预约演示
更新于:2025-05-07
Urinary tract obstruction
尿路梗阻
更新于:2025-05-07
基本信息
别名 OBSTRUCTIVE UROPATHY、OBSTRUCTIVE UROPATHY, NOS、Obstructive Uropathy + [39] |
简介 Uropathy that is caused by an impediment to flow in the urinary tract. |
关联
3
项与 尿路梗阻 相关的药物靶点- |
作用机制- |
非在研适应症- |
最高研发阶段批准上市 |
首次获批国家/地区 美国 |
首次获批日期2023-01-30 |
靶点- |
作用机制- |
非在研适应症- |
最高研发阶段批准上市 |
首次获批国家/地区 美国 |
首次获批日期1990-06-15 |
作用机制 α1A-AR拮抗剂 [+1] |
在研机构- |
在研适应症- |
最高研发阶段无进展 |
首次获批国家/地区- |
首次获批日期1800-01-20 |
50
项与 尿路梗阻 相关的临床试验NCT06921733
Non-invasive Evaluation of Kidneys in Patient With Congenital Anomalies of the Kidney and Urinary Tract (CAKUT) Using Ultrasound Localization Microscopy
This clinical study aims to non-invasively visualize perfusion and microvascularization, as well as individual glomeruli, using Ultrasound Localization Microscopy (ULM) and CEUS in patients with congenital anomalies of the kidney and urinary tract (CAKUT).
开始日期2025-04-05 |
NCT06336304
NXT Urodynamics System Post-Market Clinical Follow-up Clinical Investigation
NXT Urodynamics System Post-Market Clinical Follow-up Clinical Investigation to Confirm Ongoing Safety and Performance in Urodynamic Patient Sub-Populations
开始日期2024-06-18 |
申办/合作机构- |
NCT06037356
Prostatic Urethral Lift Versus Transurethral Resection of Prostate in Benign Prostatic Hyperplasia Patients With Urinary Retention
The study will be a prospective, randomized controlled trial comparing prostatic urethral lift (PUL) versus transurethral resection of prostate (TURP) in benign prostate hyperplasia (BPH) patients with urinary retention. The primary objective of this study is to compare the catheter-free rates of PUL vs TURP. Secondary objectives include comparison of complications rates, cost effectiveness, patient satisfactory, symptom scores, quality of life measures and urodynamic parameters.
开始日期2024-06-01 |
申办/合作机构 |
100 项与 尿路梗阻 相关的临床结果
登录后查看更多信息
100 项与 尿路梗阻 相关的转化医学
登录后查看更多信息
0 项与 尿路梗阻 相关的专利(医药)
登录后查看更多信息
7,046
项与 尿路梗阻 相关的文献(医药)2025-05-01·Urology Case Reports
Management of ureteral IgG4-Related Disease: The great masquerader
Article
作者: Hennes, David ; Yuminaga, Yuigi
2025-04-18·Cureus
Hybrid Open Anterior and Laparoscopic Repair Using Self-Gripping Mesh for Parastomal Hernia Following Ileal Conduit With Extensive Intra-abdominal Adhesions: A Case Report
Article
作者: Kitamura, Yoh ; Tsujinaka, Shingo ; Miura, Tomoya ; Shibata, Chikashi ; Sato, Yoshihiro
2025-04-04·Medicine
Ureteral orifice edema and stenting challenges in diabetic patients: Lessons from a case report
Article
作者: Guo, Shuang ; Hu, Yunxi ; Huang, Denghui ; Yang, Wenjiang ; Liu, Zhongwei
24
项与 尿路梗阻 相关的新闻(医药)2025-01-13
·动脉网
近日,在派尔特医疗、威森特医疗机器人的战略协助下,来自美国的AquaBeam水刀机器人在上海申德医院成功装机。这是AquaBeam水刀机器人产品在中国大陆市场的首次商业化落地,为中国良性前列腺增生(BPH)患者带来了无热损伤、精确高效、并发症少的创新疗法。
AquaBeam水刀机器人在上海申德医院成功装机现场
从左至右依次为:上海申德医院总经理 Vivian Lao,屹然泌尿医生集团发起人、首席专家黄翼然 教授,派尔特医疗董事长戴景萌,派尔特医疗 CEO Kerry,美国PROCEPT公司亚太总监 Kin
派尔特医疗作为中国微创外科器械耗材行业的头部企业,密切关注行业前沿技术尤其是手术机器人赛道,投资孵化了苏州威森特医疗机器人有限公司。威森特手术机器人目前已在腔镜手术机器人研发方面取得突破成就,其产品有望成为首个以大外科为核心的手术机器人。
同时,派尔特医疗在国内外医疗市场具有深厚的积淀和优良的行业资源,能够保障手术机器人的成功商业化。此次派尔特医疗、威森特手术机器人有限公司与屹然泌尿医生集团全方位战略合作,将中国第一台AquaBeam水刀机器人商业化落地于上海申德医院,是派尔特医疗在手术机器人赛道中的又一次拓展。
作为BPH专科手术机器人的行业鼻祖,美国PROCEPT BioRobotics公司(股票代码为:“PRCT”,以下简称“PROCEPT”)成立于2007年,总部位于美国加利福尼亚州雷德伍德城,致力于开发泌尿外科的变革性解决方案,推出全球独家的水刀机器人产品AuqaBeam。通过持续的技术创新和市场拓展,PROCEPT已成为泌尿外科手术机器人领域的领军企业。
AquaBeam水刀机器人
01
无热切除、范围精准,用首创水刀机器人疗法最大化保护性功能与排尿功能
良性前列腺增生症(BPH)是一种男性常见疾病,具有高发性、与年龄增长相关性。据流行病学调查数据显示,50岁以上的男性50%患有BPH,60岁男性中为60%,70岁男性中为70%。
在世界大部分地区,BPH的绝对负担正以惊人速度增长。《柳叶刀-老龄健康》(The Lancet Healthy Longevity)的研究显示,2000—2019年全球现BPH患病例数增加了70.5%。随着人均寿命延长,预计BPH的绝对负担在未来几年将继续上升。
目前,BPH的疗法主要包括药物治疗、微创手术治疗、植入物和支架以及新兴技术治疗,但都存在一定局限性:
当前BPH主要疗法的原理及局限性 动脉网制图
总的来看,上述疗法均不能很好地兼顾各种尺寸大小的前列腺增生患者的治疗与性功能以及排尿功能的保护。目前,BPH治疗主要面临五大痛点:
● 症状与治疗效果的平衡:治疗需要改善症状,同时尽量减少性功能障碍和其他并发症;
● 复发率高:许多患者在治疗后症状会逐渐复发,需要二次干预;
● 患者个性化治疗选择不足:目前方法众多,但患者病情复杂多样,无法实现“一刀切”;
●治疗率低:因为病耻感或担心影响性功能、尿失禁等并发症,很多患者不能及时治疗;
● 依从性和心理负担:长期药物治疗或术后并发症给患者带来心理压力。
为了解决上述临床痛点,为BPH患者提供更加优质的治疗选择,PROCEPT 在行业内开创了用水刀机器人治疗BPH的先河。该疗法通过AquaBeam机器人实施良性前列腺增生(BPH)切除手术,被称为“Aquablation®治疗”。
Aquablation®治疗因其无热能的切除方式、精确定制的切除范围,可以在保护性功能与排尿功能的同时,最大限度缓解不同尺寸前列腺增生患者的症状,且并发症发生概率更低,对男性性功能影响极小,有望成为下一代治疗BPH的金标准。
02
全球首个成功商业化的影像引导、机器人辅助、无热水刀的BPH疗法,已被纳入多项国际泌尿外科指南
Aquablation®治疗是全球首个也是唯一一个成功商业化的影像引导、机器人辅助、无热水刀的BPH治疗方法。同时,该疗法的核心技术支撑——AquaBeam水刀机器人系统是全球首个获得FDA批准的、具有自动化组织切除技术的手术机器人,于2017年获得欧洲CE认证、2018年获得FDA批准。
AquaBeam水刀机器人系统结合了实时多维成像、个性化治疗规划、机器人自动化无热水射流切除控制技术,能够精确且快速地去除前列腺组织。具体而言,该系统具有以下优势:
实时影像引导。AquaBeam水刀机器人结合实时超声影像(经直肠超声TRUS)和手术计划系统,可以使外科医生实时观察膀胱、尿道和前列腺的全貌,识别关键解剖结构。据悉,这种实时影像引导在BPH手术中为首创,使医生能够精准规划切除范围;此外,AquaBeam具有动态监测功能,术中实时影像监测可确保手术安全,降低损伤健康组织的风险;
个性化治疗规划。AquaBeam水刀机器人将影像系统与机器人手术计划系统无缝集成。通过影像引导,医生可根据患者前列腺的具体形状和体积定制治疗方案,避免过度或不足切除;同时,在计算机辅助规划的支持下,医生能在保护膀胱颈、精阜和外括约肌的同时,最大程度地切除病变组织;
机器人自动化无热水射流切除。AquaBeam水刀机器人具有自动化无热水射流切除控制技术,避免了术者操作个体差异和周围组织的热损伤,确保了组织的精确切除和极低的并发症发生率。手术过程中,水刀的切除路径由机器人根据医生规划自动执行,消除了手动操作可能带来的误差,减少因医生疲劳或经验不足导致的术中变数,提供了超高稳定性和可重复性。机器人技术和水动力学切除的结合,使AquaBeam成为全球首个实现完全自动化前列腺组织切除的系统。
在AquaBeam水刀机器人系统的赋能下,Aquablation®治疗显示出显著的技术先进性和优越性,已在多项临床研究中显示出安全性和有效性。研究显示,无论前列腺的大小和形状如何,该疗法均能提供持久的症状缓解,其临床优势包括:
● 无热损伤:AquaBeam通过高压水流切割组织,无需使用热能,避免了因热能引起的周围组织损伤;
● 精确性:机器人技术的应用减少了人为误差,确保了前列腺组织的精确、一致和可预测的切除;
● 高效率和短恢复时间:AquaBeam的水刀切除效率远高于传统手术,几分钟内即可完成治疗。且由于水刀的精确切割,出血量大幅降低,患者术后恢复快。相较于其他BPH治疗技术,AquaBeam手术时间短,术后患者的导尿管拔除时间显著提前、住院时间显著减少;
● 极低不可逆并发症发生率:由于采用实时超声和膀胱镜多模影像引导,外科医生能够精确地避开关键区域,减少尿失禁、射精功能障碍和勃起功能障碍等不可逆并发症的发生;
● 长期疗效:临床研究表明,Aquablation®治疗在三年内可提供长期的症状缓解。Aquablation®治疗和TURP的三年随机对照试验结果显示,Aquablation®治疗对勃起功能、整体性满意度没有影响,几乎所有BPH患者在治疗后保留射精功能,且99%的患者在治疗后没有出现尿失禁,显著优于TURP治疗。
目前,Aquablation®治疗已被纳入多项国际泌尿外科指南,在美国泌尿外科学会(AUA)、欧洲泌尿外科学会(EAU)的BPH治疗指南中均被推荐为有效的治疗选择。此外,日本泌尿学会开展的一项针对日本男性的研究表明,使用AquaBeam水刀机器人系统进行的Aquablation®治疗是可重复的、安全且有效的,为日本BPH患者提供了显著的下尿路症状和生活质量改善。
同时,Aquablation®治疗得到了全球诸多泌尿外科医生和学术机构的积极评价。
美国圣约翰健康中心(Sant John 's Health Center)的泌尿科主任Mehran Movassaghi医生指出:“该疗法提供了持久的症状缓解,并且并发症发生率低。无论患者前列腺的大小或形状如何,通过Aquablation®治疗,我们不再需要在手术的持久效果和患者术后可能出现的性功能负面影响之间做出妥协……手术后一到两周内,患者的身体状况即可恢复到健康水平。”
日本顺天堂大学医院(Juntendo Univ. Hospital)泌尿外科教授堀江茂雄博士评论道:“传统上,在良性前列腺增生手术中,外科医生通过有限的膀胱镜影像对前列腺进行可视化。在Aquablation®疗法中,我们利用实时超声成像和膀胱镜影像,能够更准确地识别关键解剖结构,制定个性化的治疗计划。”
03
已完成超50,000例BPH手术,加快在华商业化步伐
迄今,AquaBeam水刀机器人已在全球30多个国家和地区开展商业化布局,覆盖美国、德国、英国、法国、加拿大、日本、韩国等地。截至2024年,AquaBeam水刀机器人在全球范围内的装机量近500台,已成功完成了超过50,000例BPH手术。
2023年底,AquaBeam水刀机器人已获得NMPA批准,由派尔特医疗作为指定的战略合作代理商、威森特手术机器人作为中国总发货商,协助其在中国市场的商业化推广。
目前,AquaBeam水刀机器人在中国大陆参与了多次学术会议交流、完成演示手术十多例,其中BPH患者前列腺的体积最大为200克、最小为55克、平均值为87克。其切除效率高、精准低损伤、尿路梗阻症状改善显著等临床优势获得了参与专家们的高度认可。
著名泌尿外科专家、屹然泌尿医生集团发起人黄翼然教授对于首台AquaBeam水刀机器人落地表示:“新技术引进后,后续的随访、观察也很重要。我们团队能很好地完成这些工作,使得疗效更好…… 水刀机器人手术在海外开展很多、有很好的适用性。但每个手术都有它独特的地方,我们希望能够做好水刀机器人疗法的落地开花结果,更好地造福患者,让他们排尿更通畅,越做越好。”
在亚洲最早开展水刀机器人手术的专家、香港中文大学医院张源津教授:“我操刀过经尿道水蒸气热疗手术、钬激光前列腺剜除术,我觉得水刀机器人在安全性与保护性功能方面表现更好。如果向病人讲明各种手术方法的优缺点,我的大部分病人会选择水刀机器人手术。”
上海仁济医院泌尿外科孙杰教授表示:“在了解了水刀机器人后,我觉得这一疗法的另外一个优势是减少了操作者的经验、能力不同导致的疗效差异。可能操刀两年与二十几年的医生的手术切除效果是一致的,这就是这项技术比较厉害的地方…… 水刀机器人落地后可以开展真实世界研究,通过建立本地的数据进一步形成诊疗规范和培训体系,能够帮助这项创新技术更好地被中国医生们掌握,更好地服务患者。”
北京协和医院泌尿外科张玉石教授表示:“AquaBeam水刀机器人的治疗方式,不仅提高了BPH手术的精准度,还为患者提供了一种创伤更小、恢复更快的治疗选择。这项技术的应用符合我们追求卓越医疗服务的宗旨,未来有望在国内泌尿外科领域推广。”
山东大学齐鲁医院泌尿外科史本康教授评价:“水刀机器人通过人工智能和机器人技术控制高压水流,进行精准组织切除,以其精准、高效和安全的特点,显著减少了传统手术带来的并发症和术后恢复时间。”
AquaBeam水刀机器人系统参加泌尿外科学术会议并进行了表演手术直播
301海南医院史涛坪主任为一位170克超大前列腺增生患者实施了水刀机器人前列腺剜除手术。AquaBeam水刀机器人高效、精准、出血极少、疗效显著等临床优势获得高度认可。有现场专家表示,自己用激光进行同样情况的前列腺剜除手术,至少要进行一个多小时的切除操作。本次现场观看让他对AquaBeam水刀机器人的治疗确定性、极高效率、出血少等临床优势印象深刻,希望能够引进更多中国医院。
展望未来,在派尔特医疗稳步推进实施的市场化、国际化、证券化的经营战略助力下,派尔特医疗、威森特手术机器人将持续推动AquaBeam水刀机器人在中国大陆的商业化拓展,帮助国内泌尿外科医生们使用AquaBeam水刀机器人和Aquablation®疗法造福更多BPH患者。
目前,威森特手术机器人有限公司有融资需求,如果您对威森特及其产品感兴趣,请与我们取得联系:
近
期
推
荐
声明:动脉网所刊载内容之知识产权为动脉网及相关权利人专属所有或持有。未经许可,禁止进行转载、摘编、复制及建立镜像等任何使用。
动脉网,未来医疗服务平台
2024-12-11
DEERFIELD, Ill.--(
BUSINESS WIRE
)--Baxter International Inc. (NYSE:BAX), a global leader in injectables, anesthesia and drug compounding, today announced five new injectable pharmaceutical product launches in the U.S., joining the previous five launches
announced
in April of this year and marking a total of 10 U.S. injectable product launches in 2024.
“
Our Pharmaceuticals teams are relentlessly focused on bringing differentiated products to market that support our customers in helping to address vital patient needs,” said Alok Sonig, executive vice president and group president, Pharmaceuticals, at Baxter. “
We look forward to further accelerating our impact with a robust innovation pipeline across our key therapeutic areas, including critical care, anti-infectives, pain and oncology.”
The five recent product launches within Baxter’s Pharmaceuticals portfolio in the U.S. include the following. For all products, please see full Indications, including Limitations of Use, Important Risk Information and links to full Prescribing Information below.
Micafungin in 0.9% Sodium Chloride Injection single-dose container is indicated for use in adult and pediatric patients to treat Candida infections and for prophylaxis of Candida infections in patients undergoing hematopoietic stem cell transplantation for whom appropriate dosing with this formulation can be achieved. Micafungin uses Baxter’s proprietary container technology. Baxter offers Micafungin in 50 mg/50 mL, 100 mg/100 mL and 150 mg/150 mL strengths.
Cyclophosphamide Injection is an alkylating drug indicated for treatment of adults and pediatric patients with various malignant diseases and is frequently used in combination with other oncology medications. This liquid cyclophosphamide product requires further dilution before use. Baxter offers Cyclophosphamide Injection in 500 mg/2.5 mL and 1000 mg/5 mL strengths in Multiple-Dose Vials.
Pantoprazole Sodium in 0.9% Sodium Chloride Injection is a proton pump inhibitor indicated in adults for the short-term treatment (seven to 10 days) of gastroesophageal reflux disease (GERD) associated with a history of erosive esophagitis (EE) and pathological hypersecretion conditions including Zollinger-Ellison (ZE) Syndrome. Pantoprazole uses Baxter’s proprietary container technology. Baxter offers Pantoprazole in 40 mg/50 mL, 40 mg/100 mL and 80 mg/100 mL strengths.
Cefazolin in Dextrose Injection, USP now available in a new 3 g/150 mL strength, is a single-dose, first generation cephalosporin antibacterial indicated for adults and pediatric patients to treat various infections caused by susceptible organisms and for perioperative prophylaxis. Cefazolin uses Baxter’s proprietary container technology. Baxter offers Cefazolin in 1 g/50 mL, 2 g/100 mL and 3 g/150 mL strengths.
Levetiracetam in Sodium Chloride Injection is an anti-epileptic drug indicated for adjunct therapy in adult patients for partial onset seizures, myoclonic seizures, and tonic-clonic seizures. Levetiracetam uses Baxter’s proprietary container technology and is now offered in 500 mg/100 mL, 1000 mg/100 mL and 1500 mg/100 mL strengths.
Ready-to-use formats of standard concentrations of commonly prescribed drugs may offer operational efficiencies for healthcare providers. Compounding a drug for patient use is a multi-step, manual process that requires oversight by pharmacy staff. A ready-to-use product can simplify the preparation process and support patient safety by reducing the chance of contamination
2
and avoiding potential errors that may occur when medications are compounded.
3
These five newly launched products are now available for use in the U.S.
About Baxter Pharmaceuticals
Baxter is a global leader in specialty injectables, drug compounding and anesthesia that addresses unmet patient needs in the therapeutic areas of pain, critical care, anti-infectives and oncology. Baxter’s comprehensive pharmaceuticals portfolio contains injectables (including ready-to-use products), inhaled gases and compounded medications, and is designed to expand access to products that simplify medication preparation and support patient safety. Pharmaceuticals employees across the globe are focused on driving customer-centered innovation, bringing new and differentiated products and delivery platforms to market, and helping patients receive the medications they need.
About
Baxter
Every day, millions of patients, caregivers and healthcare providers rely on Baxter’s leading portfolio of diagnostic, critical care, kidney care, nutrition, hospital and surgical products used across patient homes, hospitals, physician offices and other sites of care. For more than 90 years, we’ve been operating at the critical intersection where innovations that save and sustain lives meet the healthcare providers who make it happen. With products, digital health solutions and therapies available in more than 100 countries, Baxter’s employees worldwide are now building upon the company’s rich heritage of medical breakthroughs to advance the next generation of transformative healthcare innovations. To learn more, visit
www.baxter.com
and follow us on
X
,
LinkedIn
and
Facebook
.
Micafungin in 0.9% Sodium Chloride Injection
Indications
Micafungin in Sodium Chloride Injection is an echinocandin indicated in adult and pediatric patients for:
Treatment of Candidemia, Acute Disseminated Candidiasis, Candida Peritonitis and Abscesses in adult and pediatric patients 4 months of age and older for whom appropriate dosing with this formulation can be achieved.
Treatment of Candidemia, Acute Disseminated Candidiasis, Candida Peritonitis and Abscesses without meningoencephalitis and/or ocular dissemination in pediatric patients younger than 4 months of age for whom appropriate dosing with this formulation can be achieved.
Treatment of Esophageal Candidiasis in adult and pediatric patients 4 months of age and older for whom appropriate dosing with this formulation can be achieved.
Prophylaxis of Candida Infections in adult and pediatric patients 4 months of age and older undergoing Hematopoietic Stem Cell Transplantation (HSCT) for whom appropriate dosing with this formulation can be achieved.
Limitations of Use:
The safety and effectiveness of Micafungin in Sodium Chloride Injection have
not
been established for the treatment of candidemia
with
meningoencephalitis and/or ocular dissemination in pediatric patients younger than 4 months of age as a higher dose may be needed.
Micafungin in Sodium Chloride Injection has not been adequately studied in patients with endocarditis, osteomyelitis or meningoencephalitis due to Candida.
The efficacy of Micafungin in Sodium Chloride Injection against infections caused by fungi other than Candida has not been established.
Important
Risk
Information
Contraindications: Micafungin in Sodium Chloride Injection is contraindicated in persons with known hypersensitivity to micafungin sodium, any component of Micafungin in Sodium Chloride Injection, or other echinocandins.
Hypersensitivity Reactions: Anaphylaxis and anaphylactoid reactions (including shock) have been observed. Discontinue Micafungin in Sodium Chloride Injection and administer appropriate treatment.
Hematological Effects: Acute intravascular hemolysis and hemoglobinuria was seen in a healthy volunteer during infusion of Micafungin for Injection (200 mg) and oral prednisolone (20 mg). Cases of significant hemolysis and hemolytic anemia have also been reported in patients treated with Micafungin for Injection. Patients who develop clinical or laboratory evidence of hemolysis or hemolytic anemia during therapy should be monitored closely for evidence of worsening of these conditions and evaluated for the risk/benefit of continuing therapy.
Hepatic Effects: Laboratory abnormalities in liver function tests have been seen in healthy volunteers and patients treated with Micafungin. In some patients with serious underlying conditions who were receiving Micafungin along with multiple concomitant medications, clinical hepatic abnormalities have occurred, and isolated cases of significant hepatic impairment, hepatitis, and hepatic failure have been reported. Monitor hepatic function. Discontinue if severe dysfunction occurs.
Renal Effects: Elevations in BUN and creatinine; isolated cases of renal impairment or acute renal failure have been reported. Monitor renal function.
Infusion and Injection Site Reactions: Possible histamine-mediated symptoms have been reported with Micafungin for Injection, including rash, pruritus, facial swelling, and vasodilatation. Slow the infusion rate if infusion reaction occurs. Injection site reactions, including phlebitis and thrombophlebitis have been reported, at Micafungin for Injection doses of 50 to 150 mg/day. These reactions tended to occur more often in patients receiving Micafungin for Injection via peripheral intravenous administration.
High Sodium Load: Each 50, 100, and 150 mL Galaxy container contains 200, 400, and 600 mg of sodium, respectively. Avoid use in patients with congestive heart failure, elderly patients, and patients requiring restricted sodium intake.
Adverse Reactions:
Most common adverse reactions across adult and pediatric clinical trials for all indications include diarrhea, nausea, vomiting, abdominal pain, pyrexia, thrombocytopenia, neutropenia, and headache.
In pediatric patients younger than 4 months of age, the following additional common adverse reactions were reported at an incidence rate of ≥15%: hypokalemia, acidosis, sepsis, anemia, and oxygen saturation decreased.
Drug Interactions:
Monitor for sirolimus, itraconazole or nifedipine toxicity, and dosage of sirolimus, itraconazole or nifedipine should be reduced, if necessary.
Pregnancy: Based on animal data, Micafungin in Sodium Chloride Injection may cause fetal harm. Advise pregnant women of the risk to the fetus.
Please see accompanying full Prescribing Information for
Micafungin in 0.9% Sodium Chloride Injection
.
Cyclophosphamide Injection
Indications
Cyclophosphamide Injection is an alkylating drug indicated for treatment of adult and pediatric patients with:
Malignant Diseases: malignant lymphomas: Hodgkin's disease, lymphocytic lymphoma, mixed-cell type lymphoma, histiocytic lymphoma, Burkitt's lymphoma; multiple myeloma, leukemias, mycosis fungoides, neuroblastoma, adenocarcinoma of ovary, retinoblastoma, breast carcinoma.
Important Risk Information
Contraindications
Hypersensitivity: Cyclophosphamide is contraindicated in patients who have a history of severe hypersensitivity reactions to it, any of its metabolites, or to other components of the product. Anaphylactic reactions including death have been reported. Possible cross-sensitivity with other alkylating agents can occur.
Urinary outflow obstruction
Myelosuppression, Immunosuppression, Bone Marrow Failure and Infections: Cyclophosphamide can cause myelosuppression, bone marrow failure, and severe immunosuppression which may lead to serious and sometimes fatal infections, including sepsis and septic shock. Latent infections can be reactivated. Monitoring of complete blood counts is essential during cyclophosphamide treatment so that the dose can be adjusted, if needed.
Urinary Tract and Renal Toxicity: Hemorrhagic cystitis, pyelitis, ureteritis, and hematuria have been reported with cyclophosphamide. Discontinue cyclophosphamide therapy in case of severe hemorrhagic cystitis. Urotoxicity may require interruption of cyclophosphamide treatment or cystectomy. Urotoxicity can be fatal. Before starting treatment, exclude or correct any urinary tract obstructions. Urinary sediment should be checked regularly for the presence of erythrocytes and other signs of urotoxicity and/or nephrotoxicity. Cyclophosphamide Injection should be used with caution, if at all, in patients with active urinary tract infections. Aggressive hydration with forced diuresis and frequent bladder emptying can reduce the frequency and severity of bladder toxicity. Mesna has been used to prevent severe bladder toxicity.
Cardiotoxicity: Myocarditis, myopericarditis, pericardial effusion including cardiac tamponade, and congestive heart failure, which may be fatal, have been reported with cyclophosphamide therapy. Supraventricular arrhythmias and ventricular arrhythmias have been reported after treatment with regimens that included cyclophosphamide. The risk of cardiotoxicity may be increased with high doses of cyclophosphamide, in patients with advanced age, and in patients with previous radiation treatment to the cardiac region and/or previous or concomitant treatment with other cardiotoxic agents. Monitor patients, especially those with risk factors for cardio toxicity or pre-existing cardiac disease.
Pulmonary Toxicity: Pneumonitis, pulmonary fibrosis, pulmonary veno-occlusive disease and other forms of pulmonary toxicity leading to respiratory failure have been reported during and following treatment with cyclophosphamide. Late onset pneumonitis appears to be associated with increased mortality. Monitor patients for signs and symptoms of pulmonary toxicity.
Secondary Malignancies: Cyclophosphamide is genotoxic. Secondary malignancies have been reported in patients treated with cyclophosphamide-containing regimens. The risk of bladder cancer may be reduced by prevention of hemorrhagic cystitis.
Veno-occlusive Liver Disease (VOD): VOD including fatal outcome has been reported in patients receiving cyclophosphamide-containing regimens. A cytoreductive regimen in preparation for bone marrow transplantation that consists of cyclophosphamide in combination with whole-body irradiation, busulfan, or other agents has been identified as a major risk factor. VOD has also been reported to develop gradually in patients receiving long-term low-dose immunosuppressive doses of cyclophosphamide.
Alcohol Content: The alcohol content in a dose of Cyclophosphamide Injection may affect the central nervous system. Consideration should be given to the alcohol content on the ability to drive or use machines immediately after the infusion.
Embryo-Fetal Toxicity: Cyclophosphamide Injection can cause fetal harm when administered to a pregnant woman. Exposure to cyclophosphamide during pregnancy may cause birth defects, miscarriage, fetal growth retardation, and fetotoxic effects in the newborn. Advise females of reproductive potential to use effective contraception during treatment and for up to 1 year after completion of therapy. Advise male patients with female partners of reproductive potential to use effective contraception during treatment and for 4 months after completion of therapy.
Infertility: Male and female reproductive function and fertility may be impaired in patients being treated with Cyclophosphamide Injection. Cyclophosphamide interferes with oogenesis and spermatogenesis. It may cause sterility in both sexes. Cyclophosphamide-induced sterility may be irreversible in some patients.
Adverse Reactions: Most common adverse reactions reported are neutropenia, febrile neutropenia, fever, alopecia, nausea, vomiting, and diarrhea.
Lactation: Advise not to breastfeed.
Renal Patients: Monitor for toxicity in patients with moderate and severe renal impairment.
Please see accompanying full Prescribing Information for
Cyclophosphamide Injection
.
Pantoprazole Sodium in 0.9% Sodium Chloride Injection
Indications
Pantoprazole Sodium in 0.9% Sodium Chloride Injection is a proton pump inhibitor (PPI) indicated in adults for the following:
Short-term treatment (7 to 10 days) of gastroesophageal reflux disease (GERD) associated with a history of erosive esophagitis (EE).
Pathological hypersecretion conditions including Zollinger-Ellison (ZE) Syndrome.
Important Risk Information
Contraindications
Patients with a known hypersensitivity to any component of the formulation or to substituted benzimidazoles. Hypersensitivity reactions may include anaphylaxis, anaphylactic shock, angioedema, bronchospasm, acute tubulointerstitial nephritis, and urticaria.
Patients receiving rilpivirine-containing products.
Presence of Gastric Malignancy: In adults, symptomatic response to therapy does not preclude the presence of gastric malignancy. Consider additional follow-up and diagnostic testing in adult patients who have a suboptimal response or an early symptomatic relapse after completing treatment with a PPI. In older patients, also consider an endoscopy.
Injection Site Reactions: Thrombophlebitis was reported in association with the administration of another intravenous pantoprazole sodium product.
Potential for Exacerbation of Zinc Deficiency: Pantoprazole Sodium in 0.9% Sodium Chloride Injection contains edetate disodium (the salt form of EDTA), a chelator of metal ions including zinc. Therefore, zinc supplementation should be considered in patients who are prone to zinc deficiency. Caution should be used when other EDTA containing products are also co-administered intravenously.
Acute Tubulointerstitial Nephritis (TIN): Has been observed in patients taking PPIs and may occur at any point during PPI therapy. Patients may present with varying signs and symptoms from symptomatic hypersensitivity reactions to non-specific symptoms of decreased renal function. Discontinue Pantoprazole Sodium in 0.9% Sodium Chloride Injection and evaluate patients with suspected acute TIN.
Clostridioides difficile
-associated diarrhea: Published observational studies suggest that PPI therapy may be associated with an increased risk of
Clostridioides difficile
-associated diarrhea, especially in hospitalized patients. This diagnosis should be considered for diarrhea that does not improve. Patients should use the lowest dose and shortest duration of PPI therapy appropriate to the condition being treated.
Bone Fracture: Several published observational studies suggest that PPI therapy may be associated with an increased risk for osteoporosis-related fractures of the hip, wrist, or spine. The risk of fracture was increased in patients who received high-dose, defined as multiple daily doses, and long-term PPI therapy (a year or longer). Patients should use the lowest dose and shortest duration of PPI therapy appropriate to the condition being treated. Patients at risk for osteoporosis-related fractures should be managed according to established treatment guidelines.
Severe Cutaneous Adverse Reactions: Including erythema multiforme, Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), drug reaction with eosinophilia and systemic symptoms (DRESS), and acute generalized exanthematous pustulosis (AGEP) have been reported in association with the use of PPIs. Discontinue Pantoprazole Sodium in 0.9% Sodium Chloride Injection at the first signs or symptoms of severe cutaneous adverse reactions or other signs of hypersensitivity and consider further evaluation.
Cutaneous Lupus Erythematosus (CLE) and Systemic Lupus Erythematosus (SLE): Have been reported in patients taking PPIs, including pantoprazole sodium. These events have occurred as both new onset and an exacerbation of existing autoimmune disease. The majority of PPI-induced lupus erythematous cases were CLE. Avoid administration of PPIs for longer than medically indicated. If signs or symptoms consistent with CLE or SLE are noted in patients, discontinue the drug and refer the patient to the appropriate specialist for evaluation. Most patients improve with discontinuation of the PPI alone in 4 to 12 weeks.
Hepatic Effects: Mild, transient transaminase elevations have been observed in clinical studies with another intravenous pantoprazole sodium product. The clinical significance of this finding in a large population of subjects is unknown.
Hypomagnesemia and Mineral Metabolism: Hypomagnesemia, symptomatic and asymptomatic, has been reported rarely in patients treated with PPIs for at least three months, and in most cases after a year of therapy. Serious adverse events include tetany, arrhythmias, and seizures. Hypomagnesemia may lead to hypocalcemia and/or hypokalemia and may exacerbate underlying hypocalcemia in at-risk patients. In most patients, treatment of hypomagnesemia required magnesium replacement and discontinuation of the PPI.
Fundic Gland Polyps: PPI use is associated with an increased risk of fundic gland polyps that increases with long-term use, especially beyond one year. Most PPI users who developed fundic gland polyps were asymptomatic. Use the shortest duration of PPI therapy appropriate to the condition being treated.
Adverse Reactions: Most common adverse reactions (incidence > 2%) are headache, diarrhea, nausea, abdominal pain, vomiting, flatulence, dizziness, and arthralgia.
Drug Interactions: See the full prescribing information for a list of clinically important drug interactions.
Pregnancy: Based on animal data, may cause fetal harm.
Please see accompanying full Prescribing Information for
Pantoprazole Sodium in 0.9% Sodium Chloride Injection
.
Cefazolin in Dextrose Injection, USP
Indications
Cefazolin in Dextrose Injection is a cephalosporin antibacterial indicated for:
Treatment of respiratory tract infections in adults and pediatric patients for whom appropriate dosing with this formulation can be achieved.
Limitations of Use
: Injectable benzathine penicillin is considered the drug of choice in treatment and prevention of streptococcal infections, including the prophylaxis of rheumatic fever.
Treatment of the following infections caused by susceptible isolates of the designated microorganisms in adult and pediatric patients for whom appropriate dosing with this formulation can be achieved: Urinary tract infections; Skin and skin structure infections; Biliary tract infections; Bone and joint infections; Genital infections; Septicemia; Endocarditis.
Perioperative prophylaxis in adults and pediatric patients aged 10 – 17 years old for whom appropriate dosing with this formulation can be achieved.
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Cefazolin in Dextrose Injection and other antibacterial drugs, Cefazolin in Dextrose Injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria.
Important Risk Information
Contraindications: Hypersensitivity to Cefazolin or other cephalosporin class antibacterial drugs, penicillins, or other beta-lactams.
Hypersensitivity Reactions to Cefazolin, Cephalosporins, Penicillins, or Other Beta-lactams: Serious and occasionally fatal hypersensitivity (anaphylactic) reactions have been reported in patients receiving beta-lactam antibacterial drugs. Before therapy with Cefazolin in Dextrose Injection, careful inquiry should be made to determine whether the patient has had previous immediate hypersensitivity reactions to cefazolin, cephalosporins, penicillins, or carbapenems. Exercise caution if this product is to be given to penicillin-sensitive patients because cross-hypersensitivity among betalactam antibacterial drugs may occur in up to 10% of patients with a history of penicillin allergy. If an allergic reaction occurs, discontinue the drug.
Seizures in Patients with Renal Impairment: Seizures may occur particularly in patients with renal impairment when the dosage is not reduced appropriately. Discontinue Cefazolin in Dextrose Injection if seizures occur or make appropriate dosage adjustments in patients with renal impairment. Anticonvulsant therapy should be continued in patients with known seizure disorders
Clostridioides difficile
-associated Diarrhea (CDAD): May range in severity from mild diarrhea to fatal colitis. CDAD must be considered in all patients who present with diarrhea following antibiotic use. If CDAD is suspected or confirmed, ongoing antibiotic use not directed against
C. difficile
may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibiotic treatment of
C. difficile
, and surgical evaluation should be instituted as clinically indicated.
Prothrombin Activity: Cefazolin in Dextrose Injection may be associated with a fall in prothrombin activity. Those at risk include patients with renal or hepatic impairment or poor nutritional state, as well as patients receiving a protracted course of antimicrobial therapy, and patients previously stabilized on anticoagulant therapy. Prothrombin time should be monitored in patients at risk and exogenous vitamin K administered as indicated.
Adverse Reactions:
Adult and Pediatric Patients
: Most common adverse reactions: gastrointestinal (nausea, vomiting, diarrhea), and allergic reactions (anaphylaxis, urticaria, skin rash).
Pediatric Patients with Perioperative Prophylaxis
: The most frequently reported adverse reactions (incidence ≥ 5%) were nausea, infusion site pain, and headache.
Drug Interactions:
Probenecid: The renal excretion of cefazolin is inhibited by probenecid. Co-administration of probenecid with Cefazolin in Dextrose Injection is not recommended.
Please see accompanying full Prescribing Information for
Cefazolin in Dextrose Injection, USP
.
Levetiracetam in Sodium Chloride Injection
Indications
Levetiracetam in Sodium Chloride Injection is indicated for adjunct therapy in adults (≥16 years of age) with the following seizure types when oral administration is temporarily not feasible:
Partial-onset seizures
Myoclonic seizures in patients with juvenile myoclonic epilepsy
Primary generalized tonic-clonic seizures
Important Risk Information
Contraindications: Levetiracetam in Sodium Chloride Injection is contraindicated in patients with a hypersensitivity to levetiracetam. Reactions have included anaphylaxis and angioedema.
Psychiatric Reactions: Behavioral abnormalities including psychotic symptoms, suicidal ideation, irritability, and aggressive behavior have been observed. Monitor patients for psychiatric signs and symptoms.
Somnolence and Fatigue: Monitor patients for these symptoms and advise patients not to drive or operate machinery until they have gained sufficient experience on levetiracetam.
Anaphylaxis and Angioedema: Levetiracetam can cause anaphylaxis or angioedema. In some reported cases, reactions were life-threatening and required emergency treatment. If a patient develops signs or symptoms of anaphylaxis or angioedema, levetiracetam should be discontinued and the patient should seek immediate medical attention.
Serious Dermatological Reactions: Serious reactions, including Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN), have been reported in patients treated with levetiracetam. Recurrence of the serious skin reactions following rechallenge has also been reported. Levetiracetam should be discontinued at the first sign of a rash, unless the rash is clearly not drug-related. If signs or symptoms suggest SJS/TEN, use of this drug should not be resumed, and alternative therapy should be considered.
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS)/Multiorgan Hypersensitivity: This has been reported in patients taking antiepileptic drugs, including levetiracetam. These events can be fatal or life-threatening, particularly if diagnosis and treatment do not occur as early as possible. DRESS typically, although not exclusively, presents with fever, rash, lymphadenopathy, and/or facial swelling, in association with other organ system involvement, sometimes resembling an acute viral infection. If such signs or symptoms are present, the patient should be evaluated immediately. Levetiracetam should be discontinued if an alternative etiology for the signs or symptoms cannot be established.
Coordination Difficulties: Monitor for ataxia, abnormal gait, and incoordination. Patients should be monitored for these signs and symptoms and advised not to drive or operate machinery until they have gained sufficient experience on levetiracetam.
Withdrawal Seizures: Levetiracetam must be gradually withdrawn. But if withdrawal is needed because of a serious adverse reaction, rapid discontinuation can be considered.
Hematologic Abnormalities: Hematologic abnormalities occurred in clinical trials and included decreases in white blood cell, neutrophil, and red blood cells counts; decreases in hemoglobin and hematocrit; and increases in eosinophil counts. Cases of agranulocytosis, pancytopenia, and thrombocytopenia have been reported in the post-marketing setting. A complete blood count is recommended in patients experiencing significant weakness, pyrexia, recurrent infections, or coagulation disorders.
Adverse Reactions: Most common adverse reactions (incidence in levetiracetam-treated patients is ≥5% more than in placebo-treated patients) include: somnolence, asthenia, infection, and dizziness.
Pregnancy: Plasma levels of levetiracetam may be decreased; monitor closely during pregnancy. Based on animal data, may cause fetal harm. Encourage women who are taking levetiracetam injection during pregnancy to enroll in the North American Antiepileptic Drug (NAAED) pregnancy registry.
Renal Impairment: Dosage adjustment is recommended for patients with impaired renal function and supplemental doses should be given to patients after dialysis.
Please see accompanying full Prescribing Information for
Levetiracetam in Sodium Chloride Injection
.
This release includes forward-looking statements concerning Micafungin in 0.9% Sodium Chloride Injection, Cyclophosphamide Injection, Pantoprazole Sodium in 0.9% Sodium Chloride Injection, Cefazolin in Dextrose Injection, USP and Levetiracetam in Sodium Chloride Injection, including potential benefits associated with the use of these products. The statements are based on assumptions about many important factors, including the following, which could cause actual results to differ materially from those in the forward-looking statements: demand for and market acceptance for new and existing products; product development risks; inability to create additional production capacity in a timely manner or the occurrence of other manufacturing or supply difficulties (including as a result of natural disasters, public health crises and epidemics/pandemics, regulatory actions or otherwise); satisfaction of regulatory and other requirements; actions of regulatory bodies and other governmental authorities; product quality, manufacturing or supply, or patient safety issues; changes in law and regulations; and other risks identified in Baxter's most recent filing on Form 10-K and Form 10-Q and other SEC filings, all of which are available on Baxter's website. Baxter does not undertake to update its forward-looking statements.
Baxter is a registered trademark of Baxter International Inc.
1
Includes line extensions.
2
Mercaldi CJ, Lanes S, Bradt J. Comparative risk of bloodstream infection in hospitalized patients receiving intravenous medication by open, point-of-care, or closed delivery systems. Am J Health-Syst Pharm. 2013;70:957-965.
3
Billstein-Leber M, Carrillo CJD, Cassano AT, Moline K, Robertson JJ. ASHP Guidelines on Preventing Medication Errors in Hospitals. Am J Health Syst Pharm. 2018;75(19):1493-1517.
US-PH121-240011 (v3.0) 12/2024
上市批准临床结果
2024-05-04
60% Complete Response Rate after JELMYTO Induction
25% of Patients Received a Ureteral Stent
PRINCETON, N.J.--(BUSINESS WIRE)-- UroGen Pharma Ltd. (Nasdaq: URGN), a biotech company dedicated to developing and commercializing novel solutions that treat urothelial and specialty cancers, today highlights the results of a sub-analysis from a real-world patient cohort review of JELMYTO (mitomycin) for pyelocalyceal solution presented at the American Urological Association Meeting 2024 in San Antonio, TX. The study includes a stepwise approach to retrograde administration of JELMYTO and reports that retrograde administration of JELMYTO in the clinic (n=20) produced a 60% complete response rate in patients and ureteral stents were placed in 25% of patients, which is lower than the rate reported in the pivotal OLYMPUS study.
“The retrograde instillation of JELMYTO for the clinical management of low-grade upper tract urothelial cancer is FDA-approved and offers a safe and efficacious mode of administration, and this study in a real-world population adds to the growing body of evidence supporting retrograde administration in appropriate patients,” said Khurshid Ridwan Ghani, MBChB, MS, FRCS, Professor of Urology, Director, Michigan Urological Surgery Improvement Collaborative. “The stepwise approach outlined in this study offers valuable insights into achieving positive efficacy and safety outcomes, in the clinic with the procedure done under local anesthesia, with notable durability of response observed.”
In the OLYMPUS study all enrolled patients (n=71) received retrograde administration of JELMYTO. In the OLYMPUS study 58% (n=41) of patients achieved a complete response, defined as the absence of tumor lesions 3 months after initiation of JELMYTO treatment assessed by urine cytology and ureteroscopy. Ureteric obstruction* was reported in 58% (n=41) of patients treated with JELMYTO and 88% (n=36) of that subgroup went on to receive ureteral stent placement. For additional information about the retrograde administration of JELMYTO, consult the JELMYTO Instructions for Administration accompanying the Full Prescribing Information.
In this retrospective real-world study, 20 patients with a mean tumor burden of 1.67 cm received at least one dose of JELMYTO via the retrograde mode of administration, of which 16 (80%) completed six instillations. Twelve patients (60%) had a complete response and seven patients (35%) received at least one dose of monthly maintenance therapy, of which five demonstrated durability of response (4 patients remained tumor-free at 14.25-month follow-up and 1 patient at 24 months). Six patients (30%) had an adverse event related to the urinary system. Ureteral stents were placed in five patients for stenosis (25%); of which four were transient, with no subsequent obstruction. Only one patient (5%) required permanent stenting. Three patients who were unable to tolerate the retrograde approach had antegrade administration of JELMYTO via a nephrostomy tube.
"These findings emphasize the versatility of JELMYTO administration and also underscore the significance of continuous research and innovation in elevating treatment standards for urothelial cancers,” remarked Mark Schoenberg, M.D., Chief Medical Officer at UroGen.
The limitations of this sub-analysis include the small sample size, the retrospective design, lack of a control group, and the lack of a centralized pathology review and standardized clinicopathologic assessment.
To further explore the full potential of JELMYTO for the treatment of patients with upper tract urothelial cancers (UTUC), investigators are in the process of enrolling the prospective and retrospective uTRACT Registry to capture data in a large-scale, standardized manner to report further on patient outcomes following JELMYTO treatment including longitudinal follow-up.
*Includes hydronephrosis, obstructive uropathy, pelvic-ureteric obstruction, ureteric obstruction, ureteric stenosis, and urinary tract obstruction.
About JELMYTO
JELMYTO® (mitomycin) for pyelocalyceal solution is a mitomycin-containing reverse thermal gel containing 4 mg mitomycin per mL gel indicated for the treatment of adult patients with low grade-UTUC (LG-UTUC). JELMYTO is a viscous liquid when cooled and becomes a semi-solid gel at body temperature. The drug slowly dissolves over four to six hours after instillation and is removed from the urinary tract by normal urine flow and voiding. It is approved for administration in a retrograde manner via ureteral catheter or antegrade through nephrostomy tube. The delivery system allows the initial liquid to coat and conform to the upper urinary tract anatomy. The eventual semisolid gel allows for chemoablative therapy to remain in the collecting system for four to six hours without immediately being diluted or washed away by urine flow.
About Upper Tract Urothelial Cancer (UTUC)
Urothelial cancer is the ninth most common cancer globally and the eighth most lethal neoplasm in men in the U.S. Between five percent and ten percent of primary urothelial cancers originate in the ureter or renal pelvis and are collectively referred to as UTUC. In the U.S., there are approximately 6,000 - 7,000 new or recurrent LG-UTUC patients annually. Most cases are diagnosed in patients over 70 years old, and these older patients often face comorbidities. There are limited treatment options for UTUC, with the most common being endoscopic surgery or nephroureterectomy (removal of the entire kidney and ureter). These treatments can lead to a high rate of recurrence and relapse.
About UroGen Pharma Ltd.
UroGen is a biotech company dedicated to developing and commercializing innovative solutions that treat urothelial and specialty cancers because patients deserve better options. UroGen has developed RTGel® reverse-thermal hydrogel, a proprietary sustained-release, hydrogel-based platform technology that has the potential to improve the therapeutic profiles of existing drugs. UroGen’s sustained release technology is designed to enable longer exposure of the urinary tract tissue to medications, making local therapy a potentially more effective treatment option. Our first product to treat LG-UTUC and investigational treatment UGN-102 (mitomycin) for intravesical solution for patients with low-grade non-muscle invasive bladder cancer are designed to ablate tumors by non-surgical means. UroGen is headquartered in Princeton, NJ with operations in Israel. Visit to learn more or follow us on X (Twitter), @UroGenPharma.
APPROVED USE FOR JELMYTO
JELMYTO® is a prescription medicine used to treat adults with a type of cancer of the lining of the upper urinary tract including the kidney called low-grade Upper Tract Urothelial Cancer (LG-UTUC).
IMPORTANT SAFETY INFORMATION
You should not receive JELMYTO if you have a hole or tear (perforation) of your bladder or upper urinary tract.
Before receiving JELMYTO, tell your healthcare provider about all your medical conditions, including if you:
are pregnant or plan to become pregnant. JELMYTO can harm your unborn baby. You should not become pregnant during treatment with JELMYTO. Tell your healthcare provider right away if you become pregnant or think you may be pregnant during treatment with JELMYTO. Females who are able to become pregnant: You should use effective birth control (contraception) during treatment with JELMYTO and for 6 months after the last dose. Males being treated with JELMYTO: If you have a female partner who is able to become pregnant, you should use effective birth control (contraception) during treatment with JELMYTO and for 3 months after the last dose.
are breastfeeding or plan to breastfeed. It is not known if JELMYTO passes into your breast milk. Do not breastfeed during treatment with JELMYTO and for 1 week after the last dose.
Tell your healthcare provider if you take water pills (diuretic).
How will I receive JELMYTO?
Your healthcare provider will tell you to take a medicine called sodium bicarbonate before each JELMYTO treatment.
You will receive your JELMYTO dose from your healthcare provider 1 time a week for 6 weeks. It is important that you receive all 6 doses of JELMYTO according to your healthcare provider’s instructions. If you miss any appointments, call your healthcare provider as soon as possible to reschedule your appointment. Your healthcare provider may recommend up to an additional 11 monthly doses.
JELMYTO is given to your kidney through a tube called a catheter.
During treatment with JELMYTO, your healthcare provider may tell you to take additional medicines or change how you take your current medicines.
After receiving JELMYTO:
JELMYTO may cause your urine color to change to a violet to blue color. Avoid contact between your skin and urine for at least 6 hours.
To urinate, males and females should sit on a toilet and flush the toilet several times after you use it. After going to the bathroom, wash your hands, your inner thighs, and genital area well with soap and water.
Clothing that comes in contact with urine should be washed right away and washed separately from other clothing.
JELMYTO may cause serious side effects, including:
Swelling and narrowing of the tube that carries urine from the kidney to the bladder (ureteric obstruction). If you develop swelling and narrowing, and to protect your kidney from damage, your healthcare provider may recommend the placement of a small plastic tube (stent) in the ureter to help the kidney drain. Tell your healthcare provider right away if you develop side pain or fever during treatment with JELMYTO.
Bone marrow problems. JELMYTO can affect your bone marrow and can cause a decrease in your white blood cell, red blood cell, and platelet counts. Your healthcare provider will do blood tests prior to each treatment to check your blood cell counts during treatment with JELMYTO. Your healthcare provider may need to temporarily or permanently stop JELMYTO if you develop bone marrow problems during treatment with JELMYTO.
The most common side effects of JELMYTO include: urinary tract infection, blood in your urine, side pain, nausea, trouble with urination, kidney problems, vomiting, tiredness, stomach (abdomen) pain.
You are encouraged to report negative side effects of prescription drugs to the FDA. Visit or call 1800FDA1088. You may also report side effects to UroGen Pharma at 1-855-987-6436.
Please see JELMYTO Full Prescribing Information, including the Patient Information, for additional information.
Forward-Looking Statements
This press release contains forward-looking statements as that term is defined in the Private Securities Litigation Reform Act of 1995, including, without limitation, statements regarding the results from the real-world study of the retrograde instillation of JELMYTO enhancing the prospects of favorable patient outcomes; the enrollment of the prospective and retrospective data from the uTRACT Patient Registry and plans to analyze and report on such data; the estimated patient population for UTUC and LG-UTUC; the potential of UroGen’s proprietary RTGel technology to improve therapeutic profiles of existing drugs; and UroGen’s sustained release technology making local delivery potentially more effective as compared to other treatment options. These statements are subject to a number of risks, uncertainties and assumptions, including, but not limited to: results from the real-world study of the retrograde instillation of JELMYTO may not be indicative of results that may be observed in future clinical practice and may differ from additional analysis of the data from the study or uTRACT Patient Registry; potential safety and other complications from JELMYTO use in diverse UTUC patient types; the ability to maintain regulatory approval; the ability to obtain and maintain adequate intellectual property rights and adequately protect and enforce such rights; complications associated with commercialization activities; UroGen’s RTGel technology may not perform as expected; and UroGen may not successfully develop and receive regulatory approval of any other product that incorporates UroGen’s RTGel technology. In light of these risks and uncertainties, and other risks and uncertainties that are described in the Risk Factors section of UroGen’s Annual Report on Form 10-K for the year ended December 31, 2023, filed with the SEC on March 14, 2024 (which is available at ), the events and circumstances discussed in such forward-looking statements may not occur, and UroGen’s actual results could differ materially and adversely from those anticipated or implied thereby. Any forward-looking statements speak only as of the date of this press release and are based on information available to UroGen as of the date of this release.
View source version on businesswire.com:
Contacts
INVESTOR CONTACT:
Vincent Perrone
Senior Director, Investor Relations
vincent.perrone@urogen.com
609-460-3588 ext. 1093
MEDIA CONTACT:
Cindy Romano
Director, Corporate Communications
cindy.romano@urogen.com
609-460-3583 ext. 1083
Source: UroGen Pharma Ltd.
View this news release online at:
临床结果上市批准
分析
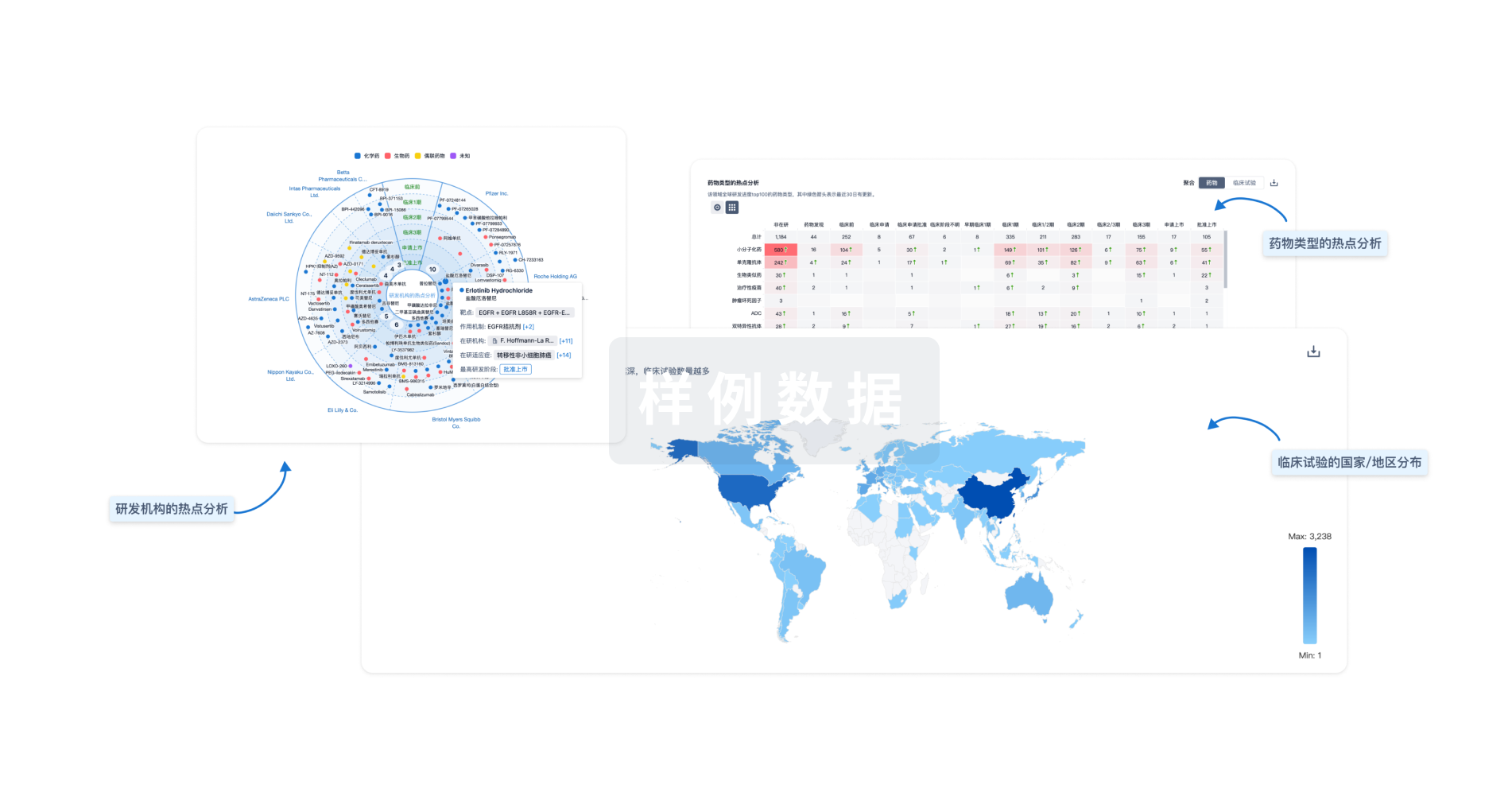
对领域进行一次全面的分析。
登录
或
生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用