预约演示
更新于:2025-05-07
Trimegestone
曲美孕酮
更新于:2025-05-07
概要
基本信息
原研机构- |
非在研机构- |
权益机构- |
最高研发阶段批准上市 |
首次获批日期 英国 (2001-02-11) |
最高研发阶段(中国)- |
特殊审评- |
登录后查看时间轴
结构/序列
分子式C22H30O3 |
InChIKeyJUNDJWOLDSCTFK-MTZCLOFQSA-N |
CAS号74513-62-5 |
关联
2
项与 曲美孕酮 相关的临床试验EUCTR2017-004664-36-DE
Characterization of ovulation inhibition of a new vaginal delivery system containing trimegestone - an open-label, single-centre study in healthy females of childbearing potential - Ovulation inhibition study with trimegestone
开始日期2018-06-19 |
申办/合作机构- |
NCT00195455
A Prospective Open Label Study to Evaluate Vasomotor Symptoms Control and Bleeding Patterns With a Continuous Regimen of a New Progestin Trimegestone 0.125 mg and 17 b Estradiol 1 mg (Totelle),as HT on Postmenopausal Women
The purpose of this study is to evaluate vasomotor symptoms (VMS) control of the continuous regimen 17 b estradiol/trimegestone combination.
开始日期2005-02-01 |
申办/合作机构 |
100 项与 曲美孕酮 相关的临床结果
登录后查看更多信息
100 项与 曲美孕酮 相关的转化医学
登录后查看更多信息
100 项与 曲美孕酮 相关的专利(医药)
登录后查看更多信息
297
项与 曲美孕酮 相关的文献(医药)2025-06-01·Clinica Chimica Acta
Quantification of trimethylamine-N-oxide (TMAO) and its main related trimethylammonium-containing compounds in human plasma by LC-MS/MS
Article
作者: Sotgia, Salvatore
2025-04-01·Reproduction in Domestic Animals
Serum Ionised Calcium and Ionised Magnesium in Dairy Cows and Their Associations With the Incidence and Severity of Postpartum Metritis
Article
作者: Ríos, José ; Elcoso, Guillermo ; Bach, Alex ; Jose‐Cunilleras, Eduard ; Bassols, Anna ; Robles‐Guirado, José Angel
2025-01-01·American Journal of Veterinary Research
Calcium and magnesium abnormalities in puppies with parvoviral enteritis
Article
作者: Celliers, Anri ; Thompson, Peter N. ; Mouton, Anneri ; Schoeman, Johan P. ; Mellanby, Richard J.
100 项与 曲美孕酮 相关的药物交易
登录后查看更多信息
研发状态
10 条最早获批的记录, 后查看更多信息
登录
| 适应症 | 国家/地区 | 公司 | 日期 |
|---|---|---|---|
| 绝经期综合征 | - | - | - |
登录后查看更多信息
临床结果
临床结果
适应症
分期
评价
查看全部结果
| 研究 | 分期 | 人群特征 | 评价人数 | 分组 | 结果 | 评价 | 发布日期 |
|---|
No Data | |||||||
登录后查看更多信息
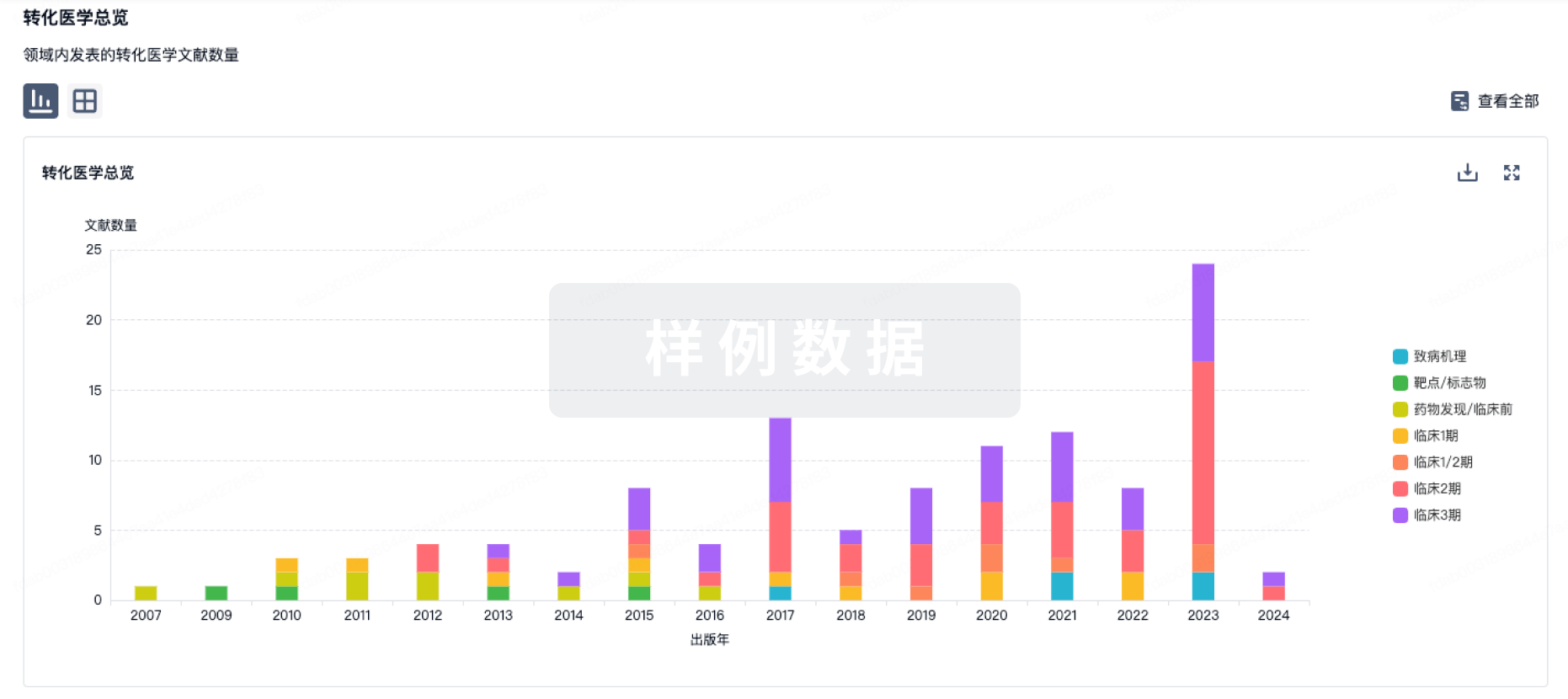
转化医学
使用我们的转化医学数据加速您的研究。
登录
或
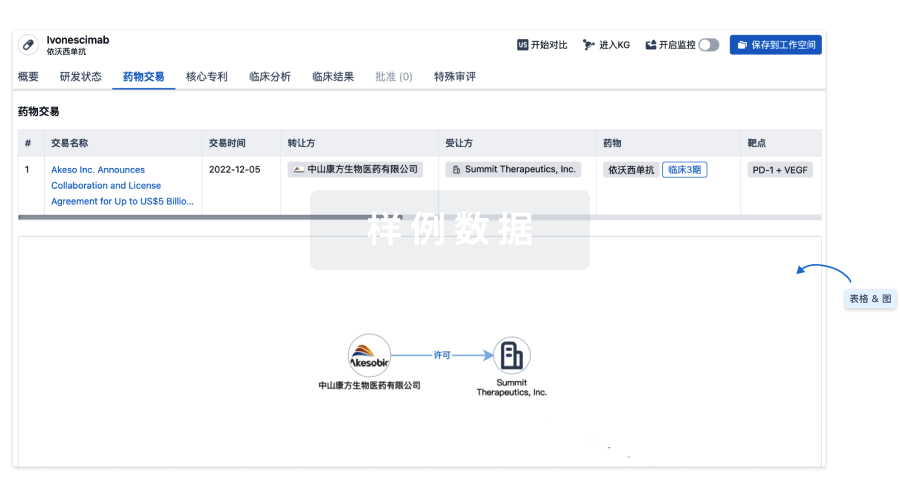
药物交易
使用我们的药物交易数据加速您的研究。
登录
或
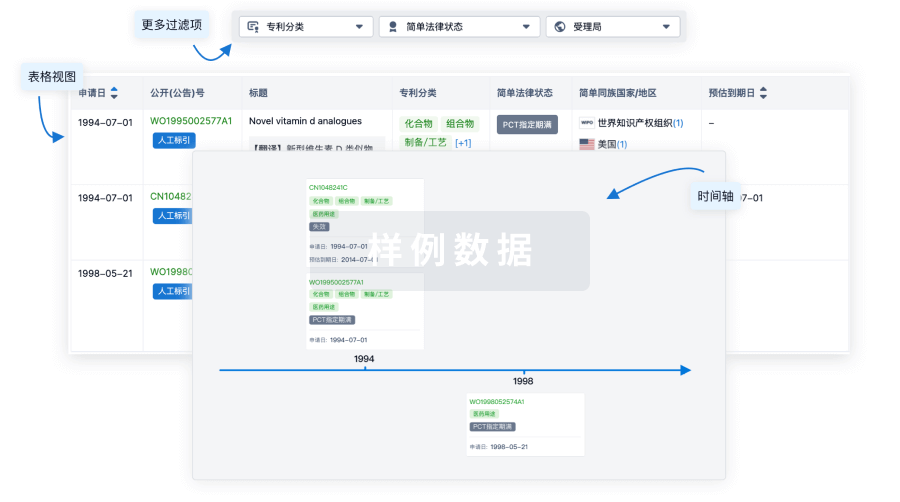
核心专利
使用我们的核心专利数据促进您的研究。
登录
或
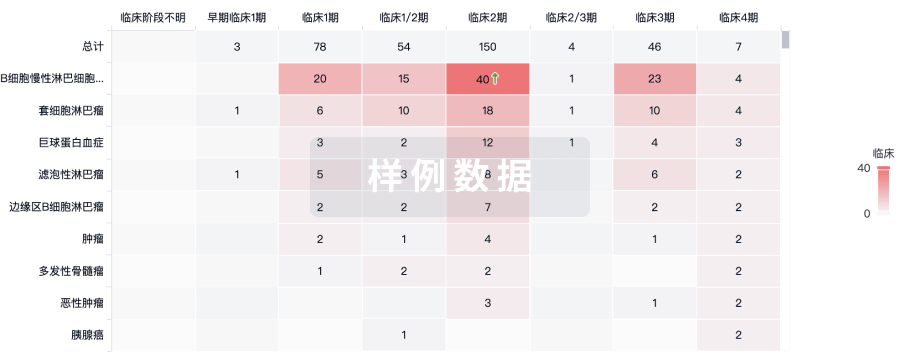
临床分析
紧跟全球注册中心的最新临床试验。
登录
或
批准
利用最新的监管批准信息加速您的研究。
登录
或
特殊审评
只需点击几下即可了解关键药物信息。
登录
或
生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用