预约演示
更新于:2025-08-11

AndroScience Corp.
更新于:2025-08-11
概览
标签
肿瘤
泌尿生殖系统疾病
神经系统疾病
小分子化药
疾病领域得分
一眼洞穿机构专注的疾病领域
暂无数据
技术平台
公司药物应用最多的技术
暂无数据
靶点
公司最常开发的靶点
暂无数据
| 排名前五的药物类型 | 数量 |
|---|---|
| 小分子化药 | 5 |
| 未知 | 1 |
| 排名前五的靶点 | 数量 |
|---|---|
| AR(雄激素受体) | 4 |
| STAT3 x STAT5A | 2 |
关联
6
项与 AndroScience Corp. 相关的药物靶点 |
作用机制 AR 降解剂 |
在研适应症 |
非在研适应症- |
最高研发阶段临床前 |
首次获批国家/地区- |
首次获批日期- |
靶点 |
作用机制 AR拮抗剂 |
在研适应症 |
非在研适应症- |
最高研发阶段临床前 |
首次获批国家/地区- |
首次获批日期- |
靶点 |
作用机制 AR拮抗剂 |
在研适应症 |
非在研适应症- |
最高研发阶段临床前 |
首次获批国家/地区- |
首次获批日期- |
2
项与 AndroScience Corp. 相关的临床试验NCT01289574
Phase 2, Multi-Center, Randomized, Double-Blind, Vehicle-Controlled Clinical Study to Evaluate the Safety and Efficacy of 0.1% and 0.025% ASC-J9 Creams Applied Topically Twice Daily for 12 Weeks for the Treatment of Facial Acne Vulgaris
To evaluate the safety and efficacy of topical 0.1% and 0.025% ASC-J9 creams applied twice daily for facial acne compared to vehicle control.
开始日期2011-02-01 |
申办/合作机构  AndroScience Corp. AndroScience Corp. [+1] |
NCT00525499
A Phase 2, Multi-Center, Randomized, Double-Blind, Vehicle-Controlled Dose-Ranging Clinical Study to Evaluate the Safety and Efficacy of ASC-J9 Cream Applied Twice Daily for 12 Weeks for the Treatment of Facial Acne Vulgaris
The purpose of this study is to evaluate if topical ASC-J9 cream is effective in treating acne.
开始日期2007-08-01 |
申办/合作机构 |
100 项与 AndroScience Corp. 相关的临床结果
登录后查看更多信息
0 项与 AndroScience Corp. 相关的专利(医药)
登录后查看更多信息
1
项与 AndroScience Corp. 相关的文献(医药)2016-05-15·Human molecular genetics2区 · 生物学
A small-molecule Nrf1 and Nrf2 activator mitigates polyglutamine toxicity in spinal and bulbar muscular atrophy
2区 · 生物学
Article
作者: Gen Sobue ; Elaine Bautista ; Laura C. Bott ; Charles C.-Y. Shih ; Ke-lian Chen ; Nisha M. Badders ; George G. Harmison ; Nico P. Dantuma ; Masahisa Katsuno ; Carlo Rinaldi ; J. Paul Taylor ; Kenneth H. Fischbeck
Spinal and bulbar muscular atrophy (SBMA, also known as Kennedy's disease) is one of nine neurodegenerative disorders that are caused by expansion of polyglutamine-encoding CAG repeats. Intracellular accumulation of abnormal proteins in these diseases, a pathological hallmark, is associated with defects in protein homeostasis. Enhancement of the cellular proteostasis capacity with small molecules has therefore emerged as a promising approach to treatment. Here, we characterize a novel curcumin analog, ASC-JM17, as an activator of central pathways controlling protein folding, degradation and oxidative stress resistance. ASC-JM17 acts on Nrf1, Nrf2 and Hsf1 to increase the expression of proteasome subunits, antioxidant enzymes and molecular chaperones. We show that ASC-JM17 ameliorates toxicity of the mutant androgen receptor (AR) responsible for SBMA in cell, fly and mouse models. Knockdown of the Drosophila Nrf1 and Nrf2 ortholog cap 'n' collar isoform-C, but not Hsf1, blocks the protective effect of ASC-JM17 on mutant AR-induced eye degeneration in flies. Our observations indicate that activation of the Nrf1/Nrf2 pathway is a viable option for pharmacological intervention in SBMA and potentially other polyglutamine diseases.
1
项与 AndroScience Corp. 相关的新闻(医药)2024-06-07
TAINAN, June 7, 2024 /PRNewswire/ -- MLB (Merry Life Biomedical Company, Ltd., Taiwan: ), a biomedical company, announced that the U.S. Food and Drug Administration (FDA) has approved IND application for
TML-6, an novel drug to treat
Alzheimer's disease (AD), enabling a Phase 1 clinical trial to be initiated this July. Advancing TML-6 into clinical trial is a critical milestone for MLB to develop a new era
multi-target drug for AD since 2018.
About TML-6
Continue Reading
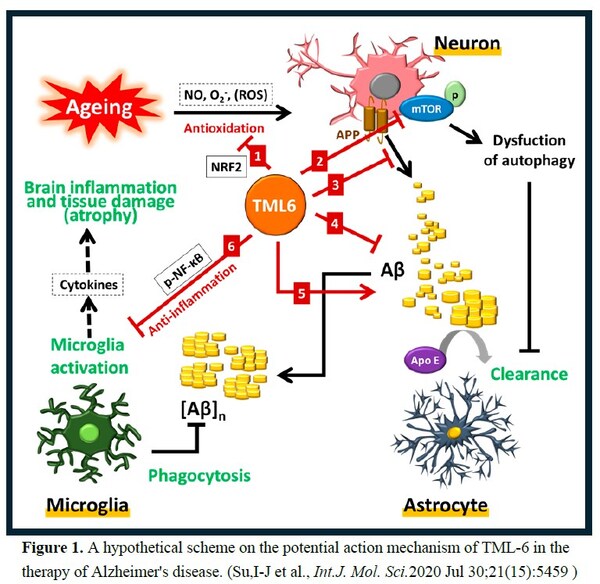
TML-6 is a novel synthetic curcumin analog. Professor Ih-Jen Su at Southern Taiwan University used a platform of 6 aging and AD biomarkers to screen the 12 compounds from Androscience (San Diego, USA) for AD candidate. Preclinical studies showed that
TML-6 (ASC-6) exhibits a
multi-target action mechanism for AD, including anti-aging, activation of autophagy (mTOR inhibitor), reducing amyloid accumulation, and anti-inflammation (Figure 1), the efficacy confirmed by 2 AD animal models.
TML-6 has a
high bioavailability through formulation and then completed preclinical toxicology and safety studies.
TML-6 should be potentially a novel drug to improve or
reverse the progression of early-stage AD.
The Design and Future Plan of TML-6 for AD Clinical Trial
s
TML-6 is developed as an oral drug and will conduct a SAD/MAD phase 1 clinical trial at Glendale Adventist Medical Center, LA, USA in 2024 Q3. Elderly cohort and CSF pharmacokinetics (PK) studies were specifically designed. For a global multi-site phase 2a clinical trial, several distinguished global AD experts provided consultation.
Blood biomarkers will be included in the phase 2a trial as the surrogate endpoint of efficacy. Furthermore, TML-6 is considering to combine with the current anti-amyloid drugs in phase 2 trial. The
drug combination could not only exhibit synergistic effects to improve AD behavior, reducing amyloid accumulation and ant-inflammation, but can also reduce antibody dosing to only 10% and avoid the adverse events (ARIAs) of anti-body drugs. MLB has successfully raised funds to conduct this global phase 2a clinical trial, scheduled to be conducted on 2025 Q3.
Contact:
Chien Hong Lin, PhD
Email: [email protected]
SOURCE Merry Life Biomedical Company, Ltd.
临床2期临床申请临床1期
100 项与 AndroScience Corp. 相关的药物交易
登录后查看更多信息
100 项与 AndroScience Corp. 相关的转化医学
登录后查看更多信息
组织架构
使用我们的机构树数据加速您的研究。
登录
或

管线布局
2026年05月18日管线快照
管线布局中药物为当前组织机构及其子机构作为药物机构进行统计,早期临床1期并入临床1期,临床1/2期并入临床2期,临床2/3期并入临床3期
临床前
6
2
其他
登录后查看更多信息
药物交易
使用我们的药物交易数据加速您的研究。
登录
或

转化医学
使用我们的转化医学数据加速您的研究。
登录
或

营收
使用 Synapse 探索超过 36 万个组织的财务状况。
登录
或

科研基金(NIH)
访问超过 200 万项资助和基金信息,以提升您的研究之旅。
登录
或

投资
深入了解从初创企业到成熟企业的最新公司投资动态。
登录
或

融资
发掘融资趋势以验证和推进您的投资机会。
登录
或

生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用