更新于:2023-04-21
Jiangxi Kelun Medical Equipment Manufacturing Co., Ltd.
更新于:2023-04-21
概览
关联
100 项与 江西科伦医疗器械制造有限公司 相关的临床结果
登录后查看更多信息
0 项与 江西科伦医疗器械制造有限公司 相关的专利(医药)
登录后查看更多信息
1
项与 江西科伦医疗器械制造有限公司 相关的文献(医药)2013-12-25·Hunan Zhongyiyao Daxue Xuebao
Evaluation of paper plastic packaging to reduce ethylene oxide residue
作者: Lan, Hai ; Qiao, Yan ; Li, Zhi-gang
According to the requirement of packaging for terminally sterilized medical devices in ISO 11607-2006, EN 868-2009 and GB/T 19633-2005, taking infusion sets for example, the applicability of paper plastic packaging was verified. The paper plastic packaging meets the quality requirement of the relevant standards and provides reliable sterile barrier. The paper plastic packaging also highly fits the requirement that "sterilization agents can reach the sterilized surface of devices and are easy to remove" in the standards The desorption time of ethylene oxide from the product is significantly less than that of the plastic packaging, and the zero residue of ethylene oxide in devices becomes possible.
380
项与 江西科伦医疗器械制造有限公司 相关的新闻(医药)2023-01-05
·赛柏蓝
来源 | 赛柏蓝作者 | 颜色 四喜推迟一个月后,“2022国家医保谈判”正式启动。 2023年1月5日,北京西城区全国人大会议中心所在街道两旁,挤满了商务车——大多是药企用车,有的前来谈判,有的则是率先打探情况。 和2020年一样,地点依然是药企熟悉的全国人大会议中心,按照2020年到场的企业代表说的,选址讲究:人大会议中心——人民的心声。 北京最低零下6摄氏度的温度,对上了今日小寒的节气。从现场来看,上午不如下午“热”。下午13:43分,大家都挤着进去,保安不得不说:“这个门是电动的,等下都挤坏了”。药企急迫的“入场”心态可见一斑。 据悉,今年大概率和去年一样,进去的企业都按要求签署“保密协议”。不过,这依旧挡不住彼此之间的相互谈论,场外的药企人士相对警惕,面前的不是自己人,往往有意避让。 (赛柏蓝摄) 场外在焦急等待,场内在“斗智斗勇”。01“红色”服饰依然是主调企业砍价也图“吉利”谈判第一天上午,呼吸类药物、抗病毒药物、麻醉镇定药物以及消化类药物等进行了谈判。 关于谈判专家组,之前就有人讨论,按照以往谈判的经验,浙江的专家最温柔,福建的专家谈判特别厉害。赛柏蓝在现场了解到,第一天,包括恒瑞、华纳制药、科伦、凯西、默沙东在内的企业出现在了谈判现场。上午,有身穿红色内搭,佩戴LV围巾的三位女性企业代表进场,她们还特地自带了计算器。有知情人士透露,外企之前有谈判经验,其实谈一次就过了,毕竟此前就知道同类产品价格,也经过了多轮测算,一般比较节省时间。也有企业老板亲自挂帅,直接入场谈判。历次医保谈判的名场面,这次也上演了。一位曾参与过医保谈判的企业人士称,一般企业会主动选择吉利的数字作为最终的报价,也体现了企业的“诚意”。02“价格还可以”、“挺好”医保谈判第一天,下午和上午相比,“热度”升了许多。根据赛柏蓝现场统计,围在现场门口的不低于60人;下午1:36分企业谈判代表陆陆续续进场,截至16:11分,已出来11家企业。有“回避型”企业,面对媒体的提问,只是挥挥手,只字不提,也有企业摘掉口罩,难掩喜悦之情,回复“开心”。 据新京报消息,一家湖南企业谈判代表谈判结束后,也开心表示,此次谈判成功,“价格还可以”,此次谈判的品种为公司旗下二类创新药物。下午南京海纳医药出来后,回复“一般、还可以,挺好”,根据国家医保局日前公布的通过初审名单药品,南京海纳医药估计此次谈判的为奥美拉唑碳酸氢钠干混悬剂。 但也有企业表面看着挺开心,嘴上却说“不好说”。有的企业则面色凝重,匆匆离开。03新冠口服药、肿瘤与罕见病药物后两日谈判医保谈判首日,气氛总体没有那么紧张,但也有人分析,后两日的谈判预计相对激烈。从知情人士获悉,谈判首日并未直接拿新冠口服药“开刀”。无论是辉瑞的新冠口服药paxlovid;还是真实生物的阿兹夫定、中成药清肺排毒颗粒后续都将面临一波砍价。 日前,辉瑞的paxlovid炒到上万一盒,阿兹夫定更是供不应求,山东一位县级民营医院医生公开发布“医院收阿兹夫定”的消息,并向赛柏蓝表示,“当地经济条件很差,基础病人很多,都是平价出售,一开始找当地医药公司调配了20瓶,但实际仍有50~100瓶的缺口”。也有接近辉瑞的代表表示,一度,辉瑞内部员工都很难搞到paxlovid。不过值得注意的是,根据获批时间,阿兹夫定的新冠适应症大概率不在谈判范围内。除了供应短缺,paxlovid的挂网价格在某些省份也有变动,从之前的2300元降至1890元/盒,此次更是面临医保谈判的砍价。值得注意的是,辉瑞的新冠口服药paxlovid此前只是临时被纳入医保,随着新冠的降级管理,新冠医保报销政策预计也会随之变化,此番能否转正还要看辉瑞的“诚意”。一位业内人士曾向赛柏蓝表示,“阿兹夫定价格300多元,辉瑞的低于700元比较合理,化学药物,成熟化工艺之后,成本很低”。 通过“国谈”初审名单的342个药品中,目录内品种有145个;目录外新进品种有198个,其中,有19个罕见病药品,被称为“天价抗癌药”的CAR-T瑞基奥仑赛注射液也首次出现在初审名单中。根据公开消息,CAR-T瑞基奥仑赛注射液价格高达129万元/针,在国家医保局针对“天价药”的“50万不谈,30万不进”的不成文规定下,其最终是否为走过场,还要看企业的决心。 此次医保谈判的亮点,除了新冠治疗类药品能否正常“转正”外,还有:建议续约品种的情况、罕见病及CAR-T的几个品种是否会进医保、PD-1的降幅、部分企业是否会遭遇“灵魂砍价”等情况,赛柏蓝也将持续跟进。就在医保谈判首日,有企业在社群内发布2022年国谈品种的招聘信息,看来企业端已经在做市场渠道等相关方面的铺垫工作。除了药企盯着医保目录,代理商们也在看,据知情人士透露,江苏一家知名药企的几个产品,虽然过了初审名单,但都最终无缘医保谈判现场,该知情人士向赛柏蓝透露,当时,初审名单出来后,很多代理商打电话,询问产品的事情,想争取代理权。显然,面对医保谈判,各方都在努力。(注:谈判结果一切以医保局最终公布结果为准)♡左下角分享,右下角赞、在看,为赛柏蓝充电
2023-01-05
2023年1月5日,早晨7点多,西皇城根北街,全国人大会议中心门口,零下6摄氏度。迟来的2022年国家医保谈判,终于缓缓拉开序幕。寒风中,一位站在路边的药企谈判代表,每两分钟擦一次自己的眼镜,雾气总不自觉地从口罩窜上镜片。无论是天气的寒冷,还是过去一年来,医药行业经历的“寒冬”,无不让人感觉到寒意浓烈。也恰恰是在这个时候,医保局的一举一动、砍价力度,更是引发着整个行业的瞩目。从首日谈判传递出的消息来看,今年似乎有些南风徐来、春意渐暖的意味。向来敏感的二级市场,似乎也捕捉到了这股暖风。截至1月5日下午收盘,有医保谈判产品的医药股普涨,包括恒瑞、科伦、复星、罗欣、百济、再鼎在内的多家药企涨幅都在4%左右。01"整体不比往年焦灼"时间来到8点钟左右,这时,全国人大会议中心门前已经聚集了很多人,媒体、企业、投资人,络绎不绝地赶来。车里等待的药企代表们,也陆续下车,径直走进会场。面色相对从容、淡定。一位在场外等候的药企管理层直言,对于药价谈判,大家渐渐习以为常,已经没有之前那么多的忐忑和紧张了。有参与谈判的药企和现场知情人士透露,上午的国谈按照药物类别大致分为5组,包括抗病毒药物、呼吸类药物、麻醉类药物、消化类药物和营养类药物等。E药经理人观察到,上午进场的药企谈判代表,大致包括恒瑞、百济、百特、科伦、大冢等10余家药企。抗感染领域或在1月5日上午率先“开跑”。科伦、苏州大冢等企业均参与其中。初审名单显示,科伦在目录外共有4款产品,包括舒更葡糖钠注射液、碳酸氢钠林格注射液、中⻓链脂肪乳/氨基酸(16)/葡萄糖(36%)注 射液、注射⽤头孢他啶/5%葡萄糖注射液。这些药品,涉及了神经肌⾁阻滞、细胞外液补充剂、肠外营养、抗感染。苏州大冢制药则携注射⽤头孢美唑钠/氯化钠注射液入场。9点56分,上午谈判的第一家企业代表走出大门,表情严肃。而后,数家企业陆续离场。从谈判人士出来后的微表情判断,有喜有忧。一位来自湖南的2类新药企业代表表示“谈判成功、价格还可以”。E药经理人根据初审名单信息推测大概率为“左奥硝唑片”的非独家竞价产品。据现场消息,国内药企和国外药企的谈判风格大不相同。外企决策速度雷厉风行,国内药企态度则更为谨慎。但总体来看,谈判过程没有往年那么焦灼,也没有出现“灵魂砍价”的意外场面。根据E药经理人了解,在科伦、大冢的抗感染产品谈判中,也没有出现“激烈交锋”的场面,反而是心态比较平和。“这是因为医保局在前期做了大量的与企业之间的沟通工作,包括在11月、12月进行了预谈判。”有企业表示,因此从现场情况可以看出,参与谈判的企业大多都表现出了相对心里“有底”的平和状态。02下午氛围明显更加紧张下午1点,不同于早上的寒冷,天气变得阳光和煦。很多药企代表也早早下车,在国谈会场门外等待、攀谈。场外观察来看,下午参与谈判的女性代表明显多于上午。半小时后,大门外人群开始拥挤,正大天晴、再鼎医药、先声药业、安斯泰来、南京海纳、北京锐业、广州百特等企业谈判代表开始陆续进场。据场外观察,药企的谈判规格一如既往的高,罗欣药业本次谈判小组由执行副总裁、首席运营官董莉君亲自带队。安斯泰来则由副总经理、大中华区政府事务及商务本部总监张鸿带队。下午的氛围也明显紧张了一些。有人猜测“非独家竞价谈判可能在陆续展开。”很多谈判代表的手上的文件袋里,多了计算器、草稿纸等计算工具。一位进场者,手里甚至还拿着一小瓶“速效救心丸”。大概一个小时后,下午第一家药企谈判代表走出会场,一脸严肃,走向马路对面,三人三车,分头离开。从“罗欣三人组”离开谈判现场的表情来看,本次谈判结果应该相对理想。罗欣此次参与谈判的有三款产品,替戈拉生片、注射⽤福沙匹坦双葡甲胺和盐酸氨溴索喷雾剂,涉及食管炎、呼吸道疾病与恶心呕吐治疗领域。安斯泰来则涉及了目录内及目录外共有2款产品,恩扎卢胺与他克莫司颗粒。其中恩扎卢胺此次为新增适应证谈判,为有高危转移风险的非转移性去势抵抗性前列腺癌。该药在2020年上市当年便成功通过医保谈判进入报销目录,造福了全国患者也为产品赢得了竞争窗口期。恩扎卢胺进入医保后增速明显,据2021年安斯泰来财报显示,在中国的业绩较纳入医保目录前增长了62%。下午的15:30分,有企业谈判代表走出人大会议中心后,突然被医保局工作人员叫回了谈判现场。据了解,被叫回的是参天制药的谈判代表。今年的医保谈判中,参天将为2022年5月获批的眼科炎症治疗新药环孢素滴眼液(III),争取医保目录的入场券。据悉,该产品曾先后被NMPA纳入临床急需境外新药名单和优先审评名单,主要适用于治疗4岁以上儿童及青少年严重性春季角膜结膜炎。随后,根据E药经理人了解到的消息,医保局是与企业在沟通,不允许对外讨论此次谈判的细节,因此代表很快又返回车中。再鼎医药谈判代表身着一袭橙红色大衣,格外显眼。据悉此次再鼎有两款产品等待进医保,一款为注射⽤甲苯磺酸奥⻢环素,另一款为瑞派替尼⽚,分别为抗感染与胃肠间质瘤两类。总体来说,一整天下来,几乎每一家药企的谈判代表出来后,都三缄其口。甚至连表情都做了统一“管理”一样严肃。对于绝大多数人,连透过表情测谈判结果的机会也没给。据了解,和往年一样,每一位入场代表都签了严格的保密协议,因而媒体获得的信息有限。在与企业的接触中,E药经理人得知,此次有企业派出了负责科研的同事参与谈判,表示希望能够将更多科学性的东西展示给谈判专家组。03备受关注的Paxlovid与PD-1本次医保谈判中,备受瞩目的是新冠口服药首次“开谈”。辉瑞的新冠口服药Paxlovid是唯一参与谈判的产品,受到了市场的高度关注。其他两款新药,真实生物的阿兹夫定以及默沙东的莫诺拉韦,皆因获批时间较晚,无缘本次医保谈判。据E药经理人现场了解,今天Paxlovid并未参与谈判,具体时间可能会在7日进行。除此之外,包括高价罕见病产品、CAR-T、国产首个双抗、PD-1等产品在内的产品有待开谈。有趣的是,在今天热闹的人群中,还有一大部分是专门来“踩点”的。其中包括了复星医药、阿斯利康、默沙东、诺和诺德、第一三共等企业。“提前来确定一下地点,感受下气氛。”一位“踩点”的药企人士笑着说。傍晚5时许,北京温度已降至1°,夜色悄然袭来,暖黄路灯次第亮起。在全国人大会议中心西一门门口,人群逐渐散去,已不如白日里那般热闹,只剩稀疏三两行人。此时才结束今天谈判的人们,已经不再需要穿过拥挤的人潮,迎接众人殷切的目光。五点半左右,大门突然再次拉开,一行三人手持文件夹,步履匆匆,其中带头走在前列的个高者,上车前长吁了一口气,车辆消失在黑夜里。而在相反方向,婆娑昏黄的光影下,刚结束今日医保谈判的四人正商量着晚餐的去处。夜幕降临,第一天的谈判尘埃落定。在此时大考后为数不多的静谧时分,他们享受着的,似乎只是谈判后片刻安宁与放松。“里面还有一波人在谈。”大考第一天结束,一切都随着夜色渐深而安静了下来。
核酸药物
2023-01-05
·米内网
精彩内容1月4日,石家庄四药|四川科伦药业联合提交了脂肪乳(10%)/氨基酸(15)/葡萄糖(20%)注射液的4类仿制上市申请。该产品近几年在中国公立医疗机构终端的销售额高速增长,2021年涨至2亿元以上,科伦在2018年拿下首仿后强势抢占市场,2022上半年的市场份额已超过八成。图1:石家庄四药|四川科伦药业最新申报的产品情况来源:CDE官网据悉,脂肪乳(10%)/氨基酸(15)/葡萄糖(20%)注射液是全球首个工业化“全合一”三腔袋产品,用于2岁以上的儿童和成人患者不能或功能不全或被禁忌经口/肠道摄取营养的营养支持。利用工业化三腔袋实现的“全合一”营养液输注方式是目前肠外营养治疗的发展趋势和公认的最佳形式,与序贯输注、多瓶串输相比,三腔袋产品配方固定,即开即混即用,可缩短配制时间、降低错配风险,减少代谢并发症及感染机率,提高用药安全性和医护效率。图2:脂肪乳(10%)/氨基酸(15)/葡萄糖(20%)注射液的批文情况来源:米内网中国上市药品(MID)数据库米内网数据显示,百特的原研药在2013年获批进入国内市场,科伦药业在2018年拿下国内首仿(现已通过一致性评价),本次石家庄四药联合四川科伦药业申报仿制上市,有望拿下国产第二家。图3:脂肪乳(10%)/氨基酸(15)/葡萄糖(20%)注射液的销售情况(单位:万元)来源:米内网中国公立医疗机构药品终端竞争格局科伦药业的脂肪乳(10%)/氨基酸(15)/葡萄糖(20%)注射液自上市以来,在中国城市公立医院、县级公立医院、城市社区中心及乡镇卫生院(简称中国公立医疗机构)终端的销售额快速飙涨,2019-2020年的增长率均达四位数,2021年突破1.6亿元,2022上半年市场份额首破80%。而百特的销售额则持续下滑,2022上半年仅剩2000万元水平。科伦药业近几年在三腔袋产品领域硕果累累,已获批上市的产品包括脂肪乳氨基酸(17)葡萄糖(11%)注射液、脂肪乳氨基酸(17)葡萄糖(19%)注射液、中长链脂肪乳/氨基酸(16)/葡萄糖(16%)注射液、脂肪乳(10%)/氨基酸(15)/葡萄糖(20%)注射液、中长链脂肪乳/氨基酸(16)/葡萄糖(30%)注射液、中长链脂肪乳/氨基酸(16)/葡萄糖(36%)注射液等,国产品牌上市后快速抢占市场。石四药集团是科伦药业重要的参股公司,据科伦药业2022年半年报显示,科伦药业(直接+间接)持有石四药集团超过20%的股权。目前科伦药业的三腔袋产品线已成熟,后续将有更多新品获批上市,石四药在科伦的助力下,即将迎来首款三腔袋产品,值得期待。数据来源:米内网数据库、公司年报注:米内网《中国公立医疗机构药品终端竞争格局》,统计范围是:中国城市公立医院、县级公立医院、城市社区中心以及乡镇卫生院,不含民营医院、私人诊所、村卫生室;上述销售额以产品在终端的平均零售价计算。数据统计如有疏漏,欢迎指正!本文为原创稿件,转载请注明来源和作者,否则将追究侵权责任。投稿及报料请发邮件到872470254@qq.com稿件要求详询米内微信首页菜单栏商务及内容合作可联系QQ:412539092【分享、点赞、在看】点一点不失联哦
上市批准一致性评价
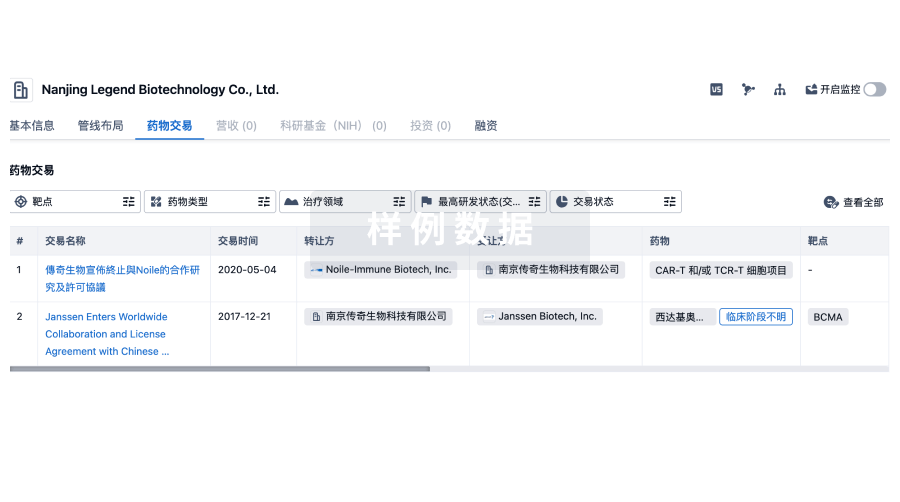
100 项与 江西科伦医疗器械制造有限公司 相关的药物交易
登录后查看更多信息
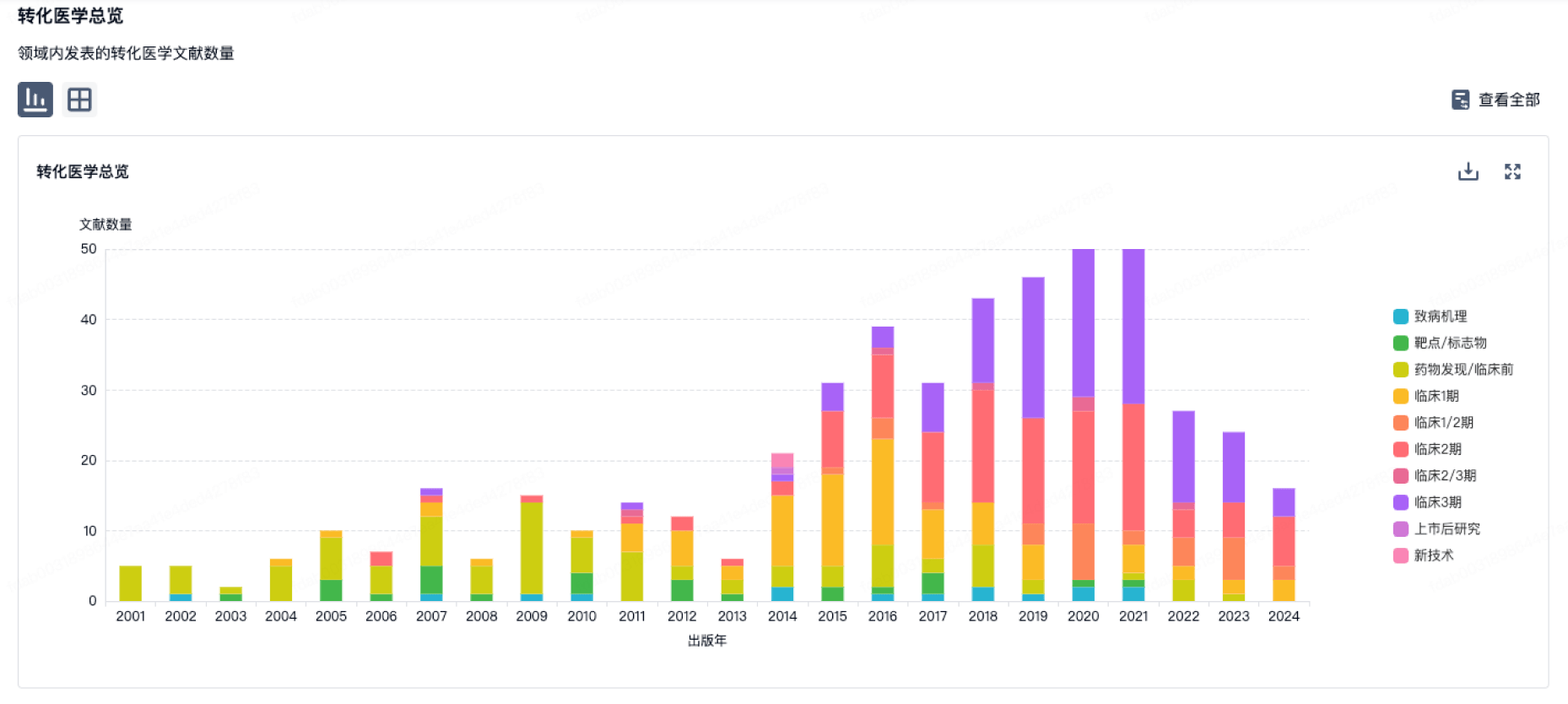
100 项与 江西科伦医疗器械制造有限公司 相关的转化医学
登录后查看更多信息
管线布局
2024年04月28日管线快照
无数据报导
登录后保持更新
药物交易
使用我们的药物交易数据加速您的研究。
登录
或
转化医学
使用我们的转化医学数据加速您的研究。
登录
或
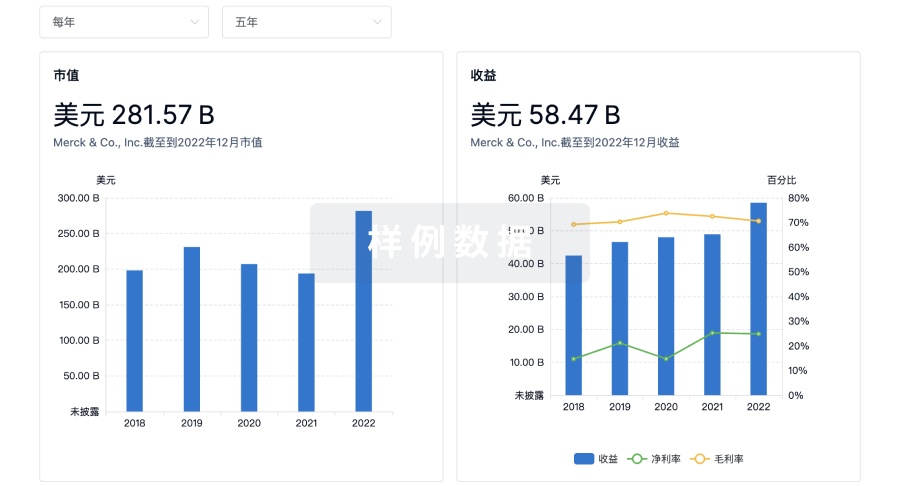
营收
使用 Synapse 探索超过 36 万个组织的财务状况。
登录
或
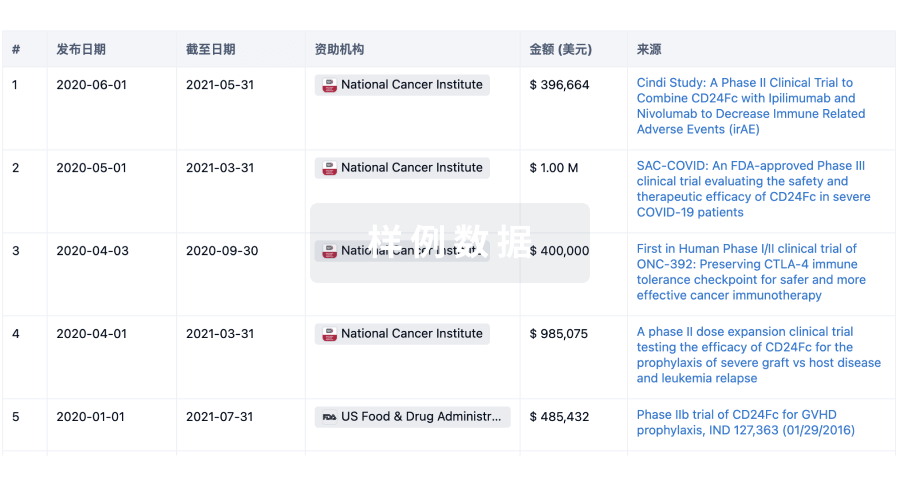
科研基金(NIH)
访问超过 200 万项资助和基金信息,以提升您的研究之旅。
登录
或
投资
深入了解从初创企业到成熟企业的最新公司投资动态。
登录
或
融资
发掘融资趋势以验证和推进您的投资机会。
登录
或
标准版
¥16800
元/账号/年
新药情报库 | 省钱又好用!
立即使用
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用