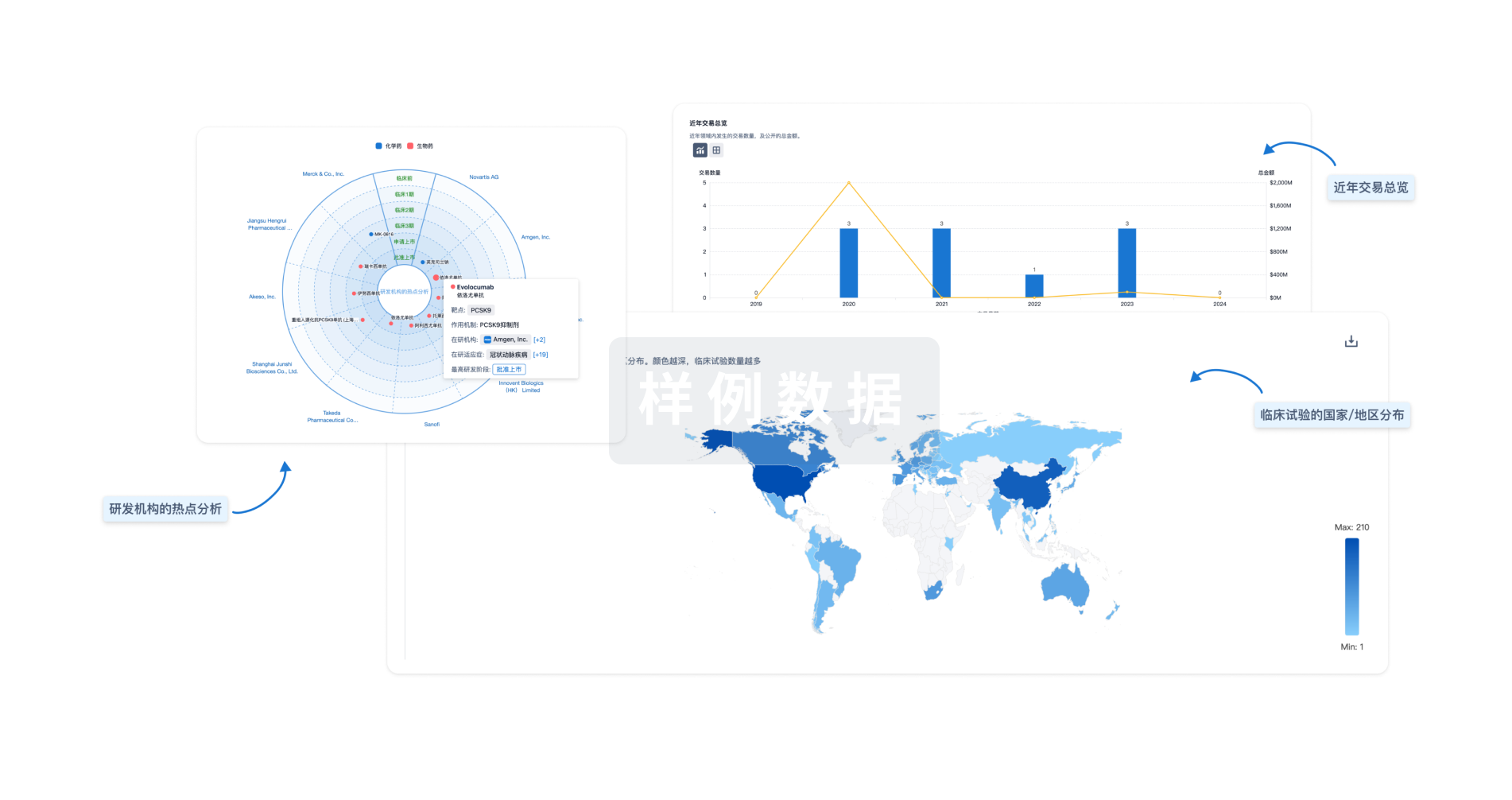
|
|
在研机构- |
|
在研适应症- |
|
最高研发阶段终止 |
首次获批国家/地区- |
首次获批日期1800-01-20 |
A Multicenter, Randomized, Double-Blind, Parallel-Group, Placebo-Controlled, Flexible-Dose Study Evaluating Efficacy, Safety, and Tolerability of Once-Daily Oral GW353162 (20-40-60mg) Versus Placebo in Subjects With Major Depressive Disorder Over an Eight-Week Treatment Period.
A Placebo Controlled Study Evaluating Efficacy, Safety and Tolerability of Medication in Patients with Major Depressive Disorder (MDD)
A Multi-Center, Randomized, Double-Blind, Parallel-Group, Placebo-Controlled, Fixed-Dose Study Evaluating the Efficacy, Safety and Tolerability of Two Doses (20mg and 60mg) of a Once-Daily Oral Formulation of GW353162 in Subjects With Major Depressive Disorder for a Treatment Period of Eight Weeks
A Placebo Controlled Study Evaluating Efficacy, Safety and Tolerability of Radafaxine in Patients with Major Depressive Disorder (MDD)
A Multicenter, Randomized, Double-Blind, Parallel-Group, Placebo-Controlled, Flexible-Dose Study Evaluating Efficacy, Safety, and Tolerability of Once-Daily Oral GW353162 (40-60mg) Versus Placebo in Subjects With Major Depressive Disorder Over an Eight-Week Treatment Period.
A Placebo Controlled Study Evaluating Efficacy, Safety and Tolerability of Medication in Patients with Major Depressive Disorder (MDD)
100 项与 nAChR α4/β2/α5 x adrenergic receptor x DAT 相关的临床结果
100 项与 nAChR α4/β2/α5 x adrenergic receptor x DAT 相关的转化医学
0 项与 nAChR α4/β2/α5 x adrenergic receptor x DAT 相关的专利(医药)