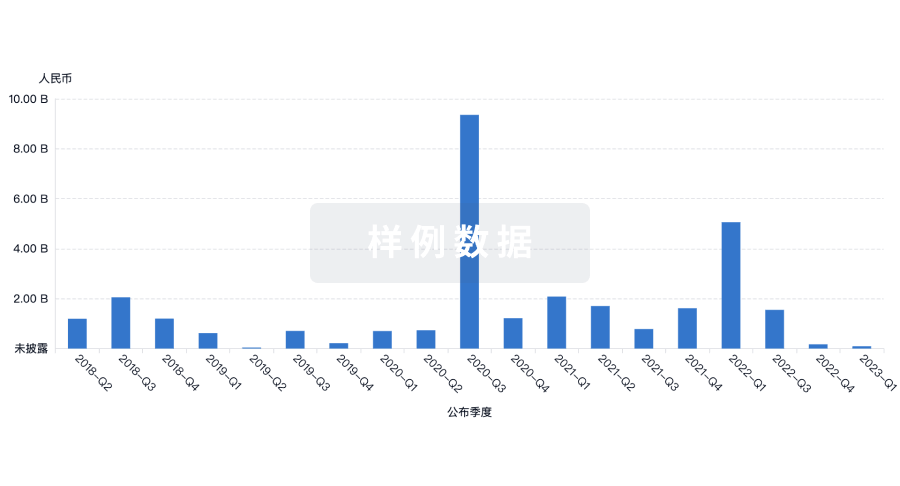
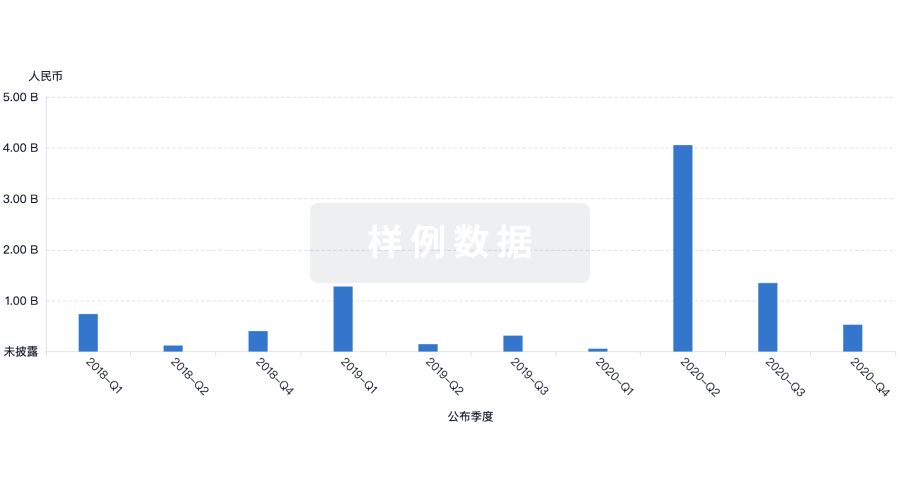
|
|
在研机构- |
|
在研适应症- |
|
最高研发阶段无进展 |
首次获批国家/地区- |
首次获批日期- |
A Randomized Open-Label Phase 2/3 Study of BT8009 as Monotherapy or in Combination in Participants With Locally Advanced or Metastatic Urothelial Cancer (Duravelo-2)
This is a global, multicenter, randomized, open-label study, with an adaptive design. The main objective of the study is to measure the efficacy and safety of BT8009 as monotherapy and in combination with pembrolizumab in participants with locally advanced or metastatic urothelial cancer (UC). The study includes a dose selection phase followed by an adaptive design continuation. The study is comprised of 2 cohorts. Cohort 1 will include participants who have not received any prior systemic therapy for locally advanced or metastatic UC and are eligible to receive platinum-based chemotherapy, whereas Cohort 2 will include participants who have received ≥ 1 prior systemic therapy for locally advanced or metastatic UC.
Phase I/II Study of the Safety, Pharmacokinetics, and Preliminary Clinical Activity of BT8009 in Patients With Nectin-4 Expressing Advanced Malignancies
This study is a Phase I/II, multicenter, first-in-human, open-label dose-escalation study of BT8009 given as a single agent and in combination with pembrolizumab in participants with advanced solid tumors associated with Nectin-4 expression or in participants with advanced solid tumor malignancies having renal insufficiency. The primary endpoints are: Dose limiting toxicities (Parts A-1 and A-2), Overall response rate per RECIST v1.1 (Part B), Safety and tolerability (Part C), and characterization of the pharmacokinetics (Part D).
Phase I/II Study of the Safety, Pharmacokinetics, and Preliminary Clinical Activity of BT5528 in Patients With Advanced Malignancies Associated With EphA2 Expression
This clinical trial is evaluating a drug called BT5528 alone and in combination with nivolumab in participants with advanced solid tumors historically known for expression of EphA2. The main goals of this study are to:
Find the recommended dose(s) of BT5528 that can be given safely to participants alone and in combination with nivolumab
Learn more about the side effects of BT5528
Learn about how effective BT5528 is for the treatment of ovarian cancer, urothelial/bladder cancer, lung cancer (NSCLC), triple-negative breast cancer, head and neck cancer (HNSCC), and gastric/upper gastrointestinal cancer.
Learn more about BT5528 therapy alone and in combination with nivolumab.
100 项与 Bicycle Tx Limited 相关的临床结果
0 项与 Bicycle Tx Limited 相关的专利(医药)
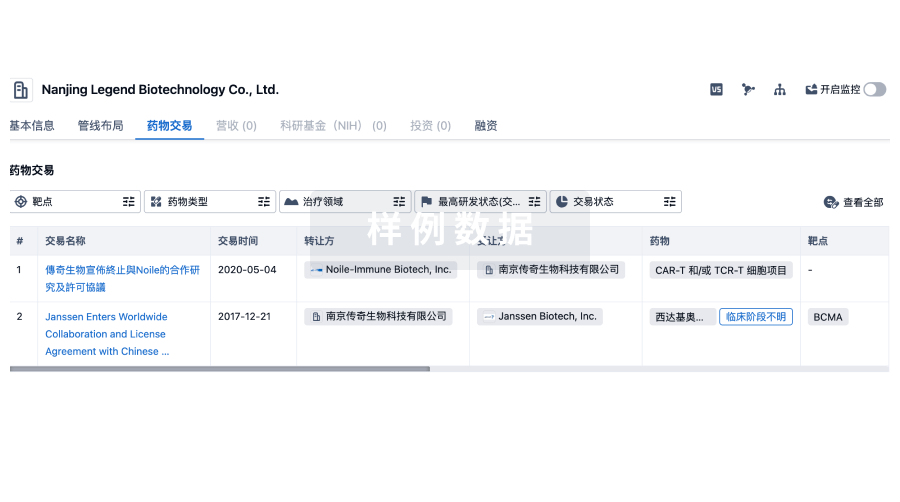
100 项与 Bicycle Tx Limited 相关的药物交易
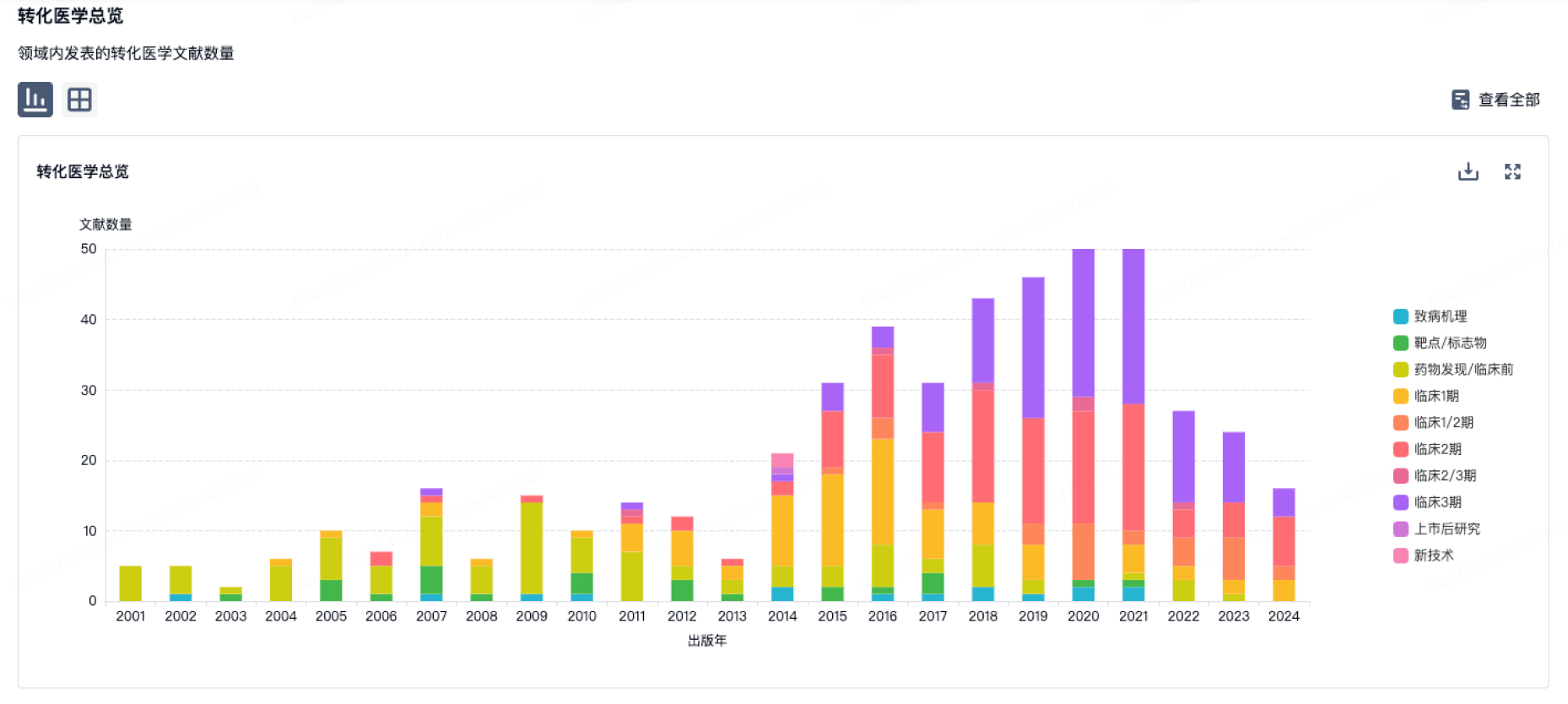
100 项与 Bicycle Tx Limited 相关的转化医学