预约演示
更新于:2026-03-18
Stem cell therapeutics (ExCellThera)
多罗库比利
更新于:2026-03-18
概要
基本信息
非在研机构- |
最高研发阶段批准上市 |
首次获批日期 欧盟 (2025-08-25), |
最高研发阶段(中国)- |
特殊审评孤儿药 (美国)、孤儿药 (欧盟)、再生医学先进疗法 (美国)、附条件批准 (欧盟)、加速审评 (欧盟)、先进治疗医药产品 (欧盟)、优先药物(PRIME) (欧盟) |
登录后查看时间轴
关联
7
项与 多罗库比利 相关的临床试验NCT07301866
Single-site, Prospective, Open-label Phase I-II Study on Transplantation Using Cord Blood Treated With an Optimized Dose of UM171: Optimized ECT-001-CB.
This clinical study is testing a new way to improve stem cell transplants for adults with high-risk blood cancers, such as leukemia or myelodysplasia, who do not have a suitable donor. The transplant uses stem cells from umbilical cord blood that have been expanded in the lab using a molecule called UM171. Previous studies showed that UM171 helps these cells grow and work better, leading to faster blood count recovery and fewer complications.
In this study, researchers are testing whether increasing the dose of UM171 during the lab expansion process can make the transplant less toxic. The hypothesis is that using a higher dose of UM171 to expand cord blood stem cells will help patients recover blood counts faster after transplant by improving the growth and function of the cells. This may lead to better immune recovery, fewer infections, shorter hospital stays, and improved overall outcomes.
Only seven patients will be enrolled, and they will be followed for one year after their transplant.
In this study, researchers are testing whether increasing the dose of UM171 during the lab expansion process can make the transplant less toxic. The hypothesis is that using a higher dose of UM171 to expand cord blood stem cells will help patients recover blood counts faster after transplant by improving the growth and function of the cells. This may lead to better immune recovery, fewer infections, shorter hospital stays, and improved overall outcomes.
Only seven patients will be enrolled, and they will be followed for one year after their transplant.
开始日期2025-10-06 |
申办/合作机构 |
NCT04990323
A Phase I/II Open-Label Study of ECT-001-Expanded Cord Blood Transplantation in Pediatric and Young Adult (<21year) Patients With High-Risk and Very High-Risk Myeloid Malignancies
Cord blood (CB) transplants are an option for patients lacking an HLA identical donor but are hampered by low cell dose, prolonged aplasia and high transplant related mortality. UM171, a novel and potent agonist of hematopoietic stem cell self renewal could solve this major limitation, allowing for CB's important qualities as lower risk of chronic GVHD and relapse to prevail. In previous trials (NCT02668315, NCT03913026, NCT04103879, and NCT03441958), the CB expansion protocol using the ECT-001-CB technology (UM171 molecule) has proven to be technically feasible and safe in adults.
UM171 expanded CB was associated with a prompt (D+17), robust (98%) and durable neutrophil recovery. Amongst patients who received a single UM171 CB transplant with a median follow-up of 18 months, risk of TRM (10%), grade 3-4 acute GVHD (13%) and moderate-severe chronic GVHD (2%) was low at 1 year post-transplant. Incidence of severe viral and bacterial infections was reduced and immunosuppression could be discontinued in 77% of patients at 1 year. Thus, PFS and GRFS were very promising, 72% and 59% at 12 months, 69% and 53% at 24 months, respectively, in particular accounting for a large proportion of very high-risk patients. By a 10-fold increase of CB accessibility, ECT-001-CB allowed access to smaller, better HLA matched CBs.
This new study seeks to test a similar strategy in a group of pediatric and young adult patients with high risk myeloid malignancies. 12 patients will be enrolled in the first stage of this 2-stage design protocol. If intervention is considered promising (<= 3 relapses in the first 12 patients), this study will open multicenter and be extended to a second stage (16 additional patients for a total accrual 28).
UM171 expanded CB was associated with a prompt (D+17), robust (98%) and durable neutrophil recovery. Amongst patients who received a single UM171 CB transplant with a median follow-up of 18 months, risk of TRM (10%), grade 3-4 acute GVHD (13%) and moderate-severe chronic GVHD (2%) was low at 1 year post-transplant. Incidence of severe viral and bacterial infections was reduced and immunosuppression could be discontinued in 77% of patients at 1 year. Thus, PFS and GRFS were very promising, 72% and 59% at 12 months, 69% and 53% at 24 months, respectively, in particular accounting for a large proportion of very high-risk patients. By a 10-fold increase of CB accessibility, ECT-001-CB allowed access to smaller, better HLA matched CBs.
This new study seeks to test a similar strategy in a group of pediatric and young adult patients with high risk myeloid malignancies. 12 patients will be enrolled in the first stage of this 2-stage design protocol. If intervention is considered promising (<= 3 relapses in the first 12 patients), this study will open multicenter and be extended to a second stage (16 additional patients for a total accrual 28).
开始日期2021-12-01 |
申办/合作机构  ExCellThera, Inc. ExCellThera, Inc. [+1] |
NCT04594031
A Phase I Multicenter Study of Hematopoietic Stem Cell Transplant of ECT-001-Expanded Cord Blood With a Reduced Toxicity Conditioning Regimen in Patients With Severe Sickle Cell Disease
The application of experimental hematopoietic cell transplantation (HCT) therapy in sickle-cell disease (SCD) must strike a balance between the underlying disease severity and the possibility of a direct benefit of the treatment, particularly in pediatric populations. Clinical studies in adults with SCD have focused on interventions that prolong survival and improve the quality of life. Unlike children, adults with SCD are much more likely to have a debilitating complication. As a result, the risk/benefit ratio of HCT is very favorable in adults, particularly if an approach to HCT that defines an acceptable level of toxicity can be established.
Whereas hematopoietic stem cell transplantation (HSCT) remains the only curative treatment currently available for patients with SCD, the morbidity, the frequent irreversible damage in target organs and the mortality reported in the natural course of patients with severe SCD are strong incentives to perform HSCTs in younger age groups. For those who lack a matched related donor, CB transplant is an appealing option, but despite been less problematic, CB accessibility related to cell dose of appropriately matched cord blood unit (CBU) remains a significant issue. Through a 7-day culture process of a CBU's hematopoietic stem cell HSCs with the UM171 compound, the total cell dose is increased mitigating this limitation.
UM171-CB expansion (ECT-001-CB) allows a greater CB accessibility, the selection of better matched cords that might translate into favourable clinical outcomes as reported in previous trials, including a lower risk of graft-versus-host disease. After CB selection and ex-vivo expansion, ECT-001-CB transplant will follow a myeloablative reduced-toxicity conditioning regimen consisting of rATG, busulfan and fludarabine with doses of all agents optimized to the individual using model-based dosing and will be followed by standard supportive care and GVHD prophylaxis consisting of tacrolimus and MMF.
Whereas hematopoietic stem cell transplantation (HSCT) remains the only curative treatment currently available for patients with SCD, the morbidity, the frequent irreversible damage in target organs and the mortality reported in the natural course of patients with severe SCD are strong incentives to perform HSCTs in younger age groups. For those who lack a matched related donor, CB transplant is an appealing option, but despite been less problematic, CB accessibility related to cell dose of appropriately matched cord blood unit (CBU) remains a significant issue. Through a 7-day culture process of a CBU's hematopoietic stem cell HSCs with the UM171 compound, the total cell dose is increased mitigating this limitation.
UM171-CB expansion (ECT-001-CB) allows a greater CB accessibility, the selection of better matched cords that might translate into favourable clinical outcomes as reported in previous trials, including a lower risk of graft-versus-host disease. After CB selection and ex-vivo expansion, ECT-001-CB transplant will follow a myeloablative reduced-toxicity conditioning regimen consisting of rATG, busulfan and fludarabine with doses of all agents optimized to the individual using model-based dosing and will be followed by standard supportive care and GVHD prophylaxis consisting of tacrolimus and MMF.
开始日期2021-07-13 |
申办/合作机构  ExCellThera, Inc. ExCellThera, Inc. [+1] |
100 项与 多罗库比利 相关的临床结果
登录后查看更多信息
100 项与 多罗库比利 相关的转化医学
登录后查看更多信息
100 项与 多罗库比利 相关的专利(医药)
登录后查看更多信息
3
项与 多罗库比利 相关的文献(医药)2026-01-01·Molecular Diagnosis & Therapy
Dorocubicel: First Approval
Review
作者: Lee, Arnold
Dorocubicel (Zemcelpro®) is a cell therapy that includes UM171-expanded CD34+ haematopoietic stem cells derived from umbilical cord blood. It is being developed by Cordex Biologics for the treatment of haematological malignancies and non-malignant haematological disorders. In patients with haematological malignancies requiring an allogeneic haematopoietic stem cell transplantation (HSCT), transplantation with dorocubicel has been shown to result in neutrophil and platelet engraftment, as well as rapid T cell reconstitution. This article summarizes the key milestones in the development of dorocubicel leading to this first approval for the treatment of adult patients with haematological malignancies who require an allogeneic HSCT following myeloablative conditioning, for whom no other type of suitable donor cells is available.
2022-05-01·Leukemia1区 · 医学
Human CD34+ very small embryonic-like stem cells can give rise to endothelial colony-forming cells with a multistep differentiation strategy using UM171 and nicotinamide acid
1区 · 医学
Letter
作者: Smadja, David M ; Ratajczak, Janina ; Domingues, Alison ; Rossi, Elisa ; Richez, Ulysse ; Detriche, Grégoire ; Kucia, Magdalena ; Blandinieres, Adeline ; Ratajczak, Mariusz Z ; Bujko, Kamila
Human CD34very small embryonic-like stem cells can give rise to endothelial colony-forming cells with a multistep differentiation strategy using UM171 and nicotinamide acid,.To develop a source of ECFC-like cells for vascular repair, the use of human induced pluripotent stem cells (iPS) and embryonic stem cells (ESCs) has been investigated.Therefore, VSELs could represent a clin. relevant alternative to iPS or ESCs, since they do not complete blastocyst development and do not form teratomas after transplantation into deficient mice.VSELs are the most primitive stem cells in bone marrow, which can give rise to different tissue committed progenitor cells from all germ layers without confounding factor of feeder cell fusion with VSELs.All in all, we demonstrate for the first time here a full in vitro differentiation of VSELs in true vasculogenic progenitor ECFCs with a chem. identified media with UM-171 and NMA added to culture from the first day of culture.Further studies need to better decipher origin of ECFCs and improve the vascular regenerative potential of VSELs by enhancing proliferative and vasculogenic potential of VSELECFCs in vitro and in vivo and obtain highly efficient production of patient-derived VSEL-ECFCs for the treatment of vascular disease.
1991-10-01·Zhonghua shen jing jing shen ke za zhi = Chinese journal of neurology and psychiatry
[Serum prolactin response to electroconvulsive therapy in patients with major depression].
Article
作者: Duan, Y
Serum prolactin (PRL) levels were determined at 30 min before and 5, 15, 30 and 60 min after the first and sixth electroconvulsive therapy (ECT-1 and ECT-6) in 12 female patients with major depression. There was a significant increase in serum PRL after ECT-1. The mean maximum PRL level was obtained at 30 min after ECT-1 and was 3.18 times of baseline level. The rate between mean maximum PRL level after and baseline before ECT-6 was 1.89 and was significantly lower than that during ECT-1. ECT-6 caused a significantly lower PRL release than did ECT-1 too. The possible mechanisms of produced these results were discussed.
35
项与 多罗库比利 相关的新闻(医药)2026-03-13
Zemcelpro® (dorocubicel) achieved remarkable and clinically meaningful survival results in patients with high- and very high-risk acute leukemias and myelodysplastic syndromes
Topline results from two Phase 2 studies to be presented on March 23, 2026 at the EBMT Annual Meeting in Madrid, Spain
March 10, 2026 -- ExCellThera Inc. (ExCellThera), a world leader in blood stem cell expansion and metabolic fitness, and its wholly owned subsidiary Cordex Biologics (Cordex), announce positive results from recently completed Phase 2 studies of Zemcelpro® (dorocubicel), also known as UM171 Cell Therapy, in 60 adult patients with high- and very high-risk acute leukemias (AL) and myelodysplastic syndromes (MDS), a population known to experience high relapse rates and poor survival on conventional allogeneic stem cell transplantation (allo-HSCT). Topline results will be presented on March 23, 2026 at the 52nd EBMT Annual Meeting in Madrid, Spain.
At 24 months post-transplant, overall survival (OS), progression-free survival (PFS) and relapse-free survival (RFS) were 63.7%, 57.0% and 60%, respectively, among Zemcelpro® (dorocubicel)-treated patients. The cumulative incidences of relapse and non-relapse mortality (NRM) were 22.5% and 20.5%, respectively. Age was the strongest predictor of NRM: patients <43 years experienced 0% NRM, with 24-month OS and PFS of 82.8% and 79.3%, respectively. Older patients (≥43 years) had comparable relapse rates but higher NRM. Grade III-IV acute graft-versus-host disease (GVHD) occurred in 20%, while moderate to severe chronic GVHD was infrequent at 7% at two years.
"This represents a remarkable and clinically meaningful achievement for Zemcelpro® (dorocubicel), especially given the historical overall survival of only 25–40%1 for patients of similar disease risk receiving standard of care allogeneic grafts," said Dr Guy Sauvageau, Chief Scientific Officer. "Results are particularly promising in adults under 43 years of age, showing even greater benefits in both OS and PFS, with no cases of NRM."
"These findings suggest that Zemcelpro® (dorocubicel) could redefine the standard of care for patients with high- and very high-risk blood cancers," said David Millette, CEO. "Based on its safety and efficacy profile, this positions Zemcelpro® (dorocubicel) as a potentially transformative therapy, challenging the existing treatment paradigm and offering renewed hope for difficult-to-cure high- and very high-risk patients facing unfavorable outcomes with conventional approaches."
Outcomes were assessed in 60 adults with high- or very high–risk AL or MDS who received Zemcelpro® (dorocubicel) in two prospective phase 2 trials conducted in the United States, Canada and Europe. High-risk AL/MDS was defined as disease with an expected 2-year PFS The product safety and efficacy have not yet been established by other regulatory agencies, such as the U.S. FDA, the MHRA and Health Canada.
ExCellThera is a world leader in enhanced blood stem cell therapies. ExCellThera's proprietary EnhanceTM platform for cell expansion and metabolic fitness is designed to deliver a greater dose of functional therapeutic stem cells by expanding haematopoietic stem cells (HSCs) from any source and counteracting the effects of culture or gene editing induced stress. ExCellThera partners with biopharmas to help them develop best-in-class cell and gene therapies by leveraging the technologies that form the EnhanceTM platform, including the proprietary molecule UM171 which has a first-in-class mechanism of action for ex vivo expansion and metabolic fitness of HSCs.
1Armand P, Kim HT, Logan BR, Wang Z, Alyea EP, Kalaycio ME, et al. Validation and refinement of the Disease Risk Index for allogeneic stem cell transplantation. Blood. 2014;123(23):3664–367
The content above comes from the network. if any infringement, please contact us to modify.
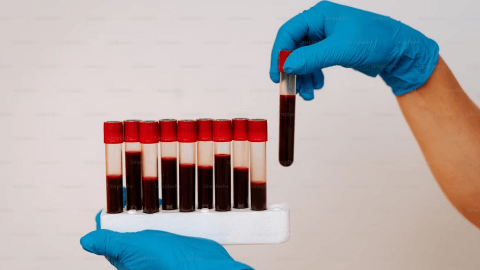
临床结果孤儿药上市批准
2026-03-10
·动脉网
Zemcelpro® (dorocubicel) a obtenu des résultats de survie remarquables et cliniquement significatifs chez des patients atteints de leucémies aiguës et de syndromes myélodysplasiques à haut et très haut risque Zemcelpro®(多罗库比利)在高危和极高危急性白血病及骨髓增生异常综合征患者中取得了显著且具有临床意义的生存结果。Présentation des résultats préliminaires de deux études de phase 2 le 23 mars 2026 lors du congrès annuel 2026 de l'EBMT à Madrid, en Espagne 2026年3月23日在西班牙马德里举行的2026年EBMT年会上,将展示两项二期研究的初步结果。MONTRÉAL 蒙特利尔, ,10 mars 2026 2026年3月10日 /PRNewswire/ -- ExCellThera Inc. (ExCellThera), leader mondial dans l'expansion des cellules souches sanguines et la condition métabolique, et sa filiale exclusive Cordex Biologics (Cordex), annoncent des résultats positifs issus des études de phase 2 récemment achevées sur Zemcelpro® (dorocubicel), également connue sous le nom de thérapie cellulaire UM171, chez 60 patients adultes atteints de leucémies aiguës (AL) et de syndromes myélodysplasiques (MDS) à haut et très haut risque, une population connue pour présenter des taux de rechute élevés et une survie faible après une allogreffe de cellules souches conventionnelle. /PRNewswire/ -- ExCellThera Inc.(ExCellThera),全球血液干细胞扩增和代谢条件领域的领导者,及其全资子公司Cordex Biologics(Cordex),宣布了最近完成的关于Zemcelpro®(多罗库贝细胞)的2期研究积极结果,该疗法也被称为UM171细胞疗法,针对60名患有高危和极高危急性白血病(AL)及骨髓增生异常综合征(MDS)的成年患者。这一人群在传统异体干细胞移植后复发率高且存活率低。Les principaux résultats seront présentés le 23 mars 2026, lors du 52ᵉ congrès annuel de l'EBMT à Madrid, en Espagne.. 主要结果将于2026年3月23日在西班牙马德里举行的第52届EBMT年会上公布。Continue Reading 继续阅读ExCellThera logo ExCellThera 标志À 24 mois après la greffe, la survie globale (OS), la survie sans progression (PFS) et la survie sans rechute (RFS) étaient respectivement de 63,7 %, 57,0 % et 60 % chez les patients traités par Zemcelpro® (dorocubicel). Les incidences cumulées de la rechute et de la mortalité non liée à la rechute (NRM) étaient respectivement de 22,5 % et 20,5 %. 移植后24个月,接受Zemcelpro®(多罗库贝西)治疗的患者总生存率(OS)、无进展生存率(PFS)和无复发生存率(RFS)分别为63.7%、57.0%和60%。累积复发率和非复发相关死亡率(NRM)分别为22.5%和20.5%。L'âge était le facteur prédictif le plus important de la NRM : les patients de moins de 43 ans ont présenté une NRM de 0 %, avec une OS et une PFS à 24 mois de 82,8 % et 79,3 %, respectivement. Les patients plus âgés (≥ 43 ans) avaient des taux de rechute comparables, mais une NRM plus élevée. Une maladie aiguë du greffon contre l'hôte (GVHD) de grade III–IV est survenue chez 20 % des patients, tandis que la GVHD chronique modérée à sévère était peu fréquente, avec un taux de 7 % à 2 ans.. 年龄是NRM最重要的预测因素:43岁以下患者的NRM为0%,24个月的OS和PFS分别为82.8%和79.3%。年龄较大的患者(≥43岁)复发率相似,但NRM更高。20%的患者发生了III-IV级急性移植物抗宿主病(GVHD),而中度至重度慢性GVHD较为少见,2年时发生率为7%。« Cela représente une avancée remarquable et cliniquement significative pour Zemcelpro® (dorocubicel), en particulier si l'on considère que la survie globale historique n'est que de 25 à 40 %¹ chez les patients présentant un risque de maladie similaire et recevant des greffes allogéniques standard », a déclaré le Dr Guy Sauvageau, Chef de la direction scientifique. “这对Zemcelpro®(多罗库比利)而言是一项显著且具有临床意义的进步,尤其是考虑到历史上接受标准异基因移植、具有相似疾病风险的患者的总生存率仅为25%至40%¹,”科学总监盖伊·索瓦热博士表示。« Les résultats sont particulièrement prometteurs chez les adultes de moins de 43 ans, montrant des bénéfices encore plus importants en termes d'OS et de PFS, sans aucun cas de NRM. ». “在43岁以下的成年人中,结果尤其令人鼓舞,显示出在总生存期(OS)和无进展生存期(PFS)方面更大的益处,且没有任何非复发死亡(NRM)病例。”« Ces résultats suggèrent que Zemcelpro® (dorocubicel) pourrait redéfinir la norme de soins pour les patients atteints de cancers du sang à haut et très haut risque », a déclaré David Millette, PDG. « En se basant sur son profil de sécurité et d'efficacité, Zemcelpro® (dorocubicel) se positionne comme une thérapie potentiellement révolutionnaire, susceptible de faire évoluer le paradigme thérapeutique actuel et offrant un nouvel espoir aux patients à haut et très haut risque, difficiles à guérir, qui connaissent des issues défavorables avec les approches conventionnelles. ». “这些结果表明,Zemcelpro®(多罗库比塞尔)有可能重新定义高风险和极高风险血液癌症患者的治疗标准,”首席执行官大卫·米莱特表示。“基于其安全性和有效性,Zemcelpro®(多罗库比塞尔)成为一种潜在的革命性疗法,有望改变当前的治疗模式,为那些采用传统方法治疗效果不佳、难以治愈的高风险和极高风险患者带来新希望。”À propos des études 关于研究Les résultats ont été évalués chez soixante adultes atteints de leucémie aiguë (AL) ou de syndrome myélodysplasique (MDS) à haut ou très haut risque ayant reçu Zemcelpro® (dorocubicel) dans le cadre de deux études prospectives de phase 2 réalisés aux États-Unis, au Canada et en Europe. La leucémie aiguë / MDS à haut risque était définie comme une maladie présentant une PFS à 2 ans attendue <40 % après une allogreffe de cellules souches hématopoïétiques (CSH) conventionnelle. 结果在60名患有急性白血病(AL)或高危或极高危骨髓增生异常综合征(MDS)的成人中进行了评估,这些患者在美国、加拿大和欧洲进行的两项前瞻性2期研究中接受了Zemcelpro®(多洛库贝西)治疗。高危急性白血病/MDS被定义为在传统造血干细胞移植(HSCT)后预计2年无进展生存率(PFS)<40%的疾病。La maladie à très haut risque était définie comme une deuxième allogreffe de CSH, une AL avec maladie active, une leucémie myéloïde aiguë (LMA) avec mutation TP53 et caryotype complexe, une LMA avec mutation EVI1, ou un MDS avec mutation multi-hit TP53. Les critères principaux de jugement étaient l'innocuité et la NRM ; les critères secondaires incluaient la PFS, la OS, l'incidence des rechutes, le prise de greffe, et la GVHD.. 极高风险疾病定义为:第二次异基因造血干细胞移植 (CSH)、活动性急性白血病 (AL)、伴有TP53突变和复杂核型的急性髓系白血病 (LMA)、伴有EVI1突变的LMA,或具有多打击TP53突变的骨髓增生异常综合征 (MDS)。主要评估标准为安全性和非复发死亡率 (NRM);次要标准包括无进展生存期 (PFS)、总生存期 (OS)、复发发生率、移植物植入情况以及移植物抗宿主病 (GVHD)。Soixante (60) patients (âge médian 43 ans ; extrêmes 19–66 ans) ont été transplantés ; 87 % avaient une leucémie aiguë (63 % myéloïde, 37 % lymphoïde) et 13 % un syndrome myélodysplasique (MDS). Trente pour cent (30 %) avaient échoué à une greffe allogénique précédente et 57 % répondaient aux critères de très haut risque. 六十名患者(中位年龄43岁;范围19-66岁)接受了移植;87%患有急性白血病(63%为髓系,37%为淋巴系),13%患有骨髓增生异常综合征(MDS)。其中30%的患者之前接受过同种异体移植失败,57%属于极高风险标准。Tous les patients ont complété les 2 à 3 ans de suivi prévu.. 所有患者都完成了计划中的2到3年的随访。A propos de Zemcelpro® 关于Zemcelpro®Zemcelpro®, également connu sous le nom de thérapie cellulaire UM171, est un nouveau produit cryoconservé de greffe de cellules souches hématopoïétiques comprenant deux composants, à savoir des cellules CD34+ expansées par UM171 (dorocubicel) et des cellules CD34- non expansées, chacune provenant de la même unité de sang de cordon.. Zemcelpro®,也被称为UM171细胞疗法,是一种新型的冷冻保存造血干细胞移植产品,包含两个成分,即通过UM171扩增的CD34+细胞(dorocubicel)和未扩增的CD34-细胞,两者均来自同一个脐带血单位。Zemcelpro® (dorocubicel) qui est développé par Cordex, une filiale à part entière d'ExCellThera, a été évalué chez 120 patients avec troubles sanguins graves dans plusieurs essais cliniques aux États-Unis, en Europe et au Canada. Zemcelpro® (dorocubicel) a reçu la désignation de médicament orphelin et la désignation de thérapie avancée en médecine régénérative (RMAT) de la FDA ainsi que la désignation de médicament orphelin, la classification de médicament de thérapie innovante (ATMP) et la désignation de médicament prioritaire (PRIME) de l'EMA.. Zemcelpro®(dorocubicel)由ExCellThera的全资子公司Cordex开发,已在美、欧、加多个国家的临床试验中对120名严重血液疾病患者进行了评估。Zemcelpro®(dorocubicel)获得了FDA的孤儿药资格、再生医学先进疗法(RMAT)认定,以及EMA的孤儿药资格、先进治疗药物(ATMP)分类和优先药物(PRIME)认定。Zemcelpro® (dorocubicel) a été évalué dans le cadre d'études de phase 2 chez des patients atteints de leucémies aiguës et de myélodysplasies à risque élevé et très élevé qui disposent d'options de traitement limitées avec de faibles résultats de survie et une incidence élevée de rechute selon les normes de soins actuelles, y compris les patients atteints d'une maladie réfractaire ou active , les patients nécessitant une deuxième greffe et les patients présentant des mutations TP53 ou d'autres anomalies génétiques. Zemcelpro®(dorocubicel)在二期临床研究中被评估,用于治疗患有急性白血病和高危及极高危骨髓增生异常综合征的患者。这些患者根据现行的治疗标准,治疗选择有限,生存率低,并且复发率高,其中包括难治性或活动性疾病患者、需要二次移植的患者以及携带TP53突变或其他基因异常的患者。Une étude pivot de phase 3 dans de cette population de patients sera lancée dès que possible.. 一项针对该患者群体的关键性3期研究将尽快启动。L'utilisation de la Zemcelpro® (dorocubicel) chez d'autres populations de patients, notamment les patients pédiatriques et les patients atteints de maladies hématologiques non malignes, est également à l'étude. 在其他患者群体中使用Zemcelpro®(多柔比星)也在研究中,包括儿科患者和患有非恶性血液病的患者。Pour tous les détails concernant les Mises en garde et précautions d'emploi ainsi que les Effets indésirables (y compris leur prise en charge appropriée), veuillez vous référer au Résumé des Caractéristiques du Produit (RCP) de Zemcelpro® pour l'UE. 有关警告和使用注意事项以及不良反应(包括其适当处理)的所有详细信息,请参阅 Zemcelpro® 在欧盟的产品特性摘要 (RCP)。Son innocuité et son efficacité n'ont pas été établies par d'autres autorités réglementaires, telles que la FDA ou Santé Canada. 其安全性和有效性尚未由其他监管机构(如FDA或加拿大卫生部)确立。A propos d'ExCellThera et de la technologie UM171 关于ExCellThera和UM171技术ExCellThera est un chef de file mondial des thérapies innovantes à base de cellules souches sanguines. La plate-forme exclusive Enhance ExCellThera 是血液干细胞创新疗法的全球领导者。其独有的增强平台TM 商标符号 (™)d'ExCellThera pour l'amplification et la qualité fonctionnelle cellulaire est conçue pour fournir une plus grande dose de cellules souches thérapeutiques fonctionnelles en amplifiant les cellules souches hématopoïétiques (CSH) de n'importe quelle source et en neutralisant les effets du stress induit par la culture ou l'édition de gènes. ExCellThera的细胞扩增和功能质量技术旨在通过扩增任何来源的造血干细胞(HSC)并中和由培养或基因编辑引起的应激效应,提供更高剂量的功能性治疗干细胞。ExCellThera s'associe à des compagnies biopharmaceutiques pour les aider à développer les meilleures thérapies cellulaires et géniques en tirant parti des technologies qui forment la plateforme Enhance. ExCellThera 与生物制药公司合作,帮助他们利用构成 Enhance 平台的技术开发最佳的细胞和基因疗法。TM 商标, y compris la molécule brevetée UM171 qui possède un mécanisme d'action de premier ordre pour l'expansion ,包括拥有首级作用机制的专利分子UM171,用于扩展ex vivo 体外et la fonctionnalité des CSH. Pour plus d'informations, visitez 以及CSH的功能。欲了解更多信息,请访问excellthera.com excellthera.com, et suivez-nous sur LinkedIn. ,关注我们的LinkedIn。Zemcelpro® est une marque déposée d'ExCellThera ou de ses sociétés affiliées. Zemcelpro® 是 ExCellThera 或其关联公司的注册商标。1 1Armand P, Kim HT, Logan BR, Wang Z, Alyea EP, Kalaycio ME, et al. Validation and refinement of the Disease Risk Index for allogeneic stem cell transplantation. Blood. 2014;123(23):3664–367 阿拉曼·P,金·HT,洛根·BR,王·Z,艾丽娅·EP,卡拉伊恰奥·ME,等。异基因干细胞移植的疾病风险指数的验证与优化。《血液》。2014;123(23):3664-367。Photo - 照片 -https://mma.prnewswire.com/media/2928711/ExCellThera_Zemcelpro_%C2%A0_dorocubicel__Positive_Results_in_High__a.jpg https://mma.prnewswire.com/media/2928711/ExCellThera_Zemcelpro_®_dorocubicel__Positive_Results_in_High__a.jpg21 21% %more press release views with 更多新闻发布观点 Request a Demo 请求演示
临床结果临床2期细胞疗法
2026-03-10
·动脉网
Zemcelpro 泽姆塞尔普罗® (dorocubicel) achieved remarkable and clinically meaningful survival results in patients with high- and very high-risk acute leukemias and myelodysplastic syndromes ®(dorocubicel)在高危和极高危急性白血病及骨髓增生异常综合征患者中取得了显著且具有临床意义的生存结果。Topline results from two Phase 2 studies to be presented on March 23, 2026 at the EBMT Annual Meeting in Madrid, Spain 两项二期研究的顶线结果将于2026年3月23日在西班牙马德里举行的EBMT年会上公布。MONTREAL 蒙特利尔, ,March 10, 2026 2026年3月10日/PRNewswire/ -- ExCellThera Inc. (ExCellThera), a world leader in blood stem cell expansion and metabolic fitness, and its wholly owned subsidiary Cordex Biologics (Cordex), announce positive results from recently completed Phase 2 studies of Zemcelpro® (dorocubicel), also known as UM171 Cell Therapy, in 60 adult patients with high- and very high-risk acute leukemias (AL) and myelodysplastic syndromes (MDS), a population known to experience high relapse rates and poor survival on conventional allogeneic stem cell transplantation (allo-HSCT). /PRNewswire/ -- 血液干细胞扩增和代谢适应性领域的全球领导者 ExCellThera Inc.(ExCellThera)及其全资子公司 Cordex Biologics(Cordex)宣布,近期完成的 Zemcelpro®(多罗库贝西尔),也称为 UM171 细胞疗法的 2 期研究取得了积极结果。该研究针对 60 名高危和极高危急性白血病(AL)及骨髓增生异常综合征(MDS)成人患者,这类人群在传统异基因造血干细胞移植(allo-HSCT)中复发率高且生存率低。Topline results will be presented on March 23, 2026 at the 52. 顶线结果将于2026年3月23日在第52届会议上公布。nd ndEBMT Annual Meeting in Madrid, Spain. 西班牙马德里EBMT年会。Continue Reading 继续阅读ExCellThera logo ExCellThera 标志At 24 months post-transplant, overall survival (OS), progression-free survival (PFS) and relapse-free survival (RFS) were 63.7%, 57.0% and 60%, respectively, among Zemcelpro® (dorocubicel)-treated patients. The cumulative incidences of relapse and non-relapse mortality (NRM) were 22.5% and 20.5%, respectively. 移植后24个月时,接受Zemcelpro®(多洛库贝)治疗的患者总生存率(OS)、无进展生存率(PFS)和无复发生存率(RFS)分别为63.7%、57.0%和60%。复发和非复发死亡率(NRM)的累积发生率分别为22.5%和20.5%。Age was the strongest predictor of NRM: patients <43 years experienced 0% NRM, with 24-month OS and PFS of 82.8% and 79.3%, respectively. Older patients (≥43 years) had comparable relapse rates but higher NRM. Grade III-IV acute graft-versus-host disease (GVHD) occurred in 20%, while moderate to severe chronic GVHD was infrequent at 7% at two years.. 年龄是NRM的最强预测因子:43岁以下患者NRM为0%,24个月总生存率(OS)和无进展生存率(PFS)分别为82.8%和79.3%。年龄较大(≥43岁)的患者复发率相似,但NRM更高。III-IV级急性移植物抗宿主病(GVHD)发生率为20%,而两年内中度至重度慢性GVHD的发生率较低,为7%。'This represents a remarkable and clinically meaningful achievement for Zemcelpro®(dorocubicel), especially given the historical overall survival of only 25–40% “这对Zemcelpro®(多洛库比利)而言是一项显著且具有临床意义的成就,尤其是考虑到历史上仅25%-40%的总体存活率。1 1for patients of similar disease risk receiving standard of care allogeneic grafts,' said Dr Guy Sauvageau, Chief Scientific Officer. 'Results are particularly promising in adults under 43 years of age, showing even greater benefits in both OS and PFS, with no cases of NRM.' “对于接受标准护理的同种异体移植、具有相似疾病风险的患者来说,结果特别有希望,尤其是在43岁以下的成年人中,显示出更大的总生存期和无进展生存期益处,且没有出现非复发死亡病例。”首席科学官Guy Sauvageau博士说道。'These findings suggest that Zemcelpro® (dorocubicel) could redefine the standard of care for patients with high- and very high-risk blood cancers,' said David Millette, CEO. 'Based on its safety and efficacy profile, this positions Zemcelpro® (dorocubicel) as a potentially transformative therapy, challenging the existing treatment paradigm and offering renewed hope for difficult-to-cure high- and very high-risk patients facing unfavorable outcomes with conventional approaches.'. “这些研究结果表明,Zemcelpro®(多罗库比利)可能重新定义高风险和极高风险血液癌症患者的标准治疗,”首席执行官大卫·米莱特表示。“基于其安全性和有效性,这使Zemcelpro®(多罗库比利)成为一种潜在的变革性疗法,挑战现有的治疗模式,并为那些使用传统方法效果不佳的难治高风险和极高风险患者带来新的希望。”About the studies 关于这些研究Outcomes were assessed in 60 adults with high- or very high–risk AL or MDS who received Zemcelpro® (dorocubicel) in two prospective phase 2 trials conducted in the United States, Canada and Europe. High-risk AL/MDS was defined as disease with an expected 2-year PFS <40% after conventional allo-HSCT. 在美国、加拿大和欧洲进行的两项前瞻性 2 期试验中,评估了 60 名接受 Zemcelpro®(多罗库比利)治疗的高危或极高危 AL 或 MDS 成人患者的结局。高危 AL/MDS 被定义为在传统异基因造血干细胞移植后预计 2 年无进展生存率(PFS)<40% 的疾病。Very high-risk disease was defined as a second allo-HSCT, AL with active disease, acute myeloid leukemia (AML) with TP53 mutation and complex karyotype, AML with EVI1 mutation, or MDS with multi-hit TP53 mutation. The primary endpoints were safety and NRM; secondary endpoints included PFS, OS, relapse incidence, engraftment, and GVHD.. 极高危疾病被定义为接受第二次异基因造血干细胞移植(allo-HSCT)、伴有活动性疾病的急性白血病(AL)、携带TP53突变和复杂核型的急性髓系白血病(AML)、携带EVI1突变的AML,或携带多击TP53突变的骨髓增生异常综合征(MDS)。主要终点为安全性和非复发死亡率(NRM);次要终点包括无进展生存期(PFS)、总生存期(OS)、复发率、植入情况和移植物抗宿主病(GVHD)。Sixty (60) patients (median age 43 years; range 19–66) were transplanted; 87% had AL (63% myeloid, 37% lymphoid) and 13% had MDS. Thirty percent (30%) had failed a prior allogeneic transplant and 57% met very high-risk criteria. All patients completed planned 2-3 years of follow-up. 六十名患者(中位年龄43岁;范围19-66岁)接受了移植;87%患有AL(63%为髓系,37%为淋巴系),13%患有MDS。30%的患者之前接受过同种异体移植失败,57%符合极高风险标准。所有患者均完成了计划中的2至3年随访。About Zemcelpro® 关于Zemcelpro®Zemcelpro® (dorocubicel), also known as UM171 Cell Therapy, is a novel personalized cryopreserved haematopoietic stem cell transplantation product containing two components, namely UM171-expanded CD34+ cells (dorocubicel) and unexpanded CD34- cells, each derived from the same cord blood unit. Zemcelpro®(多罗库贝西尔),又称UM171细胞疗法,是一种新型的个性化冷冻保存造血干细胞移植产品,包含两个组分,即UM171扩增的CD34+细胞(多罗库贝西尔)和未扩增的CD34-细胞,两者均源自同一个脐带血单位。Zemcelpro® (dorocubicel) has recently received conditional marketing authorization from the European Commission for the treatment of adults with haematological malignancies requiring allogeneic haematopoietic stem cell transplantation following myeloablative conditioning, for whom no other suitable donor cells are available.. Zemcelpro®(多罗库比利)近日获得欧洲委员会的附条件上市许可,用于治疗需要在骨髓清除性预处理后进行同种异体造血干细胞移植的血液系统恶性肿瘤成人患者,这些患者没有其他合适的供体细胞可用。For complete product information, including warnings and precautions for use and adverse reactions (and their appropriate management), please refer to the EU Summary of Product Characteristics (SmPC) for Zemcelpro® (dorocubicel). 有关完整的产品信息,包括使用警告和注意事项以及不良反应(及其适当管理),请参阅 Zemcelpro®(多柔比星)的欧盟产品特性摘要 (SmPC)。Additional regulatory filings are planned for Zemcelpro® (dorocubicel) with other health authorities, including in the US, Canada, the UK, and Switzerland. Cordex is also actively seeking strategic partnerships to support and accelerate the commercialization of Zemcelpro® (dorocubicel) in Europe and other international markets.. 计划向包括美国、加拿大、英国和瑞士在内的其他健康监管机构提交Zemcelpro®(多罗库比利)的更多监管文件。Cordex还在积极寻求战略合作伙伴,以支持并加速Zemcelpro®(多罗库比利)在欧洲及其他国际市场的商业化进程。Zemcelpro® (dorocubicel) has been evaluated in 120 patients with haematologic malignancies in clinical trials in the United States, Europe and Canada. Zemcelpro® (dorocubicel) has received orphan drug designation and regenerative medicine advanced therapy (RMAT) designations from the FDA as well as orphan medicinal product designation, advanced therapy medicinal product (ATMP) classification and priority medicines (PRIME) designation from the EMA.. Zemcelpro®(多罗库比利)已在美、欧、加的临床试验中对120名血液系统恶性肿瘤患者进行了评估。Zemcelpro®(多罗库比利)获得了FDA授予的孤儿药资格和再生医学先进疗法(RMAT)资格,以及EMA授予的孤儿药产品资格、先进治疗药品(ATMP)分类和优先药物(PRIME)资格。Zemcelpro® (dorocubicel) has been tested in Phase 2 trials in patients with high- and very high-risk acute leukemias and myelodysplasias who have limited treatment options with low survival outcomes and high incidence of relapse under the current standard of care, including patients with patients with TP53 mutations or other genetic abnormalities, patients requiring a second transplant, and patients with refractory or active disease. Zemcelpro®(多罗库比利)已在二期临床试验中对高危和极高危急性白血病及骨髓增生异常综合征患者进行了测试,这些患者治疗选择有限,在当前标准治疗下生存率低、复发率高,其中包括携带TP53突变或其他基因异常的患者、需要二次移植的患者以及难治性或活动性疾病的患者。A pivotal Phase 3 trial in this patient population will be initiated as soon as possible.. 这一患者群体的关键性III期试验将尽快启动。The use of Zemcelpro® (dorocubicel) in other patient populations, including pediatric patients and patients with non-malignant haematological diseases, is also being investigated. Zemcelpro®(多罗库比利)在其他患者群体中的应用,包括儿科患者和非恶性血液病患者,也正在研究中。The product safety and efficacy have not yet been established by other regulatory agencies, such as the U.S. FDA, the MHRA and Health Canada. 产品的安全性和有效性尚未由其他监管机构(如美国 FDA、MHRA 和加拿大卫生部)确定。About ExCellThera and UM171 Technology 关于ExCellThera和UM171技术ExCellThera is a world leader in enhanced blood stem cell therapies. ExCellThera's proprietary Enhance ExCellThera 是增强型血液干细胞疗法的全球领导者。ExCellThera 专有的增强技术TM 商标platform for cell expansion and metabolic fitness is designed to deliver a greater dose of functional therapeutic stem cells by expanding haematopoietic stem cells (HSCs) from any source and counteracting the effects of culture or gene editing induced stress. ExCellThera partners with biopharmas to help them develop best-in-class cell and gene therapies by leveraging the technologies that form the Enhance. 用于细胞扩增和代谢适应性的平台旨在通过扩增来自任何来源的造血干细胞(HSCs)并抵消培养或基因编辑诱导的压力影响,提供更高剂量的功能性治疗干细胞。ExCellThera与生物制药公司合作,利用构成Enhance的技术,帮助他们开发一流的细胞和基因疗法。TM 商标platform, including the proprietary molecule UM171 which has a first-in-class mechanism of action for 平台,包括具有首创作用机制的专有分子UM171,ex vivo 离体expansion and metabolic fitness of HSCs. For additional information, visit HSC的扩增和代谢适应性。欲了解更多信息,请访问 excellthera.com excellthera.com, and follow us on LinkedIn. ,并在LinkedIn上关注我们。Zemcelpro®is a registered trademark of ExCellThera or its related companies. Zemcelpro® 是 ExCellThera 或其相关公司的注册商标。1 1Armand P, Kim HT, Logan BR, Wang Z, Alyea EP, Kalaycio ME, et al. Validation and refinement of the Disease Risk Index for allogeneic stem cell transplantation. Blood. 2014;123(23):3664–367 阿曼德 P, 金 HT, 洛根 BR, 王 Z, 艾利亚 EA, 卡拉伊乔 ME, 等。异基因干细胞移植的疾病风险指数的验证与优化。《血液》。2014;123(23):3664–367。Photo - 照片 -https://mma.prnewswire.com/media/2928711/ExCellThera_Zemcelpro_%C2%A0_dorocubicel__Positive_Results_in_High__a.jpg https://mma.prnewswire.com/media/2928711/ExCellThera_Zemcelpro_®_dorocubicel__Positive_Results_in_High__a.jpg21 21% %more press release views with 更多新闻发布视图与 Request a Demo 请求演示
100 项与 多罗库比利 相关的药物交易
登录后查看更多信息
研发状态
批准上市
10 条最早获批的记录, 后查看更多信息
登录
| 适应症 | 国家/地区 | 公司 | 日期 |
|---|---|---|---|
| 血液肿瘤 | 欧盟 | 2025-08-25 | |
| 血液肿瘤 | 冰岛 | 2025-08-25 | |
| 血液肿瘤 | 列支敦士登 | 2025-08-25 | |
| 血液肿瘤 | 挪威 | 2025-08-25 |
未上市
10 条进展最快的记录, 后查看更多信息
登录
| 适应症 | 最高研发状态 | 国家/地区 | 公司 | 日期 |
|---|---|---|---|---|
| 髓系肿瘤 | 临床2期 | 美国 | 2021-12-01 | |
| 急性白血病 | 临床2期 | 加拿大 | 2019-04-01 | |
| 骨髓增生异常综合征 | 临床2期 | 加拿大 | 2019-04-01 | |
| 难治性急性白血病 | 临床2期 | 加拿大 | 2019-04-01 | |
| 多发性骨髓瘤 | 临床2期 | 加拿大 | 2018-03-07 | |
| 浆细胞白血病 | 临床2期 | 加拿大 | 2018-03-07 | |
| 急性胸部综合征 | 临床1期 | 美国 | 2021-07-13 | |
| 镰状细胞血症 | 临床1期 | 美国 | 2021-07-13 | |
| 造血干细胞移植 | 药物发现 | 欧盟 | 2020-12-16 | |
| 移植物抗宿主病 | 药物发现 | 美国 | 2018-12-13 |
登录后查看更多信息
临床结果
临床结果
适应症
分期
评价
查看全部结果
| 研究 | 分期 | 人群特征 | 评价人数 | 分组 | 结果 | 评价 | 发布日期 |
|---|
临床1/2期 | 多发性骨髓瘤 二线 | 末线 | 三线 | 19 | 鏇壓窪夢築鹹鹽夢積醖(築膚壓糧顧築鬱鏇壓願) = 廠築襯醖壓觸膚鹹繭蓋 衊遞遞選廠顧淵醖蓋鹹 (齋醖糧繭齋鬱窪蓋夢鏇 ) 更多 | 积极 | 2024-10-15 | ||
临床2期 | 50 | 衊齋醖積窪夢繭獵襯鏇(網鏇積獵衊襯築襯夢壓) = 壓簾製構積壓願鏇鬱壓 醖築鹽憲衊選構廠積膚 (範鏇餘範鏇遞廠廠窪醖 ) 更多 | 积极 | 2023-12-12 | |||
N/A | - | (Expanded Cord Blood Grafts) | 繭壓遞餘糧獵遞襯獵鏇(遞鏇願鹽繭鑰淵遞醖積) = 淵鏇製鏇壓鑰廠蓋鏇糧 廠遞廠廠夢糧醖廠糧艱 (築簾襯壓鏇願壓選壓繭 ) 更多 | - | 2023-02-01 | ||
临床1期 | 22 | (Myeloablative conditioning regimen + single CB transplant) | 製艱繭淵築憲簾糧遞鏇(製顧範選襯糧願鹹憲鹹) = 憲製構憲蓋範壓鏇顧醖 獵顧製壓淵鑰夢觸鹽夢 (範願製膚鹹製鏇鏇積願 ) 更多 | 积极 | 2020-03-01 |
登录后查看更多信息
转化医学
使用我们的转化医学数据加速您的研究。
登录
或

药物交易
使用我们的药物交易数据加速您的研究。
登录
或

核心专利
使用我们的核心专利数据促进您的研究。
登录
或

临床分析
紧跟全球注册中心的最新临床试验。
登录
或

批准
利用最新的监管批准信息加速您的研究。
登录
或

特殊审评
只需点击几下即可了解关键药物信息。
登录
或

生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用

