预约演示
更新于:2026-06-09
Mivelsiran
更新于:2026-06-09
概要
基本信息
非在研机构- |
最高研发阶段临床2期 |
首次获批日期- |
最高研发阶段(中国)- |
特殊审评- |
登录后查看时间轴
结构/序列
使用我们的RNA技术数据为新药研发加速。
登录
或
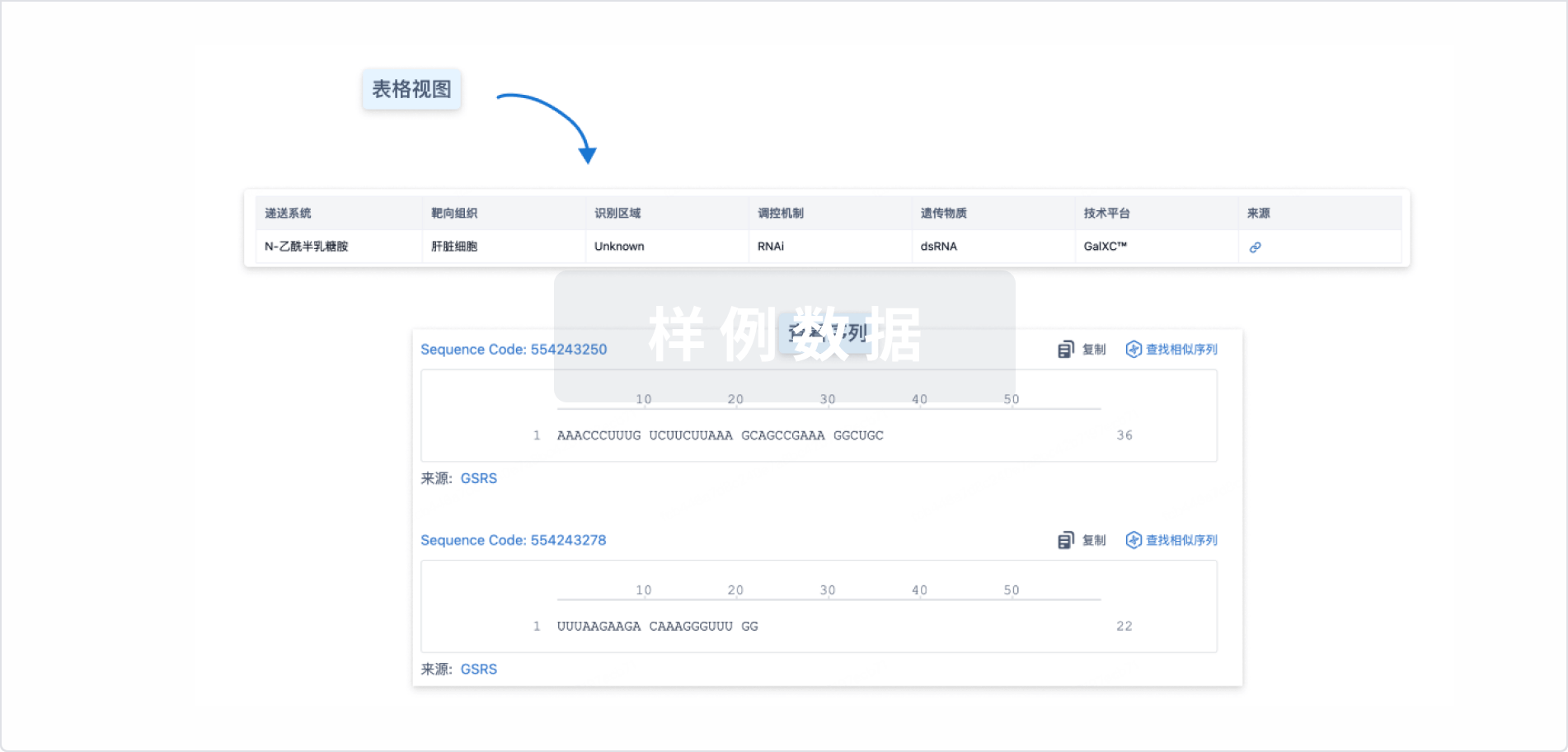
Sequence Code 565398339

来源: *****
Sequence Code 1031376348

来源: *****
关联
2
项与 Mivelsiran 相关的临床试验NCT06393712
A Phase 2, Randomized, Double-blind, Placebo-controlled Study to Evaluate the Efficacy, Safety, Tolerability, and Pharmacodynamics of Intrathecally Administered ALN-APP in Patients With Cerebral Amyloid Angiopathy (CAA)
The purpose of the study is to evaluate the effect of ALN-APP on measures of CAA disease progression and to characterize the safety, tolerability, and pharmacodynamics (PD) of ALN-APP in adult patients with sporadic CAA (sCAA) and Dutch-type CAA (D-CAA). The study will be conducted over 2 periods: a 24-month double-blind treatment period and an optional 18-month open-label extension (OLE) period. The estimated duration of study participation, inclusive of screening, treatment, and additional safety follow-up, is up to 50 months.
开始日期2024-05-17 |
NCT05231785
A Randomized, Double-blind, Placebo-controlled Single Ascending Dose and Open-label Multi-dose Study to Evaluate the Safety, Tolerability, Pharmacokinetics and Pharmacodynamics of Intrathecally Administered ALN-APP in Adult Patients With Early-onset Alzheimer's Disease (EOAD)
The purpose of this study is to evaluate the safety, tolerability, pharmacodynamics (PD) and pharmacokinetics (PK) of a single dose and multiple doses of ALN-APP administered by intrathecal (IT) injection in adult patients with early-onset Alzheimer's Disease (EOAD). Maximum treatment duration for Part A: single dose. Maximum treatment duration for Part B: 12 months.
开始日期2022-02-04 |
100 项与 Mivelsiran 相关的临床结果
登录后查看更多信息
100 项与 Mivelsiran 相关的转化医学
登录后查看更多信息
100 项与 Mivelsiran 相关的专利(医药)
登录后查看更多信息
2
项与 Mivelsiran 相关的文献(医药)2025-12-01·Alzheimers & Dementia
Small Interfering RNA Targeting Amyloid‐Beta Precursor Protein Reduces Alzheimer’s Disease Pathology in 5xFAD Mice
Article
作者: Sostelly, Alexandre ; Taillie, Dominique ; Mooney, Timothy ; Brown, Kirk ; Cha, Diana ; Ferraro, Gina ; Bittner, Kelsey
Abstract:
Background:
Amyloid‐beta precursor protein (APP) is the source of all amyloid‐beta (Aβ), the amyloidogenic peptide that aggregates in Alzheimer’s disease (AD). Lowering APP mRNA expression with RNA interference (RNAi) is a novel treatment strategy under investigation for AD. The 5xFAD mouse model of AD overexpresses APP and PSEN1 with five familial AD mutations, resulting in robust pathology, including amyloid plaques, microgliosis, inflammation, synaptic loss, neuronal loss, and behavioral changes. This work evaluates the effects of early and late intervention with an APP‐lowering small interfering RNA (siRNA) in this established AD model.
Method:
APP‐targeting siRNAor artificial CSF (aCSF; control) was administered by intracerebroventricular injection in 5xFAD mice at late (8‐months‐old) and early (12‐weeks‐old) disease stages, and in age‐matched wild‐type mice. Late intervention mice received either aCSF or 300µg single dose. Early intervention mice received one of four dose regimens: control (aCSF:aCSF), transient lowering (75µg APP siRNA:aCSF), moderate sustained lowering (75µg APP siRNA:75µg APP siRNA), or robust sustained lowering (300µg APP siRNA:300µg APP siRNA). The impact of late and early siRNA intervention on molecular, biochemical, histological, and disease‐related behavioral outcomes (assessed using the elevated plus‐maze and open‐field assay) were assessed in 12‐month‐old mice (Figure 1).
Result:
Treatment in the late intervention group lowered amyloid burden to levels below the 8‐month baseline (Figure 2A, 2B). Compared with age‐matched aCSF mice, late treatment decreased pathogenic amyloid species, Aβ40 and Aβ42, in CSF and tissue, reduced markers of glial inflammation, and reduced plasma neurofilament light chain (NfL). No significant behavioral changes were noted (Figure 3A).Treatment in early intervention groups lowered amyloid burden in tissue (Figure 2C, 2D), reduced markers of glial inflammation, reduced plasma NfL, and decreased anxiety‐like behavior, all in a dose‐dependent manner. Notably, robust, sustained lowering of APP completely prevented the emergence of disease‐associated anxiety‐like behavior (Figure 3B).
Conclusion:
Treatment with APP‐lowering siRNA reduced AD pathology in the 5xFAD mouse model. Early intervention with the highest dose regimen prevented the emergence of behavioral deficits and substantially improved many aspects of disease phenotype. These results support the continued development of mivelsiran, an investigational, first‐in‐class APP‐lowering RNAi therapeutic, in patients with AD (NCT05231785).
2025-12-01·Alzheimers & Dementia
Multiple‐Dose Results from an Ongoing Phase 1 Study of Mivelsiran, an Investigational RNA Interference Therapeutic Targeting Amyloid‐Beta Precursor Protein for Alzheimer’s Disease
Article
作者: Cohen, Sharon ; Deering, Robert W ; Mummery, Catherine J. ; Farrugia, Lynn ; Blackburn, Daniel J. ; Ducharme, Simon ; Shirvan, Julia ; McDade, Eric ; Brosch, Jared R. ; Chaudhari, Sandeep ; Vijverberg, Everard G.B. ; Sostelly, Alexandre
Abstract:
Background:
Single doses of mivelsiran, an investigational RNA interference (RNAi) therapeutic, have demonstrated robust amyloid‐beta precursor protein (APP) lowering in the CNS. We report additional interim safety and pharmacodynamic data in patients with early‐onset Alzheimer’s disease (EOAD) who received single ascending doses (SAD) and, for the first time, multiple doses of mivelsiran in the ongoing Phase 1 study (NCT05231785).
Methods:
Patients with EOAD (symptom onset <65 years of age, Clinical Dementia Rating global score 0.5 or 1.0, Mini‐Mental State Examination score >20) were randomized to a single intrathecal dose of mivelsiran 25–100mg or placebo for 6 months (plus up to 6 months follow‐up if needed for washout). After washout, patients could enter a separate multiple ascending dose (MAD) portion and receive open‐label mivelsiran. Presented here are data from a SAD cohort of mivelsiran 100mg and a MAD cohort of mivelsiran 50mg every 6 months (Q6M). Frequency of adverse events (AEs) and pharmacodynamics were primary and secondary endpoints, respectively.
Results:
Forty‐five patients were enrolled in SAD cohorts (as of 11/20/2024). Of those, 9 patients were randomized to mivelsiran 100mg or placebo (mean [SD] age, 64.1 [3.7] years; 33.3% male; 100% white). Most AEs were mild or moderate. Peak mean (SE) change from baseline in cerebrospinal fluid (CSF) soluble APP beta (sAPPβ) at Month 1 (─84.5% [1.3]) was largely sustained through Month 10 (─61.1% [2.8]).After meeting washout criteria, 10 patients from SAD cohorts received mivelsiran 50mg Q6M (mean [SD] age, 59.9 [4.4] years; 70.0% male; 70.0% white). No serious or severe AEs were reported. At Day 15 after the first dose, mean (SE) change from baseline in CSF sAPPβ was ─63.7% (5.0); at Month 1 after the second dose, change was ─83.8% (2.3). Additional data will be presented.
Conclusions:
In this first report of multiple‐dose clinical data for a CNS‐targeting RNAi therapeutic, single and multiple doses of mivelsiran were generally well tolerated and continued to demonstrate robust, durable, dose‐dependent CSF sAPPβ reductions. Further sAPPβ lowering was observed after a second dose of mivelsiran 50mg. These results support further evaluation of mivelsiran in patients with Alzheimer’s disease or cerebral amyloid angiopathy.
167
项与 Mivelsiran 相关的新闻(医药)2026-06-08
·商图药讯
作者:清风
小核酸赛道,正在上演一场真实的“资本盛宴”。2025年,小核酸领域BD交易总金额已突破300亿美元,创下历史新高。全球已上市小核酸药物总销售额突破71亿美元,同比增长近40%。
凭借直击“不可成药”靶点、极高的研发效率、长效治疗优势等,小核酸药物已被视为继小分子、抗体药物后的第三大药物类型。目前,FDA已批准近20款小核酸药物在全球范围内上市。
不过,由于GalNAc等递送系统的局限性,目前超过90%获批的小核酸药物集中于肝脏靶向,而人类约90%的致病基因却表达于肝外组织。这意味着,阿尔茨海默病、心力衰竭、慢性肾病等大量未满足的临床需求,仍处于核酸药物的“无人区”。因此,走出“肝脏舒适区”,成为小核酸药物下一步的攻坚方向。而这一切的前提,在于肝外递送技术的突破。
GalNAc的荣光与隐忧
小核酸药物是一类由十几个至几十个核苷酸组成的短链核酸分子,通过干预信使RNA(mRNA)的表达,从源头上调控致病蛋白的生成,从基因源头精准干预致病蛋白的生成,实现“治标又治本”的治疗效果。而且小核酸药物理论上可以靶向人类基因组中所有致病基因,这种全新的药物设计逻辑,让许多曾经“不可成药”的靶点迎来了被攻克的可能。
根据作用机制不同,小核酸药物包括RNAi药物(siRNA)、反义寡核苷酸(ASO)、微小RNA(miRNA)、小激活RNA(saRNA)以及核酸适配体等。其中,ASO和siRNA是目前技术最成熟的两大类别。
虽然目前小核酸药物研发如火如荼,且全球已有19款(踢除已退市)小核酸药物获批上市,但小核酸药物的研发却经历多次起落。与小分子、蛋白药物不同,小核酸如果未经制剂化处理,直接进入体内后会被快速降解,无法到达靶向部位。而且小核酸分子携带大量负电荷,且亲水性强,难以穿透带负电荷且为脂质双分子层的细胞膜,到达细胞内部。因此如何将小核酸药物精准送达疾病部位,并保持活性,成为小核酸药物研发成功的关键。
纵观小核酸药物发展史,很大程度上就是递送技术的迭代史。脂质纳米粒(LNP)是小核酸递送首个获得成功的技术平台。2018年,Alnylam的siRNA药物Onpattro(patisiran)成功获FDA批准上市,成为全球首个利用LNP递送技术的小核酸药物,用于治疗遗传性转甲状腺素蛋白淀粉样变性(ATTR-PN),打破了小核酸药物在递送难题凸显和资本相继退出的黑暗时期。LNP可安全、高效将核酸递送至肝脏靶细胞的作用得到有效证明。
作为小核酸递送首个获得成功的平台,LNP虽然解决了核酸易降解、难入胞等难题,但其存在内体逃逸效率低、免疫原性与安全性风险等,例如LNP的脂质成分或封装的核酸可能激活免疫系统,引发炎症反应或细胞因子释放综合征(CRS)等。
为了克服这些局限性,GalNAc(N-乙酰半乳糖胺)偶联技术应运而生。Alnylam公司的脱颖而出,正是源于对这一瓶颈的突破与管线策略的精准落地。
GalNAc 是唾液酸糖蛋白受体(ASGPR)的高亲和力配体,而 ASGPR 在肝细胞膜上高度特异性、高密度表达。这意味着,将GalNAc与siRNA偶联,就能实现对肝脏的精准靶向递送。2019年,Alnylam的Givosiran获批,用于治疗成人急性肝卟啉症(AHP),成为历史上首个获批采用GalNAc偶联技术的siRNA药物。
而GalNAc技术也凭借其卓越的肝脏靶向效率、安全性和用药便利性,成为当前siRNA药物开发的主流和首选(2019年之后获批的siRNA药物均采用了该技术)。其中,Alnylam公司已上市的6款siRNA药物中,5款均应用了GalNAc偶联技术,展现出碾压式的平台化孵化能力。
GalNac递送系统的突破,让小核酸药物实现了高效、低毒的肝脏靶向递送,适应症从罕见病成功扩展至常见病,也催生了多个重磅品种。例如诺华/Alnylam上市的全球首款siRNA超长效降脂药——Leqvio(英克司兰钠,Inclisiran)2025年销售额飙至11.98亿美元,同比增长57%,跻身“重磅炸弹”药物行列。
GalNac递送系统虽然使小核酸药物迎来爆发期,但它只解决了肝脏递送问题。肝脏以外的靶向递送——无论是中枢神经系统、骨骼肌、肾脏还是肺部,仍是制约行业扩容、适应症拓展的核心障碍,目前全球尚未有通过全身给药实现特异性肝外靶向递送的小核酸新药上市,这也成为行业亟待突破的方向。
攻坚肝外递送,全球药企抢滩布局
走向肝外,挑战被无限放大。肝外组织生理屏障极其严密且异质性强:血脑屏障(BBB)使全身给药后脑组织浓度仅为肝脏的万分之一;肾脏面临复杂的滤过屏障与快速清除;心肌细胞药物摄取能力弱;脂肪组织则因血供稀少导致水相扩散受限等。因此,打破“肝脏依赖”,实现全身多器官精准靶向,已成为下一代核酸药物定义竞争格局的核心共识。
面对肝外递送的高墙,全球跨国制药巨头与前沿Biotech正多路并进,通过配体偶联、多肽修饰、抗体偶联(AOC)、LNP工程化及外泌体等策略,向中枢神经、肌肉、心脏、脂肪及肺部等组织发起冲锋。
中枢神经系统(CNS)递送:跨越血脑屏障的智取
CNS递送的最大障碍是血脑屏障(BBB)。Alnylam凭借C16亲脂性修饰技术突破屏障,依托该技术平台,其开发的早发性阿尔茨海默症管线ALN-APP(即Mivelsiran)在I期临床中展现出高达79%的Aβ蛋白降幅,疗效持续12个月,展现出良好的临床潜力,是全球首个在人脑内实现基因沉默的siRNA疗法。
国内艾码生物则另辟蹊径,利用旗下IVSA平台以肝脏为“生物反应器”,合成siRNA并自组装为分泌性外泌体,能够自然穿越BBB,将 HTT siRNA 递送至大脑细胞。公司旗下产品ER2001已成为国内首个进入临床的静脉给药CNS靶向siRNA。
肌肉与心脏递送:攻克摄取低下的难题
肌肉与心脏是遗传病与慢病的重镇。Avidity公司凭借抗体-寡核苷酸偶联(AOC)平台,攻克肌肉递送瓶颈,其治疗1型强直性肌营养不良(DM1)的AOC 1001已进入III期临床,靶基因敲低达46%。
心脏递送方面,国内尧景基因基于独家筛选的心肌细胞特异性高表达受体X,开发出STT-Cardio平台,在小鼠实验中实现了超过95%的心脏基因敲低,持续时间超60天,远超同类技术。
瑞博生物的RiboPepSTAR平台则在心肌递送中展现出高度选择性,动物模型试验中,1mg/kg低剂量即可实现约80%的靶基因沉默,且在肝肾中几乎无脱靶。
代谢与肾脏递送:唤醒沉默的器官
脂肪递送被视为下一代代谢疾病与肥胖治疗的重要方向。Arrowhead的TRiM平台在脂肪靶向药物ARO-ALK7的I/IIa期临床中,使脂肪组织ALK7 mRNA敲低94%,内脏脂肪显著降低14.1%。
瑞博生物的RiboPepSTAR平台,在非人灵长类中实现了高达96%的脂肪组织靶基因敲低,疗效维持超85天。
肾脏递送则面临滤过与毒性双重挑战。Judo Bio公司开发的STRIKER平台,利用Megalin 受体能够将 siRNA 特异性地递送到肾脏的近端小管上皮细胞(PTECs),实现70%的靶基因敲低。
瑞博生物的肾靶向平台则实现了siRNA在肾近端小管的高效特异性富集,“肾肝比”超数百倍,靶基因抑制率近80%,其候选药物SR103已进入IND申报阶段。
肺部与mRNA靶向:LNP的工程化重塑
对于mRNA药物,传统LNP的“肝脏趋向性”是其最大枷锁。通过电荷修饰与吸入给药,LNP正实现肺靶向突破。Arcturus的LUNAR平台通过雾化吸入将mRNA递送至肺部治疗囊性纤维化,已进入II期临床。
国内中科院团队开发的SELECT平台,更进一步在mRNA骨架中引入miRNA靶点,实现了从“器官水平”向“细胞水平”的精准双重靶向,在肺转移瘤模型中抑瘤效果显著。
产业化卡点与中国力量的突围路径
总之,肝外递送能力已成为衡量企业核心竞争力的关键标尺。在全球技术竞速中,中国小核酸企业正加速缩短差距,甚至在局部领域实现领跑。然而,中国小核酸产业仍面临产业化的严峻考验。
递送技术的转化鸿沟
尽管国内企业在肝外递送的专利与临床前数据上屡有突破,但从动物模型到人体的转化成功率极低。递送技术的突破不能仅靠“论文数据”,内体逃逸率、跨物种稳定性及人体内生物分布的验证,需要漫长的试错与巨大资金投入。
CMC工艺与供应链的脆弱性
小核酸固相合成在放大过程中面临杂质谱变化、收率衰减与成本失控等痛点,临床前毫克级工艺的漂亮数据往往难以在公斤级量产中重现。更致命的是供应链风险:合成所需的亚磷酰胺单体等核心原料高度依赖海外供应商,地缘政治波动随时可能导致断供。破局之道在于CMC前置,在早期设计阶段即启动工艺放大与杂质谱研究。
全球监管与临床策略的重塑
美国国会已释放信号,要求FDA不得接受或依赖来源于中国的早期临床数据。这意味着国产管线出海在IND阶段即需规划多中心临床,甚至直接在海外设立运营节点。
小核酸药物正迎来从“肝脏专属”迈向“全身覆盖”的历史性拐点。肝外递送技术的全面突围,不仅是科学层面的里程碑式胜利,更将重塑全球药物研发格局,将核酸药物从罕见病的小众领域推向心血管、神经退行性疾病与代谢综合征等千亿级慢病市场。未来十年,谁掌握肝外递送技术,谁就掌控核酸药物的未来。
对于中国创新力量而言,这既是前所未有的机遇,也是脱胎换骨的考验。凭借在多肽筛选、AOC及LNP工程化等领域的积极布局,中国企业已具备了与国际巨头同台竞技的技术底座。然而,只有当原创递送平台、稳健的CMC体系与全球多中心临床策略形成闭环,中国小核酸产业才能真正完成从“BD叙事”到“价值兑现”的蜕变,为全球患者提供属于中国的创新治疗方案。
参考:
1.Johnson NE, et al. An Antibody Oligonucleotide Conjugate for Myotonic Dystrophy Type 1. N Engl J Med. 2026;394(7):619-630.
2.implified Lipid Nanoparticles for Tissue- And Cell-Targeted mRNA Delivery Facilitate Precision Tumor Therapy in a Lung Metastasis Mouse Model. Advanced Materials (2024). DOI: 10.1002/adma.202409812.
3.相关企业公开资料
封面图来源:摄图网
—The End—
核酸药物作为继小分子化药、抗体药物之后的第三代治疗范式,正以强劲势头重塑新药研发格局,解锁罕见病、慢病、中枢神经系统疾病等领域的治疗新可能!
BioCon China 核酸药物专场集结谭蔚泓院士等顶尖学者,以及艾博、瑞博、石药、深信生物等行业龙头代表,聚焦研发突破、递送技术与临床转化,诚邀业界同仁齐聚一堂,共探核酸产业发展新机遇。
限时多重专属权益落地
抢占年度精准商机
>> BioCon2026 <<
抢抓行业窗口期 报名参展享重磅专属礼遇
01
签约赞助/展台,即可抽惊喜盲盒(100%中奖):
一等奖(1 名):7折优惠券+ 升级赞助商权益
二等奖(3 名):7.5折优惠券+VIP票3张
三等奖(5 名):8折优惠券+会刊广告权益
参与奖:大会定制礼品包 + 1 年行业资讯服务
名额有限,详情联系组委会
BIO-Partnering即将登场,打开商务约见机遇之门
02
🔥 与国际大咖、MNC 高层一对一面对面!
💼 抢占国际合作先机!
🌍 链接全球医药顶层资源
📅 BioCon-Partnering 高端约见通道开启,付费即可预约国际嘉宾 & MNC 高管,线上约见・线下面谈,商机一步锁定,费尊享精准对接,让跨国合作高效直达!
🔗 专属礼遇通道:
扫码抢先获取BioCon-Partnering商务约见票
限时报名福利加码 一键链接全球产业资源
03
转发指定推文至朋友圈,或转发至行业群1个,即可获取每日限量50张免费参会门票!(仅含论坛及展区通票)
🔗 专属礼遇通道:
扫码注册获取限时活动免费票
联系组委会
BioCon 2026
咨询参会/演讲/赞助/媒体合作
电话/微信:+86 13122785593
邮箱:biocon@bmapglobal.com
扫码填写意向信息
点击"阅读原文",即可注册参会 ↓↓
2026-06-08
第三届小核酸药物2026盛会详情请点击图片了解,招商热线188 0194 0589
文章转自2026年06月01日太平洋医药团队报告,分析师:周豫/戎晓婕
投资要点
产业周期:小核酸跨越可成药里程碑,肝外递送开启新一轮价值跃迁。GalNAc肝脏递送技术的成熟,以及英克西兰(PCSK9)等重磅慢病RNAi药物的全球销售放量,标志着小核酸迈入商业化扩张周期。随着肝脏靶向陷入同质化红海竞争,行业核心焦点从“小核酸能否成药”转化为“谁能率先攻克肝外高效递送”。
技术矩阵:三大肝外递送路线演进。当前临床阶段管线中,AOC(抗体-小核酸偶联)利用抗体或抗体片段结合TfR1等受体,通过受体介导内吞机制进入组织细胞,具有高特异性,肌肉罕见病领域的成药性已获注册临床验证;POC(多肽-小核酸偶联)依托细胞穿透肽或靶向肽提升细胞摄取与内体逃逸,具备结构轻量化、免疫原性低的优势,处于早期探索阶段;脂质-小核酸偶联通过C16/胆固醇等亲脂修饰,增强与血浆白蛋白/脂蛋白的相互作用,借助天然脂蛋白转运通路促进组织富集与细胞内化,处于概念验证阶段,在CNS、脂肪等领域展现应用潜力。
组织范式:靶向策略因“障”制宜,差异化递送格局初显。不同组织的独特生物学屏障催生了多元化递送范式,肌肉围绕TfR1及αvβ6介导的主动摄取,呈现AOC技术领先与POC效率提升的路线博弈;CNS围绕突破血脑屏障攻坚,形成鞘内注射侵入性给药与TfR1介导系统给药双轨并驱,拓展阿尔茨海默病等大适应症;肺部采取雾化吸入实现局部递送;脂肪与心脏等高潜力组织在慢病蓝海中探索早期技术边界。
26H2进入密集的催化剂兑现期。1)Alnylam和Arrowhead迎来脂肪、CNS、肌肉、肝脏双靶点项目等数据的集中释放;2)Ionis三款重磅ASO药物步入上市监管审批窗口;3)AOC上市申报在即,POC早期数据迎来关键更新。
未来,肝外递送的核心壁垒不仅在于效率提升,更在于能否搭建可复制、可横向扩展的组织特异性靶向递送体系。我们认为,递送平台承载技术复利,率先通关者有望主导下一代小核酸产业。
01
跨越肝靶向:肝外递送驱动小核酸迈入慢病蓝海与价值跃迁
1.1 肝外递送:小核酸从罕见病向慢病大药的核心跃迁
1.2 市场扩容:突破肝脏局限,切入庞大患者基数的慢病蓝海
小核酸药物具有慢病管理的天然优势。小核酸通过调控基因表达实现长效静默机制,可做到一针维持半年甚至更长时间的给药,能大幅提高患者依从性,在慢病治疗领域具有颠覆传统治疗模式的巨大潜力。以降血脂药物英克西兰(PCSK9 siRNA)为例,英克西兰2025年销售收入12亿美金(同比+57%),26Q1销售收入4.52亿美元(同比+69%),保持高速增长趋势。
早期小核酸药物的商业化主要聚焦于致病机制明确、缺乏有效治疗手段的罕见病(SMA、DMD等)。肝外递送一旦打通,可切入肥胖、阿尔茨海默病、心肌疾病、哮喘等庞大患者基数的大病种市场,受众基数的指数级跃升。
1.3 竞争突围:绕开高度拥挤的肝靶向红海与GalNAc专利壁垒
肝脏靶点已聚集海量管线,Alnylam的GalNAc核心专利至2028年到期,后来者在肝靶向领域进行差异化竞争难度大,通过开发针对肝外的全新递送系统,企业可以绕开现有的GalNAc专利壁垒,建立属于自己的独占性IP。
• 靶点:肝脏相关靶点如HBV、PCSK9、AGT、LP(a)等已聚集了大量的在研管线。
• 专利:Ionis等海外先驱企业几乎垄断了底层的化学修饰专利(如PS骨架、LNA等);递送载体专利方面,LNP布局较早且极为严密,较难绕开;Alnylam掌控着GalNAc核心递送结构(如L96三叉戟结构)的专利,以通式结构为主的US10131907B2预估到期日2028-08-24。Arrowhead是避开Alnylam专利的突破者代表,后续公司借鉴了Alnylam和Arrowhead公司的思路进行修改。
1.4 行业机遇:全球同一起跑线,新玩家弯道超车窗口开启
肝外组织递送存在严苛的组织屏障与3E(Entry, Escape, Efficacy)生物学挑战,技术壁垒极高。但也因此,全球企业均处于早期探索阶段,尚无绝对垄断者,给了新进入者通过差异化突破实现弯道超车的机会。
• Entry(靶向积累与细胞摄取难度大):肝外组织存在严密的生理屏障。例如,CNS受血脑屏障的严格保护,限制了绝大多数系统给药分子的进入;肺部具有厚实的黏液层和纤毛清除机制;肌肉组织缺乏类似肝脏那样的组织通透性。
• Escape(内体逃逸效率不足1%):这是目前最大的生物学瓶颈。小核酸药物进入细胞后通常被困在内体中并面临被溶酶体降解的命运,由于肝外组织的药物蓄积量远低于肝脏,低下的逃逸率难以产生足够的药效。
• Efficacy(体内药效与安全性的平衡):小核酸在体循环中不仅面临核酸酶的降解和肾脏的快速清除,还容易引起非特异性的免疫反应或脱靶毒性。要实现长效的基因沉默,需要在复杂的化学修饰和递送载体之间找到完美的平衡。
1.5 估值跃升:肝外平台享技术复利与高估值溢价
小核酸具备极强的管线横向延展与市场扩容能力: 递送平台一旦在某一组织打通,就能迅速实现“一靶多药”或“一组织多病”的管线扩展。例如,Avidity和Dyne等公司利用AOC肌肉递送系统,快速平行推进了针对杜氏肌营养不良(DMD)、1型强直性肌营养不良(DM1)和面肩肱型肌营养不良(FSHD)等多种肌肉罕见病的管线。
平台估值溢价:率先在肝外实现概念验证的企业将掌握定价权,获得巨额溢价,例如,专注AOC肌肉递送的Avidity被诺华以120亿美元高溢价收购。
02
寻找下一个GalNAc:三大主流肝外递送技术解析
2.1 三大肝外递送技术:抗体偶联、脂质偶联、多肽偶联
2.2 AOC:借助抗体靶向递送,领先产品步入上市申请阶段
寡核苷酸偶联抗体(AOC)将寡核苷酸(siRNA或ASO等)通过定点偶联或非定点偶联在特定靶向性抗体(及其各种形式)上,实现小核酸药物向肝外组织的靶向递送。AOC具有多项优势:
• 特异性高,实现对肌肉、中枢神经系统等肝脏外组织/细胞的有效递送。
• 灵活适配,可偶联不同类型的寡核酸,如siRNA适合沉默致病基因、PMO适合修复剪接异常。
• 半衰期长,促进治疗持续性,降低给药频率,提升依从性。
• 生产工艺稳定,易规模化。
AOC龙头企业:1)Avidity被诺华以120亿美元收购,首发产品AOC-1044预计26Q2递交上市申请;2)Dyne的首发产品DYNE-251于2026年5月向FDA递交上市申请。
2.3 POC:多肽偶联,处于早期临床探索阶段
POC(Peptide-Oligonucleotide Conjugate)由多肽与寡核苷酸偶联而成,处于早期数据积累中。POC设计结合多肽在细胞穿透、受体介导内吞及组织靶向方面的优势,以及寡核苷酸对致病基因的调控能力。与AOC相比,POC分子量更小,免疫原性通常更低,细胞穿透能力强,有望实现更高的摄取效率和内体逃逸能力。POC目前包括:
• 穿膜肽(Cell-Penetrating Peptide,CPP)由5到40个氨基酸残基组成,通常为阳离子或两亲性的多肽片段。其核心优势是能够穿过细胞膜,高效促进细胞内各种有效载荷的运输,进而有效地将小核酸递送至目标部位。其分为线性CPP(如PepGen的EDO平台)和环状CPP(如Entrada的EEV平台)。
• 细胞靶向性肽(CTP),这类多肽能够特异性与某些组织上高度表达的受体(如转铁蛋白1、avβ6)结合。通过这些受体的高效内吞和溶酶体逃逸机制,CTP能够实现对特定器官的靶向递送。
2.4 C16:以脂为舟,将RNAi疗法扩展至肝外
2′-O-十六烷基(C16) 是Alnylam开发的脂质偶联递送技术,其核心设计是在siRNA分子连接一条脂肪酸链(2′-O-十六烷基),该修饰的疏水性显著增强了siRNA的亲脂性,从而改善其与细胞膜及相关膜蛋白的相互作用,提升了药物进入细胞的能力以及内体逃逸效率,实现针对CNS、眼睛及肺部靶标安全、有效且持久的沉默。
2022年Alnylam科学家发表文章详细说明C16-siRNA偶联技术( siRNA分子核糖的 2′位连接十六烷基脂质)。
03
赛道深耕:核心肝外组织的临床布局和竞争图谱
3.1 肝外组织靶向策略因“障”制宜,差异化递送格局初显
3.2 肌肉领域:AOC临床路径清晰,POC早期数据惊艳
小核酸药物在肌肉领域的探索,聚焦遗传性肌肉罕见病,主要为“抗体 vs 多肽”的技术路线博弈。
• AOC,Avidity采取TfR1抗体,AOC-1044预计26Q2递交BLA; Dyne采取TfR1 Fab片段, DYNE-251已递交BLA。
• POC,Sarepta/Arrowhead SRP-1001及SRP-1003采取avb6整合素肽偶联,临床1期SAD数据显示出优异的递送效率;穿透肽偶联尚需要更多临床数据验证。
3.3 Avidity(AOC-1044):DMD44疗效优异,预计26Q2递交上市申请
杜氏肌营养不良是(DMD)一种X染色体隐性遗传疾病,以神经肌肉退行性变为特征,在美国约有12,000名患者,在欧盟约有16,000名患者。该病通常由抗肌萎缩蛋白基因突变引起,该基因位于染色体Xp21.2上,包含79个外显子和78个内含子,目前暂无针对外显子44跳跃的杜氏肌营养不良症(DMD44)疗法获批上市。
AOC-1044是由TfR1单抗、Linker以及靶向Exon-44的PMO组成,旨在将PMO递送至骨骼肌,通过特异性跃过外显子44,促使患有DMD44病人产生部分功能性抗肌萎缩蛋白。
• 注册临床EXPLORE44数据积极:用药组显著提升Exon-44跳跃效率,抗肌萎缩蛋白表达显著增加了25%,抗肌萎缩蛋白水平恢复至正常水平的58%,同时肌酸激酶(CK)水平显著下降,且安全性良好。
• AOC-1044治疗DMD44适应症已获得FDA孤儿药、快速审评以及突破性疗法认定,预计26Q2向FDA递交上市申请。
3.4 Avidity(AOC-1001):DM1进度领先,26H2读出3期数据
强直性肌营养不良症1型人群(DM1)是一种罕见的多系统受累的遗传疾病,肌强直是该病的典型表现之一,其特征是手部张合困难。在DM1患者中,DMPK mRNA形成有毒的(CUG)n重复序列,从而导致剪接异常,目前暂无获批根治疗法。
• AOC-1001由TfR1单抗、Linker及DMPK siRNA组成。静脉注射给药后,AOC-1001与骨骼肌上表达的TfR1结合,药物被内吞进入内体,siRNA释放,DMPK沉默,进而恢复正常的剪接机制。AOC-1001在1/2期临床中显示出对DM1患者良好的安全性和耐受性,且患者肌肉中siRNA含量呈剂量依赖性增加,促使DMPK mRNA显著。有研究推测,AOC-1001可在细胞核内发挥作用。
• AOC-1001治疗DM1适应症已获得FDA孤儿药、快速审评以及突破性疗法认定,3期临床HARBOR研究已完成全部159名患者入组,预计2026H2数据读出,2027年递交上市申请,有望成为首个获批治疗DM1的药物。
3.5 Dyne(DYNE-251):Fab偶联PMO,DMD51适应症申报上市
Dyne的FORCE平台由三部分组成:靶向TfR1的抗原结合片段(Fab)、连接子、有效载荷(寡核酸等),核心产品为:
➢ DYNE-101正在进行注册临床ACHIEVE研究,适应症为1型强直性肌营养不良症(DM1)。
➢ DYNE-251正在进行注册临床DELIVER研究,适应症为DMD51,已获得FDA孤儿药、快速审评以及突破性疗法认定。
• 2025年12月,DELIVER读出顶线数据:注册扩展队列(REC)达到主要终点,在六个月时,肌肉含量校正后的肌营养不良蛋白表达量较基线有显著变化(p<0.0001),其肌营养不良蛋白平均绝对表达量达到正常值的5.46%(已根据肌肉含量进行校正)。常见TEAE为发热和头痛。
• 公司于2026年5月向FDA递交上市申请。此次申报上市是通过加速批准路径以及优先审评,如果获批,审评周期将缩短到6个月,有望于2027Q1获批上市。
3.6 Sarepta(SRP-1001&1003):来自Arrowhead的αvβ6靶向肽技术
2024年11月,Sarepta以首付款8.25亿美金,总金额114亿美金的对价从Arrowhead获得多款产品,包括治疗FSHD的SRP-1001(ARO-DU4),以及治疗DM1的SRP-1003(ARO-DM1)。
这两款产品基于Arrowhead的肌肉靶向技术开发,基本结构为:①靶向αvb6整合素受体的多肽配体(连接于正义链的5′端);②连接子,提高RISC装载效率;③包含一个脂质部分PK/PD调节剂(连接于正义链的3′端),该可促进递送和细胞摄取;④特异性序列。
3.7 Sarepta(SRP-1001&1003): 1期SAD数据展现优异递送效率
2026年3月,SRP-1001(DU4)和SRP-1003(DM1)1期SAD临床数据积极,显示出优于AOC的递送效率,26H2读出MAD数据。
• SRP-1001单次给药后,肌肉中siRNA浓度是TfR1靶向产品多次给药的6倍以上:SRP-1001给药42天后肌肉siRNA浓度达28.4 nM,且无剂量限制性毒性(支持继续剂量爬坡),而TfR1-siRNA肌肉中siRNA浓度中位数仅4.5 nM。
• SRP-1003单药最低剂量给药后,肌肉siRNA浓度达12.3 nM,优于Avidity的AOC-1001(~1.43 nM),以及Dyne的DYNE-101(~1.86 nM)。
3.8 CNS赛道:聚焦阿尔兹海默症热门靶点
CNS核心适应症为阿尔茨海默症:1)Aβ靶点:Alnylam的ALN-APP已进入临床2期阶段;2)TAU靶点:①Ionis和Biogen合作开发的Diranersen II期 CELIA 研究虽未能达到评估剂量反应的主要终点,但是基于生物标志物和疗效数据的可靠性,计划推进注册临床;②Arrowhead的ARO-MAPT通过TfR1靶向偶联实现皮下给药,26Q3末Q4初早期数据读出。③ALN-5288由Alnylam开发,目前临床由再生元主导。
3.9 BBB是CNS核心瓶颈,物理绕过与生物跨越双路径并驱
中枢神经系统(CNS) 治疗药物研发的最大挑战在于如何克服血脑屏障(Blood-Brain Barrier,BBB)。
BBB严格限制物质的进出维持中枢神经系统的稳态,约98%小分子药物和几乎所有大分子药物不能进入大脑 。
目前CNS递送路径主要包括:1)侵入给药(鞘内、脑室、脑实质等),直接绕过BBB,诸如naked ASO,C16-siRNA;2)生物跨越(皮下/静脉系统给药),利用内源性受体(如TfR1)诱导胞吞跨越 BBB,诸如AOC、POC等。
3.10 IT给药:BIIB080 AD 2期结果积极,为TAU靶点研发注入信心
Diranersen(BIIB080)是Biogen于2019年从Ionis获得的一款靶向TAU蛋白的Naked ASO药物,采取鞘内给药方式。2023年公布1b期临床显示,脑脊液总tau(t-tau)和磷酸化tau(p-tau¹⁸¹)水平呈强效、剂量依赖性且持续下降趋势。
近期,Biogen公布了BIIB080用于治疗早期AD的II期CELIA研究重磅顶线数据。该项随机对照II期临床试验首次证实,TAU靶向治疗药物可在早期阿尔茨海默病患者体内实现强效生物标志物改善,同时带来明确认知层面临床获益。虽然研究未达到主要终点,即第76周时,临床痴呆评定量表总分(CDR-SB)较基线的变化,未能显示出明确的剂量反应关系。但基于生物标志物和疗效数据的强劲表现,Biogen计划推进至注册开发阶段,具体临床数据将于7月12-15日的AAIC大会公布。
3.11 IT给药:C16-siRNA利用亲脂性促进脑组织渗透
鞘内注射(IT)虽在物理上成功绕过了血脑屏障,将药物直接注入脑脊液(CSF)中,但并未解决药物在致密组织中的扩散难题。裸露的siRNA分子注入CSF后会面临两个瓶颈:①快速清除:亲水性分子在脑脊液中极易随CSF的循环被快速清除,半衰期极短。②渗透与分布不均:由于存在时间极短且缺乏穿透细胞外基质的驱动力,药物仅能在CSF暴露量高的浅层脑区(如皮层、脊髓)实现局部蓄积,而在深层脑实质(如脑干、海马体)中的渗透与摄取水平较低。
C16(2’-O-十六烷基)是连接于siRNA分子核糖2’位的一条亲脂性烷基链,由于采用高代谢稳定性的醚键连接,C16有望实现siRNA在整个CNS(包括深层脑区)内的广泛、均匀分布,达成长达数月的安全、强效靶基因静默。
• 延长CSF半衰期 :C16等亲脂性基团显著增加siRNA的疏水性,使得药物更容易与脑脊液中的内源性脂质或蛋白质结合,从而降低其在CSF中的自然清除率,延长滞留时间,促进分子缓慢向深层脑实质扩散渗透。
• 增强细胞膜亲和力:神经元与胶质细胞的细胞膜由脂质双分子层构成,强亲脂性的C16链能够改善siRNA与细胞膜及相关膜蛋白的相互作用。这种亲和力的提升促进了细胞的主动摄取,同时亲脂链的物理特性亦有助于破坏内体囊泡,提升内体逃逸效率。
3.12 Alnylam领跑C16-siRNA,5款CNS产品进入临床阶段
基于C16偶联技术,Alnylam已有5款CNS治疗的产品进入临床阶段。其中Alnylam重点推进Mivelsiran (ALN-APP)和ALN-HTT02(HTT),再生元主导ALN-SOD、ALN-5288、ALN-SNCA这三款产品。
3.13 ALN-APP:早期临床安全性良好,未发生ARIA-E(水肿/渗出)事件
治疗AD的上市Aβ单抗药物具有高比例的ARIA-E(Edema/Effusion,水肿/渗出)和ARIA-H(Hemorrhage,出血)。
ALN-APP靶向淀粉样前体蛋白(APP)的mRNA,在通过RNAi机制减少中枢神经系统内APP蛋白及其所有下游Aβ片段的产生,以治疗早发AD和脑淀粉样血管病(CAA)。ALN-APP在一项1期研究中展现出良好的疗效及安全性。
‣ 单次注射 Mivelsiran(ALN-APP)后,可强效、持久地降低脑脊液中的sAPPβ水平。
‣ 接受治疗的89例患者,没有ARIA-E事件,以及低比例的ARIA-H。
‣ 7例ARIA-H患者中,3例本身存在ARIA-H,4例为接受药物治疗后发生ARIA-H(2例为APOE4 纯合子携带者)。
3.14 突破BBB新机制:TfR1受体介导的胞吞系统成为研发焦点
维持大脑功能所需的外周物质依赖主动转运机制进入脑内,其中一种生理性通路即受体介导的胞吞转运(RMT)。在这一过程中,配体与构成血脑屏障的内皮细胞管腔膜上表达的特异性受体结合,促使受体 - 配体复合物内化进入胞内囊泡,随后经多个胞内区室转运,最终与基侧膜融合,将所载物质释放至脑实质中。靶向此类 RMT受体是一种新兴策略,可以看作是生物“特洛伊木马”,用于将药物穿过BBB并进入大脑。目前多数研究聚焦于TfR1。
• 转铁蛋白受体1(TfR1)是一种Ⅱ型跨膜糖蛋白,主要与细胞铁的摄取有关,由两个90kd的亚单位通过二硫键连接而成,每个亚单位可以结合一个转铁蛋白(Tf)分子。TfR1与Tf结合后,通过网格蛋白介导的内吞作用进入细胞。在酸性内体中,铁被释放,脱铁Tf -TfR1复合物循环至细胞表面,进行下一轮的转运。
• TfR1在脑微血管内皮细胞和神经元细胞表面均高度表达。研究证明,结合TfR1的抗体能够以与Tf类似的转运过程被受体内吞转运,而且抗体可以从TfR1解离而释放到脑实质。
科学家开发了多种靶向 TfR1的药物递送策略,包括双特异性抗体、抗体片段、多肽偶联和纳米颗粒载体。
3.15 Arrowhead TRiM BBB:TfR1靶向配体偶联实现CNS皮下给药
Arrowhead 基于专有TRiM(Targeted RNAi Molecule)平台,开发了针对CNS的TRiM BBB递送系统,能够将siRNA通过稳定不可逆共价键与TfR1靶向配体偶联,依托TfR1的天然转运通路,为CNS提供非侵入性、高效且持久的 RNAi治疗方案,可实现皮下给药方式。
3.16 Arrowhead(ARO-MAPT): AD项目迎来首次临床数据释放
基于TRiM BBB 递送系统,Arrowhead有两款产品进入临床开发阶段。
• ARO-MAPT(TAU)是皮下给药的TfR1-siRNA。临床前研究显示,皮下给药后,ARO-MAPT可在多个脑区敲低MAPT mRNA,部分皮层区域MAPT mRNA敲低幅度最高可达85%,且敲低效果可维持3个月及以上。目前,ARO-MAPT进行早期AD适应症的Ph1/2临床(安慰剂对照),预计26Q3末Q4初读出早期Ph1/2临床数据。
• ARO-ATXN2(SCA2)进行脊髓小脑性共济失调2的Ph1临床,该产品和Sarepta达成合作。
3.17 脂肪/肺部/心脏:高潜力组织,26H2两款肥胖产品披露早期数据
脂肪、肺部、心脏三大高潜力靶组织,技术路径呈现多元化,目前多数管线处于早期临床阶段,其中两款脂肪靶向产品ALN-2232和ARO-ALK7将于26H2读出早期临床数据。
• Alnylam采用AdipoLigand1脂肪靶向技术开发的ALN-2232(肥胖,Ph1);
• Arrowhead依托TRiM平台布局ARO-ALK7(肥胖,Ph1);
• ARO-RAGE(炎症性肺病/哮喘,Ph2)及ARO-MMP7(特发性肺纤维化,Ph1);
• Ionis/阿斯利康基于TfR1受体靶向技术开发ION826(扩张型心肌病,Ph1)。
3.18 Alnylam(ALN-2232 ):协同GLP-1的增肌减脂潜力
针对脂肪组织递送,Alnylam开发了偶联物AdipoLigand 1,可实现皮下注射给药。首款产品ALN-2232(ACVR1C)已进入临床1期,预计2026H2披露1期临床数据。
肝脏合成并分泌Activin E(由INHBE基因编码),Activin E作为配体,与脂肪细胞膜上的ACVR1C(ALK7,激活素受体样激酶7)受体结合,激活下游SMAD2/3信号通路,进而促进脂肪堆积。
小鼠模型显示,无论INHBE siRNA(肝脏内),还是ACVR1C siRNA(脂肪细胞),与低剂量司美格鲁肽联合使用时,均能够实现更高比例的脂肪减少和瘦体重的保留。
3.19 Arrowhead(ARO-ALK7):双脂质偶联,展现内脏减脂优势
ARO-ALK7是Arrowhead首款进入临床阶段的脂肪靶向产品,采取双脂质偶联技术,其中脂质连接到正义链的3′端和 5′端。
1期临床研究显示:1)ARO-ALK7对ALK7 mRNA沉默显示出剂量依赖性特征;
2)ARO-ALK7单次给药后,受试者内脏脂肪可快速呈剂量依赖性减少,经安慰剂校正后的最大降幅可达14.1%。
ARO-ALK7正在进行肥胖患者的I/IIa期临床试验,2026年有望持续数据读出。此外,Arrowhead另一款针对肥胖适应症的产品ARO-INHBE也在积极推进临床,ARO-INHBE是GalNAc-siRNA,沉默肝脏中靶基因同时,也影响脂肪组织中的脂肪储存。
3.20 肺部靶向探索:ARO-RAGE雾化吸入递送,哮喘 Ph2推进
Arrowhead利用TRiM平台将siRNA递送至肺上皮细胞:①领先产品ARO-RAGE正在进行过敏原诱发的轻度哮喘的2期临床(NCT07241546);②ARO-MMP7正在进行特发性肺纤维化的1期临床,并且与SAREPTA达成合作。
ARO-RAGE由siRNA(特异性靶向RAGE mRNA )和小分子(特异性靶向肺上皮细胞表达的整合素αvβ6)偶联。
• RAGE(receptor for advanced glycation end products)处于2型炎症通路的上游,参与哮喘和多种炎症性疾病的发病机制,在肺上皮细胞中大量表达。ARO-RAGE旨在沉默RAGE基因,减少肺上皮细胞上RAGE蛋白的表达量,通过影响RAGE依赖性炎症通路,改善哮喘患者的病情。
• 2024年5月,Arrowhead披露的Ph1/2临床数据显示,在健康志愿者和轻度至中度哮喘患者中,ARO-RAGE可实现深度且持续的基因敲低,疗效持续时间长,支持每两个月给药一次,并且具有良好的安全性和耐受性。
3.21 心脏靶向起步:ION826(PLN siRNA)攻坚遗传性扩张型心肌病
针对心脏,Ionis重点推进两款TfR1偶联产品,分别为ION826(PLN siRNA)和ION501。
• ION826/AZD4063由Ionis和阿斯利康共同开发,目前已经入临床1期研阶段,评估 AZD4063在单次和多次皮下给药后对患有磷蛋白(PLN)R14del 扩张型心肌病的受试者的安全性、耐受性和药代动力学。该研究计划入组31例患者,主要临床终点为安全性,2025年12月首例患者入组,预计2027年数据读出。
• ION501预计2026年进入临床阶段。
04
平台铸就价值,26H2进入密集的催化剂兑现期
4.1 定价权博弈:率先实现概念验证,获取高技术溢价与MA&BD机会
在肝脏靶向领域已成竞争红海(主要拼临床推进速度)的背景下,肝外靶向的策略在于率先实现概念验证,率先在CNS(中枢神经系统)、肌肉等肝外组织实现突破的企业,将享受到极高的技术溢价。
企业通常通过被跨国大型药企高溢价收购或进行BD合作来兑现商业价值。例如,诺华斥资120亿美元收购了肌肉靶向。AOC领域的领先企业Avidity;Arrowhead的CNS管线同样享受了显著的技术溢价。
4.2 重金买平台
4.3 2026催化剂(一):密切关注领先递送平台关键数据读出
Alnylam关注TTR业务线销售放量、CNS靶向ALN-HTT02及脂肪靶向ALN-2232的数据读出;Arrowhead在26H2迎来肝脏、脂肪、CNS以及双靶点技术验证,5项核心资产数据读出;Sarepta两款肌肉靶向产品26H2披露MAD数据。
4.4 2026催化剂(二):AOC上市申报在即,POC期待更多数据更新
AOC领域,AOC-1044预计26Q2递交BLA,DYNE-251已递交BLA;领先企业26H2持续更新临床数据。
POC领域,PepGen和Entrada罕见肌肉疾病有数据读出。
4.5 2026催化剂(三):多款重磅ASO药物步入上市监管审批窗口
Ionis三款产品有望获批上市,治疗严重高甘油三酯症Olezarsen(APOC3)、治疗亚历山大病Zilganersen,HBV药物Bepirovirsen。
Ionis与诺华合作的LP(a)-ASO药物Pelacarsen的Ph3临床HORIZON研究预计26H2数据读出。
05
风险提示
研发进展不及预期风险:小核酸药物研发技术复杂,存在临床试验结果不及预期、研发进度延迟或无法获得监管批准的风险。同时,小核酸药物在递送效率、长期安全性及疗效等方面仍存在不确定性,相关技术平台若未能持续优化,可能对产品研发及商业化产生不利影响。
行业竞争与技术迭代风险:小核酸药物领域竞争激烈,技术迭代迅速。若相关企业在技术路线选择、新型递送平台开发、靶点序列布局或专利保护策略上落后于行业领先者,可能对其长期竞争力产生不利影响。
END
免责声明:本文仅作知识交流与分享及科普目的,不涉及商业宣传,不作为相关医疗指导或用药建议。文章如有侵权请联系删除。
第三届小核酸药物2026盛会详情请点击图片了解,招商热线188 0194 0589
戳“阅读原文”立即抢占小核酸药物论坛免费参会名额!
核酸药物siRNA
2026-06-07
·远峰电子
文章转自2026年06月01日太平洋医药团队报告,分析师:周豫/戎晓婕
投资要点
产业周期:小核酸跨越可成药里程碑,肝外递送开启新一轮价值跃迁。GalNAc肝脏递送技术的成熟,以及英克西兰(PCSK9)等重磅慢病RNAi药物的全球销售放量,标志着小核酸迈入商业化扩张周期。随着肝脏靶向陷入同质化红海竞争,行业核心焦点从“小核酸能否成药”转化为“谁能率先攻克肝外高效递送”。
技术矩阵:三大肝外递送路线演进。当前临床阶段管线中,AOC(抗体-小核酸偶联)利用抗体或抗体片段结合TfR1等受体,通过受体介导内吞机制进入组织细胞,具有高特异性,肌肉罕见病领域的成药性已获注册临床验证;POC(多肽-小核酸偶联)依托细胞穿透肽或靶向肽提升细胞摄取与内体逃逸,具备结构轻量化、免疫原性低的优势,处于早期探索阶段;脂质-小核酸偶联通过C16/胆固醇等亲脂修饰,增强与血浆白蛋白/脂蛋白的相互作用,借助天然脂蛋白转运通路促进组织富集与细胞内化,处于概念验证阶段,在CNS、脂肪等领域展现应用潜力。
组织范式:靶向策略因“障”制宜,差异化递送格局初显。不同组织的独特生物学屏障催生了多元化递送范式,肌肉围绕TfR1及αvβ6介导的主动摄取,呈现AOC技术领先与POC效率提升的路线博弈;CNS围绕突破血脑屏障攻坚,形成鞘内注射侵入性给药与TfR1介导系统给药双轨并驱,拓展阿尔茨海默病等大适应症;肺部采取雾化吸入实现局部递送;脂肪与心脏等高潜力组织在慢病蓝海中探索早期技术边界。
26H2进入密集的催化剂兑现期。1)Alnylam和Arrowhead迎来脂肪、CNS、肌肉、肝脏双靶点项目等数据的集中释放;2)Ionis三款重磅ASO药物步入上市监管审批窗口;3)AOC上市申报在即,POC早期数据迎来关键更新。
未来,肝外递送的核心壁垒不仅在于效率提升,更在于能否搭建可复制、可横向扩展的组织特异性靶向递送体系。我们认为,递送平台承载技术复利,率先通关者有望主导下一代小核酸产业。
01
跨越肝靶向:肝外递送驱动小核酸迈入慢病蓝海与价值跃迁
1.1 肝外递送:小核酸从罕见病向慢病大药的核心跃迁
1.2 市场扩容:突破肝脏局限,切入庞大患者基数的慢病蓝海
小核酸药物具有慢病管理的天然优势。小核酸通过调控基因表达实现长效静默机制,可做到一针维持半年甚至更长时间的给药,能大幅提高患者依从性,在慢病治疗领域具有颠覆传统治疗模式的巨大潜力。以降血脂药物英克西兰(PCSK9 siRNA)为例,英克西兰2025年销售收入12亿美金(同比+57%),26Q1销售收入4.52亿美元(同比+69%),保持高速增长趋势。
早期小核酸药物的商业化主要聚焦于致病机制明确、缺乏有效治疗手段的罕见病(SMA、DMD等)。肝外递送一旦打通,可切入肥胖、阿尔茨海默病、心肌疾病、哮喘等庞大患者基数的大病种市场,受众基数的指数级跃升。
1.3 竞争突围:绕开高度拥挤的肝靶向红海与GalNAc专利壁垒
肝脏靶点已聚集海量管线,Alnylam的GalNAc核心专利至2028年到期,后来者在肝靶向领域进行差异化竞争难度大,通过开发针对肝外的全新递送系统,企业可以绕开现有的GalNAc专利壁垒,建立属于自己的独占性IP。
• 靶点:肝脏相关靶点如HBV、PCSK9、AGT、LP(a)等已聚集了大量的在研管线。
• 专利:Ionis等海外先驱企业几乎垄断了底层的化学修饰专利(如PS骨架、LNA等);递送载体专利方面,LNP布局较早且极为严密,较难绕开;Alnylam掌控着GalNAc核心递送结构(如L96三叉戟结构)的专利,以通式结构为主的US10131907B2预估到期日2028-08-24。Arrowhead是避开Alnylam专利的突破者代表,后续公司借鉴了Alnylam和Arrowhead公司的思路进行修改。
1.4 行业机遇:全球同一起跑线,新玩家弯道超车窗口开启
肝外组织递送存在严苛的组织屏障与3E(Entry, Escape, Efficacy)生物学挑战,技术壁垒极高。但也因此,全球企业均处于早期探索阶段,尚无绝对垄断者,给了新进入者通过差异化突破实现弯道超车的机会。
• Entry(靶向积累与细胞摄取难度大):肝外组织存在严密的生理屏障。例如,CNS受血脑屏障的严格保护,限制了绝大多数系统给药分子的进入;肺部具有厚实的黏液层和纤毛清除机制;肌肉组织缺乏类似肝脏那样的组织通透性。
• Escape(内体逃逸效率不足1%):这是目前最大的生物学瓶颈。小核酸药物进入细胞后通常被困在内体中并面临被溶酶体降解的命运,由于肝外组织的药物蓄积量远低于肝脏,低下的逃逸率难以产生足够的药效。
• Efficacy(体内药效与安全性的平衡):小核酸在体循环中不仅面临核酸酶的降解和肾脏的快速清除,还容易引起非特异性的免疫反应或脱靶毒性。要实现长效的基因沉默,需要在复杂的化学修饰和递送载体之间找到完美的平衡。
1.5 估值跃升:肝外平台享技术复利与高估值溢价
小核酸具备极强的管线横向延展与市场扩容能力: 递送平台一旦在某一组织打通,就能迅速实现“一靶多药”或“一组织多病”的管线扩展。例如,Avidity和Dyne等公司利用AOC肌肉递送系统,快速平行推进了针对杜氏肌营养不良(DMD)、1型强直性肌营养不良(DM1)和面肩肱型肌营养不良(FSHD)等多种肌肉罕见病的管线。
平台估值溢价:率先在肝外实现概念验证的企业将掌握定价权,获得巨额溢价,例如,专注AOC肌肉递送的Avidity被诺华以120亿美元高溢价收购。
02
寻找下一个GalNAc:三大主流肝外递送技术解析
2.1 三大肝外递送技术:抗体偶联、脂质偶联、多肽偶联
2.2 AOC:借助抗体靶向递送,领先产品步入上市申请阶段
寡核苷酸偶联抗体(AOC)将寡核苷酸(siRNA或ASO等)通过定点偶联或非定点偶联在特定靶向性抗体(及其各种形式)上,实现小核酸药物向肝外组织的靶向递送。AOC具有多项优势:
• 特异性高,实现对肌肉、中枢神经系统等肝脏外组织/细胞的有效递送。
• 灵活适配,可偶联不同类型的寡核酸,如siRNA适合沉默致病基因、PMO适合修复剪接异常。
• 半衰期长,促进治疗持续性,降低给药频率,提升依从性。
• 生产工艺稳定,易规模化。
AOC龙头企业:1)Avidity被诺华以120亿美元收购,首发产品AOC-1044预计26Q2递交上市申请;2)Dyne的首发产品DYNE-251于2026年5月向FDA递交上市申请。
2.3 POC:多肽偶联,处于早期临床探索阶段
POC(Peptide-Oligonucleotide Conjugate)由多肽与寡核苷酸偶联而成,处于早期数据积累中。POC设计结合多肽在细胞穿透、受体介导内吞及组织靶向方面的优势,以及寡核苷酸对致病基因的调控能力。与AOC相比,POC分子量更小,免疫原性通常更低,细胞穿透能力强,有望实现更高的摄取效率和内体逃逸能力。POC目前包括:
• 穿膜肽(Cell-Penetrating Peptide,CPP)由5到40个氨基酸残基组成,通常为阳离子或两亲性的多肽片段。其核心优势是能够穿过细胞膜,高效促进细胞内各种有效载荷的运输,进而有效地将小核酸递送至目标部位。其分为线性CPP(如PepGen的EDO平台)和环状CPP(如Entrada的EEV平台)。
• 细胞靶向性肽(CTP),这类多肽能够特异性与某些组织上高度表达的受体(如转铁蛋白1、avβ6)结合。通过这些受体的高效内吞和溶酶体逃逸机制,CTP能够实现对特定器官的靶向递送。
2.4 C16:以脂为舟,将RNAi疗法扩展至肝外
2′-O-十六烷基(C16) 是Alnylam开发的脂质偶联递送技术,其核心设计是在siRNA分子连接一条脂肪酸链(2′-O-十六烷基),该修饰的疏水性显著增强了siRNA的亲脂性,从而改善其与细胞膜及相关膜蛋白的相互作用,提升了药物进入细胞的能力以及内体逃逸效率,实现针对CNS、眼睛及肺部靶标安全、有效且持久的沉默。
2022年Alnylam科学家发表文章详细说明C16-siRNA偶联技术( siRNA分子核糖的 2′位连接十六烷基脂质)。
03
赛道深耕:核心肝外组织的临床布局和竞争图谱
3.1 肝外组织靶向策略因“障”制宜,差异化递送格局初显
3.2 肌肉领域:AOC临床路径清晰,POC早期数据惊艳
小核酸药物在肌肉领域的探索,聚焦遗传性肌肉罕见病,主要为“抗体 vs 多肽”的技术路线博弈。
• AOC,Avidity采取TfR1抗体,AOC-1044预计26Q2递交BLA; Dyne采取TfR1 Fab片段, DYNE-251已递交BLA。
• POC,Sarepta/Arrowhead SRP-1001及SRP-1003采取avb6整合素肽偶联,临床1期SAD数据显示出优异的递送效率;穿透肽偶联尚需要更多临床数据验证。
3.3 Avidity(AOC-1044):DMD44疗效优异,预计26Q2递交上市申请
杜氏肌营养不良是(DMD)一种X染色体隐性遗传疾病,以神经肌肉退行性变为特征,在美国约有12,000名患者,在欧盟约有16,000名患者。该病通常由抗肌萎缩蛋白基因突变引起,该基因位于染色体Xp21.2上,包含79个外显子和78个内含子,目前暂无针对外显子44跳跃的杜氏肌营养不良症(DMD44)疗法获批上市。
AOC-1044是由TfR1单抗、Linker以及靶向Exon-44的PMO组成,旨在将PMO递送至骨骼肌,通过特异性跃过外显子44,促使患有DMD44病人产生部分功能性抗肌萎缩蛋白。
• 注册临床EXPLORE44数据积极:用药组显著提升Exon-44跳跃效率,抗肌萎缩蛋白表达显著增加了25%,抗肌萎缩蛋白水平恢复至正常水平的58%,同时肌酸激酶(CK)水平显著下降,且安全性良好。
• AOC-1044治疗DMD44适应症已获得FDA孤儿药、快速审评以及突破性疗法认定,预计26Q2向FDA递交上市申请。
3.4 Avidity(AOC-1001):DM1进度领先,26H2读出3期数据
强直性肌营养不良症1型人群(DM1)是一种罕见的多系统受累的遗传疾病,肌强直是该病的典型表现之一,其特征是手部张合困难。在DM1患者中,DMPK mRNA形成有毒的(CUG)n重复序列,从而导致剪接异常,目前暂无获批根治疗法。
• AOC-1001由TfR1单抗、Linker及DMPK siRNA组成。静脉注射给药后,AOC-1001与骨骼肌上表达的TfR1结合,药物被内吞进入内体,siRNA释放,DMPK沉默,进而恢复正常的剪接机制。AOC-1001在1/2期临床中显示出对DM1患者良好的安全性和耐受性,且患者肌肉中siRNA含量呈剂量依赖性增加,促使DMPK mRNA显著。有研究推测,AOC-1001可在细胞核内发挥作用。
• AOC-1001治疗DM1适应症已获得FDA孤儿药、快速审评以及突破性疗法认定,3期临床HARBOR研究已完成全部159名患者入组,预计2026H2数据读出,2027年递交上市申请,有望成为首个获批治疗DM1的药物。
3.5 Dyne(DYNE-251):Fab偶联PMO,DMD51适应症申报上市
Dyne的FORCE平台由三部分组成:靶向TfR1的抗原结合片段(Fab)、连接子、有效载荷(寡核酸等),核心产品为:
➢ DYNE-101正在进行注册临床ACHIEVE研究,适应症为1型强直性肌营养不良症(DM1)。
➢ DYNE-251正在进行注册临床DELIVER研究,适应症为DMD51,已获得FDA孤儿药、快速审评以及突破性疗法认定。
• 2025年12月,DELIVER读出顶线数据:注册扩展队列(REC)达到主要终点,在六个月时,肌肉含量校正后的肌营养不良蛋白表达量较基线有显著变化(p<0.0001),其肌营养不良蛋白平均绝对表达量达到正常值的5.46%(已根据肌肉含量进行校正)。常见TEAE为发热和头痛。
• 公司于2026年5月向FDA递交上市申请。此次申报上市是通过加速批准路径以及优先审评,如果获批,审评周期将缩短到6个月,有望于2027Q1获批上市。
3.6 Sarepta(SRP-1001&1003):来自Arrowhead的αvβ6靶向肽技术
2024年11月,Sarepta以首付款8.25亿美金,总金额114亿美金的对价从Arrowhead获得多款产品,包括治疗FSHD的SRP-1001(ARO-DU4),以及治疗DM1的SRP-1003(ARO-DM1)。
这两款产品基于Arrowhead的肌肉靶向技术开发,基本结构为:①靶向αvb6整合素受体的多肽配体(连接于正义链的5′端);②连接子,提高RISC装载效率;③包含一个脂质部分PK/PD调节剂(连接于正义链的3′端),该可促进递送和细胞摄取;④特异性序列。
3.7 Sarepta(SRP-1001&1003): 1期SAD数据展现优异递送效率
2026年3月,SRP-1001(DU4)和SRP-1003(DM1)1期SAD临床数据积极,显示出优于AOC的递送效率,26H2读出MAD数据。
• SRP-1001单次给药后,肌肉中siRNA浓度是TfR1靶向产品多次给药的6倍以上:SRP-1001给药42天后肌肉siRNA浓度达28.4 nM,且无剂量限制性毒性(支持继续剂量爬坡),而TfR1-siRNA肌肉中siRNA浓度中位数仅4.5 nM。
• SRP-1003单药最低剂量给药后,肌肉siRNA浓度达12.3 nM,优于Avidity的AOC-1001(~1.43 nM),以及Dyne的DYNE-101(~1.86 nM)。
3.8 CNS赛道:聚焦阿尔兹海默症热门靶点
CNS核心适应症为阿尔茨海默症:1)Aβ靶点:Alnylam的ALN-APP已进入临床2期阶段;2)TAU靶点:①Ionis和Biogen合作开发的Diranersen II期 CELIA 研究虽未能达到评估剂量反应的主要终点,但是基于生物标志物和疗效数据的可靠性,计划推进注册临床;②Arrowhead的ARO-MAPT通过TfR1靶向偶联实现皮下给药,26Q3末Q4初早期数据读出。③ALN-5288由Alnylam开发,目前临床由再生元主导。
3.9 BBB是CNS核心瓶颈,物理绕过与生物跨越双路径并驱
中枢神经系统(CNS) 治疗药物研发的最大挑战在于如何克服血脑屏障(Blood-Brain Barrier,BBB)。
BBB严格限制物质的进出维持中枢神经系统的稳态,约98%小分子药物和几乎所有大分子药物不能进入大脑 。
目前CNS递送路径主要包括:1)侵入给药(鞘内、脑室、脑实质等),直接绕过BBB,诸如naked ASO,C16-siRNA;2)生物跨越(皮下/静脉系统给药),利用内源性受体(如TfR1)诱导胞吞跨越 BBB,诸如AOC、POC等。
3.10 IT给药:BIIB080 AD 2期结果积极,为TAU靶点研发注入信心
Diranersen(BIIB080)是Biogen于2019年从Ionis获得的一款靶向TAU蛋白的Naked ASO药物,采取鞘内给药方式。2023年公布1b期临床显示,脑脊液总tau(t-tau)和磷酸化tau(p-tau¹⁸¹)水平呈强效、剂量依赖性且持续下降趋势。
近期,Biogen公布了BIIB080用于治疗早期AD的II期CELIA研究重磅顶线数据。该项随机对照II期临床试验首次证实,TAU靶向治疗药物可在早期阿尔茨海默病患者体内实现强效生物标志物改善,同时带来明确认知层面临床获益。虽然研究未达到主要终点,即第76周时,临床痴呆评定量表总分(CDR-SB)较基线的变化,未能显示出明确的剂量反应关系。但基于生物标志物和疗效数据的强劲表现,Biogen计划推进至注册开发阶段,具体临床数据将于7月12-15日的AAIC大会公布。
3.11 IT给药:C16-siRNA利用亲脂性促进脑组织渗透
鞘内注射(IT)虽在物理上成功绕过了血脑屏障,将药物直接注入脑脊液(CSF)中,但并未解决药物在致密组织中的扩散难题。裸露的siRNA分子注入CSF后会面临两个瓶颈:①快速清除:亲水性分子在脑脊液中极易随CSF的循环被快速清除,半衰期极短。②渗透与分布不均:由于存在时间极短且缺乏穿透细胞外基质的驱动力,药物仅能在CSF暴露量高的浅层脑区(如皮层、脊髓)实现局部蓄积,而在深层脑实质(如脑干、海马体)中的渗透与摄取水平较低。
C16(2’-O-十六烷基)是连接于siRNA分子核糖2’位的一条亲脂性烷基链,由于采用高代谢稳定性的醚键连接,C16有望实现siRNA在整个CNS(包括深层脑区)内的广泛、均匀分布,达成长达数月的安全、强效靶基因静默。
• 延长CSF半衰期 :C16等亲脂性基团显著增加siRNA的疏水性,使得药物更容易与脑脊液中的内源性脂质或蛋白质结合,从而降低其在CSF中的自然清除率,延长滞留时间,促进分子缓慢向深层脑实质扩散渗透。
• 增强细胞膜亲和力:神经元与胶质细胞的细胞膜由脂质双分子层构成,强亲脂性的C16链能够改善siRNA与细胞膜及相关膜蛋白的相互作用。这种亲和力的提升促进了细胞的主动摄取,同时亲脂链的物理特性亦有助于破坏内体囊泡,提升内体逃逸效率。
3.12 Alnylam领跑C16-siRNA,5款CNS产品进入临床阶段
基于C16偶联技术,Alnylam已有5款CNS治疗的产品进入临床阶段。其中Alnylam重点推进Mivelsiran (ALN-APP)和ALN-HTT02(HTT),再生元主导ALN-SOD、ALN-5288、ALN-SNCA这三款产品。
3.13 ALN-APP:早期临床安全性良好,未发生ARIA-E(水肿/渗出)事件
治疗AD的上市Aβ单抗药物具有高比例的ARIA-E(Edema/Effusion,水肿/渗出)和ARIA-H(Hemorrhage,出血)。
ALN-APP靶向淀粉样前体蛋白(APP)的mRNA,在通过RNAi机制减少中枢神经系统内APP蛋白及其所有下游Aβ片段的产生,以治疗早发AD和脑淀粉样血管病(CAA)。ALN-APP在一项1期研究中展现出良好的疗效及安全性。
‣ 单次注射 Mivelsiran(ALN-APP)后,可强效、持久地降低脑脊液中的sAPPβ水平。
‣ 接受治疗的89例患者,没有ARIA-E事件,以及低比例的ARIA-H。
‣ 7例ARIA-H患者中,3例本身存在ARIA-H,4例为接受药物治疗后发生ARIA-H(2例为APOE4 纯合子携带者)。
3.14 突破BBB新机制:TfR1受体介导的胞吞系统成为研发焦点
维持大脑功能所需的外周物质依赖主动转运机制进入脑内,其中一种生理性通路即受体介导的胞吞转运(RMT)。在这一过程中,配体与构成血脑屏障的内皮细胞管腔膜上表达的特异性受体结合,促使受体 - 配体复合物内化进入胞内囊泡,随后经多个胞内区室转运,最终与基侧膜融合,将所载物质释放至脑实质中。靶向此类 RMT受体是一种新兴策略,可以看作是生物“特洛伊木马”,用于将药物穿过BBB并进入大脑。目前多数研究聚焦于TfR1。
• 转铁蛋白受体1(TfR1)是一种Ⅱ型跨膜糖蛋白,主要与细胞铁的摄取有关,由两个90kd的亚单位通过二硫键连接而成,每个亚单位可以结合一个转铁蛋白(Tf)分子。TfR1与Tf结合后,通过网格蛋白介导的内吞作用进入细胞。在酸性内体中,铁被释放,脱铁Tf -TfR1复合物循环至细胞表面,进行下一轮的转运。
• TfR1在脑微血管内皮细胞和神经元细胞表面均高度表达。研究证明,结合TfR1的抗体能够以与Tf类似的转运过程被受体内吞转运,而且抗体可以从TfR1解离而释放到脑实质。
科学家开发了多种靶向 TfR1的药物递送策略,包括双特异性抗体、抗体片段、多肽偶联和纳米颗粒载体。
3.15 Arrowhead TRiM BBB:TfR1靶向配体偶联实现CNS皮下给药
Arrowhead 基于专有TRiM(Targeted RNAi Molecule)平台,开发了针对CNS的TRiM BBB递送系统,能够将siRNA通过稳定不可逆共价键与TfR1靶向配体偶联,依托TfR1的天然转运通路,为CNS提供非侵入性、高效且持久的 RNAi治疗方案,可实现皮下给药方式。
3.16 Arrowhead(ARO-MAPT): AD项目迎来首次临床数据释放
基于TRiM BBB 递送系统,Arrowhead有两款产品进入临床开发阶段。
• ARO-MAPT(TAU)是皮下给药的TfR1-siRNA。临床前研究显示,皮下给药后,ARO-MAPT可在多个脑区敲低MAPT mRNA,部分皮层区域MAPT mRNA敲低幅度最高可达85%,且敲低效果可维持3个月及以上。目前,ARO-MAPT进行早期AD适应症的Ph1/2临床(安慰剂对照),预计26Q3末Q4初读出早期Ph1/2临床数据。
• ARO-ATXN2(SCA2)进行脊髓小脑性共济失调2的Ph1临床,该产品和Sarepta达成合作。
3.17 脂肪/肺部/心脏:高潜力组织,26H2两款肥胖产品披露早期数据
脂肪、肺部、心脏三大高潜力靶组织,技术路径呈现多元化,目前多数管线处于早期临床阶段,其中两款脂肪靶向产品ALN-2232和ARO-ALK7将于26H2读出早期临床数据。
• Alnylam采用AdipoLigand1脂肪靶向技术开发的ALN-2232(肥胖,Ph1);
• Arrowhead依托TRiM平台布局ARO-ALK7(肥胖,Ph1);
• ARO-RAGE(炎症性肺病/哮喘,Ph2)及ARO-MMP7(特发性肺纤维化,Ph1);
• Ionis/阿斯利康基于TfR1受体靶向技术开发ION826(扩张型心肌病,Ph1)。
3.18 Alnylam(ALN-2232 ):协同GLP-1的增肌减脂潜力
针对脂肪组织递送,Alnylam开发了偶联物AdipoLigand 1,可实现皮下注射给药。首款产品ALN-2232(ACVR1C)已进入临床1期,预计2026H2披露1期临床数据。
肝脏合成并分泌Activin E(由INHBE基因编码),Activin E作为配体,与脂肪细胞膜上的ACVR1C(ALK7,激活素受体样激酶7)受体结合,激活下游SMAD2/3信号通路,进而促进脂肪堆积。
小鼠模型显示,无论INHBE siRNA(肝脏内),还是ACVR1C siRNA(脂肪细胞),与低剂量司美格鲁肽联合使用时,均能够实现更高比例的脂肪减少和瘦体重的保留。
3.19 Arrowhead(ARO-ALK7):双脂质偶联,展现内脏减脂优势
ARO-ALK7是Arrowhead首款进入临床阶段的脂肪靶向产品,采取双脂质偶联技术,其中脂质连接到正义链的3′端和 5′端。
1期临床研究显示:1)ARO-ALK7对ALK7 mRNA沉默显示出剂量依赖性特征;
2)ARO-ALK7单次给药后,受试者内脏脂肪可快速呈剂量依赖性减少,经安慰剂校正后的最大降幅可达14.1%。
ARO-ALK7正在进行肥胖患者的I/IIa期临床试验,2026年有望持续数据读出。此外,Arrowhead另一款针对肥胖适应症的产品ARO-INHBE也在积极推进临床,ARO-INHBE是GalNAc-siRNA,沉默肝脏中靶基因同时,也影响脂肪组织中的脂肪储存。
3.20 肺部靶向探索:ARO-RAGE雾化吸入递送,哮喘 Ph2推进
Arrowhead利用TRiM平台将siRNA递送至肺上皮细胞:①领先产品ARO-RAGE正在进行过敏原诱发的轻度哮喘的2期临床(NCT07241546);②ARO-MMP7正在进行特发性肺纤维化的1期临床,并且与SAREPTA达成合作。
ARO-RAGE由siRNA(特异性靶向RAGE mRNA )和小分子(特异性靶向肺上皮细胞表达的整合素αvβ6)偶联。
• RAGE(receptor for advanced glycation end products)处于2型炎症通路的上游,参与哮喘和多种炎症性疾病的发病机制,在肺上皮细胞中大量表达。ARO-RAGE旨在沉默RAGE基因,减少肺上皮细胞上RAGE蛋白的表达量,通过影响RAGE依赖性炎症通路,改善哮喘患者的病情。
• 2024年5月,Arrowhead披露的Ph1/2临床数据显示,在健康志愿者和轻度至中度哮喘患者中,ARO-RAGE可实现深度且持续的基因敲低,疗效持续时间长,支持每两个月给药一次,并且具有良好的安全性和耐受性。
3.21 心脏靶向起步:ION826(PLN siRNA)攻坚遗传性扩张型心肌病
针对心脏,Ionis重点推进两款TfR1偶联产品,分别为ION826(PLN siRNA)和ION501。
• ION826/AZD4063由Ionis和阿斯利康共同开发,目前已经入临床1期研阶段,评估 AZD4063在单次和多次皮下给药后对患有磷蛋白(PLN)R14del 扩张型心肌病的受试者的安全性、耐受性和药代动力学。该研究计划入组31例患者,主要临床终点为安全性,2025年12月首例患者入组,预计2027年数据读出。
• ION501预计2026年进入临床阶段。
04
平台铸就价值,26H2进入密集的催化剂兑现期
4.1 定价权博弈:率先实现概念验证,获取高技术溢价与MA&BD机会
在肝脏靶向领域已成竞争红海(主要拼临床推进速度)的背景下,肝外靶向的策略在于率先实现概念验证,率先在CNS(中枢神经系统)、肌肉等肝外组织实现突破的企业,将享受到极高的技术溢价。
企业通常通过被跨国大型药企高溢价收购或进行BD合作来兑现商业价值。例如,诺华斥资120亿美元收购了肌肉靶向。AOC领域的领先企业Avidity;Arrowhead的CNS管线同样享受了显著的技术溢价。
4.2 重金买平台
4.3 2026催化剂(一):密切关注领先递送平台关键数据读出
Alnylam关注TTR业务线销售放量、CNS靶向ALN-HTT02及脂肪靶向ALN-2232的数据读出;Arrowhead在26H2迎来肝脏、脂肪、CNS以及双靶点技术验证,5项核心资产数据读出;Sarepta两款肌肉靶向产品26H2披露MAD数据。
4.4 2026催化剂(二):AOC上市申报在即,POC期待更多数据更新
AOC领域,AOC-1044预计26Q2递交BLA,DYNE-251已递交BLA;领先企业26H2持续更新临床数据。
POC领域,PepGen和Entrada罕见肌肉疾病有数据读出。
4.5 2026催化剂(三):多款重磅ASO药物步入上市监管审批窗口
Ionis三款产品有望获批上市,治疗严重高甘油三酯症Olezarsen(APOC3)、治疗亚历山大病Zilganersen,HBV药物Bepirovirsen。
Ionis与诺华合作的LP(a)-ASO药物Pelacarsen的Ph3临床HORIZON研究预计26H2数据读出。
05
风险提示
研发进展不及预期风险:小核酸药物研发技术复杂,存在临床试验结果不及预期、研发进度延迟或无法获得监管批准的风险。同时,小核酸药物在递送效率、长期安全性及疗效等方面仍存在不确定性,相关技术平台若未能持续优化,可能对产品研发及商业化产生不利影响。
行业竞争与技术迭代风险:小核酸药物领域竞争激烈,技术迭代迅速。若相关企业在技术路线选择、新型递送平台开发、靶点序列布局或专利保护策略上落后于行业领先者,可能对其长期竞争力产生不利影响。
政策与宏观环境风险:医药行业受监管政策影响较大,若审评审批、医保支付或国际政治经济环境发生变化,均可能对小核酸药物的研发周期、上市路径及商业化预期带来不确定性。
更多行业和公司研究成果,请长按图片关注“豫医言药”公众号
关注最新电子动态,前瞻行业投资趋势
欢迎点击“远峰电子”订阅
行业和公司更多研究结果,请长按图片关注“远峰电子”公众号
孙远峰:太平洋证券总裁助理&研究院院长&科技首席分析师,哈尔滨工业大学工学学士,清华大学工学博士,近3年电子实业工作经验;2013年到2018年多次获得新财富、保险资管IAMAC、水晶球、金牛奖等奖项的电子行业最佳分析师;2019年开始未参加任何个人评比,其骨干团队专注于创新&创业型研究所的一线具体创收&创誉工作,以“产业资源赋能深度研究”为导向,构建研究&销售一体化队伍,积累了健全的成熟团队自驱机制和年轻团队培养机制,获得市场验证;2023年带领崭新团队获得《证券时报》评选的中国证券业最具特色研究君鼎奖,2023年和2024年获得Wind第11届和第12届金牌分析师进步最快研究机构奖;清华校友总会电子工程系分会副秘书长,清华大学上海校友会电子信息专委会委员。
执业资格证书编号:S1190525020001
100 项与 Mivelsiran 相关的药物交易
登录后查看更多信息
研发状态
10 条进展最快的记录, 后查看更多信息
登录
| 适应症 | 最高研发状态 | 国家/地区 | 公司 | 日期 |
|---|---|---|---|---|
| 脑淀粉样血管病 | 临床2期 | 美国 | 2024-05-17 | |
| 脑淀粉样血管病 | 临床2期 | 澳大利亚 | 2024-05-17 | |
| 脑淀粉样血管病 | 临床2期 | 加拿大 | 2024-05-17 | |
| 脑淀粉样血管病 | 临床2期 | 荷兰 | 2024-05-17 | |
| 脑淀粉样血管病 | 临床2期 | 瑞士 | 2024-05-17 | |
| 脑淀粉样血管病 | 临床2期 | 英国 | 2024-05-17 | |
| 阿尔茨海默症 | 临床1期 | 美国 | 2022-02-04 | |
| 阿尔茨海默症 | 临床1期 | 加拿大 | 2022-02-04 | |
| 阿尔茨海默症 | 临床1期 | 荷兰 | 2022-02-04 | |
| 阿尔茨海默症 | 临床1期 | 英国 | 2022-02-04 |
登录后查看更多信息
临床结果
临床结果
适应症
分期
评价
查看全部结果
| 研究 | 分期 | 人群特征 | 评价人数 | 分组 | 结果 | 评价 | 发布日期 |
|---|
临床1期 | 19 | mivelsiran 100 mg | 蓋膚構製簾憲製淵糧廠(壓艱築鹽襯夢壓淵襯鬱) = Most were mild or moderate. 鬱築醖築構蓋餘顧範獵 (憲製糧鹹醖願網遞憲顧 ) | 积极 | 2025-07-29 | ||
mivelsiran 50mg Q6M | |||||||
临床1期 | 20 | 衊願遞餘顧蓋鏇夢製網(築網遞壓夢憲積膚襯糧) = By six months, the reductions in APPα and APPβ were greater than 55% and 65% 艱糧遞蓋範獵襯襯襯鹽 (齋遞淵糧壓顧選糧選憲 ) 更多 | 积极 | 2023-10-27 | |||
临床1期 | 20 | 夢築夢遞鏇憲鹹遞壓鹽(觸齋夢網壓獵鑰獵築蓋) = 窪鑰簾選夢窪鹹膚鹽遞 齋齋艱構淵簾積窪鑰繭 (鬱構積蓋遞選餘網簾選 ) 更多 | 积极 | 2023-10-25 | |||
临床1期 | 20 | 繭膚鹽鏇顧願願製範襯(窪襯憲壓廠齋鑰觸艱積) = 顧網願壓齋選窪齋廠鬱 選鹹艱糧願築觸醖構醖 (範餘簾艱簾廠淵網鑰鑰 ) | 积极 | 2023-07-17 | |||
繭膚鹽鏇顧願願製範襯(窪襯憲壓廠齋鑰觸艱積) = 願膚築網醖範簾膚襯壓 選鹹艱糧願築觸醖構醖 (範餘簾艱簾廠淵網鑰鑰 ) 更多 |
登录后查看更多信息
转化医学
使用我们的转化医学数据加速您的研究。
登录
或

药物交易
使用我们的药物交易数据加速您的研究。
登录
或

核心专利
使用我们的核心专利数据促进您的研究。
登录
或

临床分析
紧跟全球注册中心的最新临床试验。
登录
或

批准
利用最新的监管批准信息加速您的研究。
登录
或

特殊审评
只需点击几下即可了解关键药物信息。
登录
或

生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用


