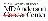更新于:2023-10-19

更新于:2023-10-19
概览
标签
免疫系统疾病
血液及淋巴系统疾病
肿瘤
细胞疗法
关联
1
项与 相关的药物51
项与 相关的临床试验Smartphone-delivered Scheduled Smoking With Compliance Facilitation as an Adjunct Cessation Therapy: a Feasibility Study
Harmony & Health: Feasibility of a Movement Intervention to Improve Psychosocial Wellbeing in Black Adults in Northeast Texas
100 项与 相关的临床结果
登录后查看更多信息
0 项与 相关的专利(医药)
登录后查看更多信息
2,639
项与 相关的文献(医药)2023-02-17·iScience
Exploiting metabolic vulnerabilities after anti-VEGF antibody therapy in ovarian cancer
Article
作者: Glassman, Deanna ; Kim, Mark S. ; Spradlin, Meredith ; Badal, Sunil ; Taki, Mana ; Bhattacharya, Pratip ; Dutta, Prasanta ; Kingsley, Charles V. ; Foster, Katherine I. ; Animasahun, Olamide ; Jeon, Jin Heon ; Achreja, Abhinav ; Jayaraman, Anusha ; Kumar, Praveen ; Nenwani, Minal ; Wuchu, Fulei ; Bayraktar, Emine ; Wu, Yutuan ; Stur, Elaine ; Mangala, Lingegowda ; Lee, Sanghoon ; Yap, Timothy A. ; Westin, Shannon N. ; Eberlin, Livia S. ; Nagrath, Deepak ; Sood, Anil K.
Despite modest clinical improvement with anti-vascular endothelial growth factor antibody (AVA) therapy in ovarian cancer, adaptive resistance is ubiquitous and additional options are limited. A dependence on glutamine metabolism, via the enzyme glutaminase (GLS), is a known mechanism of adaptive resistance and we aimed to investigate the utility of a GLS inhibitor (GLSi). Our in vitro findings demonstrated increased glutamine abundance and a significant cytotoxic effect in AVA-resistant tumors when GLSi was administered in combination with bevacizumab. In vivo, GLSi led to a reduction in tumor growth as monotherapy and when combined with AVA. Furthermore, GLSi initiated after the emergence of resistance to AVA therapy resulted in a decreased metabolic conversion of pyruvate to lactate as assessed by hyperpolarized magnetic resonance spectroscopy and demonstrated robust antitumor effects with a survival advantage. Given the increasing population of patients receiving AVA therapy, these findings justify further development of GLSi in AVA resistance.
2021-07-01·American Journal of Hematology
An effective chemotherapy-free regimen of ponatinib plus venetoclax for relapsed/refractory P hiladelphia chromosome-positive acute lymphoblastic leukemia
作者: Short, Nicholas J. ; Konopleva, Marina ; Kadia, Tapan ; Kebriaei, Partow ; Daver, Naval ; Huang, Xuelin ; Masarova, Lucia ; Cook, Robin ; Jain, Nitin ; Jabbour, Elias ; Kantarjian, Hagop ; Ravandi, Farhad
2020-11-19·New England Journal of Medicine1区 · 医学
Endometrial cancer
1区 · 医学
Review
作者: Lu, Karen H. ; Broaddus, Russell R.
A recent prospective study of progestincontaining IUDs showed that 91% of women with CAH and 54% of women with endometrial cancer had a complete response at 12 mo. Women with higher-grade tumors or tumors invading the myometrium, as seen on magnetic resonance imaging, are not candidates for conservative management.
1
项与 相关的新闻(医药)2023-09-27
SAN FRANCISCO, Sept. 27, 2023 /PRNewswire/ -- Nektar Therapeutics (Nasdaq: NKTR) announced today that it has entered into a new clinical study collaboration with Cellular Biomedicine Group Inc. ("CBMG"). The study will evaluate Nektar's NKTR-255, a novel interleukin-15 (IL-15) receptor agonist, in combination with CBMG's C-TIL051, a tumor-infiltrating lymphocyte (TIL) therapy, in advanced non-small cell lung cancer (NSCLC) patients that are relapsed or refractory to anti-PD-1 therapy.
NKTR-255 is a novel polymer-conjugated human IL-15 receptor agonist currently being studied in two separate Phase 2 studies in combination with cell therapies and immunotherapies. Preclinical and early clinical data suggest that IL-15 can improve proliferation and persistence of cellular therapies including TIL, TCR, and CAR-T therapies to increase specific anti-tumor activity.
"Early results have shown that NKTR-255 has the potential to emerge as a valuable adjuvant therapy for a range of cell therapy platforms," said Mary Tagliaferri, M.D., Chief Medical Officer at Nektar Therapeutics. "We are excited to partner with CBMG, a leader in the development of the next generation of cell therapies, to evaluate this novel combination in the clinical setting of lung cancer. Combining NKTR-255 and C-TIL051 could have the potential to deliver deep and durable objective responses with a well-tolerated regimen to patients battling relapsed or refractory NSCLC."
C-TIL051 is an autologous adoptive cell therapy being developed by CBMG that is comprised of a patient's ex vivo expanded lymphocytes using CBMG's proprietary manufacturing process. The development of C-TIL051 was based on initial NSCLC TIL studies demonstrating encouraging safety and efficacy results (NCT03215810, NCT03645928). In October of last year, CBMG received clearance of an Investigational New Drug (IND) application for C-TIL051 for late-stage NSCLC patients that are relapsed or refractory to anti-PD-1 therapy.
Under the new collaboration, CBMG will add NKTR-255 to its ongoing CBMG-sponsored Phase 1 clinical trial evaluating C-TIL051 in NSCLC patients who have relapsed on or were refractory to anti-PD-1 therapy, which is being conducted at Duke Cancer Institute (NCT05676749). The study is expected to enroll a total of 20 patients. Nektar will contribute NKTR-255 supply for the study. Nektar and CBMG will each maintain existing global rights to their respective investigational medicines.
About NKTR-255
NKTR-255 is a biologic that targets the IL-15 pathway in order to activate the body's innate and adaptive immunity. Through optimal engagement of the IL-15 receptor complex, NKTR-255 is designed to enhance functional NK cell populations and formation of long-term immunological memory, which may lead to sustained and durable anti-tumor immune response.
Preclinical and early clinical findings suggest IL-15 can improve proliferation and persistence of cellular therapies including TIL, TCR, and CAR-T therapies to increase specific anti-tumor activity.
NKTR-255 is currently being studied in two separate Phase 2 studies in combination with cell therapies and immunotherapy. A Phase 2/3 study is underway that combines NKTR-255 with approved CAR-T cell therapies in patients with diffuse large B-cell lymphoma, which is currently recruiting (NCT05664217). NKTR-255 is also being studied in a Phase 2 study in combination with avelumab as a maintenance treatment in patients with locally advanced or metastatic urothelial carcinoma in the Merck KGaA-sponsored JAVELIN Bladder Medley trial (NCT05327530), as well as in combination with durvalumab in patients with locally advanced NSCLC in an IST conducted at M.D. Anderson Cancer Center.
In addition, there are two ongoing investigator sponsored trials (ISTs) evaluating NKTR-255 as adjunct therapy following a CAR-T cell therapy. Fred Hutchinson Cancer Center is conducting a Phase 1 study evaluating NKTR-255 following lisocabtagene maraleucel treatment in patients with relapsed/refractory large B-cell lymphoma (NCT05359211), and Stanford University is conducting a Phase 1 study evaluating NKTR-255 following an investigational CD19/22 CAR-T cell therapy in patients with relapsed or refractory B-cell acute lymphoblastic leukemia (NCT03233854).
About Nektar Therapeutics
Nektar Therapeutics is a biopharmaceutical company with a robust, wholly owned R&D pipeline of investigational medicines in immunology and oncology as well as a portfolio of approved partnered medicines. Nektar is headquartered in San Francisco, California, with additional manufacturing operations in Huntsville, Alabama. Further information about the company and its drug development programs and capabilities may be found online at .
Cautionary Note Regarding Forward-Looking Statements
This press release contains forward-looking statements which can be identified by words such as: "will," "may," "evaluate," "develop," "provide," "potential" and similar references to future periods. Examples of forward-looking statements include, among others, statements regarding the therapeutic potential of, and future development plans for NKTR-255, and our other drug candidates in research programs, the prospects and plans for our collaborations with other companies, the timing of the initiation of clinical studies and the data readouts for our drug candidates. Forward-looking statements are neither historical facts nor assurances of future performance. Instead, they are based only on our current beliefs, expectations and assumptions regarding the future of our business, future plans and strategies, anticipated events and trends, the economy and other future conditions. Because forward-looking statements relate to the future, they are subject to inherent uncertainties, risks and changes in circumstances that are difficult to predict and many of which are outside of our control. Our actual results may differ materially from those indicated in the forward-looking statements. Therefore, you should not rely on any of these forward-looking statements. Important factors that could cause our actual results to differ materially from those indicated in the forward-looking statements include, among others: (i) our statements regarding the therapeutic potential of NKTR-255, and our other drug candidates are based on preclinical and clinical findings and observations and are subject to change as research and development continue; (ii) NKTR-255 and our other drug candidates are investigational agents and continued research and development for these drug candidates is subject to substantial risks, including negative safety and efficacy findings in ongoing clinical studies (notwithstanding positive findings in earlier preclinical and clinical studies); (iii) NKTR-255 and our other drug candidates are in various stages of clinical development and the risk of failure is high and can unexpectedly occur at any stage prior to regulatory approval; (iv) the timing of the commencement or end of clinical trials and the availability of clinical data may be delayed or unsuccessful due to challenges caused by the COVID-19 pandemic, regulatory delays, slower than anticipated patient enrollment, manufacturing challenges, changing standards of care, evolving regulatory requirements, clinical trial design, clinical outcomes, competitive factors, or delay or failure in ultimately obtaining regulatory approval in one or more important markets; (v) we may not achieve the expected cost savings we expect from our 2022 corporate restructuring and reorganization plan or our 2023 cost restructuring plan and we may undertake additional restructuring and cost-saving activities in the future, (vi) patents may not issue from our patent applications for our drug candidates, patents that have issued may not be enforceable, or additional intellectual property licenses from third parties may be required; and (vii) certain other important risks and uncertainties set forth in our Annual Report on Form 10-Q filed with the Securities and Exchange Commission on August 9, 2023. Any forward-looking statement made by us in this press release is based only on information currently available to us and speaks only as of the date on which it is made. We undertake no obligation to update any forward-looking statement, whether written or oral, that may be made from time to time, whether as a result of new information, future developments or otherwise.
Contact:
For Investors:
Vivian Wu of Nektar Therapeutics
628-895-0661
For Media:
David Rosen of Argot Partners
212-600-1902
[email protected]
SOURCE Nektar Therapeutics
100 项与 相关的药物交易
登录后查看更多信息
100 项与 相关的转化医学
登录后查看更多信息
组织架构
使用我们的机构树数据加速您的研究。
登录
或

管线布局
2024年09月08日管线快照
管线布局中药物为当前组织机构及其子机构作为药物机构进行统计,早期临床1期并入临床1期,临床1/2期并入临床2期,临床2/3期并入临床3期
临床2期
1
登录后查看更多信息
药物交易
使用我们的药物交易数据加速您的研究。
登录
或
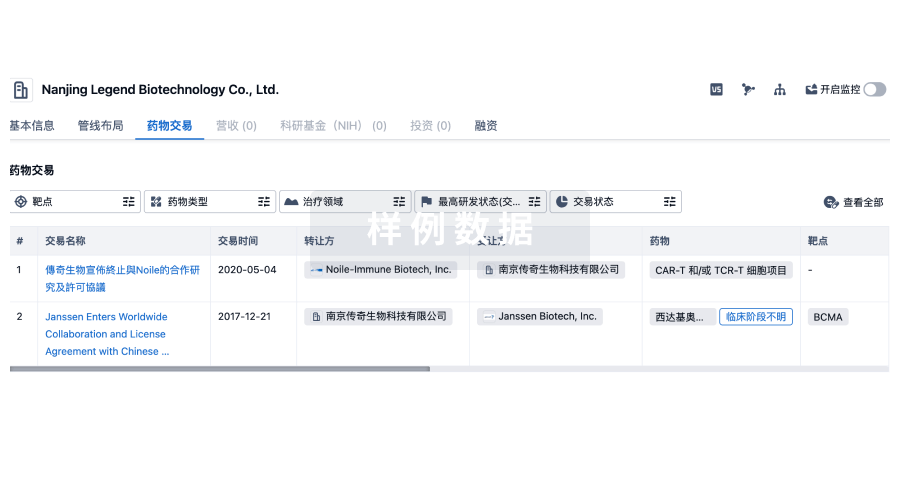
转化医学
使用我们的转化医学数据加速您的研究。
登录
或
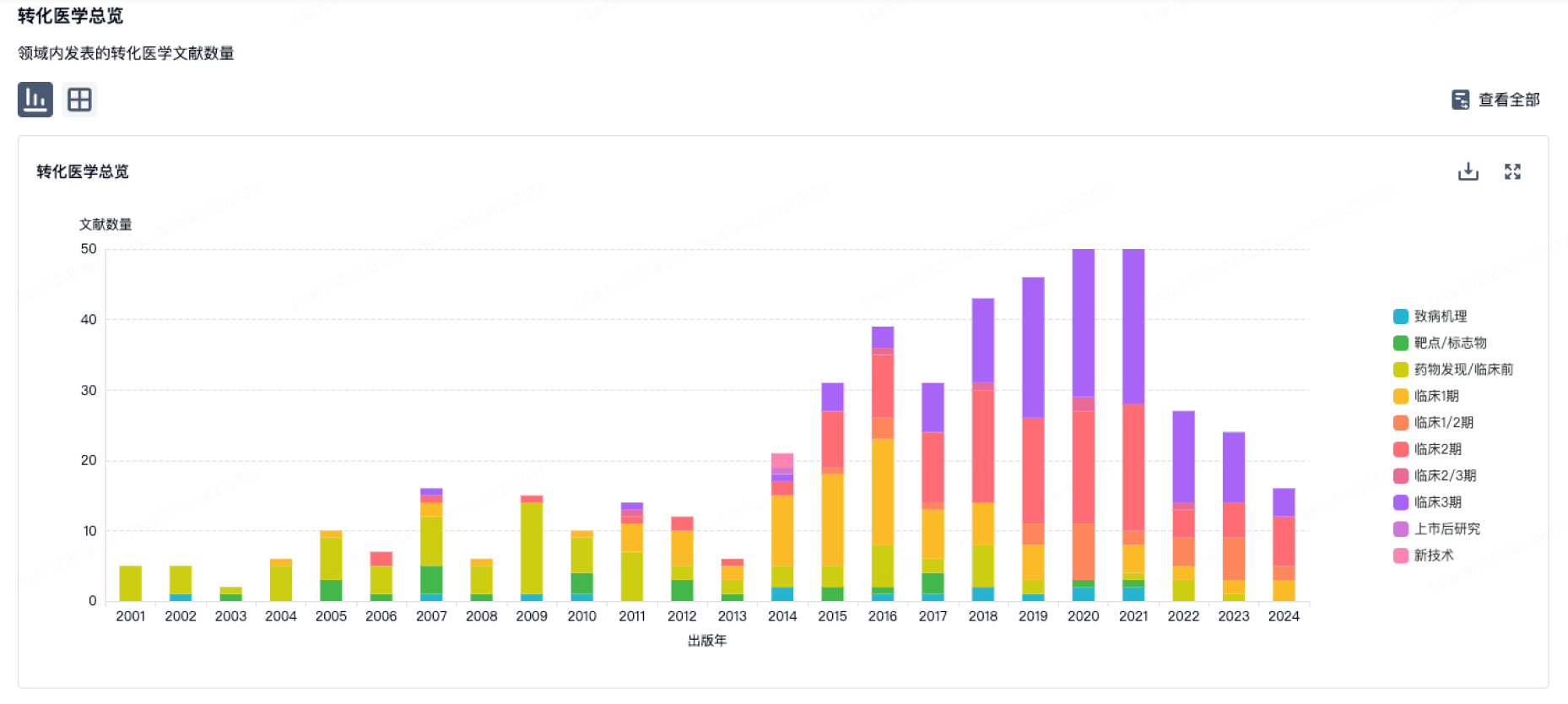
营收
使用 Synapse 探索超过 36 万个组织的财务状况。
登录
或
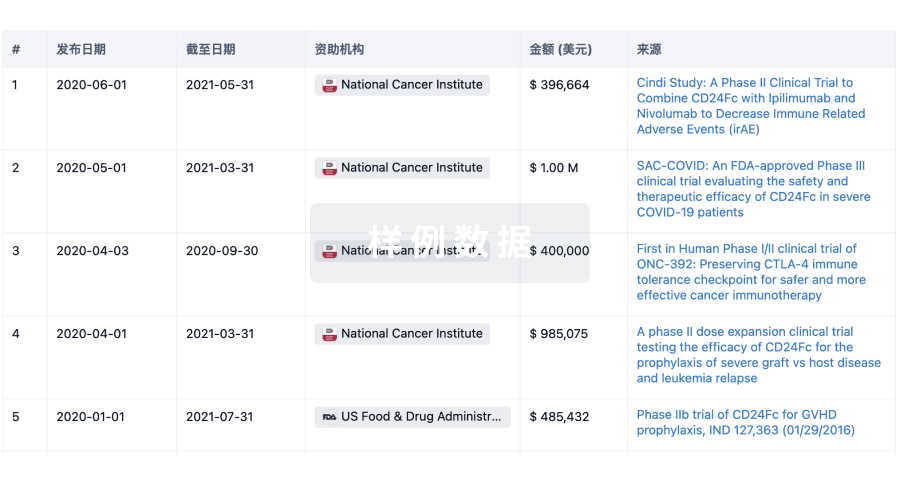
科研基金(NIH)
访问超过 200 万项资助和基金信息,以提升您的研究之旅。
登录
或
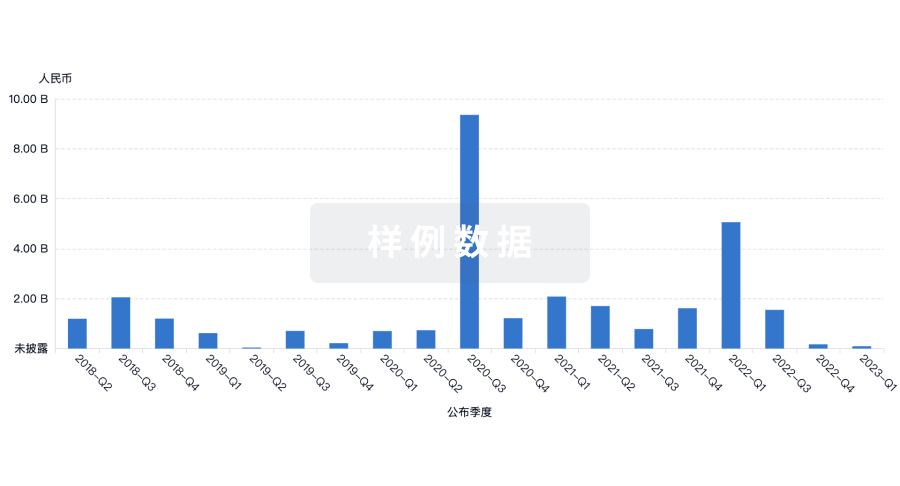
投资
深入了解从初创企业到成熟企业的最新公司投资动态。
登录
或
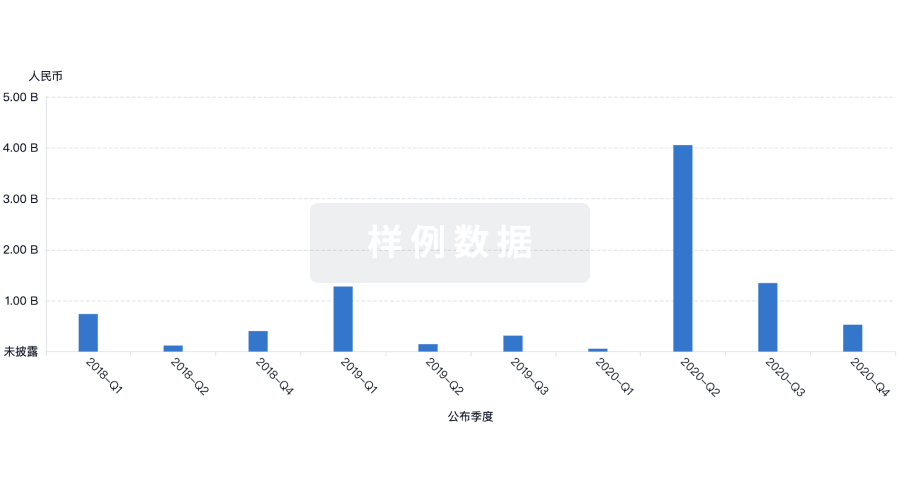
融资
发掘融资趋势以验证和推进您的投资机会。
登录
或
标准版
¥16800
元/账号/年
新药情报库 | 省钱又好用!
立即使用
来和芽仔聊天吧
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用