预约演示
更新于:2026-03-04

Oulu University Hospital
更新于:2026-03-04
概览
关联
170
项与 Oulu University Hospital 相关的临床试验NCT07416292
ListenFuture-Listening for Their Future: The Complex Associations Between Hearing, Cognition, Spoken Language, Literacy and Psychosocial Wellbeing in Deaf and Hard-of-Hearing Adolescents
ListenFuture, a nation-wide multidisciplinary project, brings new knowledge on complex associations between listening, cognition, language, literacy, and psychosocial wellbeing in deaf and hard-of-hearing (DHH) adolescents. Earlier findings indicate that DHH adolescents remain at a risk to achieving age-appropriate skills. The associations among these skills can also be language-dependent. The investigators use behavioral methods and questionnaires. The investigators study speech perception in noise, listening, and listening-related fatigue to reveal how DHH adolescents cope in today's noisy environment. The investigators study cognitive skills and executive functions, spoken language and literacy skills to find out the current state-of-knowledge. The investigators study psychosocial wellbeing, school burnout and factors associated with them. The investigators will utilize supervised and interpretable machine learning to analyse the performance domains that could best predict the outcomes of DHH adolescents. The investigators expect our project to have broad societal impact for DHH adolescents, healthcare, school, and other stakeholders.
开始日期2026-03-01 |
申办/合作机构 |
NCT07331519
Integrating Smoking Cessation With Low-dose CT-screening for Lung Cancer - a Randomized Study
The study investigates effectiveness of different smoking cessation methods and low-dose CT based lung cancer screening
开始日期2026-02-02 |
申办/合作机构 |
NCT07314398
Prospective Cohort Study on Surgical Methods and Outcomes in Emergency Groin and Ventral Hernia Repairs in Finland
Emergency hernia repairs are associated with significantly higher morbidity and mortality compared with elective hernia surgery. In Finland, approximately 500 emergency groin hernia repairs and 600 ventral hernia repairs are performed annually, but treatment practices vary widely, and high-quality evidence is lacking.
This prospective multicenter cohort study will evaluate outcomes of emergency groin and ventral hernia repairs in Finland. About 600 patients will be recruited over two years and followed for five years postoperatively. The study will collect standardized data on patient demographics, comorbidities, surgical technique, intraoperative findings, postoperative course, and long-term follow-up. Quality of life will be assessed with RAND-36, AAS, and PROMIS questionnaires.
The primary endpoint is hernia recurrence within two years after surgery. Secondary outcomes include 30- and 90-day complications, infection rates, readmissions, recovery time, and quality of life at 1, 2, and 5 years.
The results will provide robust evidence to guide clinical practice, optimize surgical techniques, and refine urgency classification and surgeon competence requirements for emergency hernia repair.
This prospective multicenter cohort study will evaluate outcomes of emergency groin and ventral hernia repairs in Finland. About 600 patients will be recruited over two years and followed for five years postoperatively. The study will collect standardized data on patient demographics, comorbidities, surgical technique, intraoperative findings, postoperative course, and long-term follow-up. Quality of life will be assessed with RAND-36, AAS, and PROMIS questionnaires.
The primary endpoint is hernia recurrence within two years after surgery. Secondary outcomes include 30- and 90-day complications, infection rates, readmissions, recovery time, and quality of life at 1, 2, and 5 years.
The results will provide robust evidence to guide clinical practice, optimize surgical techniques, and refine urgency classification and surgeon competence requirements for emergency hernia repair.
开始日期2026-02-01 |
申办/合作机构 |
100 项与 Oulu University Hospital 相关的临床结果
登录后查看更多信息
0 项与 Oulu University Hospital 相关的专利(医药)
登录后查看更多信息
5,232
项与 Oulu University Hospital 相关的文献(医药)2026-12-15·SCANDINAVIAN JOURNAL OF PRIMARY HEALTH CARE
Adolescent anorexia nervosa treated in primary care with a family-based method – long-term outcomes
Article
作者: Ronkainen, Jukka ; Nordström, Tanja ; Tossavainen, Päivi ; Jääskeläinen, Erika ; Ronkainen, Jaana
BACKGROUND:
Anorexia nervosa is a potentially lethal psychiatric disorder characterised by restrictive eating and weight loss. Adolescent patients were treated as outpatients using a novel method which involved coaching the parents to take full responsibility for their child's nutrition. In this follow-up cohort study, we compared the long-term outcomes of patients treated in primary care using the family-based coaching method (FBcM-PC) with those treated in tertiary care using traditional treatment.
METHODS:
The cohort included all adolescent patients with a restrictive eating disorder treated initially in Oulu University Hospital and/or Oulu Primary Health Care centre between 2013 and 2019. Patients (n = 168) were contacted and asked to fill out a health questionnaire. Altogether 73 (43%) responded (30 from the FBcM-PC and 43 from the traditional treatment group). The outcome variables - depression, anxiety, eating disorder symptoms and need for medical appointments - were compared between the treatment groups.
RESULTS:
Upon follow-up after a mean of 6.6 years, 77% of the FBcM-PC and 54% of the traditional treatment groups did not need medical appointments (p = 0.052). No differences were found in self-reported health, depression or anxiety between the study groups. Concerns about weight and shape were reported by 40% of both groups. 87% of the FBcM-PC and 9% of the traditional treatment group received outpatient treatment only (p < 0.001).
CONCLUSION:
Adolescent anorexia nervosa patients treated mainly in primary care had at least as favourable long-term outcomes as those treated in tertiary care. The results of this study encourage further development of outpatient treatment methods in primary care.
2026-12-15·SCANDINAVIAN JOURNAL OF PRIMARY HEALTH CARE
Multimorbidity and health related quality of life in midlife – a longitudinal study from Northern Finland Birth Cohort 1966
Article
作者: Pesonen, Paula ; Auvinen, Juha ; Junttila, Oili ; Timonen, Markku
AIM:
Multimorbidity is a major public health concern. According to previous studies, multimorbidity has been shown to be associated with health-related quality of life (HRQoL). However, existing data concerning high income countries are mainly based on cross-sectional studies. Consequently, longitudinal investigations have been called for. There exist several definitions of multimorbidity in previous population studies, and usually definition is based on self-reported symptoms or diseases or included only few diagnoses. This study aimed to examine how the number of doctor-diagnosed chronic diseases influences HRQoL over time in a population-based setting.
METHODS:
At 31 and 46 years, participants (n = 3573) of the Northern Finland Birth Cohort 1966 answered 15-Dimensional HRQoL questionnaire (15D). Mean 15D-Scores (varies between 0 and 1) were counted according to the number of 43 chosen chronic diseases. These diseases were found and the definition of multimorbidity was measured by a method using both self-reported doctor-diagnosed and register-based chronic diseases.
RESULTS:
In both genders, the mean difference of 15D-Scores increased monotonously in line with the increasing number of new emerging diseases diagnosed for 15 years of follow-up from the age of 31-46 years (p < .05). Furthermore, the more diseases the patient had at the age of 31, the more mean 15D-Scores decreased when having more new emerging diseases at the age of 46.
CONCLUSIONS:
In general, HRQoL decreased during 15-year follow-up for everyone. Furthermore, decrease in the quality of life is significantly associated with multimorbidity in this longitudinal setting.
2026-04-01·PSYCHONEUROENDOCRINOLOGY
Maternal prenatal depression and anxiety and steroid hormones in amniotic fluid
Article
作者: Homer, Natalie Z M ; Phelan, Ellie ; Heinonen, Kati ; Lahti-Pulkkinen, Marius ; Billen, Margaux ; Girchenko, Polina ; Simpson, Joanna P ; Räikkönen, Katri ; Salo, Verna ; Reynolds, Rebecca M ; Seikku, Tiina ; Tukiainen, Taru
Maternal prenatal depression and anxiety (PDA) have been associated with increased risks of adverse birth and neurodevelopmental outcomes in children. While fetal exposure to too high or low levels of steroid hormones has been proposed as a potential biological mechanism underlying these effects, few studies have directly investigated this hypothesis using fetal tissue samples, and the existing studies have been limited to examining cortisol, cortisone or testosterone. We studied associations between PDA and steroid hormones in amniotic fluid by measuring a panel of 17 steroid hormones - including progestogens, mineralocorticoids, glucocorticoids, androgens and estrogens - and their substrate-to-product ratios in 173 women with singleton pregnancies undergoing amniocentesis during second trimester. The fetuses had no chromosomal abnormalities. We defined any PDA as meeting at least one of the following criteria: reported symptoms above clinical cut off (CES-D ≥ 20 or STAI state or trait anxiety ≥ 40) during pregnancy, lifetime diagnosis (ICD-10 codes F31-33, F41-43), and/or lifetime medication purchases (ATC-codes N06A, N05B). Elastic net regression identified two glucocorticoid metabolites, 20α-dihydrocortisol and 5β-tetrahydrocortisol, with lower amniotic fluid levels in fetuses of mothers with PDA compared to those without PDA (unadjusted mean difference -0.37 SD units, 95 % CI: [-0.68, -0.07]; and -0.40 SD units, 95 % CI: [-0.70, -0.10], respectively). The model with both steroids remained significant after adjusting for maternal age, body mass index, education, smoking during pregnancy, parity, gestational age at amniocentesis and fetal sex, and in sensitivity analyses excluding mothers with diabetes and hypertensive disorders (p-values < .05) and was not moderated by fetal sex (p-value > .40). PDA was not significantly associated with any substrate-to-product ratios of the steroids, used as proxies of steroid hormone metabolizing enzymes, after correction for multiple testing. This study provides support for the prenatal programming hypothesis of PDA influencing fetal environment through suboptimal levels of steroid hormones and highlights the need to expand to a comprehensive panel of steroid metabolism.
5
项与 Oulu University Hospital 相关的新闻(医药)2026-02-27
FRIDAY, Feb. 27, 2026 -- Parental worry identifies most cases of severe illness in children and adolescents but has low specificity, according to a study published online Feb. 17 in JAMA Network Open .
Hilla Pöyry, M.D., Ph.D., from Oulu University Hospital in Finland, and colleagues evaluated whether parents accurately identify severe illnesses in their children before emergency department evaluation using a symptom questionnaire. The analysis included data from 2,375 children and adolescents visiting a pediatric emergency department from May 2024 to May 2025.
The researchers found that moderate-to-high parental worry showed the highest sensitivity (91.0 percent) but the lowest specificity (17.5 percent). There was modest diagnostic accuracy for other specific pediatric questions, but with limited additional value. The machine learning model (area under the receiver operating characteristic curve, 0.71) identified the strongest predictors of hospital admission as parental worry (feature importance score, 0.047), parent assessments of child or adolescent's general condition (feature importance score, 0.046), and need for treatment (feature importance score, 0.141).
"While parental concern may serve as an initial screening indicator, it should be complemented by clinical evaluation and objective measures to avoid unnecessary escalation of care," the authors write.
Abstract/Full Text

临床结果
2026-02-20
FRIDAY, Feb. 20, 2026 — Doctors would do well to heed any gut feelings parents might have about their child’s health, a new study says.
Nine times out of 10, children were seriously ill if their parents had a clear or strong concern about their health, researchers reported Feb. 17 in JAMA Network Open .
In fact, asking whether a parent is worried works better to identify seriously sick kids than comprehensive health questions focused on symptoms, researchers found.
“Parental concern is an important warning sign,” said lead researcher Dr. Hilla Pöyry , a pediatric specialist at the University of Oulu in Finland.
“If a parent is worried about the condition of their suddenly ill child, the child must have the opportunity to be assessed by a doctor,” Pöyry said in a news release. “A worried parent should not be left alone to make a remote assessment of their child’s condition.”
For the study, researchers tracked nearly 2,400 children and teens treated at the Oulu University Hospital’s ER. Parents completed an extensive 36-item questionnaire as part of their child’s treatment.
About 1 in 4 of the children had a severe illness requiring intensive care, surgery or a prolonged hospital stay, the study said.
The point was to see if the digital questionnaire could be a good tool for parents to use to assess the health of a sick child at home before heading to the ER.
However, the questionnaire was not accurate enough to replace an ER visit, especially if a parent was already concerned about their child’s health.
“Our results show that such tools require careful validation, and they do not yet replace the assessment of a health care professional,” Pöyry said.
Researchers found that items in the questionnaire related to parental worry were the best way to detect kids at greatest risk for serious illness.
One question alone accurately identified 91% of kids who turned out to be very sick: “As a parent, how worried are you about your child's health at the moment?”
“Although a parent may not always be able to describe the child’s symptoms in detail or accurately, they recognize a serious illness very well when asked a simple concern-based question,” Pöyry said.
However, these “gut feeling” questions weren’t very good at ruling out cases where a child wasn’t sick, the study found.
The best-performing question — “Do you feel your child is particularly ill or something is seriously wrong?” — identified healthy children only about 73% of the time.
The parental worry question identified healthy kids just 18% of the time, despite how well it worked at detecting children who were actually sick.
“While parental concern is an important red flag, it should be interpreted alongside clinical assessment to avoid unnecessary escalation of care,” the research team concluded in its paper.
Sources
University of Oulu, news release, Feb. 18, 2026
JAMA Network Open, Feb. 17, 2026

临床研究临床结果
2026-01-20
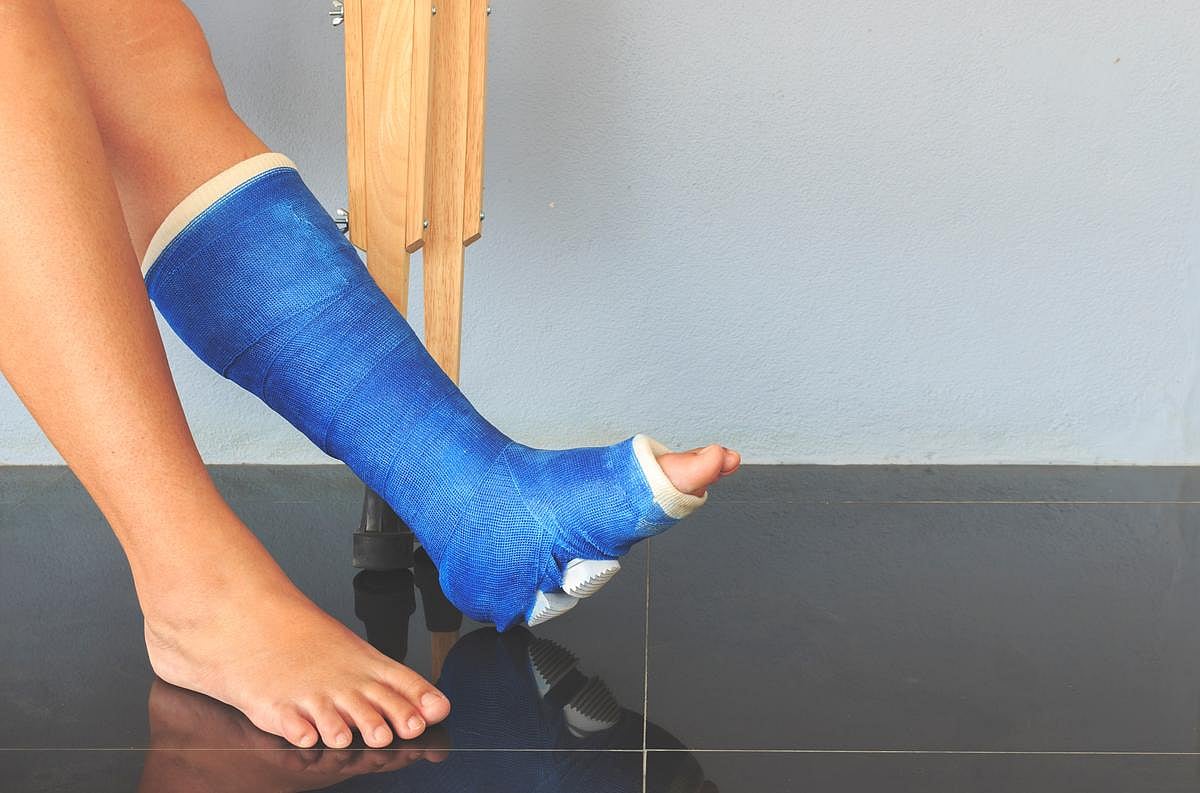
TUESDAY, Jan. 20, 2026 -- For treatment of unimalleolar Weber B ankle fractures with a congruent mortise on initial radiography but deemed unstable, cast immobilization is noninferior to surgery , according to a study published online Jan. 14 in The BMJ .
Tero Kortekangas, M.D., Ph.D., from Oulu University Hospital in Finland, and colleagues conducted a randomized, noninferiority trial to compare cast immobilization to surgery using open reduction and internal plate fixation for unimalleolar Weber B ankle fractures with a congruent but unstable ankle mortise. A total of 840 skeletally mature patients age 16 years and older with an isolated Weber B fibula fracture were assessed for fracture instability. After exclusion of those with stable fracture, mortise incongruency, or fracture dislocation, 126 with a congruent but unstable ankle mortise were randomly assigned to receive conventional cast immobilization for six weeks or surgical treatment with open reduction and internal plate fixation followed by cast immobilization for six weeks (62 and 64 patients, respectively).
In the primary intention-to-treat analysis of 121 participants with two-year follow-up, the researchers found that the mean Olerud-Molander Ankle Scores were 89 and 87 in the cast immobilization group and surgery group, respectively (mean difference, 1.3 points; 95 percent confidence interval, −4.8 to 7.3). There were no significant differences observed in any of the secondary outcomes (i.e., ankle function, pain, health-related quality of life, ankle range of motion, and radiographic outcome). Radiologic evidence of nonunion was seen for one participant in each group.
"This lends further support to the evolving concept that the treatment of ankle fractures should focus on obtaining and maintaining a congruent ankle mortise until fracture union, using the most conservative means possible," the authors write.
Abstract/Full Text
Editorial
临床结果
100 项与 Oulu University Hospital 相关的药物交易
登录后查看更多信息
100 项与 Oulu University Hospital 相关的转化医学
登录后查看更多信息
组织架构
使用我们的机构树数据加速您的研究。
登录
或

管线布局
2026年04月05日管线快照
无数据报导
登录后保持更新
药物交易
使用我们的药物交易数据加速您的研究。
登录
或

转化医学
使用我们的转化医学数据加速您的研究。
登录
或

营收
使用 Synapse 探索超过 36 万个组织的财务状况。
登录
或

科研基金(NIH)
访问超过 200 万项资助和基金信息,以提升您的研究之旅。
登录
或

投资
深入了解从初创企业到成熟企业的最新公司投资动态。
登录
或

融资
发掘融资趋势以验证和推进您的投资机会。
登录
或

生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用
