预约演示
更新于:2025-05-07
Acidaemia
有机酸血症
更新于:2025-05-07
基本信息
别名 ACIDEMIA、Acidaemia、Acidemia + [8] |
简介 An abnormally low blood pH (usually defined as less than 7.35). [PMID:24381489] |
关联
7
项与 有机酸血症 相关的药物靶点- |
作用机制- |
非在研适应症- |
最高研发阶段临床前 |
首次获批国家/地区- |
首次获批日期1800-01-20 |
靶点- |
作用机制- |
在研适应症 |
最高研发阶段临床前 |
首次获批国家/地区- |
首次获批日期1800-01-20 |
作用机制 PANK2 agonists [+1] |
在研机构- |
在研适应症 |
最高研发阶段临床前 |
首次获批国家/地区- |
首次获批日期1800-01-20 |
16
项与 有机酸血症 相关的临床试验NCT06953505
Pilot Study of At Home Ammonia Monitoring in Patients With an Inborn Error of Ammonia Metabolism
The goal of this observational study is to learn if people with certain ammonia metabolism disorders will measure their ammonia levels at home.
The main question it aims to answer is:
• Will participants measure their ammonia every day?
Participants will be asked to:
* Attend three in-person study visits at the clinic.
* Measure temperature, heart rate, and blood oxygen every day.
* Complete a short survey every day.
* Measure ammonia every day.
The main question it aims to answer is:
• Will participants measure their ammonia every day?
Participants will be asked to:
* Attend three in-person study visits at the clinic.
* Measure temperature, heart rate, and blood oxygen every day.
* Complete a short survey every day.
* Measure ammonia every day.
开始日期2025-06-01 |
申办/合作机构 |
CTR20242574
一项评价卡谷氨酸分散片治疗N-乙酰谷氨酸合成酶缺乏症及有机酸血症(丙酸血症、甲基丙二酸血症或异戊酸血症)引起的高氨血症的安全性、有效性和药代动力学的单臂、开放性、多中心临床研究
1. 高氨血症中国患者中评价卡谷氨酸分散片的AE发生率和严重程度。
2. 评价卡谷氨酸分散片在缓解高氨血症方面的有效性。
3. 评价卡谷氨酸分散片在高氨血症中国患者中的药代动力学特征并探索协变量(如年龄、体重、性别和其他)对卡谷氨酸PK的影响。
开始日期2024-09-27 |
申办/合作机构 锐康迪(北京)医药有限公司 [+2] |
NCT05921955
Effect of Maternal Oxygen Supplementation by High-flow Nasal Oxygen Compared With Room Air on Fetal Acidemia
The study propose that the using the high-flow nasl oxygen to provide oxygen for maternal can improve the fetal acidemia and the neonatal outcomes during cesarean section with combined spinal-epidural anesthesia.
开始日期2023-07-01 |
申办/合作机构 |
100 项与 有机酸血症 相关的临床结果
登录后查看更多信息
100 项与 有机酸血症 相关的转化医学
登录后查看更多信息
0 项与 有机酸血症 相关的专利(医药)
登录后查看更多信息
2,819
项与 有机酸血症 相关的文献(医药)2025-12-31·The Journal of Maternal-Fetal & Neonatal Medicine
Letter to the editor: “Diagnostic capacity and interobserver variability in FIGO, ACOG, NICE and Chandraharan cardiotocographic guidelines to predict neonatal acidemia” The authors should reconsider their conclusions
Article
作者: Karassane, Roselene ; Dap, Matthieu ; Bertholdt, Charline ; Fijean, Anne-Laure ; Morel, Olivier
2025-04-01·Acta Obstetricia et Gynecologica Scandinavica
Frequency of fetal blood sampling, delivery mode and neonatal outcome after revised CTG ‐classification and updated lactate meter in Sweden: An observational study
Article
作者: Johansson, Kari ; Gyllencreutz, Erika ; Gröndal, Klara ; Wretler, Stina ; Holzmann, Malin
2025-04-01·American Journal of Kidney Diseases
Renal Tubular Acidosis: Core Curriculum 2025
Review
作者: Bonner, Ryan ; Hladik, Gerald
53
项与 有机酸血症 相关的新闻(医药)2025-04-28
*本内容仅供医疗卫生专业人士阅读参考前言您的血糖管理跟上了吗?2型糖尿病(T2DM)患者的稳态控糖之旅犹如翻越万里长城,沿途布满了重重难关与挑战,每一步都考验着决心与智慧......FRC控糖小队再启新程!本期是“宁启新程·控糖有道——2025长城病例闯关赛”第三期,由南通大学附属医院顾云娟教授担任守关人,由南通大学附属医院欧阳嵘医生担任领队,带领FRC控糖小队来到了控糖挑战第三关居庸关......(照例,文末互动哦~)闯关不易,他们能否成功?Some days later根据FRC控糖小队的降糖方案,降糖效果到底如何呢?那么,FRC控糖小队在欧阳医生的带领下能否顺利通关,拿到通关令牌?一起来看看守关人顾主任怎么说~守关人:大家分析到位,思路清晰。面对患者较为年轻、患病时间短却已出现并发症、伴发肝功能异常等,且多种药物联合治疗依从性差的情况,采取“先强化、后简化”的治疗策略,其中甘精利司的精准选择更是关键,整个方案的制定和实施都非常出色!基础胰岛素与GLP-1RA从联合治疗到复方制剂(FRC),在机制互补、增效减副的基础上,可进一步简化治疗方案,在指南中的地位也逐步提升。《中国2型糖尿病防治指南(2020年版)》[1]指出,FRC在胰岛素使用剂量相同或更低的情况下,降糖效果优于基础胰岛素,并且能减少低血糖风险,避免胰岛素治疗带来的体重增加等不良反应。《中国糖尿病防治指南(2024版)》表示[2],FRC可简化治疗方案、增加患者依从性,并首次将FRC纳入诊疗路径,作为T2DM高血糖治疗路径中的二联推荐以及胰岛素治疗路径中的起始推荐。在中国T2DM人群中的研究证实,甘精利司对于口服药(OADs)治疗不佳患者具有显著疗效且安全性较好。LixiLan-O-AP[3]研究纳入以中国人群为主的亚太地区成人受试者,经OADs治疗血糖控制不佳添加甘精利司治疗,HbA1c降幅高达1.9%,HbA1c达标率近80%,显著优于添加甘精胰岛素或利司那肽治疗组;治疗12周葡萄糖目标范围内时间(TIR)即可达到85%,较甘精胰岛素增重显著更少,低血糖发生率相似。SoliD研究[4]显示,在OADs治疗血糖控制不佳的中国T2DM患者中,相比德谷门冬,甘精利司自基线至24周的HbA1c降幅达到了非劣与优效,两组差值-0.2%,具有统计学意义;在两组FPG降幅相当的情况下,甘精利司三餐后平均血糖较德谷门冬进一步下降25%;较德谷门冬,甘精利司总体低血糖事件率减少29%,并实现-1.5 kg的体重获益,以更少胰岛素剂量实现近2倍复合终点达标率。值得一提的是,作为甘精利司中的GLP-1RA利司那肽在降低餐后血糖方面表现十分优异。国外一项综述分析,将不同降糖药物对PPG影响的数据进行了汇总,结果显示在不同降糖方案中,利司那肽可强效降低PPG,降幅高达3.1-5.9 mmol/L[5]。此外,在部分人群中的研究显示利司那肽还具有肝脏保护的作用,包括可有效改善ALT、肝功能和肝纤维化等[6-7]。总之,甘精利司作为新型FRC制剂,涵盖T2DM多重病理生理机制,可以起到一针双效、简化治疗、多重获益的作用,是OADs控制不佳患者起始胰岛素治疗的优选方案。前路紫荆关,血糖暗流涌动望与宁同行,稳扎稳打破难关“居庸关”闯关互动扫描二维码进入签到并答题(连续集齐6期通关令牌,即可赢取学术好礼)参考文献:(上下滑动查看更多)[1]中华医学会糖尿病学分会.中国2型糖尿病防治指南(2020年版)[J].中华内分泌代谢杂志,2021,37(4):311-398.DOI:10.3760/cma.j.cn311282-20210304-00142.[2]中华医学会糖尿病学分会. 中国糖尿病防治指南(2024版). 中华糖尿病杂志,2025,17(01):16-139. DOI:10.3760/cma.j.cn115791-20241203-00705.[3]Yang W, Dong X, Li Q, et al. Efficacy and safety benefits of iGlarLixi versus insulin glargine 100 U/mL or lixisenatide in Asian Pacific people with suboptimally controlled type 2 diabetes on oral agents: The LixiLan-O-AP randomized controlled trial. Diabetes Obes Metab. 2022 Aug;24(8):1522-1533. doi: 10.1111/dom.14722. Epub 2022 May 12. PMID: 35441412.[4]Liu M, Gu W, Chen L, et al. The efficacy and safety of iGlarLixi versus IDegAsp in Chinese people with type 2 diabetes suboptimally controlled with oral antidiabetic drugs: The Soli-D randomized controlled trial. Diabetes Obes Metab. 2024 Sep;26(9):3791-3800. doi: 10.1111/dom.15724. Epub 2024 Jun 22. PMID: 38922731.[5]Hershon KS, Hirsch BR, Odugbesan O. Importance of Postprandial Glucose in Relation to A1C and Cardiovascular Disease. Clin Diabetes. 2019 Jul;37(3):250-259. doi: 10.2337/cd18-0040. PMID: 31371856; PMCID: PMC6640888.[6]Gluud LL, Knop FK, Vilsbøll T. Effects of lixisenatide on elevated liver transaminases: systematic review with individual patient data meta-analysis of randomised controlled trials on patients with type 2 diabetes. BMJ Open. 2014 Dec 19;4(12):e005325. doi: 10.1136/bmjopen-2014-005325. PMID: 25526792; PMCID: PMC4275683.[7]Abstracts of 52nd EASD Annual Meeting. Diabetologia. 2016 Aug;59 Suppl 1(Suppl 1):1-581. doi: 10.1007/s00125-016-4046-9. PMID: 27539147; PMCID: PMC7096097.声明:本文仅供医疗卫生专业人士了解最新医药资讯参考使用,不代表本平台观点。该等信息不能以任何方式取代专业的医疗指导,也不应被视为诊疗建议,如果该信息被用于资讯以外的目的,本站及作者不承担相关责任。MAT-CN-2507811最新《国际糖尿病》读者专属微信交流群建好了,快快加入吧!扫描左边《国际糖尿病》小助手二维码(微信号:guojitnb),回复“国际糖尿病读者”,ta会尽快拉您入群滴!(来源:《国际糖尿病》编辑部)版权声明版权属《国际糖尿病》所有。欢迎个人转发分享。其他任何媒体、网站未经授权,禁止转载。
临床3期临床结果临床失败
2025-04-23
·梅斯医学
27岁的小李(化名)身材清瘦、性格开朗,但过去四年,他反复出现“奇怪”的发作:剧烈运动后就会呕吐、腹泻,甚至昏迷不醒。第一次是在一次登山后,随之而来的还有高氨血症、代谢性酸中毒和心衰……经过抢救才转危为安。此后,小李的生活彻底变了。只要稍微一动多了,就可能出现恶心、无力、呕吐,甚至胸闷气短。多次就诊后,只能以“周围神经病”“高氨血症”来暂时解释症状,但病情始终反复。直到最近一次发作——轻微活动后突发胸闷、气短、心电图异常,心肌酶升高,再次住院。医生在进一步回顾病史、评估症状、并结合氨升高、神经病变、心肌损伤等表现后,终于高度怀疑并进行遗传代谢病相关检测,最终确诊为:迟发型丙酸血症。常染色体隐性遗传病丙酸血症(propionic acidemia,PA)是支链氨基酸和偶数链脂肪酸代谢异常的一种较常见的有机酸血症,为常染色体隐性遗传病,是由于丙酰CoA酶羧化酶活性缺乏,导致体内丙酸及其代谢产物前体异常蓄积,而出现一 系列生化异常、神经系统和其他脏器损害症状。不同地区有较大差异,为1/280 000~1/90 000,中国约为1/200 000。丙酸血症虽属于罕见病,但是有机酸血症中常见疾病,患者临床表现缺乏特异性,个体差异较大,可自新生儿到成年发病。临床表现丙酸血症可发生于新生儿至成年,分为早发型(≤3个月)和晚发型(>3个月),以早发型最常见。早发型多在出生后数天内出现代谢失代偿,表现为喂养困难、呕吐、嗜睡、低肌张力等,易误诊为围生期脑损伤等。若不及时治疗,易发展为昏迷甚至死亡,并遗留神经及多器官损害。晚发型常因感染、饥饿、手术等应激诱发,表现为代谢危象及认知障碍。部分患者起病隐匿,出现进食困难、智力运动发育迟缓、癫痫等症状。常见并发症包括脑基底核损伤、心肌病、血细胞减少、肝胰损害、视听障碍及骨质疏松。个别病例表现为肾损伤或孤立性心肌病,提示该病临床谱仍在扩展。诊断对于有脑发育异常、发育迟缓、癫痫、脑瘫、不明原因呕吐、昏迷、酸中毒、心肌病或有相关家族史的高危患儿,应警惕丙酸血症等代谢病,尽早进行血氨、血气、血糖、肝肾功能、心肌酶谱等基础检查,并结合特殊生化分析明确诊断。1. 新生儿筛查与生化分析:通过液相色谱-串联质谱(LC-MS/MS)检测血酯酰肉碱谱,典型患者表现为C3、C3/Co、C3/C2升高,游离肉碱(Co)和甘氨酸水平异常。由于可能存在假阳性或假阴性,应结合尿液有机酸谱(GC-MS)检查,若3-羟基丙酸、甲基枸橼酸、丙酰甘氨酸升高,且甲基丙二酸正常,可提示丙酸血症。2. 酶活性检测:可通过特定方法测定皮肤成纤维细胞或外周血淋巴细胞中的丙酰辅酶A羧化酶活性,辅助诊断。但因检测条件复杂,不适用于常规筛查。3. 基因分析:通过Sanger测序、靶向或全外显子测序检测PCCA或PCCB基因的致病突变可确诊。PCCA突变多为点突变(约80%),PCCB点突变比例更高(约97%)。若未发现双等位基因突变,需进一步做拷贝数变异分析(如qPCR、MLPA、array-CGH)以排除大段缺失或重复。治疗 急性期治疗:丙酸血症急性期治疗应以生命支持为核心,纠正酸中毒、电解质紊乱,限制蛋白摄入,积极补充热量(1.5倍基础需求),抑制分解代谢。使用左卡尼汀促进毒物排出,重度高氨血症时可用精氨酸类药物,必要时行透析。特殊配方奶粉应在短期内替代天然蛋白,但应在48小时内恢复部分天然蛋白摄入。 稳定期治疗与管理:饮食管理: 高热量、低蛋白饮食,补充特殊配方奶粉,天然蛋白摄入量随年龄调整。鼓励母乳喂养,避免饥饿,确保微量营养素摄入。药物治疗: 持续口服左卡尼汀维持正常水平;短期使用甲硝唑减少肠道丙酸产生;苯甲酸钠类药物降血氨;必要时使用生长激素。应密切监测代谢指标和生长发育情况,个体化管理。 肝移植与新治疗探索:对于反复发作、治疗效果差的患儿,肝移植可显著改善代谢控制和生活质量,提升蛋白耐受力。新兴疗法如酶替代、基因治疗、抗氧化剂及回补疗法仍在研究中,未来或为治疗提供更多选择。参考资料:1.单纯型甲基丙二酸尿症饮食治疗与营养管理专家共识 . 中国实用儿科杂志,2018,33(7):481-486.2. 高明,张强强,王欣阳,等. 成人迟发型丙酸血症合并心肌病1例. 中国临床案例成果数据库,2024,06(01):E2452-E2452.3.刘怡,杨艳玲. 丙酸血症的筛查、诊断与治疗. 中华实用儿科临床杂志,2019,34(20):1531-1534. DOI:10.3760/cma.j.issn.2095-428X.2019.20.003来源 | 梅斯医学编辑 | wanny神经系统罕见病交流群↓点击下方“阅读原文”,下载梅斯医学APP吧!
2025-04-19
*本内容仅供医疗卫生专业人士阅读参考编者按2025年4月7日至10日,国际糖尿病联盟(IDF)世界糖尿病大会在泰国曼谷盛大举行。作为全球糖尿病领域的顶级学术盛会,本次大会以“塑造糖尿病的未来”为主题,发布了多项具有里程碑意义的成果,包括第11版《IDF全球糖尿病地图》、《IDF 2025全球2型糖尿病临床实践指南》等,此外在基础胰岛素/胰高血糖素样肽-1受体激动剂(GLP-1RA)复方制剂(FRC)领域的多项研究成果引发关注。为更好传递本次大会精粹,《国际糖尿病》特别邀请到南京市第一医院内分泌科李倩教授,带领我们一起深入解析本届IDF大会精彩亮点和重磅研究发现。寰宇全球糖尿病领域迎来重大革新在本次大会上,第11版《IDF全球糖尿病地图》[1]正式发布,揭示了当前全球糖尿病的严峻现状和未来趋势,为糖尿病防治工作提供了重要参考依据。新版地图显示,中国以约1.5亿的糖尿病患者数位居全球首位,成为糖尿病患者人数最多的国家。糖尿病是一种慢性疾病,若未能得到有效控制,会引发一系列严重并发症,包括心血管疾病、肾脏疾病、视网膜病变、神经病变等。2024年,全球约有340万人因糖尿病或其并发症死亡,占20~79岁成年人全因死亡人数的9.3%。与此同时,《IDF 2025全球2型糖尿病临床实践指南》[2]时隔8年重磅更新。新版IDF指南指出,全球范围内,2型糖尿病(T2DM)患者普遍血糖控制不佳,改善血糖控制仍是未被满足的需求和糖尿病治疗的重点。早期及时强化治疗对于克服临床惰性至关重要,当非胰岛素治疗无法达到血糖控制目标时,胰岛素是T2DM治疗的关键选择之一。长期结局研究已证实胰岛素在改善血糖控制方面的疗效,以及在减少T2DM微血管和大血管并发症方面的遗留效应。同时,新版IDF指南表示,胰岛素联合其他药物,如GLP-1RA(包括固定剂量组合)或SGLT2i可能增强血糖控制、促进体重减轻、减少胰岛素用量和降低低血糖风险。并且强调,定期重新评估胰岛素治疗是必要的,应定期考虑胰岛素去强化或简化,尤其是在老年或虚弱的个体中,以最大限度降低低血糖风险,减少治疗负担,提高生活质量。聚焦重磅将MDI转为FRC控糖更有优势正如新版IDF指南所言,对于T2DM患者,应定期考虑胰岛素去强化治疗。每日多次胰岛素注射(MDI)虽是复杂且高效的胰岛素治疗方案,但往往会导致体重增加、低血糖风险升高以及生活质量降低。在本次大会上,来自捷克共和国布拉格临床和实验医学研究所的Martin Haluzik教授,汇报了IDEAL随机对照试验的结果(摘要号:BA2025-1479)[3],证实将MDI转为FRC甘精胰岛素利司那肽注射液(以下简称甘精利司)是一种有效且安全的治疗选择,引发广泛关注。IDEAL研究是一项为期24周的多中心、开放标签、平行组、4期随机对照试验(RCT),旨在评估将MDI去强化为每日一次甘精利司治疗T2DM的疗效和安全性。纳入年龄18~80岁、每日胰岛素总剂量(TDD)≤0.8 U/kg,且空腹C肽水平高于正常下限的T2DM患者,1:1随机分配至每日一次给药的甘精利司组或继续接受MDI治疗组。主要终点是24周时糖化血红蛋白(HbA1c)的变化。次要终点包括:体重、TDD、空腹血糖(FPG)、餐后血糖(PPG)、低血糖、持续葡萄糖监测(CGM)衍生指标以及生活质量的变化。共纳入了91例患者,其中女性占22%(20/91),平均年龄为66±9岁,平均糖尿病病程为17±9年,平均体质指数(BMI)为33.5±5.5 kg/m²。主要结果如下:甘精利司组平均HbA1c变化非劣效于MDI组(-0.47±0.91% vs. -0.37±0.77%,非劣效性检验P=0.01);平均体重降幅高于MDI组(-4.8 kg vs. -0.5 kg,P<0.001)。甘精利司组TDD降低(P<0.0001),而MDI组TDD保持不变(P=0.233)。甘精利司组报告低血糖的研究访视比例低于MDI组(5.5% vs. 9.6%,P=0.029)。24周时,甘精利司组的中位葡萄糖目标范围内时间(TIR)自基线的70%增加至80%(P=0.039),而MDI组TIR无显著变化(78% vs. 82%,P=0.728)。24周时,甘精利司组的糖尿病治疗满意度调查问卷-当前版本(DTSQs)评分高于MDI组(35.0 vs. 29.0,P<0.001)。研究表明,对于T2DM患者而言,甘精利司不仅能够提供与MDI相当的血糖控制水平,而且还可以减少体重、降低TDD、减少胰岛素注射次数,且不增加低血糖风险,以及提高患者对糖尿病治疗的满意度。立足实际FRC多项真实世界数据出炉除上述的RCT外,本次大会上还有多项关于FRC的真实世界研究结果出炉,这些研究更贴合临床的实际情况,为优化临床治疗提供了更好的借鉴。真实世界研究1FRC简化胰岛素治疗可行安全一项研究探讨了甘精利司简化胰岛素治疗方案在血糖控制相对良好T2DM患者中的应用(摘要号:BA2025-0159)[4]。这项回顾性单中心研究评估了2017年1月至2023年6月期间,将基础-餐时或预混胰岛素方案替换为甘精利司治疗患者的真实世界数据,这些患者基线HbA1c≤7.5%,至少有1次随访。转换为甘精利司后平均5个月和11个月时分别进行了第一次和第二次随访(V1和V2),分别有59名和41名患者完成了随访。结果显示,基线时HbA1c为6.42±0.69%,在V1和V2时分别变化为6.31±0.77%和6.27±0.93%(P=0.456)。基线时体重为87.1±17 kg,在V1和V2时分别降至84.2±17.6 kg(P<0.0001)和84.9±19.2 kg(P=0.032)。在基线、V1和V2时报告至少发生一次低血糖事件的患者比例分别为52.5%、30.5%和22%。在V1和V2时,甘精利司的平均剂量分别为21.8±8.3 U和22.1±8.7 U。研究提示,在日常临床实践中,对于血糖控制相对良好的T2DM患者,用甘精利司替换复杂胰岛素方案是可行且安全的。这种简化治疗方案能够提供相似甚至更好的血糖控制,同时可降低胰岛素剂量、减少每日注射次数、降低低血糖风险,并对体重管理产生有利影响。真实世界研究2FRC长期疗效获得验证另一项真实世界研究Soli-Durability则评估了甘精利司在美国T2DM患者中使用24个月的长期有效性和安全性(摘要号:BA2025-0629)[5]。纳入既往接受过口服降糖药(OADs)、基础胰岛素±OADs或GLP-1RA±OADs,并在2017年1月1日至2020年3月31日新起始甘精利司治疗的成年T2DM患者。共纳入了1685例起始甘精利司治疗的患者(平均年龄:58.4岁;52.6%为女性)。在整个人群中(包括估算的24个月值),24个月时平均HbA1c降低0.9%。与观察数据(n=535)平均HbA1c降低1.0%相似。在24个月治疗期间,共有592例(35.1%)患者达到HbA1c<7%的目标。从起始治疗到随访,平均体重降低0.2 kg。基线时低血糖事件发生率为16.1/100人年;24个月时降至11.4/100人年。研究表明,在真实世界分析中,起始甘精利司治疗与临床上显著的HbA1c降低、体重减轻以及低血糖发生率的下降相关,且这种效果在3个月时显现,并在24个月内得以维持。真实世界研究3FRC是口服不佳患者强化治疗的实用选择此外,还有一项研究评估了甘精利司在仅使用OADs治疗控制不佳的T2DM患者中的真实世界疗效和安全性(摘要号:BA2025-1060)[6]。这项前瞻性观察研究纳入了至少使用三种OADs(包括磺脲类药物和二甲双胍)治疗且经过剂量优化,但HbA1c>7.5%的成年T2DM患者。治疗方案从OADs转换为甘精利司,剂量根据基线HbA1c和临床需求个体化调整。结果显示,第3个月时,参与者平均FPG和PPG水平分别降低2.91 mmol/L和5.11 mmol/L,平均HbA1c降低1.73%,平均体重降低3.62 kg。2例患者分别经历了1次有记录的轻度低血糖发生。研究显示,甘精利司在使用OADs治疗失败的T2DM患者中有效改善了血糖控制,且具有可管理的安全性和极低的严重低血糖风险。这些发现支持将甘精利司作为T2DM治疗强化的实用选择。指南指引,循证为基FRC全方位优化血糖管理从最新公布的《IDF全球糖尿病地图》中可以看到,在全球范围内糖尿病防控都面临严峻挑战,尤其是在中国。《IDF 2025全球2型糖尿病临床实践指南》明确指出,血糖控制仍是糖尿病治疗的重点,早期及时强化治疗至关重要,并推荐FRC作为血糖管理的选择之一。事实上,与全球同步,在中国FRC也已被多项指南纳入T2DM的诊疗路径,并作为胰岛素治疗的起始推荐方案[7-8]。FRC在胰岛素去强化或简化治疗中发挥重要作用。本次会议上公布的IDEAL研究证实,将MDI去强化转换为甘精利司治疗方案在控糖效果相似的同时,在体重管理、减少胰岛素剂量、注射次数和低血糖风险控制等方面具有显著优势,为临床治疗提供了更优的选择。此外,多项真实世界研究也证明了甘精利司在简化胰岛素治疗、长期疗效以及OADs控糖不佳强化治疗方面表现优异。这些进展不仅为临床医生提供了新的治疗思路,也为糖尿病患者带来了更多希望。参考文献:(上下滑动查看更多)[1]IDF Diabetes Atlas 11th Edition.https://diabetesatlas.org/resources/idf-diabetes-atlas-2025/[2] International Diabetes Federation. IDF Global Clinical Practice Recommendations for Managing Type 2 Diabetes – 2025. https://idf.org/t2d-cpr-2025[3]P. NOVODVORSKY, L. Thieme, I. Lankova, et al.Insulin Therapy De-intensification with iGlarLixi in People with Type 2 Diabetes: the IDEAL Randomised Controlled Trial.IDF 2025: BA2025-1479.[4]Z.Taybani, B. Bótyik, K. Fehértemplomi,et al.Insulin treatment simplification with iGlarLixi in relatively well-controlled patients with type 2 diabetes.IDF 2025: BA2025-0159.[5]Z.R. Malik, J. Anderson, R. Giuseppina,et al.Real-world study evaluating long-term effectiveness of iGlarLixi in people with T2D in the USA: Soli-Durability study.IDF 2025:BA2025-0629.[6]Z.S. Samrat Samajdar, S.R. Joshi, B. Saboo, et al.Fixed-Dose Glargine and Lixisenatide: A Practical Option to Minimize Hypoglycemia and Weight Gain in Type 2 Diabetes.IDF 2025: BA2025-1060.[7]中华医学会糖尿病学分会. 中国糖尿病防治指南(2024版). 中华糖尿病杂志,2025,17(01):16-139. DOI:10.3760/cma.j.cn115791-20241203-00705.[8]国家老年医学中心,中华医学会老年医学分会,中国老年保健协会糖尿病专业委员会.中国老年糖尿病诊疗指南(2024版)[J].中华糖尿病杂志, 2024, 16(02):147-189. DOI:10.3760/cma.j.cn115791-20240112-00020.李倩 教授南京市第一医院主任医师,南京医科大学教授、博士生导师,南京市第一医院内分泌科主任,南京市医学会内分泌学分会副主任委员,江苏省医学会糖尿病学分会委员,江苏省医师协会内分泌学分会委员,江苏省六大高峰人才。曾先后在美国霍普金斯大学医学院和南京医科大学公共卫生学院博士后工作,主要研究早期糖尿病胰岛修复治疗,糖尿病并发血管神经病变等方面的研究,先后主持国家自然科学基金、江苏省自然基金等科研项目。声明:本文仅供医疗卫生专业人士了解最新医药资讯参考使用,不代表本平台观点。该等信息不能以任何方式取代专业的医疗指导,也不应被视为诊疗建议,如果该信息被用于资讯以外的目的,本站及作者不承担相关责任。MAT-CN-2507660最新《国际糖尿病》读者专属微信交流群建好了,快快加入吧!扫描左边《国际糖尿病》小助手二维码(微信号:guojitnb),回复“国际糖尿病读者”,ta会尽快拉您入群滴!(来源:《国际糖尿病》编辑部)版权声明版权属《国际糖尿病》所有。欢迎个人转发分享。其他任何媒体、网站未经授权,禁止转载。
临床研究临床结果基因疗法
分析
对领域进行一次全面的分析。
登录
或
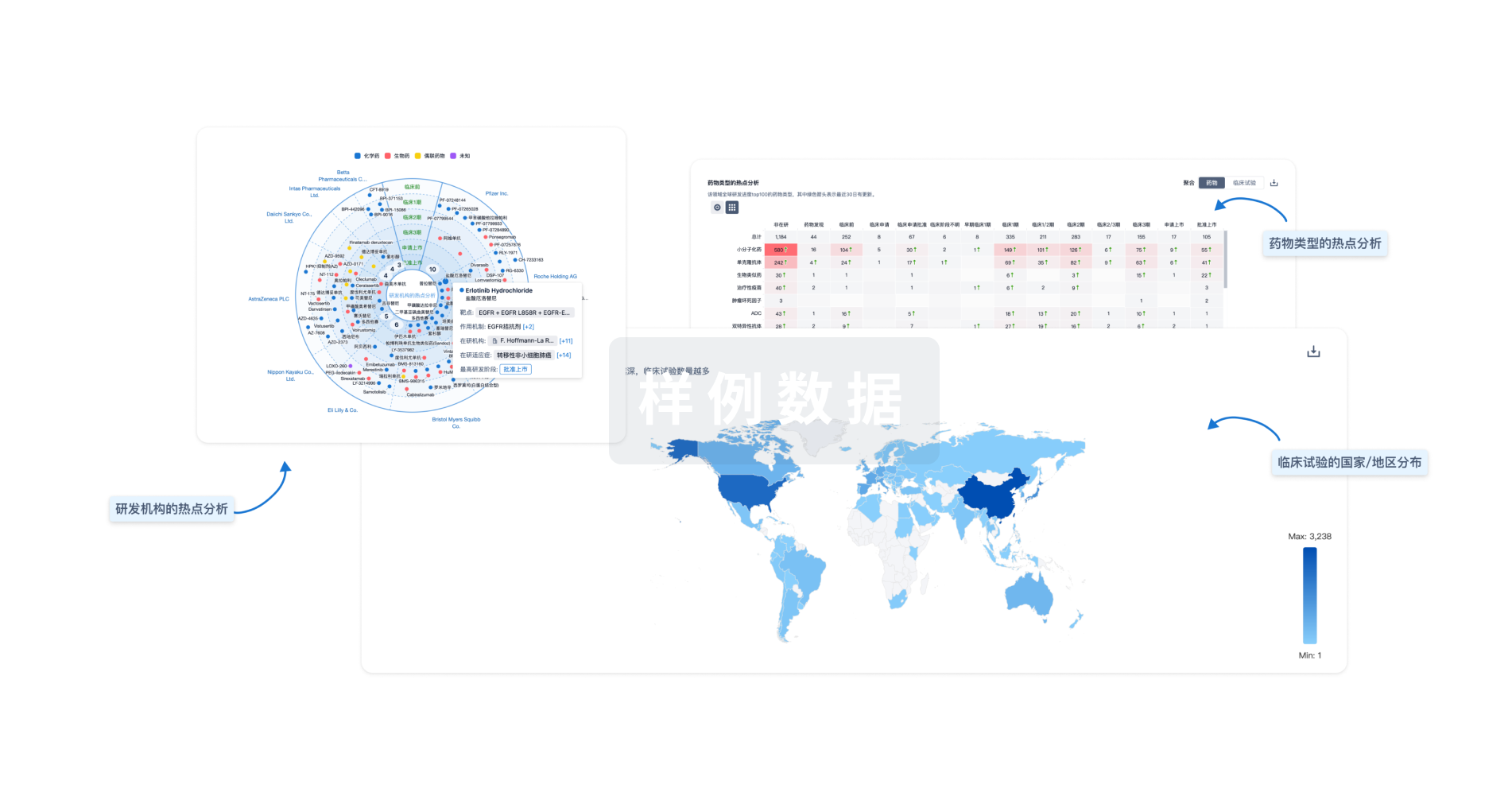
生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用




