靶点- |
作用机制- |
在研机构- |
|
在研适应症- |
|
最高研发阶段无进展 |
首次获批国家/地区- |
首次获批日期- |
靶点- |
作用机制- |
在研机构- |
|
在研适应症- |
|
最高研发阶段无进展 |
首次获批国家/地区- |
首次获批日期- |
The Safety and Efficacy Assessment of Allogeneic γδ T Cells Combined With Targeted Therapy and Immunotherapy in Hepatocellular Carcinoma Patients
The purpose of this study is to evaluate the safety and efficacy of allogeneic γδ T cells combined with targeted therapy and PD-1 monoclonal antibody in patients with hepatocellular carcinoma resistant to PD-1 monoclonal antibody.
Hepatocellular Carcinoma
The Safety and Efficacy Assessment of Allogeneic γδ T Cells Combined With Targeted Therapy and Immunotherapy in Hepatocellular Carcinoma Patients
The purpose of this study is to evaluate the safety and efficacy of allogeneic γδ T cells combined with targeted therapy and PD-1 monoclonal antibody in first-line treatment of patients with hepatocellular carcinoma.
The Safety and Efficacy of PD-1 Antibody Combined With Pegylated Interferon-α Therapy to Promote the Clinical Cure in Nucleoside (Acid) Analogues-suppressed Chronic Hepatitis B Patients: A Protocol for the Prospective Pilot Study
This is a prospective, open-labled, randomized controlled study to assess efficacy and safety of treatment with Sintilimab (PDL-1 antibody) combined Peg-IFNα-2b in CHB patients on stable NAs treatment.
100 项与 No 302 Hospital Of The People Liberation Army Of China 相关的临床结果
0 项与 No 302 Hospital Of The People Liberation Army Of China 相关的专利(医药)
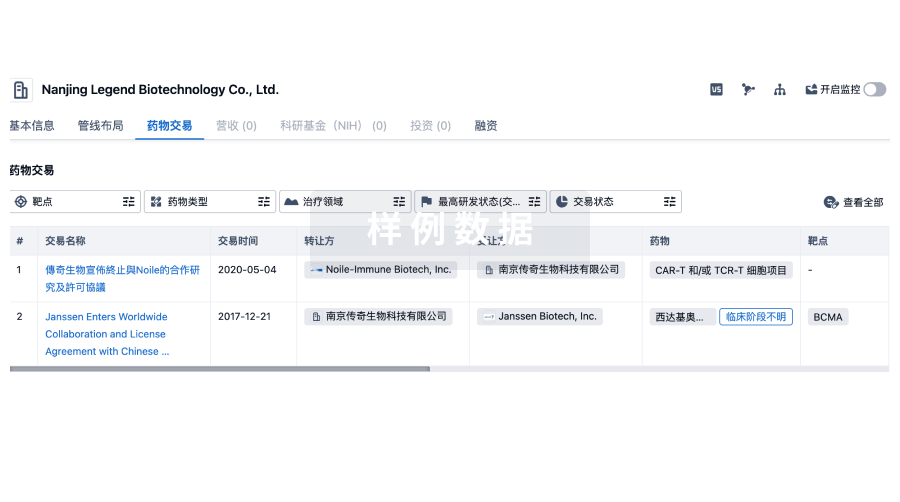
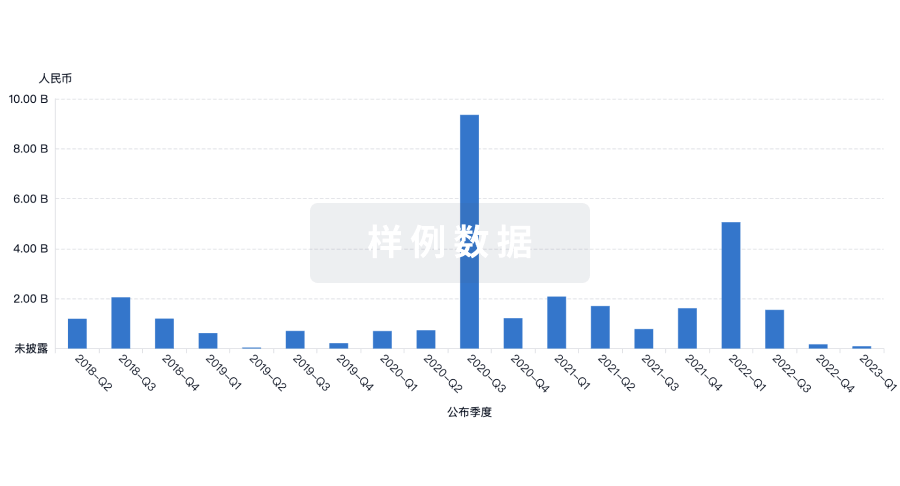
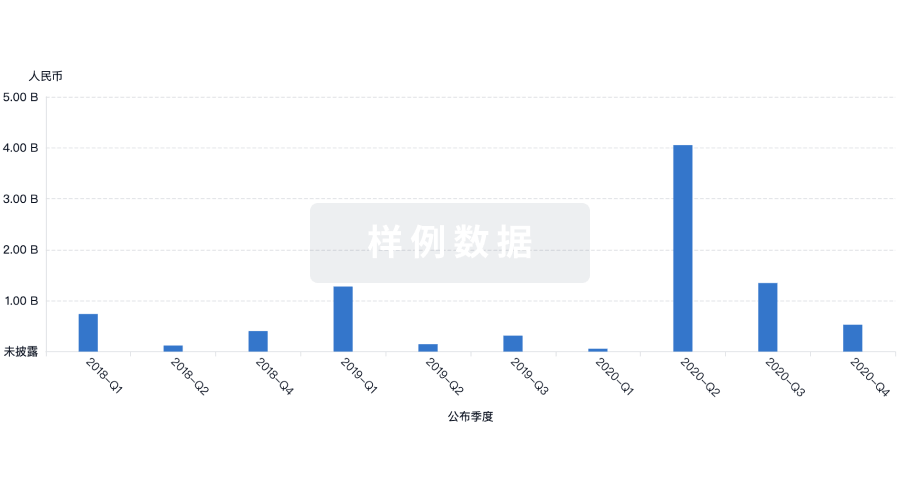
100 项与 No 302 Hospital Of The People Liberation Army Of China 相关的药物交易
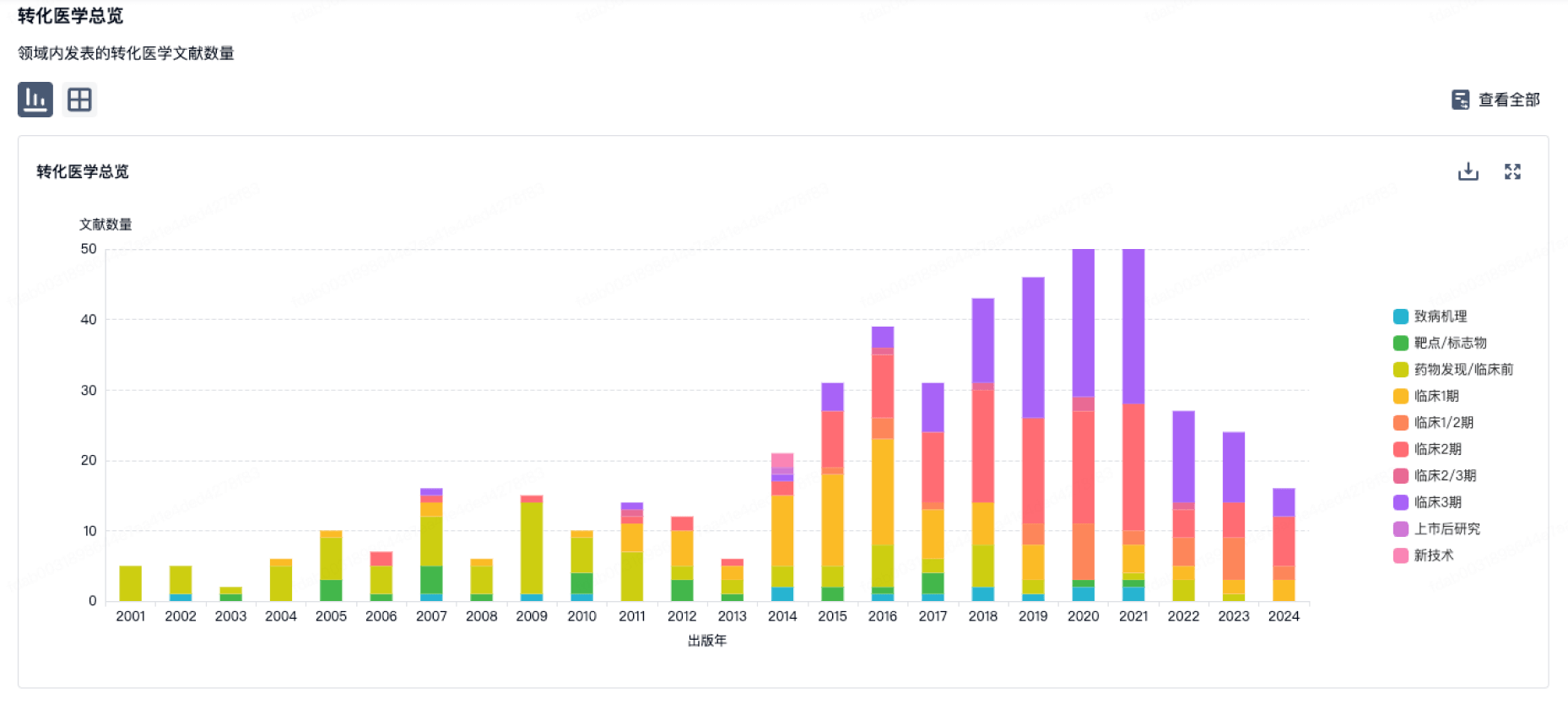
100 项与 No 302 Hospital Of The People Liberation Army Of China 相关的转化医学