现血压控制,显著增加了不良血管事件的发生风险。目前尚不清楚,以移动健康为核心、护士主导的干预措施,能否有效控制非洲资源有限地区近期卒中患者的高血压。
方法: 本项PINGS试验对比了常规护理与为期12个月的综合干预方案的疗效和安全性,干预方案包括:家庭血压自我监测、护士对血压升高患者的个案管理、电话闹钟用药提醒、护士每周通过加纳方言语音电话开展心血管风险降低教育。本研究为多中心、随机、开放标签、终点盲法评估试验,于2020年10月23日至2024年4月5日在10家医院开展,共纳入500例≥18岁、发病1个月内的卒中患者,且患者血压≥140/90mmHg。主要结局为12个月时收缩压<140mmHg,采用意向性治疗分析;次要结局包括主要不良心血管事件和严重不良事件。
结果: 244例患者被分配至干预组,256例分配至常规护理组,女性占43%,平均年龄58(11)岁。干预组12个月收缩压较基线平均下降5.5mmHg(95%CI:-9.6~-1.4mmHg;P=0.008)。干预组主要结局达标率为67%(163例),常规护理组为43%(109例),组间差异24%(95%CI:15%~33%;P<0.001)。两组主要不良心血管事件次要结局、用药依从性关键中介指标无显著差异;干预组严重不良事件发生率11.1%(27/244),常规护理组7.0%(18/256),P=0.12。
结论: 在常规护理基础上,采用简易移动健康技术并将诊疗任务转移给护士,可安全改善低资源地区卒中幸存者的血压控制情况,未来仍需进一步研究验证该结果并明确疗效驱动因素。
血液系统恶性肿瘤成人患者的心血管疾病风险:一项基于关联数据库的人群队列研究
期刊: J Am Coll CardiolPMID: 41949515
简述: 本项基于荷兰国家数据库的人群队列研究,纳入174984例血液系统恶性肿瘤患者与855085名健康对照,评估11种心血管结局的发病风险。结果显示所有肿瘤亚型患者的心力衰竭风险均显著升高,骨髓增生异常综合征与多发性骨髓瘤风险最高;静脉血栓栓塞风险在诊断后第一年达峰值,且所有亚型患者的风险持续升高5年,不同肿瘤亚型的心血管疾病风险存在显著差异。摘要翻译:
背景: 近几十年来,血液系统恶性肿瘤患者的生存率大幅提升,治疗相关不良反应和长期健康并发症(包括心血管疾病)受到广泛关注。这类并发症会降低癌症治疗耐受性,增加患者的发病率和死亡率。
目的: 评估不同类型血液系统恶性肿瘤患者心血管疾病的短期和长期发病风险。
方法: 选取1995年至2023年荷兰癌症登记处中,确诊12种最常见血液系统恶性肿瘤的成年患者(≥18岁),并匹配普通人群对照。将研究数据与国家住院登记、死亡原因登记相关联,明确11种心血管结局。采用泊松回归模型和Fine-Gray竞争风险模型,评估心血管疾病的绝对发病风险和相对发病风险。
结果: 研究纳入174984例血液系统恶性肿瘤患者和855085名对照,所有肿瘤亚型患者的心力衰竭发病率均升高,每1000人年的超额发病率以骨髓增生异常综合征(37.75;95%CI:35.60~39.90)和多发性骨髓瘤(24.68;95%CI:23.46~25.90)最高。静脉血栓栓塞风险在确诊后第一年内达到峰值,所有肿瘤亚型患者的风险持续升高5年;深静脉血栓形成的第一年风险比,慢性淋巴细胞白血病为3.52(95%CI:2.58~4.80),霍奇金淋巴瘤高达34.04(95%CI:19.83~58.44)。
结论: 与普通人群相比,血液系统恶性肿瘤患者及幸存者的心血管疾病短期和长期风险均显著升高,且风险水平因肿瘤亚型不同存在显著差异。明确患者、疾病和治疗相关的风险驱动因素,对制定针对性的预防和管理策略、改善患者预后至关重要。
基于风箱模型物理信息神经网络的心血管数字孪生技术
期刊: NPJ Digit MedPMID: 41965786
简述: 心血管数字孪生是精准医疗的核心技术,现有构建方法存在有创、依赖大数据等缺陷。本研究提出风箱模型物理信息神经网络框架,将风箱模型的控制微分方程嵌入神经网络训练过程,通过无创生物阻抗可穿戴设备估算动脉顺应性、外周阻力等个性化参数,预测血压波形,误差较传统深度学习模型降低12%-25%,为无创、可解释的心血管数字孪生构建奠定基础。摘要翻译:
心血管数字孪生技术通过构建心血管系统虚拟模型,实现个体化诊疗分析、实时监测和心血管动力学个性化模拟,有望变革精准医疗领域。精准构建虚拟模型需要准确估算动脉顺应性、外周阻力等个性化参数,但现有方法操作繁琐、多为有创检查或依赖大规模数据集。为解决上述局限,本研究提出风箱模型物理信息神经网络框架,该框架联合风箱模型与物理信息神经网络,通过无创生物阻抗可穿戴设备估算个性化心血管参数、预测血压波形。风箱模型物理信息神经网络将风箱模型的控制微分方程嵌入训练过程,仅需少量真实数据即可实现可解释、高精度的血压预测。研究通过健康人群和高血压患者的生物阻抗数据集验证该框架,预测误差较传统数据驱动深度学习模型降低12%-25%;同时,该框架可高精度估算动脉顺应性和外周阻力,在合成心血管波形数据集中误差仅0.77%-6.07%。本研究证实风箱模型物理信息神经网络是无创、可解释心血管数字孪生框架的可靠基础。
一条新型G13-RAPGEF2-RAP1信号通路对血小板黏附至关重要
期刊: BloodPMID: 41949994
简述: 本研究证实血小板中存在新型G13-RAPGEF2-RAP1信号通路,RAPGEF2作为低表达的鸟苷酸交换因子,在G13蛋白下游调控RAP1信号与整合素激活,尤其在高剪切应力条件下发挥关键作用,弥补了既往仅关注CalDAG-GEFI的研究空白,深化了对血小板G蛋白信号与整合素功能的认知,为心血管疾病的抗血小板靶向治疗研发提供新方向。摘要翻译:
止血与血栓形成高度依赖血小板在剪切应力条件下快速激活整合素受体并牢固黏附于损伤部位的独特能力。整合素激活的核心是小GTP酶RAP1,其自身由鸟苷酸交换因子(GEFs)激活。CalDAG-GEFI(RASGRP2)是血小板中表达量最高且功能占主导地位的RAP1鸟苷酸交换因子。然而,一项全基因组关联研究也提示,低表达的RAP1鸟苷酸交换因子RAPGEF2(PDZ-GEFI)在人体血小板聚集中具有重要作用。本研究使用巨核细胞特异性敲除RAPGEF2(Rapgef2mKO)、CalDAG-GEFI敲除(Caldaggef1-/-)以及二者双敲除(DKO)的小鼠,探究RAPGEF2信号通路对血小板功能、止血与血栓形成的作用。研究在小鼠及人体血小板中均检测到RAPGEF2蛋白。与对照组或Caldaggef1-/-血小板相比,DKO血小板的RAP1激活与整合素αIIbβ3介导的聚集均显著减弱。与对照组相比,Rapgef2mKO血小板的整合素激活能力降低,聚集反应可逆性增强,且在体外及体内剪切应力条件下的黏附功能受损。机制研究强烈提示,RAPGEF2作用于与异源三聚体G蛋白G13(GNA13)偶联的受体下游,如整合素αIIbβ3与血栓烷受体。综上,本研究从遗传学角度证实,血小板中的RAPGEF2作为G13下游的重要调控因子,参与RAP1信号传导与整合素激活,尤其在高剪切应力条件下作用显著。这些发现显著深化了我们对血小板G蛋白信号与整合素功能的理解,对研发更优化的心血管疾病抗血小板靶向治疗具有潜在意义。
Tonlamarsen治疗未控制高血压患者的疗效:KARDINAL 2期随机临床试验
期刊: J Am Coll CardiolPMID: 41949512
简述: 本项随机、安慰剂对照的2期KARDINAL试验评估了每月皮下注射90mg Tonlamarsen持续5个月对比单次Tonlamarsen注射后序贯安慰剂治疗未控制高血压患者的安全性与疗效,结果显示每月给药可更显著降低血浆血管紧张素原水平,但两组在诊室收缩压降低方面无额外差异,严重不良事件发生率低且组间相似。摘要翻译:
背景:血管紧张素原的生成是肾素-血管紧张素-醛固酮系统激活的限速步骤。Tonlamarsen是一种靶向肝脏血管紧张素原合成的在研反义寡核苷酸,其在高血压患者中的疗效与安全性尚不明确。
目的:本研究旨在评估每月皮下注射90mg Tonlamarsen持续5个月,对比单次Tonlamarsen注射后序贯安慰剂治疗的安全性与疗效。
方法:这项随机、安慰剂对照试验纳入了接受2-5种降压药物治疗后诊室收缩压>135mmHg的成年高血压患者。受试者进入为期3部分的治疗期,每月给予研究药物,包括4周安慰剂导入期、随后4周单次Tonlamarsen注射的活性导入期,之后随机分组接受额外4次Tonlamarsen注射或匹配安慰剂治疗,共16周。共同主要终点为从基线至第20周,两组间血浆血管紧张素原水平变化的组间差异,以及诊室收缩压变化的组间差异。
结果:共计279名受试者完成安慰剂导入期,206名在活性导入期接受90mg Tonlamarsen,198名被随机分组。随机分组受试者在安慰剂导入期前后的平均血压分别为147/90mmHg与147/89mmHg。Tonlamarsen活性导入期后,平均血压为140/87mmHg。首次注射Tonlamarsen后20周,单次Tonlamarsen注射序贯安慰剂组血浆血管紧张素原水平的最小二乘(LS)平均百分比变化为-23.0%(95%CI:-27.8%至-18.2%),每月Tonlamarsen给药组为-67.2%(95%CI:-71.9%至-62.4%),LS平均差异为-44.1%(97.5%CI:-51.9%至-36.4%;P<0.0001)。单次Tonlamarsen注射序贯安慰剂组诊室收缩压的LS平均变化为-6.7mmHg(95%CI:-9.8至-3.5mmHg),每月Tonlamarsen治疗组为-6.7mmHg(95%CI:-9.8至-3.6mmHg),LS平均差异为-0.1mmHg(95%CI:-4.5至4.4mmHg;P=0.97)。严重不良事件发生率较低,且治疗组间相似。
结论:在未控制高血压患者中,每月给予Tonlamarsen较单次给药可更有效降低血浆血管紧张素原水平,但未带来额外的血压降低获益。
经皮冠状动脉介入治疗与冠状动脉旁路移植术治疗无保护左主干狭窄的对比:随机、开放标签、非劣效性NOBLE试验10年最终结果
期刊: LancetPMID: 41936368
简述: NOBLE试验10年最终结果显示,对于无保护左主干冠状动脉疾病且无其他复杂病变的患者,经皮冠状动脉介入治疗(PCI,应用新一代药物洗脱支架)与冠状动脉旁路移植术(CABG)的10年全因死亡率无显著差异,证实PCI在符合两种治疗条件的患者中安全性与CABG相当,为心脏团队制定个体化患者诊疗策略提供依据。摘要翻译:
背景:对于存在显著无保护左主干冠状动脉疾病的患者,指南推荐冠状动脉旁路移植术(CABG)优于经皮冠状动脉介入治疗(PCI)。本研究旨在提供对比新一代药物洗脱支架PCI与CABG的长期结局数据,此类长期数据目前较为匮乏。
方法:这项已发表的前瞻性、随机、开放标签、非劣效性试验在丹麦、爱沙尼亚、芬兰、德国、拉脱维亚、立陶宛、挪威、瑞典及英国的36家医院纳入无保护左主干冠状动脉狭窄患者。入组资格由多学科心脏团队评估确定,临床标准为慢性或急性冠状动脉综合征且预期寿命>1年,造影标准为左主干冠状动脉直径狭窄≥50%或左主干开口、中段或分叉处血流储备分数≤0.80。
排除24小时内发生ST段抬高型心肌梗死、或被判定CABG/PCI风险过高的患者。经造影证实存在显著左主干冠状动脉疾病的患者通过在线系统按1:1比例随机分配至PCI组或CABG组,分组依据研究中心、性别、左主干远端分叉病变及糖尿病进行分层。
主要终点为意向治疗(ITT)人群的10年全因死亡率差异,采用Kaplan-Meier估计与未校正Cox回归进行分析。患者在死亡、移民、退出或失访日期进行截尾。在预设亚组中评估全因死亡率的差异。本试验已在ClinicalTrials.gov注册,注册号NCT01496651(试验进行中,不再招募受试者)。
结果:2008年12月9日至2015年1月21日,1201名患者被随机分配至PCI组(n=598)或CABG组(n=603)。17名患者在1年内失访,每组各592名患者纳入ITT人群。PCI组平均年龄66.2岁(标准差9.9),CABG组平均年龄66.2岁(标准差9.4)。1184名受试者中256名(22%)为女性,928名(78%)为男性。两组10年全因死亡率无差异(PCI组592人中136人[23%],CABG组592人中145人[25%];风险比0.93[95%CI 0.74-1.18];P=0.56)。基于SYNTAX评分的全因死亡率亦未发现显著差异。
解读:对于无保护左主干冠状动脉疾病且无其他复杂病变的患者,PCI与CABG的10年全因死亡率无显著差异,提示在符合两种治疗条件的患者中,PCI的安全性与CABG相当。该结果将助力心脏团队制定以患者为中心的个体化诊疗策略。
通过REWIND研究的插补安慰剂分析评估替尔泊肽在SURPASS-CVOT研究中真实的主要不良心血管事件获益
期刊: Diabetes CarePMID: 41940793
简述: 本研究通过联合SURPASS-CVOT与REWIND研究数据,采用预设分析方法对比替尔泊肽与插补安慰剂的主要不良心血管事件治疗效应,经倾向评分校正后开展间接比较,结果显示替尔泊肽可显著降低2型糖尿病合并动脉粥样硬化性心血管疾病患者的主要不良心血管事件、心血管死亡或心力衰竭事件及全因死亡风险,各项敏感性分析结果均保持一致。摘要翻译:
目的: 在预设分析中,利用SURPASS-CVOT与REWIND研究数据,评估替尔泊肽对比插补安慰剂的主要不良心血管事件治疗效应。
研究设计与方法: 针对SURPASS-CVOT研究的主要终点(3项主要不良心血管事件)与次要终点开展与安慰剂的间接比较。分析纳入REWIND研究中符合SURPASS-CVOT入组标准的受试者,以及SURPASS-CVOT研究的全部受试者。采用倾向评分估计校正两项研究间受试者特征的差异。替尔泊肽对比安慰剂的治疗效应间接分析结果,通过SURPASS-CVOT研究中替尔泊肽与度拉糖肽的3项主要不良心血管事件风险比,乘以REWIND研究中度拉糖肽对比安慰剂的风险比计算得出。开展敏感性分析,包括未校正分析(纳入筛选后的REWIND受试者与全部REWIND受试者),以及全部REWIND受试者的校正分析;事后敏感性分析采用近期纳入REWIND研究的胰高血糖素样肽1受体激动剂荟萃分析数据。
结果: 分析纳入REWIND研究9901名受试者中的2055人,以及SURPASS-CVOT研究的全部13165名受试者。间接治疗效应比较显示,替尔泊肽对比安慰剂与更低的3项主要不良心血管事件风险相关(风险比0.72;95%置信区间0.55~0.94),同时降低心血管死亡或心力衰竭事件风险(风险比0.70;95%置信区间0.51~0.96)与全因死亡风险(风险比0.61;95%置信区间0.45~0.82)。包括未校正分析、全部REWIND队列数据或胰高血糖素样肽1受体激动剂荟萃分析数据在内的敏感性分析结果总体一致。
结论: 在这项预设的探索性间接比较中,在2型糖尿病合并确诊动脉粥样硬化性心血管疾病的受试者中,替尔泊肽对比插补安慰剂与心血管结局及全因死亡率降低相关。
2026-04-10
本文涉及大量转载内容,版权归属原作者,如涉侵权,请联系删除。
兆维
事业发展部
——
Hongene
Biotech B&D
INDUSTRY NEWS
核酸产业要闻半月报
01 国内药物研发进展
<
舶望制药 siRNA 疗法获 FDA 快速通道认定
3 月 16 日,舶望制药宣布其在研 siRNA 疗法 BW-20805 获得美国食品药品监督管理局(FDA)授予快速通道资格(Fast Track Designation, FTD),用于治疗遗传性血管性水肿(hereditary angioedema, HAE)。
BW-20805 是一种靶向前激肽释放酶 (prekallikrein, PKK)的 siRNA 疗法,通过靶向 PKK mRNA 而抑制 PKK 的表达,有望为 HAE 患者提供长效的发作预防。PKK 是目前 HAE 治疗领域已获得充分验证的作用靶点。舶望制药目前在 HAE 成人患者中开展 II 期临床研究,预计将于 2026 年下半年完成 II 期临床试验,并随后启动 III 期临床研究。
参考资料:
https://www.argobiopharma.com/cn/news/163.html
前沿生物 siRNA 新药获批临床,针对 IgA 肾病
3 月 16 日,中国国家药监局药品审评中心(CDE)官网公示显示,前沿生物 1 类新药 FB7013 注射液获批临床,拟用于原发性 IgA 肾病的治疗。根据公示信息,FB7013 注射液是靶向 MASP-2 的 GalNAc 偶联的 siRNA 药物,抑制 MASP-2 可能对 IgA 肾病的肾小球和小管间质都有保护作用。
FB7013 注射液是一款靶向补体系统凝集素途径关键蛋白 MASP-2 靶点的 siRNA 药物。该产品通过特异性抑制 MASP-2 活性阻断凝集素途径异常激活,减少补体介导的肾脏组织损伤;基于作用机制,未来可拓展至膜性肾病、糖尿病肾病等多个补体异常激活相关疾病领域。
参考资料:
https://www.cde.org.cn/
https://www.frontierbiotech.com/detail/1562.html
GSK 治疗乙肝的 ASO 疗法在中国拟纳入优先审评
3 月 17 日,中国国家药监局药品审评中心(CDE)官网公示显示,葛兰素史克(GSK)申报的贝普若韦生注射液(Bepirovirsen solution for injection)拟纳入优先审评,受理号为:JXHS2600033。
该产品用于慢性乙型肝炎病毒感染者的有限疗程治疗,适用于以下人群:核苷(酸)类似物在治、HBsAg ≤ 3000IU/mL、无肝硬化的成人慢性乙型肝炎病毒(HBV)感染者。公开资料显示,这是 GSK 与 Ionis Pharmaceuticals 合作开发的在研反义寡核苷酸(ASO)疗法。
Bepirovirsen 是一种具有三重作用机制的在研 ASO 疗法,旨在识别并破坏乙肝病毒的遗传成分(即 RNA),从而可能使患者的免疫系统重新获得控制病毒感染的能力。Bepirovirsen 可抑制体内病毒复制,降低血液中 HBsAg 水平,并激活免疫系统,从而提高获得持久应答的机会。GSK 从 Ionis Pharmaceuticals 获得 Bepirovirsen 授权,并与其合作推进该药物的开发。
参考资料:
https://www.cde.org.cn/
https://mp.weixin.qq.com/s/aEiBTcbp2bVbH6wjfqCg9w
深信生物 mRNA 疗法获 FDA 临床试验许可
3 月 18 日,深信生物宣布,美国 FDA 已批准其 mRNA 候选药物 IN026 的临床试验(IND)申请。IN026 是一款针对难治性痛风的在研 mRNA 疗法。此次 IND 获批后,公司将在 I 期临床试验中系统评估 IN026 在这一治疗需求未被满足的患者群体中的安全性、耐受性、药代动力学及药效学特征。
IN026 是一款旨在治疗难治性痛风的在研 mRNA 疗法。通过将编码尿酸氧化酶(UOX)的 mRNA 递送至肝脏,利用表达的 UOX 促进体内尿酸的系统性降解。依托深信生物自主开发、专为重复给药和长期疾病控制而设计的 mRNA-LNP(信使核糖核酸-脂质纳米颗粒)技术平台,IN026 有望成为针对难治性痛风及其他慢性代谢疾病的潜在 mRNA 蛋白替代疗法。
参考资料:
https://finance.sina.com.cn/wm/2026-03-18/doc-inhrkfva3455651.shtml
信立泰自研 siRNA 药物 SAL0145 获批临床
3 月 25 日,信立泰发布公告,公司自主研发的创新药物 SAL0145 注射液获批开展 MASH 适应症的临床试验。
SAL0145 是一种小干扰核酸(siRNA)药物,临床前研究显示,SAL0145 具有治疗 MASH 的潜力,若能研发成功并获批上市,将有望为患者提供新的用药选择,满足未被满足的临床需求,并进一步丰富公司慢病领域的创新产品管线。
参考资料:
https://www.salubris.com/NewsUpdates/info_itemid_5668.html
安龙生物与阳光诺和合作开发的 siRNA 新药 ABA001 正式获批临床
4 月 3 日,安龙生物与阳光诺和共同宣布,双方合作开发的治疗高血压 siRNA 创新药 ABA001(受理号:CXHL2600091)已正式获得国家药品监督管理局(NMPA)药物临床试验批准通知书。
ABA001 是一款靶向血管紧张素原的 GalNAc-siRNA 药物,属于 1 类创新药。该药物旨在利用前沿的 RNA 干扰技术,为全球高血压患者提供一种安全有效、单次注射即可实现超长期平稳降压的革命性治疗方案。
参考资料:
https://www.cde.org.cn/
http://www.sun-novo.com/newsinfo/9018214.html
星核迪赛 DSL201 注射液获中国 CDE 批准,实现中美双报里程碑
4 月 3 日,苏州星核迪赛生物技术有限公司宣布,公司自主研发的全球首款基于 mRNA 编码 A 型肉毒毒素蛋白的创新型药物 DSL201 注射液,正式获得中国国家药品监督管理局药品审评中心(CDE)的临床试验批准,受理号为 CXSL2501184。该药物将用于评估其在中度至重度眉间纹受试者中的安全性、耐受性、免疫原性及初步有效性。此次获批标志着 DSL201 注射液已成功获得美国食品药品监督管理局(FDA)和中国 CDE 的双重许可,正式迈入中美双报的临床研究阶段。
DSL201 注射液的活性成分为 mRNA 编码的 A 型肉毒毒素,通过星核迪赛独有的局部表达平台 DScSLOTH️®️ 与低免疫原性平台 DScBISON️®️ 联合开发的靶向递送系统,实现了药物在注射部位的精准、高效表达,同时具备优异的安全性。临床前研究已充分证实,DSL201 注射液作为一款 mRNA-LNP 药物,展现出良好的安全性与长效性,有望为求美者带来全新的治疗选择。
参考资料:
https://www.cde.org.cn/
02 国内投融资及合作信息
<
百洋医药战略投资思合基因
3 月 19 日晚,百洋医药发布公告,公司拟与思合基因(北京)生物科技有限公司签署《投资协议》,约定以现金方式向思合基因投资 2700 万元,投资后公司持有思合基因 10% 股权。通过此次合作,百洋医药战略性锁定了思合基因所有在研管线的全球优先受让权与商业化权益。这标志着百洋医药正式切入小核酸黄金赛道,开启下一代创新疗法布局的新篇章。
参考资料:
http://www.baiyyy.com/news/details/460.html
炫景生物完成超 2 亿元 Pre-A 轮融资
3 月 20 日,炫景生物完成的超 2 亿元 Pre-A 轮融资由康君资本领投,德联资本、华泰金斯瑞基金、金易赋新、国海创新资本、联想创投和现有股东元希海河共同参与投资。
参考资料:
https://www.rigerna.com/gsxw/class/
先衍生物完成 1.5 亿元 A 轮融资
3 月 20 日,先衍生物完成的 1.5 亿元 A 轮融资由道远资本领投,启申创投联合领投,嘉乐资本、成都科创投集团、四川省中试研发有限公司、厦门高新投、增锐铭禾等跟投,成都先导、银满基金、怀格资本、钧天创投、曹家铭、华博器械等老股东持续追投。
本轮融资将主要用于推动核心小核酸产品管线的临床开发、加速肝外递送等关键技术平台的持续优化,并进一步扩充临床前产品布局、核心团队建设。
先衍生物专注于寡聚核酸药物研发,已成功搭建三大拥有自主知识产权的小核酸药物研发核心技术平台:NEATL(新颖递送及核酸增强修饰技术)、LOCATED(脂质偶联肝外递送技术)和 DIRECT(单分子双靶点干扰技术)。
参考资料:
https://news.pedaily.cn/202603/561892.shtml
峰肽药业宣布完成 8900 万美元 B 轮融资,并获超额认购
3 月 26 日,峰肽药业(Pinnacle Medicines)宣布完成 8900 万美元 B 轮融资,并获超额认购。本轮融资由 LAV 与 Foresite Capital 共同领投,泉创资本、汉康资本、RA Capital Management、Logos Capital 以及现有投资人奥博资本和启明创投共同参与。
本轮融资所得将用于支持公司在自身免疫和心血管代谢疾病领域推进其项目加速进入临床阶段,同时进一步拓展公司专有的多肽研发平台并提升研发能力。峰肽药业正在开发新一代口服多肽药物,旨在实现口服给药便利性的同时达到与抗体相当的疗效和安全性。
峰肽药业是一家专注于开发新一代口服多肽疗法的生物技术公司,旨在通过口服给药就能够实现媲美抗体的治疗效果。公司正在构建差异化的药物发现平台以加速多肽药物的设计和优化。该平台深度融合了基于物理学的分子模拟、人工智能驱动的药物设计以及先进的多肽化学技术。峰肽药业正在推进一系列具有潜力的“同类首创”(First-in-class)和“同类最优”(Best-in-class)口服多肽项目管线。公司管线聚焦在自身免疫和心血管代谢疾病中已获得充分临床数据验证的靶点通路。
参考资料:
https://www.prnewswire.com/news-releases/pinnacle-medicines-a-pioneer-of-oral-peptide-therapeutics-announces-oversubscribed-89-million-series-b-financing-to-advance-pipeline-into-clinical-trials-302724900.html
元思生肽完成 B 轮融资
3 月 31 日,智能化平台驱动的大环肽药物研发领军企业元思生肽(Syneron Bio)宣布完成 1.5 亿美元 B 轮融资。本轮融资由一家国际生物科技基金领投,并由德诚资本与鼎晖 VGC 联合领投,阿布扎比投资局(ADIA)旗下全资子公司、淡马锡独立全资子公司淡明资本、启明创投、博远资本及知名产业投资机构跟投。同时,原有股东阿斯利康、礼来亚洲基金、创新工场、五源资本、高瓴创投(GL Ventures)、Biotech Development Fund 及联想创投等机构持续加持。本轮募集资金将重点用于深化其自主研发的大环肽发现平台 Synova™ 的智能化迭代,并全力加速公司多元化创新药管线向临床阶段转化。
元思生肽(Syneron Bio)是一家致力于开发新型大环肽药物的生物科技公司,公司利用独有的 Synova™ 平台技术建立了丰富而差异化的大环肽药物管线,为全球肿瘤、自身免疫疾病及罕见病领域提供突破性的治疗方案。Syneron Bio 核心团队具有丰富的药物研发经验及国际一流的数据科学技术能力。公司成立四年以来完成了多轮股权融资,投资人涵盖多家知名产业基金及风险投资基金。
参考资料:
https://finance.sina.com.cn/tech/roll/2026-03-31/doc-inhswpms3287953.shtml
03 国外药物研发进展
<
Sarepta 公布两项 siRNA 疗法积极临床结果
3 月 25 日,Sarepta Therapeutics 公布了其两项针对神经肌肉疾病的 siRNA 项目的首次临床结果。来自 SRP-1001(用于面肩肱型肌营养不良 1 型,FSHD1)和 SRP-1003(用于强直性肌营养不良 1 型,DM1)的 I/II 期递增剂量研究的早期数据显示,这两款候选疗法在肌肉组织中实现了剂量依赖性的暴露,并在生物标志物层面展现出早期作用信号,同时整体耐受性良好。研究还显示,单次给药后即可观察到靶蛋白或 mRNA 水平的降低,提示其在靶向机制上的初步有效性。此外,两项研究中大多数不良事件为轻至中度,且未呈现剂量依赖性,进一步支持其安全性。根据新闻稿,这些结果亦显示其基于 αvβ6 整合素靶向递送平台具有差异化效果。
从机制上看,FSHD 由 4 号染色体上 DUX4 基因异常激活所致,其表达的 DUX4 蛋白对肌肉细胞具有毒性并驱动肌肉退行性变化,目前尚无治愈方法或疾病修饰疗法。SRP-1001 通过 RNAi 机制靶向并降低骨骼肌中 DUX4 蛋白的生成,旨在从源头干预疾病进程。另一方面,DM1 是最常见的成人发病型肌营养不良,由 DMPK 基因重复序列扩增引起。SRP-1003 通过靶向并沉默 DMPK mRNA,可潜在缓解相关疾病症状。
参考资料:
https://www.businesswire.com/news/home/20260325621936/en/Sarepta-Announces-First-Clinical-Data-from-siRNA-Pipeline-Targeting-FSHD1-and-DM1
Wave life sciences INHBE siRNA 疗效不及预期
3 月 26 日,Wave Life Sciences 公布 INHBE siRNA 新药 WVE-007 I 期临床最新数据。240mg WVE-007 单次给药 3 个月内脏脂肪减少 7.8%,6 个月减少 14.3%,总脂肪分别减少 5.1%、5.3%,肌肉增加 2.2%、2.4%。400mg WVE-007 单次给药 3 个月内脏脂肪减少 5.0%,总脂肪减少 0.7%,肌肉减少 0.2%。Wave Life Sciences 指出 400mg 队列基线脂肪放量更少,BMI 更低。对于内脏脂肪 500g 以上亚组分析,内脏脂肪减少 7.8%,与 240mg 队列的数据相当。
受此消息影响,Wave Life Sciences 股价盘前大跌 58%,市值仅剩下 10 亿美元。WVE-007 的安全性优势尤为明显,去年 12 月披露的数据也显示出一定的疗效潜力,但此次更新数据没有看到明确的量效关系,让资本市场对于 INHBE siRNA 的想象空间和热情有所下降。
参考资料:
https://ir.wavelifesciences.com/news-releases/news-release-details/wave-life-sciences-announces-positive-interim-phase-1-data
https://mp.weixin.qq.com/s/qF3nFtRbGREUrXKgLqZPiA
Beam Therapeutics 公布 BEAM-302 临床试验数据
3 月 26 日,Beam Therapeutics 公布了其在研基因编辑疗法 BEAM-302 用于治疗 α1 抗胰蛋白酶缺乏症(AATD)的 I/II 期临床试验最新安全性与有效性数据。
数据显示,在 60mg 剂量下,患者体内总 AAT 水平稳定维持在平均 16.1µM,所有患者在最长 12 个月随访中均持续高于 11µM 的保护阈值。同时,经纠正的 M-AAT 占总 AAT 的 94%,并伴随致病突变型 Z-AAT 水平降低 84%。在功能层面,治疗后患者在呼吸道感染期间仍可诱导 AAT 表达,其中一例患者总 AAT 水平升至约 30µM,且维持 95% 的 M-AAT 比例,提示该疗法不仅可恢复基础蛋白水平,还具备生理调节能力。安全性方面,BEAM-302 在最高 75mg 单次给药下整体耐受性良好,各剂量组之间安全性表现一致。
基于在单剂量队列中观察到的良好安全性与疗效数据,公司已确定 60mg 为最佳剂量,并计划于 2026 年下半年启动全球关键性研究,以支持潜在的加速批准。
BEAM-302 基于一种靶向肝脏的脂质纳米颗粒(LNP)递送体系,通过碱基编辑技术直接纠正导致严重 α1 抗胰蛋白酶缺乏症的致病突变。该疗法通过一次性碱基编辑,有望同时减少错误折叠 Z-AAT 蛋白在肝脏的聚集、提升功能性 M-AAT 水平,并改善循环中总 AAT 水平,从病因层面干预 AATD 相关的肝脏与肺部病理过程。
参考资料:
https://www.globenewswire.com/news-release/2026/03/25/3262054/0/en/Beam-Therapeutics-Announces-Compelling-Updated-Clinical-Data-from-the-Ongoing-Phase-1-2-Trial-of-BEAM-302-in-Alpha-1-Antitrypsin-Deficiency-AATD-to-Support-Advancement-to-Pivotal-D.html
Biogen 宣布 Nusinersen 获美国 FDA 批准高剂量给药方案
3 月 30 日,渤健(Biogen)宣布,美国 FDA 已批准其 ASO 疗法 Spinraza(nusinersen)高剂量给药方案,用于治疗脊髓性肌萎缩症。Spinraza 是渤健和 Ionis Pharmaceuticals 联合开发的一款 ASO 药物。它通过与 SMN2 基因转录形成的 mRNA 相结合,改变 RNA 的剪接过程,从而增加正常 SMN 蛋白的表达量。因此,这一疗法可以在 SMN1 基因失活的 SMA 患者身上增加正常 SMN 蛋白的水平,从而维持运动神经元的生存。2016 年底,Spinraza 获得 FDA 批准上市,成为首款治疗 SMA 的疗法。该疗法曾获 2017 年美国盖伦奖(Prix Galien USA Awards)中的最佳生物技术产品奖。
此次获批的高剂量方案包括 50mg/5mL 和 28mg/5mL 两种剂量,是在低剂量(12mg)Spinraza基础上开发而成。通过在起始剂量期和维持期提供更高药物浓度,高剂量方案旨在为 SMA 患者群体提供新的治疗选择,以更好满足持续存在的治疗需求。
该批准主要基于 II/III 期 DEVOTE 研究数据,其中关键队列结果显示,在此前未接受治疗且出现症状的婴儿患者中,高剂量 Spinraza 在运动功能改善方面达到统计学显著差异。以 CHOP-INTEND 评分评估,与 ENDEAR 研究中预设的未治疗匹配对照组相比,治疗组平均差异为 26.19 分(+15.1 vs. -11.1, p<0.0001)。
参考资料:
https://investors.biogen.com/news-releases/news-release-details/biogen-announces-second-positive-phase-2-litifilimab-trial
Alltrna tRNA 疗法获批临床
3 月 31 日,Alltrna 宣布,其自主研发的 AP003 的 I 期临床试验已获澳大利亚批准,这是全球首个获批进入临床试验的 tRNA 疗法,用于治疗由精氨酸-TGA(Arg-TGA)提前终止密码子导致的疾病。
AP003 是一种经过化学修饰、工程化的 tRNA 疗法,采用已获临床验证的肝靶向脂质纳米粒(LNP)包裹递送。Arg-TGA 变异的产生机制为:编码精氨酸的密码子 CGA 或 AGA 发生单碱基突变,变为 UGA(DNA 中为 TGA)终止密码子,导致翻译过程无法插入对应氨基酸。AP003 可在 mRNA 翻译为蛋白质的过程中对提前终止密码子进行通读,在多肽链中重新插入精氨酸,从而恢复蛋白质正常合成。
参考资料:
https://www.alltrna.com/press/alltrna-announces-approval-to-initiate-first-in-human-clinical-trial-of-ap003-the-first-trna-therapeutic-to-enter-clinical-trials
Kardigan 公布 Tonlamarsen 的 II 期临床试验结果
4 月 2 日,Kardigan 公司公布了其靶向血管紧张素原(AGT)的 ASO 药物 Tonlamarsen的 II 期临床试验 KARDINAL 结果。Tonlamarsen 是一款每月一次皮下注射的在研药物,通过抑制肝脏中 AGT 的生成,从源头调节肾素-血管紧张素-醛固酮系统(RAAS),旨在为急性重度高血压患者提供一种潜在的血压管理方案。Tonlamarsen 由 Ionis Pharmaceuticals 公司发现和开发,Kardigan 从 Ionis Pharmaceuticals 获得了该药的全球独家开发和商业化授权。
此次公布的结果显示,从基线到接受 Tonlamarsen 治疗后第 20 周,患者的血浆 AGT 水平显著下降,oSBP(office systolic blood pressure)也有临床意义上的降低(-6.7mmHg)。每月给药组患者在第 20 周时平均 AGT 水平降低 67%,而单次给药组患者的平均 AGT 水平降低了 23%。在血压控制方面,基线血压较高(>150mmHg)的患者 oSBP 的降幅最大,为 -8.9mmHg。在安全性上,Tonlamarsen 总体耐受性良好,未出现有临床意义的治疗相关低血压或高钾血症,其良好的疗效和安全性数据支持了公司在急性重度高血压患者中启动 IIb 期临床试验的计划。
参考资料:
https://www.businesswire.com/news/home/20260328084287/en/Kardigan-Announces-Positive-Phase-2-Data-for-Tonlamarsen-in-Patients-with-Uncontrolled-Hypertension-Presented-as-Late-Breaker-at-ACC.26-and-Simultaneously-Published-in-JACC
作者:李昱
排版/校对:Levi
审核:Shirley & Vivian
2026-04-04
·药明康德
近期,全球多肽和寡核苷酸(TIDES)领域迎来系列进展。每月一次皮下注射的反义寡核苷酸(ASO)药物tonlamarsen的2期临床试验结果积极,治疗后第20周,患者的门诊收缩压(oSBP)出现具有临床意义的下降。美国FDA批准ASO疗法Spinraza(nusinersen)高剂量给药方案,用于治疗脊髓性肌萎缩症(SMA)。信达生物公布了玛仕度肽注射液在中国肥胖受试者中开展的2期9 mg剂量的研究结果,在第24周时较安慰剂校正后的减重幅度达15.4%。工程化tRNA疗法AP003在澳大利亚获批启动1期临床试验。本文将节选其中部分重要进展做简单介绍,仅供读者参阅。
Tonlamarsen:公布2期临床试验数据
Kardigan公司公布了其靶向血管紧张素原(AGT)的ASO药物tonlamarsen的2期临床试验KARDINAL结果。Tonlamarsen是一款每月一次皮下注射的在研药物,通过抑制肝脏中AGT的生成,从源头调节肾素-血管紧张素-醛固酮系统(RAAS),旨在为急性重度高血压患者提供一种潜在的血压管理方案。Tonlamarsen由Ionis Pharmaceuticals公司发现和开发,Kardigan从Ionis Pharmaceuticals获得了该药的全球独家开发和商业化授权。
此次公布的结果显示,从基线到接受tonlamarsen治疗后第20周,患者的血浆AGT水平显著下降,oSBP也有临床意义上的降低(-6.7 mmHg)。每月给药组患者在第20周时平均AGT水平降低67%,而单次给药组患者的平均AGT水平降低了23%。在血压控制方面,基线血压较高(>150 mmHg)的患者oSBP的降幅最大,为-8.9 mmHg。在安全性上,tonlamarsen总体耐受性良好,未出现有临床意义的治疗相关低血压或高钾血症,其良好的疗效和安全性数据支持了公司在急性重度高血压患者中启动2b期临床试验的计划。
Nusinersen:获美国FDA批准高剂量给药方案
渤健(Biogen)宣布,美国FDA已批准其ASO疗法Spinraza(nusinersen)高剂量给药方案,用于治疗脊髓性肌萎缩症。Spinraza是渤健和Ionis Pharmaceuticals联合开发的一款ASO药物。它通过与SMN2基因转录形成的mRNA相结合,改变RNA的剪接过程,从而增加正常SMN蛋白的表达量。因此,这一疗法可以在SMN1基因失活的SMA患者身上增加正常SMN蛋白的水平,从而维持运动神经元的生存。2016年底,Spinraza获得FDA批准上市,成为首款治疗SMA的疗法。该疗法曾获2017年美国盖伦奖(Prix Galien USA Awards)中的最佳生物技术产品奖。
此次获批的高剂量方案包括50 mg/5 mL和28 mg/5 mL两种剂量,是在低剂量Spinraza(12 mg)基础上开发而成。通过在起始剂量期和维持期提供更高药物浓度,高剂量方案旨在为SMA患者群体提供新的治疗选择,以更好满足持续存在的治疗需求。该批准主要基于2/3期DEVOTE研究数据,其中关键队列结果显示,在此前未接受治疗且出现症状的婴儿患者中,高剂量Spinraza在运动功能改善方面达到统计学显著差异。以CHOP-INTEND评分评估,与ENDEAR研究中预设的未治疗匹配对照组相比,治疗组平均差异为26.19分(+15.1 vs. -11.1,p<0.0001)。
玛仕度肽:公布2期临床试验数据
信达生物公布了玛仕度肽注射液在中国肥胖受试者中开展的2期9 mg剂量的临床研究结果。玛仕度肽是信达生物与礼来(Eli Lilly and Company)共同推进的一款胰高血糖素(GCG)/胰高血糖素样肽-1(GLP-1)双受体激动剂。作为一种哺乳动物胃泌酸调节素(OXM)类似物,玛仕度肽除了通过激动GLP-1受体促进胰岛素分泌、降低血糖和减轻体重外,还可通过激动GCG受体增加能量消耗增强减重疗效,同时改善肝脏脂肪代谢。玛仕度肽已在多项临床研究中展现出优秀的减重和降糖疗效,能够降低腰围、血脂、血压、血尿酸、肝酶及肝脏脂肪含量,并改善胰岛素敏感性,带来多重代谢获益。玛仕度肽注射液高剂量9 mg用于成人中重度肥胖患者长期体重控制的上市申请已于2025年11月获中国国家药品监督管理局(NMPA)药品审评中心(CDE)受理。
此次公布的2期结果显示,玛仕度肽9 mg治疗组在第24周时较安慰剂校正后的减重幅度达15.4%,心血管代谢指标改善,83.3%的受试者肝脏脂肪含量转至正常(低于5%)。第48周时减重疗效进一步提升,经安慰剂校正后的减重幅度可达18.6%,心血管代谢指标基本维持。安全性方面,玛仕度肽9 mg的耐受性良好,无受试者因不良事件提前终止治疗。
AP003:在澳大利亚获批启动1期临床试验
Alltrna公司宣布,澳大利亚治疗用品管理局(TGA)批准其启动AP003的1期临床试验。AP003是一款经过化学修饰的工程化tRNA疗法,封装在肝脏靶向脂质纳米颗粒中,正在开发用于治疗携带精氨酸转TGA(Arg-TGA)无义突变的肝脏终止密码子疾病患者。该候选疗法旨在通过读取提前终止密码子(PTC),在RNA翻译为蛋白质时重新引入精氨酸到生长中的多肽链中,恢复蛋白质生成。新闻稿指出,AP003是全球首个进入临床试验阶段的tRNA疗法。
在临床前模型中,AP003已被证明能够在由共享PTC驱动的疾病环境中恢复全长蛋白表达和下游功能活性。此外,临床前安全性评估显示,AP003的安全性特征与现有的寡核苷酸及脂质纳米颗粒疗法一致,支持其在人体中的进一步临床评估。这些数据支持Alltrna在多种由共享PTC驱动的疾病中评估AP003的广泛临床策略。
一体化平台助力寡核苷酸药物开发
在寡核苷酸药物快速发展的背景下,其复杂的化学修饰形式、多阶段代谢过程以及体内低暴露水平,使生物分析面临显著高于传统小分子的技术挑战。围绕这一需求,药明康德DMPK团队建立了基于液相色谱-串联质谱(LC-MS/MS)的系统化分析平台,用于寡核苷酸药物及其活性代谢产物的高灵敏检测与高分辨分离,支持从早期研究到临床前阶段的连续研究需求。LC-MS/MS结合液相色谱优异的分离能力与质谱检测的高选择性,可在复杂生物基质中实现对母体寡核苷酸及其代谢片段的精准定量与结构解析,为药代动力学与代谢机制研究提供关键数据基础。
针对寡核苷酸分子强亲水性、离子化效率受限以及易形成金属加合物等分析难点,药明康德通过优化样品前处理流程、离子对色谱体系及质谱参数设置,建立了适用于不同修饰类型寡核苷酸的分析策略,包括流动相组成调控与梯度洗脱优化,从而显著提升目标分子与代谢产物之间的分离度与检测灵敏度。同时,通过对多电荷离子信号的系统调控与方法学验证,团队能够在低浓度样品中保持良好的重复性与定量准确性,为PK、PD及安全性研究提供可靠数据支持。依托成熟的寡核苷酸DMPK研究经验与标准化生物分析流程,药明康德可实现母药与活性代谢产物的同步检测与关联分析,帮助研发团队深入理解体内代谢转化路径与暴露特征,为核酸药物的结构优化与开发决策提供坚实的分析基础。
参考资料:
[1] 全年减重超30斤,玛仕度肽9mg剂量在大体重中国人群中开展的II期临床研究结果在Cell子刊MED发布. Retrieved April 2, 2026, from https://mp.weixin.qq.com/s/cIH-3bJCoJuA_vQH0J_K_A
[2] Protagonist Announces Presentation of One-Year Phase 3 Data for ICOTYDE(TM) in Moderate-to-Severe Plaque Psoriasis at the 2026 American Academy of Dermatology (AAD) Annual Meeting. Retrieved March 30, 2026, from https://feeds.issuerdirect.com/news-release.html?newsid=8572882090629309&symbol=PTGX
[3] 中国国家药监局药品审评中心(CDE)官网. Retrieved April 2, 2026, from https://www.cde.org.cn/main/xxgk/listpage/4b5255eb0a84820cef4ca3e8b6bbe20c
[4] Biogen Announces Second Positive Phase 2 Litifilimab Trial in Cutaneous Lupus Erythematosus at 2026 American Academy of Dermatology Annual Meeting, Showing a Significant Reduction in Skin Disease Activity. Retrieved March 30, 2026 from https://investors.biogen.com/news-releases/news-release-details/biogen-announces-second-positive-phase-2-litifilimab-trial
[5] Avacta announces first patient treated in Phase 1 FOCUS-01 trial of FAP-Exd (AVA6103) - a sustained-release pre|CISION® exatecan peptide drug conjugate. Retrieved April 2, 2026, from https://avacta.com/avacta-announces-first-patient-treated-in-phase-1-focus-01-trial-of-fap-exd-ava6103-a-sustained-release-precision-exatecan-peptide-drug-conjugate/
[6] 元思生肽(Syneron Bio)完成1.5亿美元B轮融资,引领大环肽药物研发新范式. Retrieved April 2, 2026, from https://www.vbdata.cn/newsDetail/f7fe90c62cd311f1960800163e034e34
[7] Kardigan Announces Positive Phase 2 Data for Tonlamarsen in Patients with Uncontrolled Hypertension Presented as Late-Breaker at ACC.26 and Simultaneously Published in JACC. Retrieved April 2, 2026, from https://www.businesswire.com/news/home/20260328084287/en/Kardigan-Announces-Positive-Phase-2-Data-for-Tonlamarsen-in-Patients-with-Uncontrolled-Hypertension-Presented-as-Late-Breaker-at-ACC.26-and-Simultaneously-Published-in-JACC
[8] Alltrna Announces Approval to Initiate First-in-Human Clinical Trial of AP003, the First tRNA Therapeutic to Enter Clinical Trials. Retrieved April 2, 2026, from https://www.prnewswire.com/news-releases/alltrna-announces-approval-to-initiate-first-in-human-clinical-trial-of-ap003-the-first-trna-therapeutic-to-enter-clinical-trials-302728901.html
免责声明:本文仅作信息交流之目的,文中观点不代表药明康德立场,亦不代表药明康德支持或反对文中观点。本文也不是治疗方案推荐。如需获得治疗方案指导,请前往正规医院就诊。
版权说明:欢迎个人转发至朋友圈,谢绝媒体或机构未经授权以任何形式转载至其他平台。转载授权请在「药明康德」微信公众号回复“转载”,获取转载须知。
分享,点赞,在看,聚焦全球生物医药健康创新
100 项与 Tonlamarsen 相关的药物交易
登录后查看更多信息
研发状态
10 条进展最快的记录, 后查看更多信息
登录
| 适应症 | 最高研发状态 | 国家/地区 | 公司 | 日期 |
|---|---|---|---|---|
| 高血压,严重 | 临床2期 | 美国 | 2026-04-20 | |
| 高血压 | 临床2期 | 美国 | 2022-04-29 |
登录后查看更多信息
临床结果
临床结果
适应症
分期
评价
查看全部结果
| 研究 | 分期 | 人群特征 | 评价人数 | 分组 | 结果 | 评价 | 发布日期 |
|---|
No Data | |||||||
登录后查看更多信息
转化医学
使用我们的转化医学数据加速您的研究。
登录
或

药物交易
使用我们的药物交易数据加速您的研究。
登录
或

核心专利
使用我们的核心专利数据促进您的研究。
登录
或

临床分析
紧跟全球注册中心的最新临床试验。
登录
或

批准
利用最新的监管批准信息加速您的研究。
登录
或

特殊审评
只需点击几下即可了解关键药物信息。
登录
或

生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用

