预约演示
更新于:2025-05-07
Mount Sinai Hospital (Canada)
更新于:2025-05-07
概览
关联
1
项与 Mount Sinai Hospital (Canada) 相关的药物作用机制 NK2R调节剂 [+1] |
在研机构- |
在研适应症- |
非在研适应症 |
最高研发阶段无进展 |
首次获批国家/地区- |
首次获批日期- |
219
项与 Mount Sinai Hospital (Canada) 相关的临床试验NCT06907732
Fetoscopic Robotic Opens Spina Bifida Treatment - a Pilot Feasibility Study
Fetal spina bifida is a common birth defect that results in hydrocephalus, motor-, bowel-, bladder- and sexual dysfunction in the child. The condition is progressive in utero. Fetal surgery between 22-26 weeks gestation has been shown to stop the gradual fetal deterioration observed in this disease and improve infant outcomes. Children with spina bifida who have undergone fetal surgery have a lower need for hydrocephalus treatment (80%->40%) and twice the chance to walk independently by the age of 3 years (20%->40%). These benefits are also sustained in the longer term.
The traditional 'open' fetal surgical approach, however, as currently offered clinically at the Ontario Fetal Centre, comes with significant risks: it increases the risk of preterm birth, carries significant maternal morbidity and results in important uterine scarring. The latter comes with a risk of uterine rupture and fetal death both in the index pregnancy and future pregnancies.
To overcome these down sides of open fetal surgery, different centers have attempted a fetoscopic approach to the surgery. Fetoscopy indeed avoids uterine scarring and is likely protective against uterine rupture but is technically complex. This results in long surgical learning curves, poor dissemination of the surgery amongst centers worldwide, longer procedures and suboptimal surgical results which translate in decreased infant benefits - particularly with regards to motor function.
The investigators have developed a fetoscopic robotic approach where they leverage the dexterity of robotic instruments to perform these complex surgeries. The team expects that this will result in easier and faster procedures with better surgical outcomes and therefore fetal benefits comparable to open fetal surgery, while at the same time avoiding the need for hysterotomy.
In this prospective exploratory phase 1 study, the investigators propose to assess the feasibility of such a robotic approach, as developed and trained on a high-fidelity phantom, in 15 patients. The research team will collect maternal and fetal safety and efficacity data to inform later studies.
The traditional 'open' fetal surgical approach, however, as currently offered clinically at the Ontario Fetal Centre, comes with significant risks: it increases the risk of preterm birth, carries significant maternal morbidity and results in important uterine scarring. The latter comes with a risk of uterine rupture and fetal death both in the index pregnancy and future pregnancies.
To overcome these down sides of open fetal surgery, different centers have attempted a fetoscopic approach to the surgery. Fetoscopy indeed avoids uterine scarring and is likely protective against uterine rupture but is technically complex. This results in long surgical learning curves, poor dissemination of the surgery amongst centers worldwide, longer procedures and suboptimal surgical results which translate in decreased infant benefits - particularly with regards to motor function.
The investigators have developed a fetoscopic robotic approach where they leverage the dexterity of robotic instruments to perform these complex surgeries. The team expects that this will result in easier and faster procedures with better surgical outcomes and therefore fetal benefits comparable to open fetal surgery, while at the same time avoiding the need for hysterotomy.
In this prospective exploratory phase 1 study, the investigators propose to assess the feasibility of such a robotic approach, as developed and trained on a high-fidelity phantom, in 15 patients. The research team will collect maternal and fetal safety and efficacity data to inform later studies.
开始日期2025-07-01 |
申办/合作机构 |
NCT06849596
A Cluster Crossover Randomized Controlled Trial of Manual T-piece Versus Ventilator Positive Pressure Ventilation During Resuscitation of Extremely Premature Neonates: The MVP Trial
Many extremely premature infants, born before 28 weeks' gestation age, require immediate help with breathing after birth. Positive pressure ventilation (PPV) using a device called a T-piece resuscitator is a common method. PPV is needed to establish proper lung function, improve gas exchange, and encourage the infant to breathe spontaneously. However, T-piece resuscitators have limitations, like a lack of visual feedback and variable settings, which may result in reduced effectiveness of PPV. Improving PPV effectiveness may reduce the need for more invasive procedures, such as intubation, which pose an increased risk of complications and death for these fragile infants. A novel approach, that may overcome the above limitations and deliver PPV with precise settings through a nasal mask, is to use a ventilator to deliver PPV (V-PPV) using a respiratory mode called nasal intermittent positive pressure ventilation (NIPPV). While NIPPV is commonly used in neonatal intensive care units to support breathing in premature infants, the impact of V-PPV use during immediate post-birth stabilization needs to be studied. Preliminary data from our recent single-center study confirmed the feasibility of using V-PPV for resuscitation of extremely premature babies and indicated its potential superiority with a 28% decrease in the need for intubation compared to historical use of T-piece. This promising innovation may enhance outcomes for these vulnerable infants by refining the way we provide respiratory support in their critical first moments. The research objective is to compare the clinical outcomes of extremely premature infants receiving manual T-piece versus V-PPV during immediate post-birth stabilization. The primary aim is to evaluate the impact of V-PPV on major health complications or death. This study seeks to provide insights into improving the care and outcomes of these infants during a critical stage of transition from fetus to newborn.
开始日期2025-04-01 |
申办/合作机构 |
NCT04931173
Is Mechanical Bowel Preparation Necessary to Reduce Surgical Site Infection Following Colon Surgery: a Randomized Controlled Trial
Following colon surgery, surgical site infection (SSI) is the most common complication and leads to longer recovery time for patients and increased health care costs. Currently, there is high quality evidence to show that the combination of intravenous antibiotics (IVA), mechanical bowel preparation (MBP) and oral antibiotics (OA) is the most effective bowel preparation to reduce SSI. However, there are no studies comparing IVA+MBP+OA and IVA+OA. This is an important question because if IVA+OA works the same as IVA+MBP+OA, then MBP may be safely omitted as part of the bowel preparation to reduce SSI and patients would avoid the side effects of MBP prior to their surgery. Therefore, the objective is to perform a trial to determine if IVA+OA works the same as IVA+MBP+OA to reduce SSI following colon surgery.
开始日期2025-03-01 |
100 项与 Mount Sinai Hospital (Canada) 相关的临床结果
登录后查看更多信息
0 项与 Mount Sinai Hospital (Canada) 相关的专利(医药)
登录后查看更多信息
3
项与 Mount Sinai Hospital (Canada) 相关的新闻(医药)2025-01-25
RAHWAY, NJ & NUTLEY, NJ, USA I January 24, 2025 I
Merck (NYSE: MRK), known as MSD outside of the United States and Canada, and Eisai today announced results from the Phase 3 LEAP-015 trial evaluating KEYTRUDA
®
(pembrolizumab), Merck’s anti-PD-1 therapy, plus LENVIMA
®
(lenvatinib), the orally available multiple receptor tyrosine kinase inhibitor (TKI) discovered by Eisai, in combination with chemotherapy (KEYTRUDA plus LENVIMA-based regimen), for the first-line treatment of patients with locally advanced unresectable or metastatic human epidermal growth factor receptor 2 (HER2)-negative gastroesophageal adenocarcinoma. At an interim analysis, the KEYTRUDA plus LENVIMA-based regimen demonstrated a statistically significant improvement in progression-free survival (PFS), one of the study’s dual primary endpoints, and objective response rate (ORR), a key secondary endpoint, compared to standard of care chemotherapy. The study continued, and at the final analysis, it did not meet its other primary endpoint of overall survival (OS). The safety profile of the KEYTRUDA plus LENVIMA-based regimen was consistent with that observed in previously reported studies evaluating the combination. A full evaluation of the data from this study is ongoing, and Merck and Eisai will present these results at an upcoming medical meeting.
“Locally advanced unresectable or metastatic gastroesophageal adenocarcinoma remains a challenging disease to treat and a leading cause of cancer death worldwide,” said Dr. Gregory Lubiniecki, Vice President, Global Clinical Development, Merck Research Laboratories. “These study results add to our understanding of this combination and will inform our future research as we strive to improve outcomes for more patients with cancer.”
“Gastric and gastroesophageal cancers continue to present challenges due to their heterogeneity and generally poor prognoses,” said Dr. Corina Dutcus, Senior Vice President, Oncology Global Clinical Development Lead at Eisai Inc. “While the LEAP-015 trial did not show a statistically significant increase in overall survival, we were pleased to observe an improvement in progression-free survival and objective response rate for patients treated with KEYTRUDA plus LENVIMA in combination with chemotherapy. These results contribute to the scientific community’s collective understanding of these complex diseases and add to the body of knowledge in oncology research. We are deeply grateful to the patients, caregivers and investigators who participated in this study.”
KEYTRUDA plus LENVIMA is approved in the U.S., the EU, Japan and other countries for the treatment of advanced renal cell carcinoma (RCC) and certain types of advanced endometrial carcinoma. Lenvatinib is marketed as KISPLYX for advanced RCC in the EU. Merck and Eisai are studying the KEYTRUDA plus LENVIMA combination through the LEAP (
LE
nvatinib
A
nd
P
embrolizumab) clinical program in hepatocellular carcinoma and esophageal cancer across multiple clinical trials.
In gastric cancer, KEYTRUDA is approved in combination with fluoropyrimidine- and platinum-containing chemotherapy for the first-line treatment of adults with locally advanced unresectable or metastatic HER2-negative gastric or gastroesophageal junction (GEJ) adenocarcinoma based on OS and other data from the Phase 3 KEYNOTE-859 trial. KEYTRUDA is also approved in combination with trastuzumab, fluoropyrimidine- and platinum-containing chemotherapy, for the first-line treatment of adults with locally advanced unresectable or metastatic HER2-positive gastric or GEJ adenocarcinoma, whose tumors express PD-L1 (Combined Positive Score [CPS] ≥1) as determined by an FDA-approved test. This indication is approved under accelerated approval based on tumor response rate and durability of response data from the Phase 3 KEYNOTE-811 trial. Continued approval of this indication may be contingent upon verification and description of clinical benefit in the confirmatory trials.
In esophageal cancer, KEYTRUDA is approved in combination with platinum- and fluoropyrimidine-based chemotherapy for the treatment of patients with locally advanced or metastatic esophageal or GEJ (tumors with epicenter 1 to 5 centimeters above the GEJ) carcinoma that is not amenable to surgical resection or definitive chemoradiation. This approval is based on OS and other data from the Phase 3 KEYNOTE-590 trial.
Results from the LEAP-015 trial do not affect the current approved indications for KEYTRUDA plus LENVIMA or other ongoing trials from the LEAP clinical program.
About LEAP-015
LEAP-015 is a randomized, open-label, Phase 3 trial (ClinicalTrials.gov,
NCT04662710
) evaluating KEYTRUDA plus LENVIMA in combination with chemotherapy versus chemotherapy alone for the first-line treatment of patients with locally advanced unresectable or metastatic HER2-negative gastroesophageal adenocarcinoma. There are two parts of the study: a safety run-in (Part 1) and the main study (Part 2). In Part 2, the primary endpoints are OS and PFS as assessed by blinded independent central review (BICR) per Response Evaluation Criteria in Solid Tumors version 1.1 (RECIST v1.1) in patients whose tumors express PD-L1 (Combined Positive Score [CPS] ≥1) and in all patients. Secondary endpoints are ORR and duration of response (DOR) as assessed by BICR per RECIST v1.1 in both patients whose tumors express PD-L1 (CPS ≥1) as well as in all patients, and safety. In Part 2, up to 880 patients were randomized 1:1 to receive:
About gastric cancer
Gastric (stomach) cancer tends to develop slowly over many years and rarely causes early symptoms, resulting in most cases going undetected until an advanced stage. More than 70% of patients with gastric cancer develop advanced-stage disease. Most gastric cancers are adenocarcinomas (about 90% to 95%), which develop from cells in the innermost lining of the stomach (known as the mucosa). Gastric cancer is the fifth most diagnosed cancer and the fifth leading cause of cancer death worldwide, with approximately 969,000 patients diagnosed and 660,000 deaths from the disease globally in 2022. In the U.S., it is estimated there will be approximately 26,890 patients diagnosed with gastric cancer and 10,880 deaths from the disease in 2024. The five-year relative survival rate for patients diagnosed with gastric cancer at a distant stage is 7% in the U.S.
About esophageal cancer
Esophageal cancer is the 11th most commonly diagnosed cancer and the seventh leading cause of death from cancer worldwide. It is estimated there were 511,000 new cases of esophageal cancer diagnosed and about 445,000 deaths resulting from the disease worldwide in 2022. In the U.S., it is estimated there will be approximately 22,370 patients diagnosed with esophageal cancer and 16,130 deaths from the disease in 2024. The five-year relative survival rate for patients diagnosed with advanced esophageal cancer is 6% in the U.S. Cancers that start in gland cells (cells that make mucus) are called adenocarcinomas and are often found in the lower third of the esophagus (lower thoracic esophagus). Adenocarcinoma is the most common form of esophageal cancer in the U.S. and its incidence is rapidly increasing in other parts of the world.
About KEYTRUDA
®
(pembrolizumab) injection, 100 mg
KEYTRUDA is an anti-programmed death receptor-1 (PD-1) therapy that works by increasing the ability of the body’s immune system to help detect and fight tumor cells. KEYTRUDA is a humanized monoclonal antibody that blocks the interaction between PD-1 and its ligands, PD-L1 and PD-L2, thereby activating T lymphocytes which may affect both tumor cells and healthy cells.
Merck has the industry’s largest immuno-oncology clinical research program. There are currently more than 1,600 trials studying KEYTRUDA across a wide variety of cancers and treatment settings. The KEYTRUDA clinical program seeks to understand the role of KEYTRUDA across cancers and the factors that may predict a patient’s likelihood of benefitting from treatment with KEYTRUDA, including exploring several different biomarkers.
Selected KEYTRUDA
®
(pembrolizumab) Indications in the U.S.
Gastric Cancer
KEYTRUDA, in combination with trastuzumab, fluoropyrimidine- and platinum-containing chemotherapy, is indicated for the first-line treatment of adults with locally advanced unresectable or metastatic HER2-positive gastric or gastroesophageal junction (GEJ) adenocarcinoma whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-approved test.
This indication is approved under accelerated approval based on tumor response rate and durability of response. Continued approval of this indication may be contingent upon verification and description of clinical benefit in the confirmatory trials.
KEYTRUDA, in combination with fluoropyrimidine- and platinum-containing chemotherapy, is indicated for the first-line treatment of adults with locally advanced unresectable or metastatic HER2-negative gastric or gastroesophageal junction (GEJ) adenocarcinoma.
Esophageal Cancer
KEYTRUDA is indicated for the treatment of patients with locally advanced or metastatic esophageal or gastroesophageal junction (GEJ) (tumors with epicenter 1 to 5 centimeters above the GEJ) carcinoma that is not amenable to surgical resection or definitive chemoradiation either:
Melanoma
KEYTRUDA is indicated for the treatment of patients with unresectable or metastatic melanoma.
KEYTRUDA is indicated for the adjuvant treatment of adult and pediatric (12 years and older) patients with Stage IIB, IIC, or III melanoma following complete resection.
Non-Small Cell Lung Cancer
KEYTRUDA, in combination with pemetrexed and platinum chemotherapy, is indicated for the first-line treatment of patients with metastatic nonsquamous non-small cell lung cancer (NSCLC), with no EGFR or ALK genomic tumor aberrations.
KEYTRUDA, in combination with carboplatin and either paclitaxel or paclitaxel protein-bound, is indicated for the first-line treatment of patients with metastatic squamous NSCLC.
KEYTRUDA, as a single agent, is indicated for the first-line treatment of patients with NSCLC expressing PD-L1 [Tumor Proportion Score (TPS) ≥1%] as determined by an FDA-approved test, with no EGFR or ALK genomic tumor aberrations, and is:
KEYTRUDA, as a single agent, is indicated for the treatment of patients with metastatic NSCLC whose tumors express PD-L1 (TPS ≥1%) as determined by an FDA-approved test, with disease progression on or after platinum-containing chemotherapy. Patients with EGFR or ALK genomic tumor aberrations should have disease progression on FDA-approved therapy for these aberrations prior to receiving KEYTRUDA.
KEYTRUDA is indicated for the treatment of patients with resectable (tumors ≥4 cm or node positive) NSCLC in combination with platinum-containing chemotherapy as neoadjuvant treatment, and then continued as a single agent as adjuvant treatment after surgery.
KEYTRUDA, as a single agent, is indicated as adjuvant treatment following resection and platinum-based chemotherapy for adult patients with Stage IB (T2a ≥4 cm), II, or IIIA NSCLC.
Malignant Pleural Mesothelioma
KEYTRUDA, in combination with pemetrexed and platinum chemotherapy, is indicated for the first-line treatment of adult patients with unresectable advanced or metastatic malignant pleural mesothelioma (MPM).
Head and Neck Squamous Cell Cancer
KEYTRUDA, in combination with platinum and fluorouracil (FU), is indicated for the first-line treatment of patients with metastatic or with unresectable, recurrent head and neck squamous cell carcinoma (HNSCC).
KEYTRUDA, as a single agent, is indicated for the first-line treatment of patients with metastatic or with unresectable, recurrent HNSCC whose tumors express PD-L1 [Combined Positive Score (CPS) ≥1] as determined by an FDA-approved test.
KEYTRUDA, as a single agent, is indicated for the treatment of patients with recurrent or metastatic HNSCC with disease progression on or after platinum-containing chemotherapy.
Classical Hodgkin Lymphoma
KEYTRUDA is indicated for the treatment of adult patients with relapsed or refractory classical Hodgkin lymphoma (cHL).
KEYTRUDA is indicated for the treatment of pediatric patients with refractory cHL, or cHL that has relapsed after 2 or more lines of therapy.
Primary Mediastinal Large B-Cell Lymphoma
KEYTRUDA is indicated for the treatment of adult and pediatric patients with refractory primary mediastinal large B-cell lymphoma (PMBCL), or who have relapsed after 2 or more prior lines of therapy. KEYTRUDA is not recommended for treatment of patients with PMBCL who require urgent cytoreductive therapy.
Urothelial Cancer
KEYTRUDA, in combination with enfortumab vedotin, is indicated for the treatment of adult patients with locally advanced or metastatic urothelial cancer.
KEYTRUDA, as a single agent, is indicated for the treatment of patients with locally advanced or metastatic urothelial carcinoma:
KEYTRUDA, as a single agent, is indicated for the treatment of patients with Bacillus Calmette-Guerin (BCG)-unresponsive, high-risk, non-muscle invasive bladder cancer (NMIBC) with carcinoma in situ (CIS) with or without papillary tumors who are ineligible for or have elected not to undergo cystectomy.
Microsatellite Instability-High or Mismatch Repair Deficient Cancer
KEYTRUDA is indicated for the treatment of adult and pediatric patients with unresectable or metastatic microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR) solid tumors, as determined by an FDA-approved test, that have progressed following prior treatment and who have no satisfactory alternative treatment options.
Microsatellite Instability-High or Mismatch Repair Deficient Colorectal Cancer
KEYTRUDA is indicated for the treatment of patients with unresectable or metastatic MSI-H or dMMR colorectal cancer (CRC) as determined by an FDA-approved test.
Cervical Cancer
KEYTRUDA, in combination with chemoradiotherapy (CRT), is indicated for the treatment of patients with FIGO 2014 Stage III-IVA cervical cancer.
KEYTRUDA, in combination with chemotherapy, with or without bevacizumab, is indicated for the treatment of patients with persistent, recurrent, or metastatic cervical cancer whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-approved test.
KEYTRUDA, as a single agent, is indicated for the treatment of patients with recurrent or metastatic cervical cancer with disease progression on or after chemotherapy whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-approved test.
Hepatocellular Carcinoma
KEYTRUDA is indicated for the treatment of patients with hepatocellular carcinoma (HCC) secondary to hepatitis B who have received prior systemic therapy other than a PD-1/PD-L1-containing regimen.
Biliary Tract Cancer
KEYTRUDA, in combination with gemcitabine and cisplatin, is indicated for the treatment of patients with locally advanced unresectable or metastatic biliary tract cancer (BTC).
Merkel Cell Carcinoma
KEYTRUDA is indicated for the treatment of adult and pediatric patients with recurrent locally advanced or metastatic Merkel cell carcinoma (MCC).
Renal Cell Carcinoma
KEYTRUDA, in combination with axitinib, is indicated for the first-line treatment of adult patients with advanced renal cell carcinoma (RCC).
KEYTRUDA, in combination with lenvatinib, is indicated for the first-line treatment of adult patients with advanced RCC.
KEYTRUDA is indicated for the adjuvant treatment of patients with RCC at intermediate-high or high risk of recurrence following nephrectomy, or following nephrectomy and resection of metastatic lesions.
Endometrial Carcinoma
KEYTRUDA, in combination with carboplatin and paclitaxel, followed by KEYTRUDA as a single agent, is indicated for the treatment of adult patients with primary advanced or recurrent endometrial carcinoma.
KEYTRUDA, in combination with lenvatinib, is indicated for the treatment of adult patients with advanced endometrial carcinoma that is mismatch repair proficient (pMMR) as determined by an FDA-approved test or not MSI-H, who have disease progression following prior systemic therapy in any setting are not candidates for curative surgery or radiation.
KEYTRUDA, as a single agent, is indicated for the treatment of adult patients with advanced endometrial carcinoma that is MSI-H or dMMR, as determined by an FDA-approved test, who have disease progression following prior systemic therapy in any setting and are not candidates for curative surgery or radiation.
Tumor Mutational Burden-High Cancer
KEYTRUDA is indicated for the treatment of adult and pediatric patients with unresectable or metastatic tumor mutational burden-high (TMB-H) [≥10 mutations/megabase (mut/Mb)] solid tumors, as determined by an FDA-approved test, that have progressed following prior treatment and who have no satisfactory alternative treatment options.
This indication is approved under accelerated approval based on tumor response rate and durability of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in the confirmatory trials. The safety and effectiveness of KEYTRUDA in pediatric patients with TMB-H central nervous system cancers have not been established.
Cutaneous Squamous Cell Carcinoma
KEYTRUDA is indicated for the treatment of patients with recurrent or metastatic cutaneous squamous cell carcinoma (cSCC) or locally advanced cSCC that is not curable by surgery or radiation.
Triple-Negative Breast Cancer
KEYTRUDA is indicated for the treatment of patients with high-risk early-stage triple-negative breast cancer (TNBC) in combination with chemotherapy as neoadjuvant treatment, and then continued as a single agent as adjuvant treatment after surgery.
KEYTRUDA, in combination with chemotherapy, is indicated for the treatment of patients with locally recurrent unresectable or metastatic TNBC whose tumors express PD-L1 (CPS ≥10) as determined by an FDA-approved test.
Please see Prescribing Information for KEYTRUDA (pembrolizumab) at
http://www.merck.com/product/usa/pi_circulars/k/keytruda/keytruda_pi.pdf
and Medication Guide for KEYTRUDA at
http://www.merck.com/product/usa/pi_circulars/k/keytruda/keytruda_mg.pdf
.
About LENVIMA
®
(lenvatinib); available as 10 mg and 4 mg capsules
LENVIMA, discovered and developed by Eisai, is an orally available multiple receptor tyrosine kinase inhibitor that inhibits the kinase activities of vascular endothelial growth factor (VEGF) receptors VEGFR1 (FLT1), VEGFR2 (KDR), and VEGFR3 (FLT4). LENVIMA inhibits other kinases that have been implicated in pathogenic angiogenesis, tumor growth, and cancer progression in addition to their normal cellular functions, including fibroblast growth factor (FGF) receptors FGFR1-4, the platelet derived growth factor receptor alpha (PDGFRA), KIT, and RET. In syngeneic mouse tumor models, LENVIMA decreased tumor-associated macrophages, increased activated cytotoxic T cells, and demonstrated greater antitumor activity in combination with an anti-PD-1 monoclonal antibody compared to either treatment alone.
LENVIMA
®
(lenvatinib) Indications in the U.S.
Please see Prescribing Information for LENVIMA (lenvatinib) at
http://www.lenvima.com/pdfs/prescribing-information.pdf
.
About the Merck and Eisai strategic collaboration
In March 2018, Eisai and Merck, known as MSD outside of the United States and Canada, through an affiliate, entered into a strategic collaboration for the worldwide co-development and co-commercialization of LENVIMA. Under the agreement, the companies jointly develop, manufacture and commercialize LENVIMA, both as monotherapy and in combination with Merck’s anti-PD-1 therapy KEYTRUDA. Eisai and Merck are studying the KEYTRUDA plus LENVIMA combination through the LEAP (
LE
nvatinib
A
nd
P
embrolizumab) clinical program in hepatocellular carcinoma and esophageal cancer across multiple clinical trials.
Merck’s focus on cancer
Every day, we follow the science as we work to discover innovations that can help patients, no matter what stage of cancer they have. As a leading oncology company, we are pursuing research where scientific opportunity and medical need converge, underpinned by our diverse pipeline of more than 25 novel mechanisms. With one of the largest clinical development programs across more than 30 tumor types, we strive to advance breakthrough science that will shape the future of oncology. By addressing barriers to clinical trial participation, screening and treatment, we work with urgency to reduce disparities and help ensure patients have access to high-quality cancer care. Our unwavering commitment is what will bring us closer to our goal of bringing life to more patients with cancer. For more information, visit
https://www.merck.com/research/oncology
.
About Merck
At Merck, known as MSD outside of the United States and Canada, we are unified around our purpose: We use the power of leading-edge science to save and improve lives around the world. For more than 130 years, we have brought hope to humanity through the development of important medicines and vaccines. We aspire to be the premier research-intensive biopharmaceutical company in the world – and today, we are at the forefront of research to deliver innovative health solutions that advance the prevention and treatment of diseases in people and animals. We foster a diverse and inclusive global workforce and operate responsibly every day to enable a safe, sustainable and healthy future for all people and communities. For more information, visit
www.merck.com
and connect with us on
X (formerly Twitter)
,
Facebook
,
Instagram
,
YouTube
and
LinkedIn
.
Eisai’s focus on cancer
Eisai acknowledges “Oncology” as one of its key strategic areas, and will continue to focus on the discovery and development of anti-cancer drugs within drug discovery domains including “microenvironment”, “proteostasis disruption”, and “cell lineage and cell differentiation” under the Deep Human Biology Learning (DHBL) drug discovery and development organization. Eisai aspires to discover innovative new drugs with new targets and mechanisms of action from these domains, with the aim of contributing to the cure of cancers.
About Eisai
Eisai’s Corporate Concept is “to give first thought to patients and people in the daily living domain, and to increase the benefits that health care provides.” Under this Concept [also known as our
human health care
(
hhc
) Concept], we aim to effectively achieve social good in the form of relieving anxiety over health and reducing health disparities. With a global network of R&D facilities, manufacturing sites and marketing subsidiaries, we strive to create and deliver innovative products to target diseases with high unmet medical needs, with a particular focus in our strategic areas of Neurology and Oncology.
In addition,
our continued commitment to the elimination of neglected tropical diseases (NTDs)
,
which is a target (3.3) of the United Nations Sustainable Development Goals (SDGs)
, is demonstrated by our work on various activities together with global partners.
For more information about Eisai, please visit
www.eisai.com
(for global headquarters: Eisai Co., Ltd.),
us.eisai.com
(for U.S. headquarters: Eisai Inc.) or
www.eisai.eu
(for Europe, Middle East, Africa, Russia, Australia, and New Zealand headquarters: Eisai Europe Ltd.), and connect with us on Twitter (
U.S.
and
global
) and LinkedIn (for
U.S.
and
EMEA
).
SOURCE:
Merck
临床结果临床3期上市批准加速审批免疫疗法
2024-12-16
December 16, 2024 4:30 pm ET
RAHWAY, N.J.--(BUSINESS WIRE)--
Merck (NYSE: MRK), known as MSD outside of the United States and Canada, today announced the discontinuation of the clinical development programs for vibostolimab, an anti-TIGIT antibody, and favezelimab, an anti-LAG-3 antibody. Vibostolimab is being evaluated as an investigational fixed-dose combination with pembrolizumab (KEYTRUDA
®
) in the KeyVibe program. Favezelimab is being evaluated as an investigational fixed-dose combination with pembrolizumab in the KEYFORM program.
Merck is discontinuing the Phase 3 KeyVibe-003 and KeyVibe-007 trials, which are evaluating the fixed-dose combination of vibostolimab and pembrolizumab in certain patients with non-small cell lung cancer (NSCLC), based on the recommendation of an independent Data Monitoring Committee (DMC). In a pre-planned analysis, both trials met the pre-specified futility criteria for the primary endpoint of overall survival. In these studies, the safety profile of vibostolimab/pembrolizumab was consistent with that observed for vibostolimab and pembrolizumab in previously reported studies, with no new safety signals identified. As expected with dual checkpoint inhibitor therapy, more immune-related adverse events were observed with the fixed-dose combination than with pembrolizumab. Considering the totality of data from the Phase 3 KeyVibe studies, including the efficacy outcomes from KeyVibe-003 and KeyVibe-007, the company has decided to discontinue the Phase 3 KeyVibe-006 trial and other vibostolimab studies.
Separately, Merck has decided to end the favezelimab clinical development program, and will stop enrollment in the Phase 3 KEYFORM-008 trial evaluating the fixed-dose combination of favezelimab and pembrolizumab in patients with relapsed or refractory classical Hodgkin lymphoma (cHL) whose disease has progressed following prior anti-PD-1 therapy. Patients currently in this trial may continue on therapy until study completion. KEYFORM-008 is the only Phase 3 study in the KEYFORM clinical development program for which results are not available. The company has made this decision after a thorough evaluation of data from the favezelimab clinical program and will prioritize the development of other candidates in its comprehensive and diversified oncology pipeline. This decision is not based on any concerns about the safety of this fixed-dose combination.
Merck is informing study investigators for these clinical trials and advises patients to speak to their study team and physician regarding next steps and treatment options. Data analyses for the Phase 3 trials are ongoing, and the results will be shared with the scientific community.
"Following a careful analysis of the data, the decision has been made to discontinue development of these candidates to prioritize other ongoing programs. We are grateful to all the patients, caregivers and investigators for their many contributions that made these studies possible," said Dr. Marjorie Green, senior vice president and head of oncology, global clinical development, Merck Research Laboratories. "We continue to pursue the most promising science with a focus on agents with the greatest potential to improve outcomes for more patients with cancer."
About KeyVibe-003
KeyVibe-003 is a randomized, double-blind Phase 3 trial (ClinicalTrials.gov,
NCT04738487
) evaluating the fixed-dose combination of vibostolimab and pembrolizumab (MK-7684A) versus pembrolizumab monotherapy, as a first-line treatment for patients with PD-L1 positive metastatic NSCLC. The primary endpoint is overall survival (OS) in participants with PD-L1 TPS ≥50%. Secondary endpoints include OS in participants with PD-L1 TPS ≥1% and TPS 1-49%, progression-free survival (PFS), overall response rate (ORR), duration of response (DOR), safety and quality of life. The trial enrolled 1,264 patients who were randomized (1:1) to receive:
Vibostolimab/pembrolizumab fixed-dose combination (pembrolizumab 200 mg and vibostolimab 200 mg intravenously [IV] every three weeks [Q3W] for up to 35 administrations); or
Pembrolizumab (200 mg IV Q3W for up to 35 administrations)
About KeyVibe-007
KeyVibe-007 is a randomized, double-blind Phase 3 trial (ClinicalTrials.gov,
NCT05226598
) evaluating the fixed-dose combination of vibostolimab and pembrolizumab with chemotherapy in treatment-naïve patients with metastatic NSCLC. The primary endpoint is OS in participants with PD-L1 TPS ≥1%. Secondary endpoints include OS in all participants, PFS, ORR and DOR in TPS ≥ 1% and all participants, safety and patient reported outcomes. The trial enrolled 739 patients who were randomized (1:1) to receive:
Vibostolimab/pembrolizumab fixed-dose combination (pembrolizumab 200mg and vibostolimab 200 mg IV) plus platinum doublet chemotherapy (Q3W for 4 cycles); then vibostolimab/pembrolizumab (200mg/200mg IV) for up to 31 cycles (plus pemetrexed 500mg/m
2
Q3W maintenance for nonsquamous histology)
Pembrolizumab (200 mg IV) plus platinum doublet chemotherapy (Q3W for 4 cycles); then pembrolizumab (200mg IV) for up to 31 cycles (plus pemetrexed 500mg/m
2
Q3W maintenance for nonsquamous histology)
About KeyVibe-006
KeyVibe-006 is a randomized, open-label Phase 3 trial (ClincialTrials.gov,
NCT05298423
) evaluating the fixed-dose combination of vibostolimab and pembrolizumab with concurrent chemoradiotherapy followed by vibostolimab and pembrolizumab versus concurrent chemoradiotherapy followed by durvalumab in patients with stage III NSCLC. The primary endpoints are PFS and OS for all participants and for participants with TPS ≥ 1%. The secondary endpoints are ORR, DOR, safety and patient reported outcomes. The trial enrolled approximately 580 patients who were randomized (1:1) to receive:
Vibostolimab/pembrolizumab (200mg/200mg IV) plus platinum doublet (1 cycle); then platinum doublet plus vibostolimab/pembrolizumab (2 cycles) plus thoracic radiotherapy; then vibostolimab/pembrolizumab (200mg/200mg IV Q3W for 17 cycles); or
Platinum doublet (1 cycle); then platinum doublet (2 cycles) plus thoracic radiotherapy; then durvalumab (10 mg/kg Q2W for 26 cycles)
About KEYFORM-008
KEYFORM-008
is a randomized, open-label Phase 3 trial (ClinicalTrials.gov,
NCT05508867
) evaluating the fixed-dose combination of favezelimab and pembrolizumab (MK-4280A) versus physician's choice chemotherapy for the treatment of patients with PD-1 relapsed or refractory classical Hodgkin lymphoma. The primary endpoint is PFS per Lugano Response Criteria as assessed by Blinded Independent Central Review (BICR). The secondary endpoints are OS, ORR, DOR and safety. The trial enrolled 169 patients who were randomized (1:1) to receive:
Favezelimab/pembrolizumab (800 mg/200 mg IV on day 1, then Q3W for up to 35 infusions); or
Physician's choice of either bendamustine (between 90-120 mg/m
2
IV on day 1 and day 2 of either a 3- or 4-week cycle for up to 6 cycles); or gemcitabine (between 800-1,200 mg/m
2
IV on day 1 and day 8 of a Q3W cycle for up to 6 cycles)
About vibostolimab
Vibostolimab (MK-7684) is an investigational humanized anti-TIGIT antibody discovered and developed by Merck. Vibostolimab restores antitumor activity by blocking the TIGIT receptor from binding to its ligands (CD112 and CD155), thereby activating T lymphocytes that help destroy tumor cells.
About favezelimab
Favezelimab (MK-4280) is an investigational anti-lymphocyte activation gene-3 (LAG-3) antibody. LAG-3 is a cell surface immunomodulatory receptor expressed on various immune cells that down-regulates T cell proliferation and activation. Favezelimab aims to restore T cell effector function by preventing LAG-3 from binding to its primary ligand, major histocompatibility complex (MHC) class II molecules.
About KEYTRUDA
®
(pembrolizumab) injection, 100 mg
KEYTRUDA is an anti-programmed death receptor-1 (PD-1) therapy that works by increasing the ability of the body’s immune system to help detect and fight tumor cells. KEYTRUDA is a humanized monoclonal antibody that blocks the interaction between PD-1 and its ligands, PD- L1 and PD-L2, thereby activating T lymphocytes which may affect both tumor cells and healthy cells.
Merck has the industry’s largest immuno-oncology clinical research program. There are currently more than 1,600 trials studying KEYTRUDA across a wide variety of cancers and treatment settings. The KEYTRUDA clinical program seeks to understand the role of KEYTRUDA across cancers and the factors that may predict a patient's likelihood of benefitting from treatment with KEYTRUDA, including exploring several different biomarkers.
Selected KEYTRUDA
®
(pembrolizumab) Indications in the U.S.
Non-Small Cell Lung Cancer
KEYTRUDA, in combination with pemetrexed and platinum chemotherapy, is indicated for the first-line treatment of patients with metastatic nonsquamous non-small cell lung cancer (NSCLC), with no EGFR or ALK genomic tumor aberrations.
KEYTRUDA, in combination with carboplatin and either paclitaxel or paclitaxel protein-bound, is indicated for the first-line treatment of patients with metastatic squamous NSCLC.
KEYTRUDA, as a single agent, is indicated for the first-line treatment of patients with NSCLC expressing PD-L1 [Tumor Proportion Score (TPS) ≥1%] as determined by an FDA-approved test, with no EGFR or ALK genomic tumor aberrations, and is:
Stage III where patients are not candidates for surgical resection or definitive chemoradiation, or
metastatic.
KEYTRUDA, as a single agent, is indicated for the treatment of patients with metastatic NSCLC whose tumors express PD-L1 (TPS ≥1%) as determined by an FDA-approved test, with disease progression on or after platinum-containing chemotherapy. Patients with EGFR or ALK genomic tumor aberrations should have disease progression on FDA-approved therapy for these aberrations prior to receiving KEYTRUDA.
KEYTRUDA is indicated for the treatment of patients with resectable (tumors ≥4 cm or node positive) NSCLC in combination with platinum-containing chemotherapy as neoadjuvant treatment, and then continued as a single agent as adjuvant treatment after surgery.
KEYTRUDA, as a single agent, is indicated as adjuvant treatment following resection and platinum-based chemotherapy for adult patients with Stage IB (T2a ≥4 cm), II, or IIIA NSCLC.
Classical Hodgkin Lymphoma
KEYTRUDA is indicated for the treatment of adult patients with relapsed or refractory classical Hodgkin lymphoma (cHL).
KEYTRUDA is indicated for the treatment of pediatric patients with refractory cHL, or cHL that has relapsed after 2 or more lines of therapy.
See additional selected KEYTRUDA indications in the U.S. after the Selected Important Safety Information.
Selected Important Safety Information for KEYTRUDA
Severe and Fatal Immune-Mediated Adverse Reactions
KEYTRUDA is a monoclonal antibody that belongs to a class of drugs that bind to either the programmed death receptor-1 (PD-1) or the programmed death ligand 1 (PD-L1), blocking the PD-1/PD-L1 pathway, thereby removing inhibition of the immune response, potentially breaking peripheral tolerance and inducing immune-mediated adverse reactions. Immune-mediated adverse reactions, which may be severe or fatal, can occur in any organ system or tissue, can affect more than one body system simultaneously, and can occur at any time after starting treatment or after discontinuation of treatment. Important immune-mediated adverse reactions listed here may not include all possible severe and fatal immune-mediated adverse reactions.
Monitor patients closely for symptoms and signs that may be clinical manifestations of underlying immune-mediated adverse reactions. Early identification and management are essential to ensure safe use of anti–PD-1/PD-L1 treatments. Evaluate liver enzymes, creatinine, and thyroid function at baseline and periodically during treatment. For patients with TNBC treated with KEYTRUDA in the neoadjuvant setting, monitor blood cortisol at baseline, prior to surgery, and as clinically indicated. In cases of suspected immune-mediated adverse reactions, initiate appropriate workup to exclude alternative etiologies, including infection. Institute medical management promptly, including specialty consultation as appropriate.
Withhold or permanently discontinue KEYTRUDA depending on severity of the immune-mediated adverse reaction. In general, if KEYTRUDA requires interruption or discontinuation, administer systemic corticosteroid therapy (1 to 2 mg/kg/day prednisone or equivalent) until improvement to Grade 1 or less. Upon improvement to Grade 1 or less, initiate corticosteroid taper and continue to taper over at least 1 month. Consider administration of other systemic immunosuppressants in patients whose adverse reactions are not controlled with corticosteroid therapy.
Immune-Mediated Pneumonitis
KEYTRUDA can cause immune-mediated pneumonitis. The incidence is higher in patients who have received prior thoracic radiation. Immune-mediated pneumonitis occurred in 3.4% (94/2799) of patients receiving KEYTRUDA, including fatal (0.1%), Grade 4 (0.3%), Grade 3 (0.9%), and Grade 2 (1.3%) reactions. Systemic corticosteroids were required in 67% (63/94) of patients. Pneumonitis led to permanent discontinuation of KEYTRUDA in 1.3% (36) and withholding in 0.9% (26) of patients. All patients who were withheld reinitiated KEYTRUDA after symptom improvement; of these, 23% had recurrence. Pneumonitis resolved in 59% of the 94 patients.
Pneumonitis occurred in 8% (31/389) of adult patients with cHL receiving KEYTRUDA as a single agent, including Grades 3-4 in 2.3% of patients. Patients received high-dose corticosteroids for a median duration of 10 days (range: 2 days to 53 months). Pneumonitis rates were similar in patients with and without prior thoracic radiation. Pneumonitis led to discontinuation of KEYTRUDA in 5.4% (21) of patients. Of the patients who developed pneumonitis, 42% interrupted KEYTRUDA, 68% discontinued KEYTRUDA, and 77% had resolution.
Pneumonitis occurred in 7% (41/580) of adult patients with resected NSCLC who received KEYTRUDA as a single agent for adjuvant treatment of NSCLC, including fatal (0.2%), Grade 4 (0.3%), and Grade 3 (1%) adverse reactions. Patients received high-dose corticosteroids for a median duration of 10 days (range: 1 day to 2.3 months). Pneumonitis led to discontinuation of KEYTRUDA in 26 (4.5%) of patients. Of the patients who developed pneumonitis, 54% interrupted KEYTRUDA, 63% discontinued KEYTRUDA, and 71% had resolution.
Immune-Mediated Colitis
KEYTRUDA can cause immune-mediated colitis, which may present with diarrhea. Cytomegalovirus infection/reactivation has been reported in patients with corticosteroid-refractory immune-mediated colitis. In cases of corticosteroid-refractory colitis, consider repeating infectious workup to exclude alternative etiologies. Immune-mediated colitis occurred in 1.7% (48/2799) of patients receiving KEYTRUDA, including Grade 4 (<0.1%), Grade 3 (1.1%), and Grade 2 (0.4%) reactions. Systemic corticosteroids were required in 69% (33/48); additional immunosuppressant therapy was required in 4.2% of patients. Colitis led to permanent discontinuation of KEYTRUDA in 0.5% (15) and withholding in 0.5% (13) of patients. All patients who were withheld reinitiated KEYTRUDA after symptom improvement; of these, 23% had recurrence. Colitis resolved in 85% of the 48 patients.
Hepatotoxicity and Immune-Mediated Hepatitis
KEYTRUDA as a Single Agent
KEYTRUDA can cause immune-mediated hepatitis. Immune-mediated hepatitis occurred in 0.7% (19/2799) of patients receiving KEYTRUDA, including Grade 4 (<0.1%), Grade 3 (0.4%), and Grade 2 (0.1%) reactions. Systemic corticosteroids were required in 68% (13/19) of patients; additional immunosuppressant therapy was required in 11% of patients. Hepatitis led to permanent discontinuation of KEYTRUDA in 0.2% (6) and withholding in 0.3% (9) of patients. All patients who were withheld reinitiated KEYTRUDA after symptom improvement; of these, none had recurrence. Hepatitis resolved in 79% of the 19 patients.
KEYTRUDA With Axitinib
KEYTRUDA in combination with axitinib can cause hepatic toxicity. Monitor liver enzymes before initiation of and periodically throughout treatment. Consider monitoring more frequently as compared to when the drugs are administered as single agents. For elevated liver enzymes, interrupt KEYTRUDA and axitinib, and consider administering corticosteroids as needed. With the combination of KEYTRUDA and axitinib, Grades 3 and 4 increased alanine aminotransferase (ALT) (20%) and increased aspartate aminotransferase (AST) (13%) were seen at a higher frequency compared to KEYTRUDA alone. Fifty-nine percent of the patients with increased ALT received systemic corticosteroids. In patients with ALT ≥3 times upper limit of normal (ULN) (Grades 2-4, n=116), ALT resolved to Grades 0-1 in 94%. Among the 92 patients who were rechallenged with either KEYTRUDA (n=3) or axitinib (n=34) administered as a single agent or with both (n=55), recurrence of ALT ≥3 times ULN was observed in 1 patient receiving KEYTRUDA, 16 patients receiving axitinib, and 24 patients receiving both. All patients with a recurrence of ALT ≥3 ULN subsequently recovered from the event.
Immune-Mediated Endocrinopathies
Adrenal Insufficiency
KEYTRUDA can cause primary or secondary adrenal insufficiency. For Grade 2 or higher, initiate symptomatic treatment, including hormone replacement as clinically indicated. Withhold KEYTRUDA depending on severity. Adrenal insufficiency occurred in 0.8% (22/2799) of patients receiving KEYTRUDA, including Grade 4 (<0.1%), Grade 3 (0.3%), and Grade 2 (0.3%) reactions. Systemic corticosteroids were required in 77% (17/22) of patients; of these, the majority remained on systemic corticosteroids. Adrenal insufficiency led to permanent discontinuation of KEYTRUDA in <0.1% (1) and withholding in 0.3% (8) of patients. All patients who were withheld reinitiated KEYTRUDA after symptom improvement.
Hypophysitis
KEYTRUDA can cause immune-mediated hypophysitis. Hypophysitis can present with acute symptoms associated with mass effect such as headache, photophobia, or visual field defects. Hypophysitis can cause hypopituitarism. Initiate hormone replacement as indicated. Withhold or permanently discontinue KEYTRUDA depending on severity. Hypophysitis occurred in 0.6% (17/2799) of patients receiving KEYTRUDA, including Grade 4 (<0.1%), Grade 3 (0.3%), and Grade 2 (0.2%) reactions. Systemic corticosteroids were required in 94% (16/17) of patients; of these, the majority remained on systemic corticosteroids. Hypophysitis led to permanent discontinuation of KEYTRUDA in 0.1% (4) and withholding in 0.3% (7) of patients. All patients who were withheld reinitiated KEYTRUDA after symptom improvement.
Thyroid Disorders
KEYTRUDA can cause immune-mediated thyroid disorders. Thyroiditis can present with or without endocrinopathy. Hypothyroidism can follow hyperthyroidism. Initiate hormone replacement for hypothyroidism or institute medical management of hyperthyroidism as clinically indicated. Withhold or permanently discontinue KEYTRUDA depending on severity. Thyroiditis occurred in 0.6% (16/2799) of patients receiving KEYTRUDA, including Grade 2 (0.3%). None discontinued, but KEYTRUDA was withheld in <0.1% (1) of patients.
Hyperthyroidism occurred in 3.4% (96/2799) of patients receiving KEYTRUDA, including Grade 3 (0.1%) and Grade 2 (0.8%). It led to permanent discontinuation of KEYTRUDA in <0.1% (2) and withholding in 0.3% (7) of patients. All patients who were withheld reinitiated KEYTRUDA after symptom improvement. Hypothyroidism occurred in 8% (237/2799) of patients receiving KEYTRUDA, including Grade 3 (0.1%) and Grade 2 (6.2%). It led to permanent discontinuation of KEYTRUDA in <0.1% (1) and withholding in 0.5% (14) of patients. All patients who were withheld reinitiated KEYTRUDA after symptom improvement. The majority of patients with hypothyroidism required long-term thyroid hormone replacement. The incidence of new or worsening hypothyroidism was higher in 1185 patients with HNSCC, occurring in 16% of patients receiving KEYTRUDA as a single agent or in combination with platinum and FU, including Grade 3 (0.3%) hypothyroidism. The incidence of new or worsening hypothyroidism was higher in 389 adult patients with cHL (17%) receiving KEYTRUDA as a single agent, including Grade 1 (6.2%) and Grade 2 (10.8%) hypothyroidism. The incidence of new or worsening hyperthyroidism was higher in 580 patients with resected NSCLC, occurring in 11% of patients receiving KEYTRUDA as a single agent as adjuvant treatment, including Grade 3 (0.2%) hyperthyroidism. The incidence of new or worsening hypothyroidism was higher in 580 patients with resected NSCLC, occurring in 22% of patients receiving KEYTRUDA as a single agent as adjuvant treatment (KEYNOTE-091), including Grade 3 (0.3%) hypothyroidism.
Type 1 Diabetes Mellitus (DM), Which Can Present With Diabetic Ketoacidosis
Monitor patients for hyperglycemia or other signs and symptoms of diabetes. Initiate treatment with insulin as clinically indicated. Withhold KEYTRUDA depending on severity. Type 1 DM occurred in 0.2% (6/2799) of patients receiving KEYTRUDA. It led to permanent discontinuation in <0.1% (1) and withholding of KEYTRUDA in <0.1% (1) of patients. All patients who were withheld reinitiated KEYTRUDA after symptom improvement.
Immune-Mediated Nephritis With Renal Dysfunction
KEYTRUDA can cause immune-mediated nephritis. Immune-mediated nephritis occurred in 0.3% (9/2799) of patients receiving KEYTRUDA, including Grade 4 (<0.1%), Grade 3 (0.1%), and Grade 2 (0.1%) reactions. Systemic corticosteroids were required in 89% (8/9) of patients. Nephritis led to permanent discontinuation of KEYTRUDA in 0.1% (3) and withholding in 0.1% (3) of patients. All patients who were withheld reinitiated KEYTRUDA after symptom improvement; of these, none had recurrence. Nephritis resolved in 56% of the 9 patients.
Immune-Mediated Dermatologic Adverse Reactions
KEYTRUDA can cause immune-mediated rash or dermatitis. Exfoliative dermatitis, including Stevens-Johnson syndrome, drug rash with eosinophilia and systemic symptoms, and toxic epidermal necrolysis, has occurred with anti– PD-1/PD-L1 treatments. Topical emollients and/or topical corticosteroids may be adequate to treat mild to moderate nonexfoliative rashes. Withhold or permanently discontinue KEYTRUDA depending on severity. Immune-mediated dermatologic adverse reactions occurred in 1.4% (38/2799) of patients receiving KEYTRUDA, including Grade 3 (1%) and Grade 2 (0.1%) reactions. Systemic corticosteroids were required in 40% (15/38) of patients. These reactions led to permanent discontinuation in 0.1% (2) and withholding of KEYTRUDA in 0.6% (16) of patients. All patients who were withheld reinitiated KEYTRUDA after symptom improvement; of these, 6% had recurrence. The reactions resolved in 79% of the 38 patients.
Other Immune-Mediated Adverse Reactions
The following clinically significant immune-mediated adverse reactions occurred at an incidence of <1% (unless otherwise noted) in patients who received KEYTRUDA or were reported with the use of other anti–PD-1/PD-L1 treatments. Severe or fatal cases have been reported for some of these adverse reactions. Cardiac/Vascular: Myocarditis, pericarditis, vasculitis; Nervous System: Meningitis, encephalitis, myelitis and demyelination, myasthenic syndrome/myasthenia gravis (including exacerbation), Guillain-Barré syndrome, nerve paresis, autoimmune neuropathy; Ocular: Uveitis, iritis and other ocular inflammatory toxicities can occur. Some cases can be associated with retinal detachment. Various grades of visual impairment, including blindness, can occur. If uveitis occurs in combination with other immune-mediated adverse reactions, consider a Vogt-Koyanagi-Harada-like syndrome, as this may require treatment with systemic steroids to reduce the risk of permanent vision loss; Gastrointestinal: Pancreatitis, to include increases in serum amylase and lipase levels, gastritis, duodenitis; Musculoskeletal and Connective Tissue: Myositis/polymyositis, rhabdomyolysis (and associated sequelae, including renal failure), arthritis (1.5%), polymyalgia rheumatica; Endocrine: Hypoparathyroidism; Hematologic/Immune: Hemolytic anemia, aplastic anemia, hemophagocytic lymphohistiocytosis, systemic inflammatory response syndrome, histiocytic necrotizing lymphadenitis (Kikuchi lymphadenitis), sarcoidosis, immune thrombocytopenic purpura, solid organ transplant rejection, other transplant (including corneal graft) rejection.
Infusion-Related Reactions
KEYTRUDA can cause severe or life-threatening infusion-related reactions, including hypersensitivity and anaphylaxis, which have been reported in 0.2% of 2799 patients receiving KEYTRUDA. Monitor for signs and symptoms of infusion-related reactions. Interrupt or slow the rate of infusion for Grade 1 or Grade 2 reactions. For Grade 3 or Grade 4 reactions, stop infusion and permanently discontinue KEYTRUDA.
Complications of Allogeneic Hematopoietic Stem Cell Transplantation (HSCT)
Fatal and other serious complications can occur in patients who receive allogeneic HSCT before or after anti–PD-1/PD-L1 treatments. Transplant-related complications include hyperacute graft-versus-host disease (GVHD), acute and chronic GVHD, hepatic veno-occlusive disease after reduced intensity conditioning, and steroid-requiring febrile syndrome (without an identified infectious cause). These complications may occur despite intervening therapy between anti–PD-1/PD-L1 treatments and allogeneic HSCT. Follow patients closely for evidence of these complications and intervene promptly. Consider the benefit vs risks of using anti–PD-1/PD-L1 treatments prior to or after an allogeneic HSCT.
Increased Mortality in Patients With Multiple Myeloma
In trials in patients with multiple myeloma, the addition of KEYTRUDA to a thalidomide analogue plus dexamethasone resulted in increased mortality. Treatment of these patients with an anti–PD-1/PD-L1 treatment in this combination is not recommended outside of controlled trials.
Embryofetal Toxicity
Based on its mechanism of action, KEYTRUDA can cause fetal harm when administered to a pregnant woman. Advise women of this potential risk. In females of reproductive potential, verify pregnancy status prior to initiating KEYTRUDA and advise them to use effective contraception during treatment and for 4 months after the last dose.
Adverse Reactions
In KEYNOTE-006, KEYTRUDA was discontinued due to adverse reactions in 9% of 555 patients with advanced melanoma; adverse reactions leading to permanent discontinuation in more than one patient were colitis (1.4%), autoimmune hepatitis (0.7%), allergic reaction (0.4%), polyneuropathy (0.4%), and cardiac failure (0.4%). The most common adverse reactions (≥20%) with KEYTRUDA were fatigue (28%), diarrhea (26%), rash (24%), and nausea (21%).
In KEYNOTE-054, when KEYTRUDA was administered as a single agent to patients with stage III melanoma, KEYTRUDA was permanently discontinued due to adverse reactions in 14% of 509 patients; the most common (≥1%) were pneumonitis (1.4%), colitis (1.2%), and diarrhea (1%). Serious adverse reactions occurred in 25% of patients receiving KEYTRUDA. The most common adverse reaction (≥20%) with KEYTRUDA was diarrhea (28%). In KEYNOTE-716, when KEYTRUDA was administered as a single agent to patients with stage IIB or IIC melanoma, adverse reactions occurring in patients with stage IIB or IIC melanoma were similar to those occurring in 1011 patients with stage III melanoma from KEYNOTE-054.
In KEYNOTE-189, when KEYTRUDA was administered with pemetrexed and platinum chemotherapy in metastatic nonsquamous NSCLC, KEYTRUDA was discontinued due to adverse reactions in 20% of 405 patients. The most common adverse reactions resulting in permanent discontinuation of KEYTRUDA were pneumonitis (3%) and acute kidney injury (2%). The most common adverse reactions (≥20%) with KEYTRUDA were nausea (56%), fatigue (56%), constipation (35%), diarrhea (31%), decreased appetite (28%), rash (25%), vomiting (24%), cough (21%), dyspnea (21%), and pyrexia (20%).
In KEYNOTE-407, when KEYTRUDA was administered with carboplatin and either paclitaxel or paclitaxel protein-bound in metastatic squamous NSCLC, KEYTRUDA was discontinued due to adverse reactions in 15% of 101 patients. The most frequent serious adverse reactions reported in at least 2% of patients were febrile neutropenia, pneumonia, and urinary tract infection. Adverse reactions observed in KEYNOTE-407 were similar to those observed in KEYNOTE-189 with the exception that increased incidences of alopecia (47% vs 36%) and peripheral neuropathy (31% vs 25%) were observed in the KEYTRUDA and chemotherapy arm compared to the placebo and chemotherapy arm in KEYNOTE-407.
In KEYNOTE-042, KEYTRUDA was discontinued due to adverse reactions in 19% of 636 patients with advanced NSCLC; the most common were pneumonitis (3%), death due to unknown cause (1.6%), and pneumonia (1.4%). The most frequent serious adverse reactions reported in at least 2% of patients were pneumonia (7%), pneumonitis (3.9%), pulmonary embolism (2.4%), and pleural effusion (2.2%). The most common adverse reaction (≥20%) was fatigue (25%).
In KEYNOTE-010, KEYTRUDA monotherapy was discontinued due to adverse reactions in 8% of 682 patients with metastatic NSCLC; the most common was pneumonitis (1.8%). The most common adverse reactions (≥20%) were decreased appetite (25%), fatigue (25%), dyspnea (23%), and nausea (20%).
In KEYNOTE-671, adverse reactions occurring in patients with resectable NSCLC receiving KEYTRUDA in combination with platinum-containing chemotherapy, given as neoadjuvant treatment and continued as single-agent adjuvant treatment, were generally similar to those occurring in patients in other clinical trials across tumor types receiving KEYTRUDA in combination with chemotherapy.
The most common adverse reactions (reported in ≥20%) in patients receiving KEYTRUDA in combination with chemotherapy were fatigue/asthenia, nausea, constipation, diarrhea, decreased appetite, rash, vomiting, cough, dyspnea, pyrexia, alopecia, peripheral neuropathy, mucosal inflammation, stomatitis, headache, weight loss, abdominal pain, arthralgia, myalgia, insomnia, palmar-plantar erythrodysesthesia, urinary tract infection, and hypothyroidism.
In the neoadjuvant phase of KEYNOTE-671, when KEYTRUDA was administered in combination with platinum-containing chemotherapy as neoadjuvant treatment, serious adverse reactions occurred in 34% of 396 patients. The most frequent (≥2%) serious adverse reactions were pneumonia (4.8%), venous thromboembolism (3.3%), and anemia (2%). Fatal adverse reactions occurred in 1.3% of patients, including death due to unknown cause (0.8%), sepsis (0.3%), and immune-mediated lung disease (0.3%). Permanent discontinuation of any study drug due to an adverse reaction occurred in 18% of patients who received KEYTRUDA in combination with platinum-containing chemotherapy; the most frequent adverse reactions (≥1%) that led to permanent discontinuation of any study drug were acute kidney injury (1.8%), interstitial lung disease (1.8%), anemia (1.5%), neutropenia (1.5%), and pneumonia (1.3%).
Of the KEYTRUDA-treated patients who received neoadjuvant treatment, 6% of 396 patients did not receive surgery due to adverse reactions. The most frequent (≥1%) adverse reaction that led to cancellation of surgery in the KEYTRUDA arm was interstitial lung disease (1%).
In the adjuvant phase of KEYNOTE-671, when KEYTRUDA was administered as a single agent as adjuvant treatment, serious adverse reactions occurred in 14% of 290 patients. The most frequent serious adverse reaction was pneumonia (3.4%). One fatal adverse reaction of pulmonary hemorrhage occurred. Permanent discontinuation of KEYTRUDA due to an adverse reaction occurred in 12% of patients who received KEYTRUDA as a single agent, given as adjuvant treatment; the most frequent adverse reactions (≥1%) that led to permanent discontinuation of KEYTRUDA were diarrhea (1.7%), interstitial lung disease (1.4%), increased aspartate aminotransferase (1%), and musculoskeletal pain (1%).
Adverse reactions observed in KEYNOTE-091 were generally similar to those occurring in other patients with NSCLC receiving KEYTRUDA as a single agent, with the exception of hypothyroidism (22%), hyperthyroidism (11%), and pneumonitis (7%). Two fatal adverse reactions of myocarditis occurred.
In KEYNOTE-048, KEYTRUDA monotherapy was discontinued due to adverse events in 12% of 300 patients with HNSCC; the most common adverse reactions leading to permanent discontinuation were sepsis (1.7%) and pneumonia (1.3%). The most common adverse reactions (≥20%) were fatigue (33%), constipation (20%), and rash (20%).
In KEYNOTE-048, when KEYTRUDA was administered in combination with platinum (cisplatin or carboplatin) and FU chemotherapy, KEYTRUDA was discontinued due to adverse reactions in 16% of 276 patients with HNSCC. The most common adverse reactions resulting in permanent discontinuation of KEYTRUDA were pneumonia (2.5%), pneumonitis (1.8%), and septic shock (1.4%). The most common adverse reactions (≥20%) were nausea (51%), fatigue (49%), constipation (37%), vomiting (32%), mucosal inflammation (31%), diarrhea (29%), decreased appetite (29%), stomatitis (26%), and cough (22%).
In KEYNOTE-012, KEYTRUDA was discontinued due to adverse reactions in 17% of 192 patients with HNSCC. Serious adverse reactions occurred in 45% of patients. The most frequent serious adverse reactions reported in at least 2% of patients were pneumonia, dyspnea, confusional state, vomiting, pleural effusion, and respiratory failure. The most common adverse reactions (≥20%) were fatigue, decreased appetite, and dyspnea. Adverse reactions occurring in patients with HNSCC were generally similar to those occurring in patients with melanoma or NSCLC who received KEYTRUDA as a monotherapy, with the exception of increased incidences of facial edema and new or worsening hypothyroidism.
In KEYNOTE-204, KEYTRUDA was discontinued due to adverse reactions in 14% of 148 patients with cHL. Serious adverse reactions occurred in 30% of patients receiving KEYTRUDA; those ≥1% were pneumonitis, pneumonia, pyrexia, myocarditis, acute kidney injury, febrile neutropenia, and sepsis. Three patients died from causes other than disease progression: 2 from complications after allogeneic HSCT and 1 from unknown cause. The most common adverse reactions (≥20%) were upper respiratory tract infection (41%), musculoskeletal pain (32%), diarrhea (22%), and pyrexia, fatigue, rash, and cough (20% each).
In KEYNOTE-087, KEYTRUDA was discontinued due to adverse reactions in 5% of 210 patients with cHL. Serious adverse reactions occurred in 16% of patients; those ≥1% were pneumonia, pneumonitis, pyrexia, dyspnea, GVHD, and herpes zoster. Two patients died from causes other than disease progression: 1 from GVHD after subsequent allogeneic HSCT and 1 from septic shock. The most common adverse reactions (≥20%) were fatigue (26%), pyrexia (24%), cough (24%), musculoskeletal pain (21%), diarrhea (20%), and rash (20%).
In KEYNOTE-170, KEYTRUDA was discontinued due to adverse reactions in 8% of 53 patients with PMBCL. Serious adverse reactions occurred in 26% of patients and included arrhythmia (4%), cardiac tamponade (2%), myocardial infarction (2%), pericardial effusion (2%), and pericarditis (2%). Six (11%) patients died within 30 days of start of treatment. The most common adverse reactions (≥20%) were musculoskeletal pain (30%), upper respiratory tract infection and pyrexia (28% each), cough (26%), fatigue (23%), and dyspnea (21%).
In KEYNOTE-A39, when KEYTRUDA was administered in combination with enfortumab vedotin to patients with locally advanced or metastatic urothelial cancer (n=440), fatal adverse reactions occurred in 3.9% of patients, including acute respiratory failure (0.7%), pneumonia (0.5%), and pneumonitis/ILD (0.2%). Serious adverse reactions occurred in 50% of patients receiving KEYTRUDA in combination with enfortumab vedotin; the serious adverse reactions in ≥2% of patients were rash (6%), acute kidney injury (5%), pneumonitis/ILD (4.5%), urinary tract infection (3.6%), diarrhea (3.2%), pneumonia (2.3%), pyrexia (2%), and hyperglycemia (2%). Permanent discontinuation of KEYTRUDA occurred in 27% of patients. The most common adverse reactions (≥2%) resulting in permanent discontinuation of KEYTRUDA were pneumonitis/ILD (4.8%) and rash (3.4%). The most common adverse reactions (≥20%) occurring in patients treated with KEYTRUDA in combination with enfortumab vedotin were rash (68%), peripheral neuropathy (67%), fatigue (51%), pruritus (41%), diarrhea (38%), alopecia (35%), weight loss (33%), decreased appetite (33%), nausea (26%), constipation (26%), dry eye (24%), dysgeusia (21%), and urinary tract infection (21%).
In KEYNOTE-052, KEYTRUDA was discontinued due to adverse reactions in 11% of 370 patients with locally advanced or metastatic urothelial carcinoma. Serious adverse reactions occurred in 42% of patients; those ≥2% were urinary tract infection, hematuria, acute kidney injury, pneumonia, and urosepsis. The most common adverse reactions (≥20%) were fatigue (38%), musculoskeletal pain (24%), decreased appetite (22%), constipation (21%), rash (21%), and diarrhea (20%).
In KEYNOTE-045, KEYTRUDA was discontinued due to adverse reactions in 8% of 266 patients with locally advanced or metastatic urothelial carcinoma. The most common adverse reaction resulting in permanent discontinuation of KEYTRUDA was pneumonitis (1.9%). Serious adverse reactions occurred in 39% of KEYTRUDA-treated patients; those ≥2% were urinary tract infection, pneumonia, anemia, and pneumonitis. The most common adverse reactions (≥20%) in patients who received KEYTRUDA were fatigue (38%), musculoskeletal pain (32%), pruritus (23%), decreased appetite (21%), nausea (21%), and rash (20%).
In KEYNOTE-057, KEYTRUDA was discontinued due to adverse reactions in 11% of 148 patients with high-risk NMIBC. The most common adverse reaction resulting in permanent discontinuation of KEYTRUDA was pneumonitis (1.4%). Serious adverse reactions occurred in 28% of patients; those ≥2% were pneumonia (3%), cardiac ischemia (2%), colitis (2%), pulmonary embolism (2%), sepsis (2%), and urinary tract infection (2%). The most common adverse reactions (≥20%) were fatigue (29%), diarrhea (24%), and rash (24%).
Adverse reactions occurring in patients with MSI-H or dMMR CRC were similar to those occurring in patients with melanoma or NSCLC who received KEYTRUDA as a monotherapy.
In KEYNOTE-158 and KEYNOTE-164, adverse reactions occurring in patients with MSI-H or dMMR cancer were similar to those occurring in patients with other solid tumors who received KEYTRUDA as a single agent.
In KEYNOTE-811, when KEYTRUDA was administered in combination with trastuzumab, fluoropyrimidine- and platinum-containing chemotherapy, KEYTRUDA was discontinued due to adverse reactions in 6% of 217 patients with locally advanced unresectable or metastatic HER2+ gastric or GEJ adenocarcinoma. The most common adverse reaction resulting in permanent discontinuation was pneumonitis (1.4%). In the KEYTRUDA arm vs placebo, there was a difference of ≥5% incidence between patients treated with KEYTRUDA vs standard of care for diarrhea (53% vs 44%) and nausea (49% vs 44%).
In KEYNOTE-859, when KEYTRUDA was administered in combination with fluoropyrimidine- and platinum-containing chemotherapy, serious adverse reactions occurred in 45% of 785 patients. Serious adverse reactions in >2% of patients included pneumonia (4.1%), diarrhea (3.9%), hemorrhage (3.9%), and vomiting (2.4%). Fatal adverse reactions occurred in 8% of patients who received KEYTRUDA, including infection (2.3%) and thromboembolism (1.3%). KEYTRUDA was permanently discontinued due to adverse reactions in 15% of patients. The most common adverse reactions resulting in permanent discontinuation of KEYTRUDA (≥1%) were infections (1.8%) and diarrhea (1.0%). The most common adverse reactions (reported in ≥20%) in patients receiving KEYTRUDA in combination with chemotherapy were peripheral neuropathy (47%), nausea (46%), fatigue (40%), diarrhea (36%), vomiting (34%), decreased appetite (29%), abdominal pain (26%), palmar-plantar erythrodysesthesia syndrome (25%), constipation (22%), and weight loss (20%).
In KEYNOTE-590, when KEYTRUDA was administered with cisplatin and fluorouracil to patients with metastatic or locally advanced esophageal or GEJ (tumors with epicenter 1 to 5 centimeters above the GEJ) carcinoma who were not candidates for surgical resection or definitive chemoradiation, KEYTRUDA was discontinued due to adverse reactions in 15% of 370 patients. The most common adverse reactions resulting in permanent discontinuation of KEYTRUDA (≥1%) were pneumonitis (1.6%), acute kidney injury (1.1%), and pneumonia (1.1%). The most common adverse reactions (≥20%) with KEYTRUDA in combination with chemotherapy were nausea (67%), fatigue (57%), decreased appetite (44%), constipation (40%), diarrhea (36%), vomiting (34%), stomatitis (27%), and weight loss (24%).
Adverse reactions occurring in patients with esophageal cancer who received KEYTRUDA as a monotherapy were similar to those occurring in patients with melanoma or NSCLC who received KEYTRUDA as a monotherapy.
In KEYNOTE-A18, when KEYTRUDA was administered with CRT (cisplatin plus external beam radiation therapy [EBRT] followed by brachytherapy [BT]) to patients with FIGO 2014 Stage III-IVA cervical cancer, fatal adverse reactions occurred in 1.4% of 292 patients, including 1 case each (0.3%) of large intestinal perforation, urosepsis, sepsis, and vaginal hemorrhage. Serious adverse reactions occurred in 30% of patients; those ≥1% included urinary tract infection (2.7%), urosepsis (1.4%), and sepsis (1%). KEYTRUDA was discontinued for adverse reactions in 7% of patients. The most common adverse reaction (≥1%) resulting in permanent discontinuation was diarrhea (1%). For patients treated with KEYTRUDA in combination with CRT, the most common adverse reactions (≥10%) were nausea (56%), diarrhea (50%), vomiting (33%), urinary tract infection (32%), fatigue (26%), hypothyroidism (20%), constipation (18%), decreased appetite and weight loss (17% each), abdominal pain and pyrexia (12% each), hyperthyroidism, dysuria, rash (11% each), and pelvic pain (10%).
In KEYNOTE-826, when KEYTRUDA was administered in combination with paclitaxel and cisplatin or paclitaxel and carboplatin, with or without bevacizumab (n=307), to patients with persistent, recurrent, or first-line metastatic cervical cancer regardless of tumor PD-L1 expression who had not been treated with chemotherapy except when used concurrently as a radio-sensitizing agent, fatal adverse reactions occurred in 4.6% of patients, including 3 cases of hemorrhage, 2 cases each of sepsis and due to unknown causes, and 1 case each of acute myocardial infarction, autoimmune encephalitis, cardiac arrest, cerebrovascular accident, femur fracture with perioperative pulmonary embolus, intestinal perforation, and pelvic infection. Serious adverse reactions occurred in 50% of patients receiving KEYTRUDA in combination with chemotherapy with or without bevacizumab; those ≥3% were febrile neutropenia (6.8%), urinary tract infection (5.2%), anemia (4.6%), and acute kidney injury and sepsis (3.3% each).
KEYTRUDA was discontinued in 15% of patients due to adverse reactions. The most common adverse reaction resulting in permanent discontinuation (≥1%) was colitis (1%).
For patients treated with KEYTRUDA, chemotherapy, and bevacizumab (n=196), the most common adverse reactions (≥20%) were peripheral neuropathy (62%), alopecia (58%), anemia (55%), fatigue/asthenia (53%), nausea and neutropenia (41% each), diarrhea (39%), hypertension and thrombocytopenia (35% each), constipation and arthralgia (31% each), vomiting (30%), urinary tract infection (27%), rash (26%), leukopenia (24%), hypothyroidism (22%), and decreased appetite (21%).
For patients treated with KEYTRUDA in combination with chemotherapy with or without bevacizumab, the most common adverse reactions (≥20%) were peripheral neuropathy (58%), alopecia (56%), fatigue (47%), nausea (40%), diarrhea (36%), constipation (28%), arthralgia (27%), vomiting (26%), hypertension and urinary tract infection (24% each), and rash (22%).
In KEYNOTE-158, KEYTRUDA was discontinued due to adverse reactions in 8% of 98 patients with previously treated recurrent or metastatic cervical cancer. Serious adverse reactions occurred in 39% of patients receiving KEYTRUDA; the most frequent included anemia (7%), fistula, hemorrhage, and infections [except urinary tract infections] (4.1% each). The most common adverse reactions (≥20%) were fatigue (43%), musculoskeletal pain (27%), diarrhea (23%), pain and abdominal pain (22% each), and decreased appetite (21%).
In KEYNOTE-394, KEYTRUDA was discontinued due to adverse reactions in 13% of 299 patients with previously treated hepatocellular carcinoma. The most common adverse reaction resulting in permanent discontinuation of KEYTRUDA was ascites (2.3%). The most common adverse reactions in patients receiving KEYTRUDA (≥10%) were pyrexia (18%), rash (18%), diarrhea (16%), decreased appetite (15%), pruritus (12%), upper respiratory tract infection (11%), cough (11%), and hypothyroidism (10%).
In KEYNOTE-966, when KEYTRUDA was administered in combination with gemcitabine and cisplatin, KEYTRUDA was discontinued for adverse reactions in 15% of 529 patients with locally advanced unresectable or metastatic biliary tract cancer. The most common adverse reaction resulting in permanent discontinuation of KEYTRUDA (≥1%) was pneumonitis (1.3%). Adverse reactions leading to the interruption of KEYTRUDA occurred in 55% of patients. The most common adverse reactions or laboratory abnormalities leading to interruption of KEYTRUDA (≥2%) were decreased neutrophil count (18%), decreased platelet count (10%), anemia (6%), decreased white blood cell count (4%), pyrexia (3.8%), fatigue (3.0%), cholangitis (2.8%), increased ALT (2.6%), increased AST (2.5%), and biliary obstruction (2.3%).
In KEYNOTE-017 and KEYNOTE-913, adverse reactions occurring in patients with MCC (n=105) were generally similar to those occurring in patients with melanoma or NSCLC who received KEYTRUDA as a single agent.
In KEYNOTE-426, when KEYTRUDA was administered in combination with axitinib, fatal adverse reactions occurred in 3.3% of 429 patients. Serious adverse reactions occurred in 40% of patients, the most frequent (≥1%) were hepatotoxicity (7%), diarrhea (4.2%), acute kidney injury (2.3%), dehydration (1%), and pneumonitis (1%). Permanent discontinuation due to an adverse reaction occurred in 31% of patients; KEYTRUDA only (13%), axitinib only (13%), and the combination (8%); the most common were hepatotoxicity (13%), diarrhea/colitis (1.9%), acute kidney injury (1.6%), and cerebrovascular accident (1.2%). The most common adverse reactions (≥20%) were diarrhea (56%), fatigue/asthenia (52%), hypertension (48%), hepatotoxicity (39%), hypothyroidism (35%), decreased appetite (30%), palmar-plantar erythrodysesthesia (28%), nausea (28%), stomatitis/mucosal inflammation (27%), dysphonia (25%), rash (25%), cough (21%), and constipation (21%).
In KEYNOTE-564, when KEYTRUDA was administered as a single agent for the adjuvant treatment of renal cell carcinoma, serious adverse reactions occurred in 20% of patients receiving KEYTRUDA; the serious adverse reactions (≥1%) were acute kidney injury, adrenal insufficiency, pneumonia, colitis, and diabetic ketoacidosis (1% each). Fatal adverse reactions occurred in 0.2% including 1 case of pneumonia. Discontinuation of KEYTRUDA due to adverse reactions occurred in 21% of 488 patients; the most common (≥1%) were increased ALT (1.6%), colitis (1%), and adrenal insufficiency (1%). The most common adverse reactions (≥20%) were musculoskeletal pain (41%), fatigue (40%), rash (30%), diarrhea (27%), pruritus (23%), and hypothyroidism (21%).
In KEYNOTE-868, when KEYTRUDA was administered in combination with chemotherapy (paclitaxel and carboplatin) to patients with advanced or recurrent endometrial carcinoma (n=382), serious adverse reactions occurred in 35% of patients receiving KEYTRUDA in combination with chemotherapy, compared to 19% of patients receiving placebo in combination with chemotherapy (n=377). Fatal adverse reactions occurred in 1.6% of patients receiving KEYTRUDA in combination with chemotherapy, including COVID-19 (0.5%) and cardiac arrest (0.3%). KEYTRUDA was discontinued for an adverse reaction in 14% of patients. Adverse reactions occurring in patients treated with KEYTRUDA and chemotherapy were generally similar to those observed with KEYTRUDA alone or chemotherapy alone, with the exception of rash (33% all Grades; 2.9% Grades 3-4).
Adverse reactions occurring in patients with MSI-H or dMMR endometrial carcinoma who received KEYTRUDA as a single agent were similar to those occurring in patients with melanoma or NSCLC who received KEYTRUDA as a single agent.
Adverse reactions occurring in patients with TMB-H cancer were similar to those occurring in patients with other solid tumors who received KEYTRUDA as a single agent.
Adverse reactions occurring in patients with recurrent or metastatic cSCC or locally advanced cSCC were similar to those occurring in patients with melanoma or NSCLC who received KEYTRUDA as a monotherapy.
In KEYNOTE-522, when KEYTRUDA was administered with neoadjuvant chemotherapy (carboplatin and paclitaxel followed by doxorubicin or epirubicin and cyclophosphamide) followed by surgery and continued adjuvant treatment with KEYTRUDA as a single agent (n=778) to patients with newly diagnosed, previously untreated, high-risk early-stage TNBC, fatal adverse reactions occurred in 0.9% of patients, including 1 each of adrenal crisis, autoimmune encephalitis, hepatitis, pneumonia, pneumonitis, pulmonary embolism, and sepsis in association with multiple organ dysfunction syndrome and myocardial infarction. Serious adverse reactions occurred in 44% of patients receiving KEYTRUDA; those ≥2% were febrile neutropenia (15%), pyrexia (3.7%), anemia (2.6%), and neutropenia (2.2%). KEYTRUDA was discontinued in 20% of patients due to adverse reactions. The most common reactions (≥1%) resulting in permanent discontinuation were increased ALT (2.7%), increased AST (1.5%), and rash (1%). The most common adverse reactions (≥20%) in patients receiving KEYTRUDA were fatigue (70%), nausea (67%), alopecia (61%), rash (52%), constipation (42%), diarrhea and peripheral neuropathy (41% each), stomatitis (34%), vomiting (31%), headache (30%), arthralgia (29%), pyrexia (28%), cough (26%), abdominal pain (24%), decreased appetite (23%), insomnia (21%), and myalgia (20%).
In KEYNOTE-355, when KEYTRUDA and chemotherapy (paclitaxel, paclitaxel protein-bound, or gemcitabine and carboplatin) were administered to patients with locally recurrent unresectable or metastatic TNBC who had not been previously treated with chemotherapy in the metastatic setting (n=596), fatal adverse reactions occurred in 2.5% of patients, including cardio-respiratory arrest (0.7%) and septic shock (0.3%). Serious adverse reactions occurred in 30% of patients receiving KEYTRUDA in combination with chemotherapy; the serious reactions in ≥2% were pneumonia (2.9%), anemia (2.2%), and thrombocytopenia (2%). KEYTRUDA was discontinued in 11% of patients due to adverse reactions. The most common reactions resulting in permanent discontinuation (≥1%) were increased ALT (2.2%), increased AST (1.5%), and pneumonitis (1.2%). The most common adverse reactions (≥20%) in patients receiving KEYTRUDA in combination with chemotherapy were fatigue (48%), nausea (44%), alopecia (34%), diarrhea and constipation (28% each), vomiting and rash (26% each), cough (23%), decreased appetite (21%), and headache (20%).
Lactation
Because of the potential for serious adverse reactions in breastfed children, advise women not to breastfeed during treatment and for 4 months after the last dose.
Pediatric Use
In KEYNOTE-051, 173 pediatric patients (65 pediatric patients aged 6 months to younger than 12 years and 108 pediatric patients aged 12 years to 17 years) were administered KEYTRUDA 2 mg/kg every 3 weeks. The median duration of exposure was 2.1 months (range: 1 day to 25 months).
Adverse reactions that occurred at a ≥10% higher rate in pediatric patients when compared to adults were pyrexia (33%), leukopenia (30%), vomiting (29%), neutropenia (28%), headache (25%), abdominal pain (23%), thrombocytopenia (22%), Grade 3 anemia (17%), decreased lymphocyte count (13%), and decreased white blood cell count (11%).
Geriatric Use
Of the 564 patients with locally advanced or metastatic urothelial cancer treated with KEYTRUDA in combination with enfortumab vedotin, 44% (n=247) were 65-74 years and 26% (n=144) were 75 years or older. No overall differences in safety or effectiveness were observed between patients 65 years of age or older and younger patients. Patients 75 years of age or older treated with KEYTRUDA in combination with enfortumab vedotin experienced a higher incidence of fatal adverse reactions than younger patients. The incidence of fatal adverse reactions was 4% in patients younger than 75 and 7% in patients 75 years or older.
Additional Selected KEYTRUDA Indications in the U.S.
Melanoma
KEYTRUDA is indicated for the treatment of patients with unresectable or metastatic melanoma.
KEYTRUDA is indicated for the adjuvant treatment of adult and pediatric (12 years and older) patients with Stage IIB, IIC, or III melanoma following complete resection.
Malignant Pleural Mesothelioma
KEYTRUDA, in combination with pemetrexed and platinum chemotherapy, is indicated for the first-line treatment of adult patients with unresectable advanced or metastatic malignant pleural mesothelioma (MPM).
Head and Neck Squamous Cell Cancer
KEYTRUDA, in combination with platinum and fluorouracil (FU), is indicated for the first-line treatment of patients with metastatic or with unresectable, recurrent head and neck squamous cell carcinoma (HNSCC).
KEYTRUDA, as a single agent, is indicated for the first-line treatment of patients with metastatic or with unresectable, recurrent HNSCC whose tumors express PD-L1 [Combined Positive Score (CPS) ≥1] as determined by an FDA-approved test.
KEYTRUDA, as a single agent, is indicated for the treatment of patients with recurrent or metastatic HNSCC with disease progression on or after platinum-containing chemotherapy.
Primary Mediastinal Large B-Cell Lymphoma
KEYTRUDA is indicated for the treatment of adult and pediatric patients with refractory primary mediastinal large B-cell lymphoma (PMBCL), or who have relapsed after 2 or more prior lines of therapy. KEYTRUDA is not recommended for treatment of patients with PMBCL who require urgent cytoreductive therapy.
Urothelial Cancer
KEYTRUDA, in combination with enfortumab vedotin, is indicated for the treatment of adult patients with locally advanced or metastatic urothelial cancer.
KEYTRUDA, as a single agent, is indicated for the treatment of patients with locally advanced or metastatic urothelial carcinoma:
who are not eligible for any platinum-containing chemotherapy, or
who have disease progression during or following platinum-containing chemotherapy or within 12 months of neoadjuvant or adjuvant treatment with platinum-containing chemotherapy.
KEYTRUDA, as a single agent, is indicated for the treatment of patients with Bacillus Calmette-Guerin (BCG)-unresponsive, high-risk, non-muscle invasive bladder cancer (NMIBC) with carcinoma in situ (CIS) with or without papillary tumors who are ineligible for or have elected not to undergo cystectomy.
Microsatellite Instability-High or Mismatch Repair Deficient Cancer
KEYTRUDA is indicated for the treatment of adult and pediatric patients with unresectable or metastatic microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR) solid tumors, as determined by an FDA-approved test, that have progressed following prior treatment and who have no satisfactory alternative treatment options.
Microsatellite Instability-High or Mismatch Repair Deficient Colorectal Cancer
KEYTRUDA is indicated for the treatment of patients with unresectable or metastatic MSI-H or dMMR colorectal cancer (CRC) as determined by an FDA-approved test.
Gastric Cancer
KEYTRUDA, in combination with trastuzumab, fluoropyrimidine- and platinum-containing chemotherapy, is indicated for the first-line treatment of adults with locally advanced unresectable or metastatic HER2-positive gastric or gastroesophageal junction (GEJ) adenocarcinoma whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-approved test.
This indication is approved under accelerated approval based on tumor response rate and durability of response. Continued approval of this indication may be contingent upon verification and description of clinical benefit in the confirmatory trials.
KEYTRUDA, in combination with fluoropyrimidine- and platinum-containing chemotherapy, is indicated for the first-line treatment of adults with locally advanced unresectable or metastatic HER2-negative gastric or gastroesophageal junction (GEJ) adenocarcinoma.
Esophageal Cancer
KEYTRUDA is indicated for the treatment of patients with locally advanced or metastatic esophageal or gastroesophageal junction (GEJ) (tumors with epicenter 1 to 5 centimeters above the GEJ) carcinoma that is not amenable to surgical resection or definitive chemoradiation either:
in combination with platinum- and fluoropyrimidine-based chemotherapy, or
as a single agent after one or more prior lines of systemic therapy for patients with tumors of squamous cell histology that express PD-L1 (CPS ≥10) as determined by an FDA-approved test.
Cervical Cancer
KEYTRUDA, in combination with chemoradiotherapy (CRT), is indicated for the treatment of patients with FIGO 2014 Stage III-IVA cervical cancer.
KEYTRUDA, in combination with chemotherapy, with or without bevacizumab, is indicated for the treatment of patients with persistent, recurrent, or metastatic cervical cancer whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-approved test.
KEYTRUDA, as a single agent, is indicated for the treatment of patients with recurrent or metastatic cervical cancer with disease progression on or after chemotherapy whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-approved test.
Hepatocellular Carcinoma
KEYTRUDA is indicated for the treatment of patients with hepatocellular carcinoma (HCC) secondary to hepatitis B who have received prior systemic therapy other than a PD-1/PD-L1-containing regimen.
Biliary Tract Cancer
KEYTRUDA, in combination with gemcitabine and cisplatin, is indicated for the treatment of patients with locally advanced unresectable or metastatic biliary tract cancer (BTC).
Merkel Cell Carcinoma
KEYTRUDA is indicated for the treatment of adult and pediatric patients with recurrent locally advanced or metastatic Merkel cell carcinoma (MCC).
Renal Cell Carcinoma
KEYTRUDA, in combination with axitinib, is indicated for the first-line treatment of adult patients with advanced renal cell carcinoma (RCC).
KEYTRUDA is indicated for the adjuvant treatment of patients with RCC at intermediate-high or high risk of recurrence following nephrectomy, or following nephrectomy and resection of metastatic lesions.
Endometrial Carcinoma
KEYTRUDA, in combination with carboplatin and paclitaxel, followed by KEYTRUDA as a single agent, is indicated for the treatment of adult patients with primary advanced or recurrent endometrial carcinoma.
KEYTRUDA, as a single agent, is indicated for the treatment of adult patients with advanced endometrial carcinoma that is MSI-H or dMMR, as determined by an FDA-approved test, who have disease progression following prior systemic therapy in any setting and are not candidates for curative surgery or radiation.
Tumor Mutational Burden-High Cancer
KEYTRUDA is indicated for the treatment of adult and pediatric patients with unresectable or metastatic tumor mutational burden-high (TMB-H) [≥10 mutations/megabase (mut/Mb)] solid tumors, as determined by an FDA-approved test, that have progressed following prior treatment and who have no satisfactory alternative treatment options.
This indication is approved under accelerated approval based on tumor response rate and durability of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in the confirmatory trials. The safety and effectiveness of KEYTRUDA in pediatric patients with TMB-H central nervous system cancers have not been established.
Cutaneous Squamous Cell Carcinoma
KEYTRUDA is indicated for the treatment of patients with recurrent or metastatic cutaneous squamous cell carcinoma (cSCC) or locally advanced cSCC that is not curable by surgery or radiation.
Triple-Negative Breast Cancer
KEYTRUDA is indicated for the treatment of patients with high-risk early-stage triple-negative breast cancer (TNBC) in combination with chemotherapy as neoadjuvant treatment, and then continued as a single agent as adjuvant treatment after surgery.
KEYTRUDA, in combination with chemotherapy, is indicated for the treatment of patients with locally recurrent unresectable or metastatic TNBC whose tumors express PD-L1 (CPS ≥10) as determined by an FDA-approved test.
Merck’s focus on cancer
Every day, we follow the science as we work to discover innovations that can help patients, no matter what stage of cancer they have. As a leading oncology company, we are pursuing research where scientific opportunity and medical need converge, underpinned by our diverse pipeline of more than 25 novel mechanisms. With one of the largest clinical development programs across more than 30 tumor types, we strive to advance breakthrough science that will shape the future of oncology. By addressing barriers to clinical trial participation, screening and treatment, we work with urgency to reduce disparities and help ensure patients have access to high-quality cancer care. Our unwavering commitment is what will bring us closer to our goal of bringing life to more patients with cancer. For more information, visit
https://www.merck.com/research/oncology
.
About Merck
At Merck, known as MSD outside of the United States and Canada, we are unified around our purpose: We use the power of leading-edge science to save and improve lives around the world. For more than 130 years, we have brought hope to humanity through the development of important medicines and vaccines. We aspire to be the premier research-intensive biopharmaceutical company in the world – and today, we are at the forefront of research to deliver innovative health solutions that advance the prevention and treatment of diseases in people and animals. We foster a diverse and inclusive global workforce and operate responsibly every day to enable a safe, sustainable and healthy future for all people and communities. For more information, visit
www.merck.com
and connect with us on
X (formerly Twitter)
,
Facebook
,
Instagram
,
YouTube
and
LinkedIn
.
Forward-Looking Statement of Merck & Co., Inc., Rahway, N.J., USA
This news release of Merck & Co., Inc., Rahway, N.J., USA (the “company”) includes “forward-looking statements” within the meaning of the safe harbor provisions of the U.S. Private Securities Litigation Reform Act of 1995. These statements are based upon the current beliefs and expectations of the company’s management and are subject to significant risks and uncertainties. There can be no guarantees with respect to pipeline candidates that the candidates will receive the necessary regulatory approvals or that they will prove to be commercially successful. If underlying assumptions prove inaccurate or risks or uncertainties materialize, actual results may differ materially from those set forth in the forward-looking statements.
Risks and uncertainties include but are not limited to, general industry conditions and competition; general economic factors, including interest rate and currency exchange rate fluctuations; the impact of pharmaceutical industry regulation and health care legislation in the United States and internationally; global trends toward health care cost containment; technological advances, new products and patents attained by competitors; challenges inherent in new product development, including obtaining regulatory approval; the company’s ability to accurately predict future market conditions; manufacturing difficulties or delays; financial instability of international economies and sovereign risk; dependence on the effectiveness of the company’s patents and other protections for innovative products; and the exposure to litigation, including patent litigation, and/or regulatory actions.
The company undertakes no obligation to publicly update any forward-looking statement, whether as a result of new information, future events or otherwise. Additional factors that could cause results to differ materially from those described in the forward-looking statements can be found in the company’s Annual Report on Form 10-K for the year ended December 31, 2023 and the company’s other filings with the Securities and Exchange Commission (SEC) available at the SEC’s Internet site (
www.sec.gov
).
Please see Prescribing Information for KEYTRUDA (pembrolizumab) at
http://www.merck.com/product/usa/pi_circulars/k/keytruda/keytruda_pi.pdf
and Medication Guide for KEYTRUDA at
http://www.merck.com/product/usa/pi_circulars/k/keytruda/keytruda_mg.pdf
.
Media Contacts:
Robert Josephson
(203) 914-2372
Sienna Choi
(908) 873-4311
Investor Contacts:
Peter Dannenbaum
(732) 594-1579
Steven Graziano
(732) 594-1583
Source: Merck & Co., Inc.
临床3期临床结果
2022-10-18
CHICAGO and FORT WORTH, Texas, Oct. 18, 2022 (GLOBE NEWSWIRE) -- Actuate Therapeutics, Inc., a Chicago-based clinical-stage biopharmaceutical company advancing lead drug elraglusib (a novel GSK-3 inhibitor), today announced the addition of eminent cancer research leaders Roger B. Cohen, MD, Lee M. Ellis, MD, and John L. Marshall, MD to their Scientific Advisory Board. Dr. Roger Cohen is Professor of Medicine in the Hematology-Oncology Division at the University of Pennsylvania. He is a graduate of Harvard Medical School and completed internal medicine and hematology training at Mount Sinai Hospital (NY) followed by research fellowships at the Memorial Sloan-Kettering Cancer Center and National Institutes of Health and a medical oncology fellowship at the National Cancer Institute. He was a medical officer at the FDA Center for Biologics from 1989-1994 where he was Deputy Director, Division of Monoclonal Antibodies. Prior to his arrival at Penn, Dr. Cohen was Director of the Clinical Trials Office at the University of Virginia Cancer Center in Charlottesville and then Director of the Phase 1 Program at the Fox Chase Cancer Center where he also served as interim Medical Oncology Department Chair for more than 2 years. Dr. Cohen just completed a 12-year term as Associate Director Clinical Research for Penn’s Abramson Cancer Center. He continues to be an active clinical investigator with research interests that focus on evaluation of novel therapies, including monoclonal antibodies, immune therapies, and small molecule cell-signaling pathway inhibitors. He primarily sees patients with lung and head and neck cancer. Dr. Lee Ellis is the Ruben Distinguished Chair in Gastroenterology Cancer Research and a professor in the Departments of Surgical Oncology, and Molecular and Cellular Oncology at the University of Texas MD Anderson Cancer Center (MDACC). He recently completed serving as a member of the ASCO Board of Directors and he was an inaugural co-chair of the National Cancer Institute’s (NCI) National Clinical Trials Network Core Correlative Sciences Committee serving in the role until May, 2022. Dr. Ellis is Vice Chair for Translational Research for SWOG, a position he has held since 2015. He serves on the ECOG Data Monitoring Committee, a position he has held since 2005. In 2015, Ellis was named the Society of Surgical Oncology John Wayne Clinical Cancer Research Lecturer, and he received the Flance-Karl Award from the American Surgical Association in 2017 for his “seminal contributions in understanding colorectal cancer biology.” Recently, Dr. Ellis was named as the 2022 recipient of the AACR Daniel D. Von Hoff Award for Outstanding Contributions to Education and Training in Cancer Research. He currently serves on several data safety monitoring committees and is a deputy editor for JAMA Oncology. Dr. John Marshall received his training at Duke University, the University of Louisville, and Georgetown University. Dr. Marshall is an internationally recognized expert in new drug development for GI cancer, with expertise in phase I, II, and III trial design, and has served as Principal Investigator for more than one hundred clinical trials throughout his career. While he has an interest in many areas of cancer research, his primary focus is on the use of precision medicine in treating cancer. Dr. Marshall has become an outspoken advocate for GI cancer patients and the importance of clinical research participation. In 2009, he established the Otto J. Ruesch Center for the Cure of GI Cancers, an organization solely focused on improving the lives of GI cancer patients through innovative research, personalized medicine, and focused advocacy. In 2015, Dr. Marshall both established and directed the Precision Oncology Alliance, a national alliance established to study the impact of molecular profiling on cancer research, value and outcomes. He currently is serving as the Oncology CMO for Indivumed, creating a global precision medicine research network. In 2020, he and his wife published “Off Our Chests, A Candid Tour Through the World of Cancer”. In 2022, he became the inaugural Physician Executive Director of the MedStar Washington DC Integrated Hematology Oncology Division. Drs. Cohen, Ellis and Marshall join world-class cancer research experts Daniel Billadeau, PhD and Patricia LoRusso, DO on Actuate’s SAB: Dr. Daniel Billadeau is Professor and Chair of the Department of Immunology, and Deputy Director for Basic Science in the Mayo Clinic Comprehensive Cancer Center. Dr. Billadeau obtained his graduate degree from the University of Minnesota and did his postdoctoral training in molecular immunology/biochemistry before beginning his independent research career in the Division of Oncology Research at Mayo Clinic. Dr. Billadeau’s research efforts have focused on identifying molecular targets in pancreatic ductal adenocarcinoma (PDAC). His group was the first to demonstrate that glycogen synthase kinase-3β (GSK-3) is overexpressed and localized to the nucleus in PDAC where it regulates tumor cell survival and chemoresistance. His group is actively engaged in understanding the role of GSK-3 in tumor development in vivo and is working toward defining mechanisms of resistance to elraglusib. Dr. Patricia LoRusso is Associate Director of Developmental Therapeutics, Yale Cancer Center. Dr. LoRusso has been a practicing academic medical oncologist performing clinical/translational research in early phase clinical trials for over 30 years. She has had continuous NIH/NCI peer review funding for 28 years, having held a U-grant for early phase clinical trials through the NCI Cancer Therapy Evaluation Program (CTEP) for 26 years. She has also collaborated on numerous other grants and have been an investigator in P01 and P30 funding mechanisms. Understanding the need for team science, she has participated in P50 mechanisms and has been awarded team science grants through such organizations as Stand Up to Cancer (Co-Leader: Melanoma Dream Team), the Department of Defense (DOD) and the Komen Foundation (Co-leader, KG111063: Targeting Stem Cells in Triple-Negative Breast Cancer (TNBC) in Different Racial Populations). Dr. LoRusso has also been involved in many service disciplines at the NCI. In addition, Dr. LoRusso has served in leadership positions of several other organizations, including the Board of Directors and numerous scientific and education committees of the American Association for Cancer Research (AACR), the education and scientific committees of the American Society of Clinical Oncology (ASCO), and the steering committee for the Food and Drug Administration (FDA) Accelerating Anticancer Agent Development and Validation Workshop. She has worked closely with Cancer Research United Kingdom (CRUK), a UK Welcome Trust - the second largest funding agency for cancer research. She is currently serving a 3-year term as the chair of their New Agents Committee (NAC). Working closely over the past 3 decades with patients suffering from advanced malignancies, Dr. LoRusso has become an advocate, not only for cancer researchers and clinicians, but more importantly for the patients and their caregivers. Having experienced at a young age the death of her own parents from cancer, she understands the urgent need for new cancer discoveries and the potential for longevity and quality of life. The Actuate SAB was established to shepherd the development and advancement of Actuate’s best-in-class GSK-3β inhibitors into and through clinical trials in patients with advanced cancer. With elraglusib (9-ING-41), Actuate’s lead program, advancing into Phase 2 and registration path clinical trials, the Advisory Board will play a larger role in advising on target diseases for prioritization and clinical trial design. "We are honored and pleased to collaborate with an Advisory Board that possesses such a high caliber of research and development expertise and accomplishment. The engagement of our talented and experienced scientific advisors in the design and execution of our clinical development plans will maximize the potential impact of elraglusib for the benefit of our patients” said Andrew P. Mazar, Ph.D., Chief Operating Officer of Actuate Therapeutics. “Each of the SAB members is a well-known thought leader in the development of new oncology therapeutics, and we appreciate the expertise they bring to our team." “The members of our SAB are among the premier oncology researchers and clinicians in the world” said Daniel Schmitt, President & CEO of Actuate Therapeutics. “We view their interest in Actuate Therapeutics’ programs as strong validation of the importance and potential impact our compounds could play in the treatment of advanced cancer.” About Actuate Therapeutics, Inc.Actuate is a clinical-stage biopharmaceutical company focused on the development and commercialization of novel therapeutics for high impact cancers and inflammatory diseases. Actuate’s lead drug, elraglusib (a novel GSK-3 inhibitor), targets molecular pathways in cancer that are involved in promoting tumor growth and resistance to conventional cancer drugs such as chemotherapy. Elraglusib is also emerging as a mediator of anti-tumor immunity through the inhibition of multiple immune checkpoints and the regulation of immune cell function. For additional information, please visit the Company’s website at http://www.actuatetherapeutics.com Public Relations Dept.,Actuate Therapeutics, Inc.+1 (847) 986-4190info@actuatetherapeutics.com
抗体小分子药物ASCO会议AACR会议合作
100 项与 Mount Sinai Hospital (Canada) 相关的药物交易
登录后查看更多信息
100 项与 Mount Sinai Hospital (Canada) 相关的转化医学
登录后查看更多信息
组织架构
使用我们的机构树数据加速您的研究。
登录
或

管线布局
2026年06月09日管线快照
管线布局中药物为当前组织机构及其子机构作为药物机构进行统计,早期临床1期并入临床1期,临床1/2期并入临床2期,临床2/3期并入临床3期
其他
1
登录后查看更多信息
当前项目
| 药物(靶点) | 适应症 | 全球最高研发状态 |
|---|---|---|
Biological isolate (United Paragon Associates) ( NK2R x mGluRs ) | 重度抑郁症 更多 | 无进展 |
登录后查看更多信息
药物交易
使用我们的药物交易数据加速您的研究。
登录
或
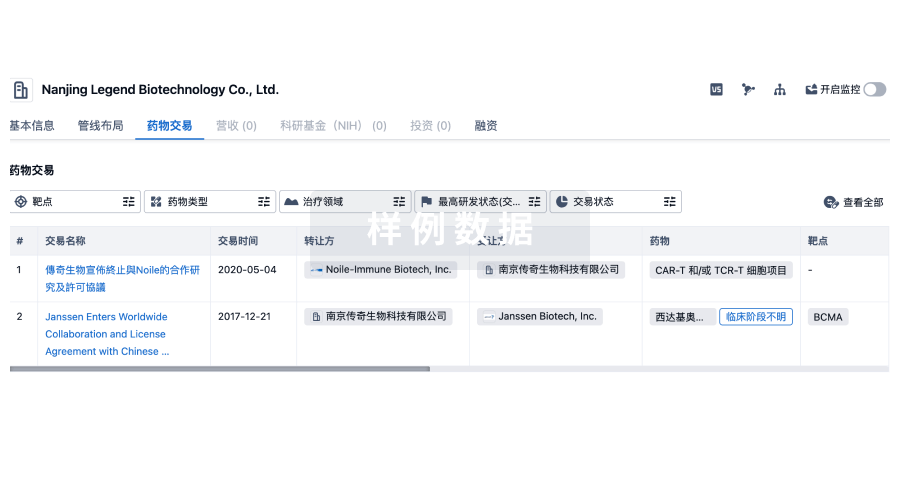
转化医学
使用我们的转化医学数据加速您的研究。
登录
或
营收
使用 Synapse 探索超过 36 万个组织的财务状况。
登录
或
科研基金(NIH)
访问超过 200 万项资助和基金信息,以提升您的研究之旅。
登录
或
投资
深入了解从初创企业到成熟企业的最新公司投资动态。
登录
或
融资
发掘融资趋势以验证和推进您的投资机会。
登录
或
生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用