A Randomized, Open-label, Multicenter Phase 3 Study of SKB264 Versus Treatment of Physician's Choice (TPC) in Patients With Unresectable Locally Advanced, Recurrent or Metastatic HR+/HER2- Breast Cancer Who Had Failed at Least One Line of Chemotherapy
The purpose of this study is to evaluate the efficacy and safety of SKB264 in patients with unresectable locally advanced, recurrent, or metastatic HR+/HER2- breast cancer.
A Randomized, Open-Label, Multicenter Phase 3 Study to Evaluate SKB264 Monotherapy Versus Pemetrexed in Combination With Platinum in Patients With Locally Advanced or Metastatic Non-squamous Non-small Cell Lung Cancer With EGFR Mutation Who Have Failed to Epidermal Growth Factor Receptor Tyrosine Kinase Inhibitor (EGFR-TKI) Therapy
This is a randomized, open-label, multicenter Phase 3 clinical study to evaluate SKB264 monotherapy versus pemetrexed in combination with platinum in subjects with locally advanced or metastatic non-squamous NSCLC with EGFR mutation who have failed to EGFR-TKI therapy.
A Multicenter, Open-label, Phase 2 Study to Evaluate the Efficacy and Safety of SKB264 Monotherapy in Selected Subjects With Advanced Solid Tumors
The purpose of this study is to assess the safety and efficacy of SKB264 monotherapy in subjects with selected advanced solid tumors.The study is divided into two parts: the Part Ⅰ consists of 5 cohorts, and the Part Ⅱ for expansion. Eligible subjects will receive SKB264 monotherapy, until there is no longer clinical benefit, intolerable toxicity, discontinuation of study treatment required by the subject, or other protocol-specified treatment discontinuation criteria, whichever occurs first.
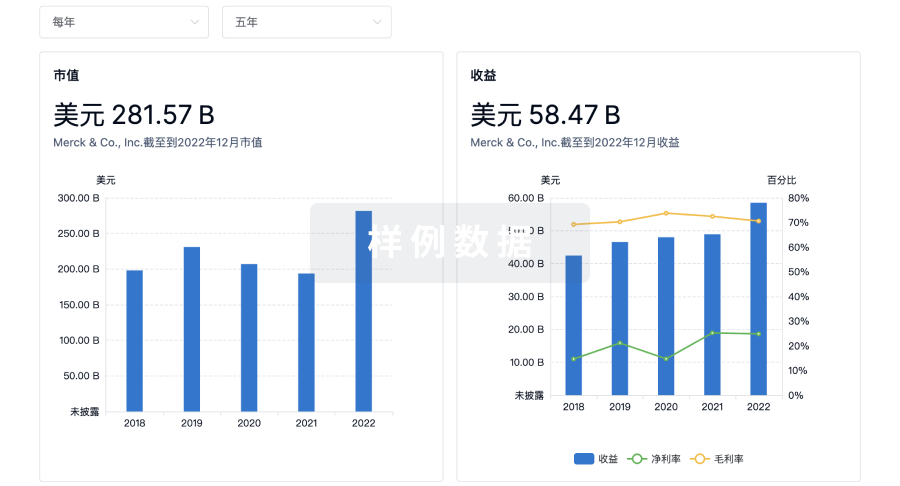
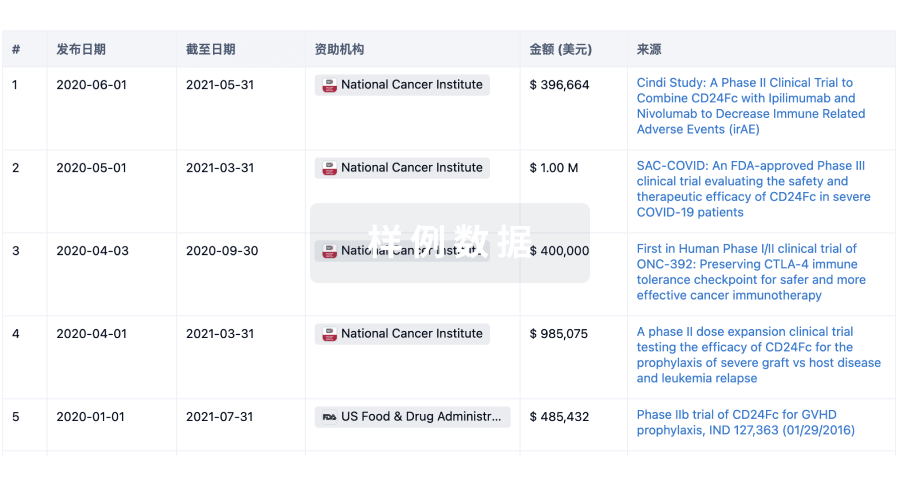
100 项与 Sichuan Kelun Pharmaceutical Research Institute Co., Ltd. 相关的临床结果
0 项与 Sichuan Kelun Pharmaceutical Research Institute Co., Ltd. 相关的专利(医药)
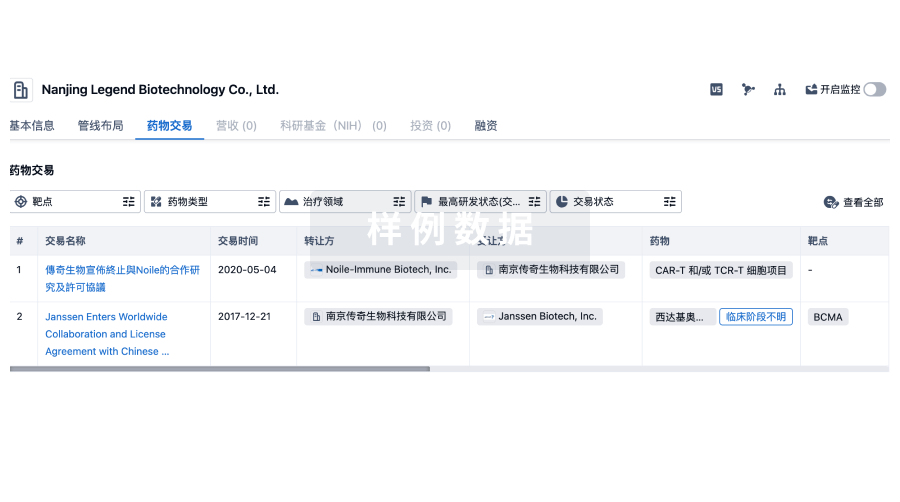
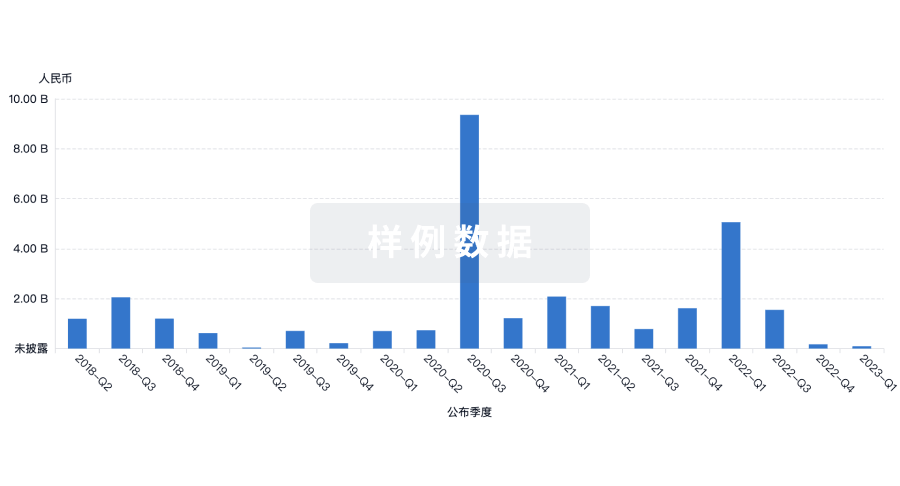
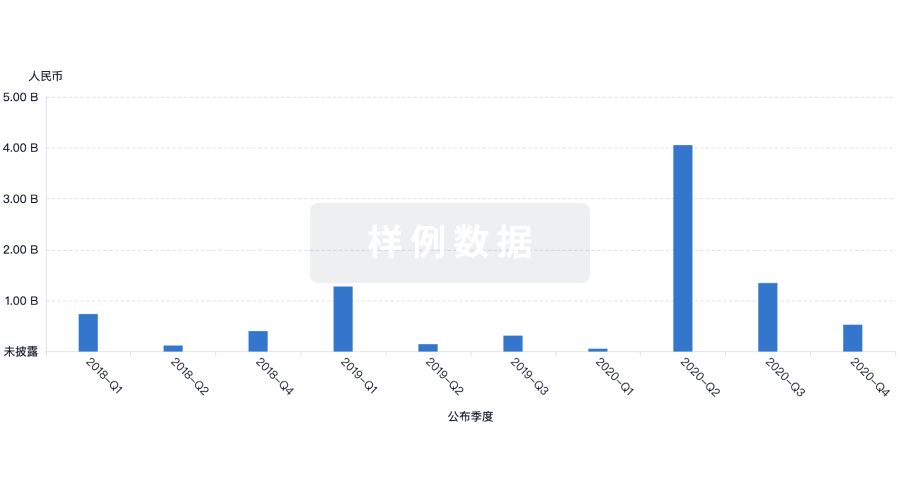
100 项与 Sichuan Kelun Pharmaceutical Research Institute Co., Ltd. 相关的药物交易
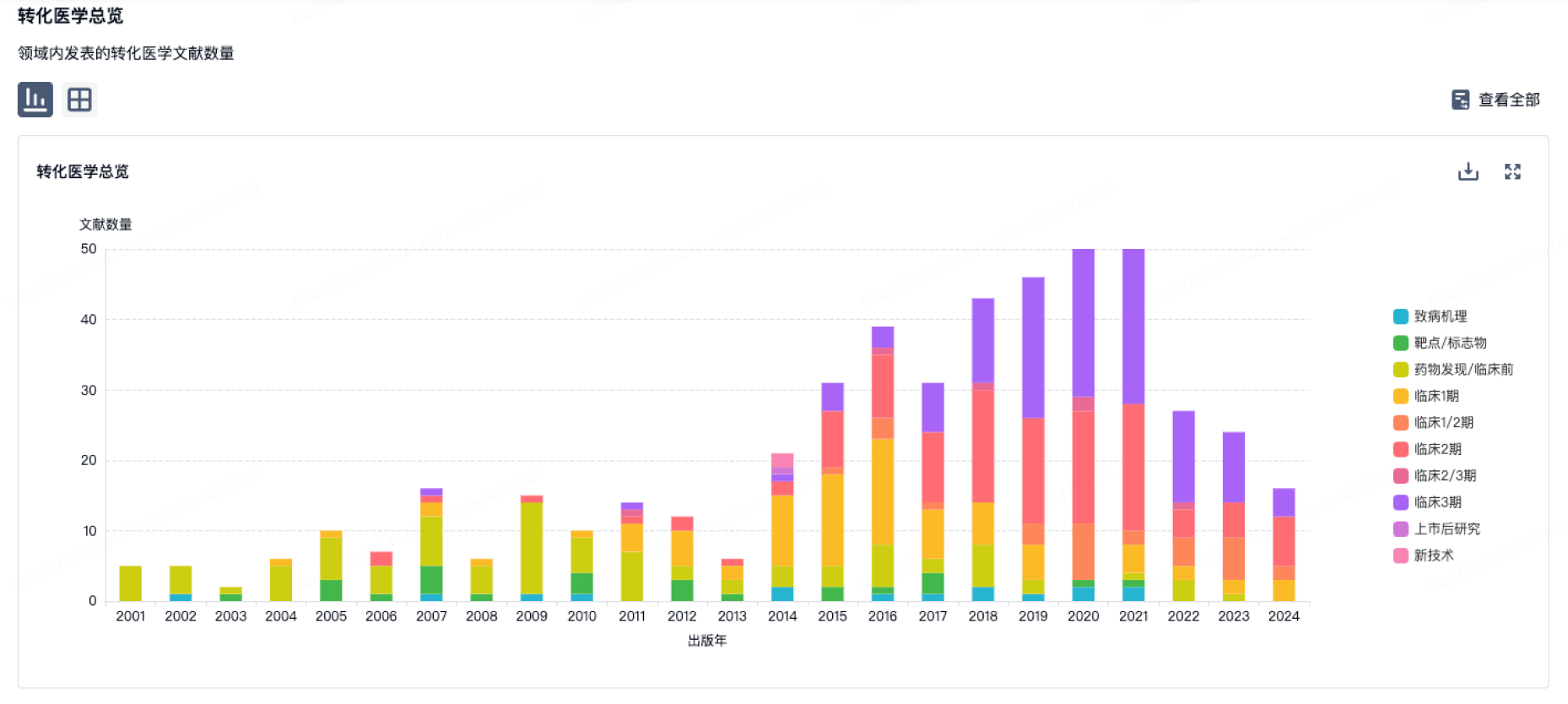
100 项与 Sichuan Kelun Pharmaceutical Research Institute Co., Ltd. 相关的转化医学