预约演示
更新于:2026-04-03
ARO-HSD
更新于:2026-04-03
概要
基本信息
药物类型 siRNA |
别名 GSK-4532990 |
作用方式 抑制剂 |
作用机制 17β-HSD13抑制剂(17β-羟基类固醇脱氢酶13抑制剂)、RNA干扰 |
治疗领域 |
非在研适应症- |
非在研机构- |
最高研发阶段临床2期 |
首次获批日期- |
最高研发阶段(中国)- |
特殊审评- |
登录后查看时间轴
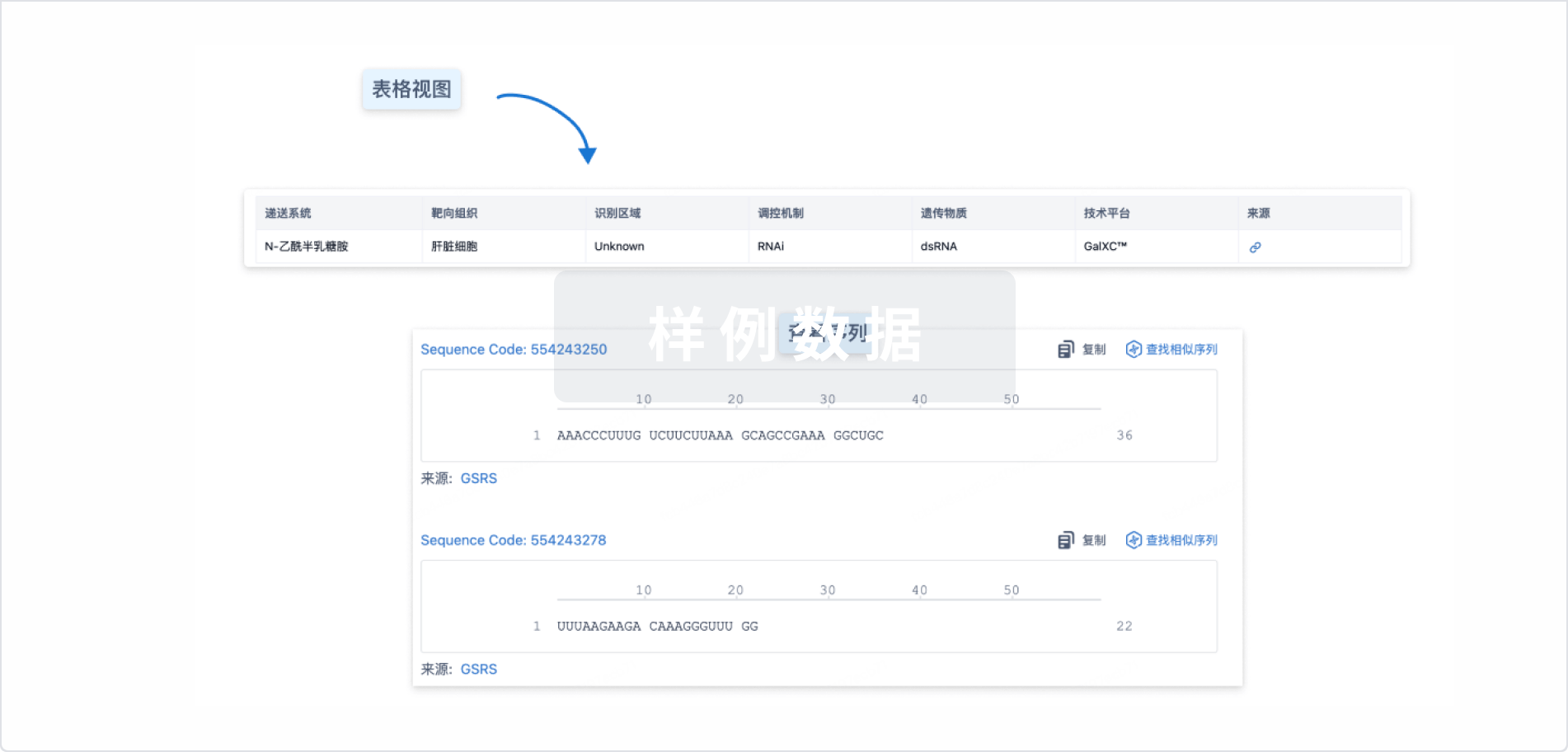
结构/序列
使用我们的RNA技术数据为新药研发加速。
登录
或
关联
5
项与 ARO-HSD 相关的临床试验NCT06613698
A Dose-Finding, Double-Blind, Placebo-Controlled Phase 2 Study to Evaluate the Efficacy and Safety of GSK4532990 for Steatohepatitis in Adults With Alcohol-related Liver Disease (ALD)
The goal of this study is to assess the safety and efficacy of GSK4532990 in participants with alcohol-related liver disease.
开始日期2024-09-27 |
申办/合作机构 |
NCT06104319
A Phase 2a, Single Dose, Open-label, Dose Exploration Study to Assess the PK-PD Activity, Safety, and Tolerability of GSK4532990 in Adult. Participants With NASH or Suspected NASH
The purpose of this study is to understand how the drug GSK4532990 is processed in the body (pharmacokinetics) and how it works in the liver (pharmacodynamics) as well as to ensure it is safe and well-tolerated. The total study duration for each participant will be approximately 24-36 weeks.
开始日期2024-01-22 |
申办/合作机构 |
CTRI/2023/03/051029
17 B-Hydroxysteroid Dehydrogenase type 13 Minimization for the treatment of NASH (HORIZON): A Double-Blind, Placebo Controlled Phase 2b Study to Evaluate the Efficacy and Safety of GSK4532990 in Adults with Pre-Cirrhotic Non-Alcoholic Steatohepatitis. - HORIZON
开始日期2023-04-06 |
申办/合作机构- |
100 项与 ARO-HSD 相关的临床结果
登录后查看更多信息
100 项与 ARO-HSD 相关的转化医学
登录后查看更多信息
100 项与 ARO-HSD 相关的专利(医药)
登录后查看更多信息
1
项与 ARO-HSD 相关的文献(医药)2023-04-01·Journal of hepatology
A phase I/II study of ARO-HSD, an RNA interference therapeutic, for the treatment of non-alcoholic steatohepatitis
Article
作者: Rohit Loomba ; Jeong Heo ; Russell Scott ; Bruce D. Given ; Ki Tae Yoon ; Dawn R. Christianson ; Eric Garcia-Medel ; Min Yi ; Martin Weltman ; Ed Gane ; Lung-Yi Mak ; Young Oh Kweon ; Jeong-Hoon Lee ; Kenneth Cusi ; Christian Schwabe ; James Hamilton ; Brent A. Neuschwander-Tetri ; Jung Il Lee ; Man-Fung Yuen ; Stephen A. Harrison
BACKGROUND & AIMS:
Loss-of-function HSD17β13 mutations protect against the development of chronic liver disease. HSD17β13 inhibition represents a potential approach to treat liver diseases, such as non-alcoholic steatohepatitis (NASH). ARO-HSD is an RNA interference (RNAi) therapeutic designed to selectively reduce expression of HSD17β13 mRNA in hepatocytes. In this study, we evaluated the effects of ARO-HSD in normal healthy volunteers (NHVs) and patients with confirmed or clinically suspected NASH.
METHODS:
The safety, tolerability, and pharmacodynamics of ARO-HSD were evaluated in 32 NHVs and 18 patients with confirmed/clinically suspected NASH. Double-blind NHV cohorts received single escalating doses of ARO-HSD (25, 50, 100, or 200 mg) or placebo subcutaneously on Day 1. Open-label patient cohorts received ARO-HSD (25, 100, or 200 mg) subcutaneously on Days 1 and 29. Liver biopsy was performed pre-dose and on Day 71 to evaluate expression levels of HSD17β13 mRNA and protein.
RESULTS:
ARO-HSD treatment was well tolerated with no treatment-related serious adverse events or drug discontinuations. The most frequently reported treatment-emergent adverse events were mild injection site reactions, which were short in duration. Mean changes in hepatic HSD17β13 mRNA from baseline to Day 71 were: -56.9% (25 mg), -85.5% (100 mg), and -93.4% (200 mg). The mean HSD17β13 mRNA reduction was 78.6% (p <0.0001) across pooled cohorts. Hepatic HSD17β13 protein levels were similarly reduced across doses. In patients, mean changes in alanine aminotransferase from baseline to Day 71 were -7.7% (25 mg), -39.3% (100 mg), and -42.3% (200 mg) (p <0.001 for pooled cohorts).
CONCLUSIONS:
ARO-HSD was well tolerated at doses ≤200 mg. This proof-of-concept study demonstrated that short-term treatment with ARO-HSD reduces hepatic HSD17β13 mRNA and protein expression, which is accompanied by reductions in alanine aminotransferase.
CLINICALTRIALS:
GOV NUMBER:
NCT04202354.
IMPACTS AND IMPLICATIONS:
There is an unmet medical need for new therapies to treat alcohol-related and non-alcoholic liver disease. ARO-HSD is a small-interfering RNA designed to silence HSD17β13 expression and hence to phenocopy the protective effect seen in individuals with HSD17β13 loss-of-function. The reductions in HSD17β13 expression and in transaminases seen with ARO-HSD administration represent an initial step towards clinical validation of HSD17β13, a drug target with substantial genetic validation, as an important modulator of human liver disease.
55
项与 ARO-HSD 相关的新闻(医药)2026-03-19
GSK said Thursday that a drug it recently agreed to out-license to Alfasigma has been greenlit by the FDA to treat debilitating itch in patients with primary biliary cholangitis (PBC). Lynavoy (linerixibat), an oral IBAT inhibitor, becomes the first drug approved in the US for cholestatic pruritus linked to PBC (see – Spotlight On: Key PDUFA dates to watch in 1Q26).The filing was backed by the Phase III GLISTEN trial, which met its primary and key secondary endpoints, with Lynavoy demonstrating rapid, significant and sustained improvement over placebo at alleviating cholestatic pruritus and itch-related sleep interference.However, Lynavoy may struggle to compete in a market that has been reshaped by the introduction of PPAR agonists Livdelzi (seladelpar) from Gilead Sciences and Iqirvo (elafibranor) from Ipsen; both were approved for PBC in 2024 and have been shown to also significantly improve itch in PBC patients.Alfasigma made its entry into the PBC space through its 2023 takeout of Intercept Pharmaceuticals for around $794 million, gaining access to the FXR agonist Ocaliva (obeticholic acid) in the process. That treatment, however, was pulled from the US market last year at the FDA's request following years of safety concerns and regulatory scrutiny.GSK's licensing deal earlier this month included an upfront payment of $300 million from Alfasigma, along with another $100 million upon FDA approval. GSK is also eligible to receive $20 million linked to approvals of the drug in the EU and UK, where it is currently under review, and up to $270 million in sales-based milestones plus tiered double-digit sales royalties. Marketing applications have also been filed in China and Canada."For many patients, cholestatic pruritus remains a persistent, poorly addressed condition," Kaivan Khavandi, GSK's R&D head for respiratory, immunology and inflammation, said in a company release. "This is the first liver medicine from our pipeline to receive approval."However, with the Alfasigma deal, the UK drugmaker will be concentrating on other liver disease spaces it sees as larger commercial opportunities, such as hepatitis B and metabolic dysfunction-associated steatohepatitis (MASH).It is currently investigating the FGF21 analogue efimosfermin alfa (GSK6519754) in the Phase III ZENITH-1 and ZENITH-2 studies for MASH, while the siRNA oligonucleotide GSK4532990 is in Phase II development for MASH as well as for alcohol-related liver disease. Its most advanced candidate for hepatitis B infection is the antisense oligonucleotide bepirovirsen (GSK3228836), which is being tested in the Phase III B-Well 1 and B-Well 2 studies.
临床3期临床结果临床2期引进/卖出上市批准
2026-02-27
2 月 27 日晚间,前沿生物(688221.SH)披露 2025 年度业绩快报,去年公司实现营收 1.44 亿元,同比增长 10.96%;扣非后净利润亏损 2.96 亿元,较上年亏损收窄 3107.03 万元。业绩快报再次提到上市公司小核酸(以下又称 siRNA)管线产品达成重要国际合作一事。2 月 23 日(春节假期休市最后一天)晚间,上市公司已披露,其与葛兰素史克公司(下简称 GSK)签署独家授权许可协议,将两款小核酸管线产品的全球独家开发、生产及商业化权利授予对方。根据协议,前沿生物将获得 4000 万美元首付款、1300 万美元近期里程碑付款,公司还将额外在两个项目中累计获得最高 9.5 亿美元的基于成功开发、监管及商业化里程碑的付款。也就是说,交易总金额突破 10 亿美元。然而,这一重磅 BD(商务拓展)并未提振前沿生物股价。在春节假期后的首个交易日(2 月 24 日),前沿生物股价大涨 9.29%。但此后三个交易日中,公司股价上演"过山车"行情,累计下跌 14.17%,回吐了此前的所有涨幅。从大涨到下跌,这笔看似救急的交易,似乎还不足以驱散市场的疑虑。"" 前沿生物股价 5 日走势图 图片来源:百度截图前沿生物书面回复《每日经济新闻》称,本次交易对公司财务层面产生了积极且切实的影响。首付款直接增加公司现金流,为核心管线研发提供资金支撑;中长期的里程碑付款与销售提成,为公司提供可预期的收入来源,有望持续带来现金流。 GSK 为何看中了前沿生物?根据公告,GSK 将获得前沿生物旗下两款小核酸管线产品在全球范围内的独家开发、生产及商业化权利,其中一款候选药物已进入新药临床试验申请(IND)阶段。前沿生物方面并未在公告中详细披露上述两款小核酸产品的靶点、适应证等信息。根据公司官网披露的研发管线进展,前沿生物目前共有 7 款小核酸药物产品在研,治疗领域涵盖肝炎、高尿酸血症与痛风、肿瘤、高脂血症等。其中提交 IND 的仅有一款代号为 FB7013 的产品,适应证为 IgA 肾病。"" 图片来源:公司官网截图另据前沿生物企业公众号,GSK 呼吸、免疫与炎症研发高级副总裁兼全球负责人 Kaivan Khavandi 提及:"该协议进一步强化了我们的免疫学管线,新增了两款具有潜在全球首创价值的寡核苷酸(寡核苷酸包括小核酸)疗法,为改善多种肾病患者预后带来重要机遇。"本次前沿生物与 GSK 达成授权合作的两款产品均处于早期研发阶段,这与去年以来达成的多笔重磅 BD 呈现出相似特征。有分析师向记者表示,此类合作本质是跨国药企以相对较低的前期成本锁定潜在技术平台或创新靶点。同时,跨国药企对中国创新药资产的关注,正从"临床验证后的管线"转向"尚未成熟的创新分子"和底层研发平台能力。资料显示,相较于传统小分子和生物药,以 siRNA 药物为代表的核酸疗法可精准沉默致病蛋白,高效降解且作用时间长,目前已成为新药开发热门方向。据 Evaluate 和 BCG 预测,2026 年全球寡核苷酸类药物市场规模将超 150 亿美元,2020 年至 2025 年复合年增长率 35%。从 GSK 的角度看,其对小核酸药物的布局实则谋划已久,并已然形成较为系统的合作版图:早在 2021 年 11 月,GSK 便与 Arrowhead 公司达成合作,引进后者治疗非酒精性脂肪性肝炎(NASH)的 RNAi 疗法 ARO-HSD。2022 年 12 月,GSK 又与 Wave Life Sciences 公司签署总额约 33 亿美元的合作协议,共同推进寡核苷酸药物的研究。近两年,GSK 的合作重心进一步向具体适应证延伸。2025 年 10 月,GSK 宣布与 Empirico 公司合作开发针对慢阻肺的 siRNA 项目 EMP-012,总交易额约 7.45 亿美元。而此次与前沿生物的合作,则延伸至肾病方向。这恰恰也是国内小核酸药物的优势领域。据 Cytiva 发布的《中国小核酸药物行业发展报告》,截至 2024 年 1 月,我国开展的小核酸药物研发项目共 35 项,大多数处于临床早期阶段。与国外不同的是,我国药企基本直接跳过了小核酸药物罕见病适应症的研发,而是聚焦心血管系统疾病和肝病等常见慢性疾病上,两者占比合计 68.3% 左右。主要原因在于我国慢性疾病患病人数庞大且发病总体呈上升趋势,市场发展空间大,这也使得国内药企聚焦小核酸药物在慢性疾病方面的研发。 前沿生物 5 年累计亏损超 17 亿元进入 2026 年,创新药领域的 BD 热潮仍在持续。但与此前"一个 BD 公告拉出多个涨停板"的情形不同,近期多起重磅合作公布后,相关公司股价反应较平淡,甚至后续出现明显回调。对于这一现象,CIC 灼识咨询总监卢李康在 1 月末接受《每日经济新闻》记者采访时分析称,资本市场对近期大额 BD 交易反应平淡,是多种因素共同作用的结果。一方面,可能是市场预期已提前消化。在交易正式公布前,市场可能已有传闻或预期,利好消息在公告时已被部分消化;另一方面,资本市场对标的资产长期价值的审慎评估,使得市场越来越关注 BD 交易背后的产品临床数据优劣、靶点创新性、市场格局等实质价值,而非单纯交易金额。具体到前沿生物的这笔交易中,两款小核酸药物管线均处于早期阶段,后续研究、审批至商业化周期漫长,不确定性极高。一个有力的参照是,就在本月,GSK 刚刚"退货"了一款数据更为成熟的在研小核酸药物。2 月初,Wave Life Sciences 公司宣布从 GSK 手中收回其罕见病药物 WVE-006 的全部权利。值得注意的是,GSK 退回 WVE-006 并非因为产品失败,而是源于公司的战略调整。双方达成共识,认为该罕见病项目应该由更专注该领域的公司主导推进,而 GSK 则将资源集中于患者基数更大的核心治疗领域。回溯这笔交易,GSK 在 2022 年 12 月为获得 WVE-006 的全球独家许可,曾支付了高达 1.7 亿美元的预付款。连这样一款曾被寄予厚望、拥有积极临床数据且交易对价不菲的药物,都可能因跨国药企的战略转向而被放弃,这无疑为前沿生物的合作前景敲响了警钟。股价"过山车"的背后,还有前沿生物的基本面现状。曾被一些媒体称为"抗艾第一股"的前沿生物,自 2020 年 10 月登陆科创板以来,始终未能实现盈利。财务数据显示,2020 年至 2024 年,公司的扣非净利润持续亏损,累计亏损金额超过 16 亿元。"" 前沿生物主要财务数据 图片来源:东方财富网页截图另就现金流情况而言,截至 2025 年三季度末,前沿生物的期末现金及现金等价物仅为 0.97 亿元。"" 图片来源:公告截图前沿生物方面向记者表示,公司早在数年前便聚焦小核酸赛道,并看好该领域的临床应用与市场前景。未来 3 到 5 年,公司将聚焦三大板块:一是深耕 HIV(艾滋病)治疗主业,依托在该领域积累的研发团队、技术与人才优势,围绕差异化临床需求布局新产品,争取推进一款新的创新药早研产品进入 PCC(临床前候选化合物)阶段;二是推进高端仿制药上市,弥补公司现金流,为创新研发提供支撑;三是重点布局小核酸创新药领域,同时探索与公司技术能力、产能布局具备协同性的新品种。每日经济新闻
2026-02-12
编者按伴随着肥胖的流行,代谢功能障碍相关脂肪性肝炎(MASH)已成为全球公共卫生领域的重要挑战。近期,随着瑞美替罗与司美格鲁肽的相继获批,MASH治疗领域正经历一场深刻变革。这不仅体现在治疗手段的日益丰富,更在于治疗理念的系统性升级——从以生活方式干预为主导的模式,逐步转向融合了精准靶向、全身代谢调控及个体化治疗的综合管理新阶段。为全面把握MASH治疗的最新进展,本文基于近期发表于Lancet. Gastroenterology & Hepatology的一篇综述,对该领域进行系统解读,从流行病学现状、关键治疗突破、临床实践挑战及未来发展方向等多个维度展开分析,以期为临床决策提供循证参考,助力患者管理的优化与提升。被忽略的“代谢性杀手”:从良性肝病到多系统健康威胁代谢功能障碍相关脂肪性肝炎(MASH)长期以来被视为一种良性的肝脏病变,初期往往无明显症状,导致患者及部分医务人员对其危害认识不足,诊断和治疗严重滞后。近年来,随着大规模流行病学研究和临床随访数据的积累,人们逐渐认识到MASH远非一种静止或无害的疾病,而是一个具有明确进展风险、影响多系统健康的“代谢性杀手”。根据近期流行病学调查数据,MASH已成为全球范围内影响最为广泛的慢性肝病之一。近三十年来,其患病率已上升超过50%,约38.2%的成年人群受到不同程度的影响,成为不容忽视的公共卫生问题。更为关键的是,MASH具有明确的进展风险。临床观察显示,约有20%的患者会由单纯脂肪变性发展为肝纤维化,而在这些患者中,又有约20%可能进一步进展为肝硬化或肝细胞癌(图1)。这种由代谢紊乱驱动、逐步恶化的病程,凸显了早期干预与全程管理的重要性。图1. MASH病程进展从预后角度看,MASH患者的主要死亡原因并非单一肝脏问题,而是多系统受累的综合体现:心血管疾病居首位,其次为肝外恶性肿瘤,肝脏相关并发症(如肝硬化失代偿)位列第三。这一死亡谱系清晰表明,MASH管理必须超越肝脏本身,采取兼顾代谢、心血管及肿瘤风险的综合治疗策略。在此背景下,肝纤维化分期已成为评估疾病风险、指导临床决策的核心指标。特别是处于F2及以上分期的高危MASH患者,其未来发生肝脏失代偿、肝癌等严重事件的风险显著升高,因此成为当前新药研发和临床试验的重点关注人群,也是临床实践中需要优先干预的目标群体。治疗新时代:双路径突破重构MASH治疗格局随着对MASH发病机制的深入理解,药物研发取得了突破性进展。2024~2025年间,瑞美替罗和司美格鲁肽相继获批用于MASH治疗,两者具有完全不同的作用机制(图2)。 图2. 瑞美替罗和司美格鲁肽对MASH不同的作用靶点1. 瑞美替罗: 肝脏靶向疗法2024年,全球首个专门针对MASH的药物——甲状腺激素受体β(THR-β)激动剂瑞美替罗获得FDA批准,标志着肝脏靶向治疗新时代的开启。瑞美替罗高度选择性地激活肝脏中的THR-β受体,通过优化线粒体功能,促进脂肪酸β氧化,从细胞层面逆转肝脏脂质堆积的病理过程。MAESTRO-NASH试验显示,为期12个月的瑞美替罗治疗后,患者实现了病理改善与生活质量改善的双重获益。肝脏组织学改善:MASH消退率达到25.9%~29.9%(安慰剂组仅9.7%),纤维化分期改善至少一个级别的患者比例为24.2%~25.9%(安慰剂组14.2%)。纤维化改善的患者同时报告了具有临床意义的生活质量提升,2. 司美格鲁肽:代谢重塑疗法2025年,胰高血糖素样肽-1(GLP-1)受体激动剂——司美格鲁肽获批用于MASH治疗,开启了通过代谢调控治疗肝脏疾病的新范式。司美格鲁肽通过系统性代谢调节发挥多重效应:一方面通过中枢性食欲抑制和延缓胃排空实现显著的体重减轻,另一方面通过改善胰岛素敏感性、调节脂质代谢等途径,从根源上改善导致MASH的代谢紊乱状态。 ESSENCE试验结果显示,经过72周治疗后,司美格鲁肽组在组织学改善方面表现出色:MASH消退且纤维化无恶化的比例高达62.9%(安慰剂组34.1%),纤维化改善且MASH无恶化的比例为37.0%(安慰剂组22.5%)。这两个药物的获批,代表了MASH治疗的两种不同但互补的策略路径:一是直接作用于肝脏的特异性靶向治疗,二是通过改善全身代谢状态间接获益的系统性治疗,共同构成了当前MASH药物治疗的基石。治疗策略多元化:从代谢调节到精准靶向的协同突破随着对MASH复杂发病机制的深入理解,其药物研发格局已从过去单一靶点的探索,演变为多靶点、多策略并行的多元化时代。这些进展主要聚焦于两大类创新路径:针对全身性代谢紊乱的代谢调节疗法与直接作用于肝脏病理核心的肝脏靶向疗法,并辅以新兴的基因靶向疗法,共同构成了从根源调控到病灶精准干预的协同突破体系。1.代谢调节疗法——调控全身性代谢紊乱此类疗法旨在纠正驱动MASH发生发展的全身性代谢失衡。多靶点肠促胰素受体激动剂:以Tirzepatide(GIP/GLP-1双重激动剂)和Retatrutide(GLP-1/GIP/胰高血糖素三重激动剂)为代表。它们超越了单一GLP-1受体激动剂的作用,通过协同激活多个肠促胰素通路,在强效降糖、减重的基础上,对肝脏脂肪代谢和炎症显示出更全面的调控潜力。例如,在SYNERGY NASH Ⅱ期研究中,替尔泊肽使高达62%的患者实现MASH缓解且纤维化无恶化。成纤维细胞生长因子21(FGF21)类似物:如Efruxifermin、Pegozafermin 和Efimospherin α。FGF21是调节能量代谢的关键激素。这些工程化类似物通过增强肝脏脂肪酸β-氧化、抑制脂肪新生、改善胰岛素敏感性等多重作用,在Ⅱ期临床试验中显著改善了MASH组织学活动和纤维化,目前均已进入Ⅲ期研究阶段。过氧化物酶体增殖物激活受体(PPAR)激动剂:以Lanifibranor为代表,能同时激活PPAR-α、PPAR-γ和PPAR-δ亚型。这种多重激活带来了协同效应:调节脂质代谢、改善胰岛素敏感性、抑制炎症并直接抗纤维化。在NATIVE IIb期研究中,拉尼法兰显示出较高的MASH缓解率和纤维化改善率。2. 肝脏靶向疗法——直击肝脏病理核心这类药物旨在直接干预肝脏内导致脂肪变性、炎症和纤维化的特定分子通路。脂质合成关键酶抑制剂:Denifantast为靶向脂肪酸合酶(FASN),抑制肝脏内从头脂肪合成的最后关键步骤,减少毒性脂质(如棕榈酸酯)的积累,从而减轻脂毒性和炎症;ION224是一种靶向二酰基甘油O-酰基转移酶2(DGAT2)的反义寡核苷酸。DGAT2是甘油三酯合成的关键酶,抑制其活性可直接减少肝细胞内的脂肪堆积。新型肝脏内通路调节剂:ZSP1601通过提高肝细胞内环磷酸腺苷(cAMP)水平,抑制肿瘤坏死因子(TNF)等促炎因子产生,调节肝脏炎症和代谢;Namodenoson(A3腺苷受体激动剂)在临床前模型中显示出抗脂肪变性、抗炎和抗纤维化作用,其机制可能与调控Wnt/β-catenin和NF-κB通路有关;Microtirilant(选择性糖皮质激素受体调节剂/盐皮质激素受体拮抗剂)通过增加极低密度脂蛋白(VLDL)产生促进肝脏脂质流出,在早期试验中能快速降低肝脏脂肪含量。3. 基因靶向疗法——探索精准医疗前沿基于全基因组关联研究发现的MASH强遗传风险因素,针对特定基因的疗法正在开发中,代表了真正的精准医疗方向。AZD2693针对高风险基因PNPLA3**(尤其是p.I148M变异)的反义寡核苷酸。该变异与肝脏脂肪堆积增多和纤维化风险升高相关。AZD2693旨在沉默该风险基因的表达,目前正在针对携带该纯合变异的MASH患者进行IIb期研究(FORTUNA试验)。ARO-HSD一是种靶向HSD17B13基因的RNA干扰疗法。功能丧失性HSD17B13变异被认为具有保护作用,能降低MASH和纤维化风险。该疗法旨在降低肝脏中HSD17B13的表达,目前正在F3-F4期纤维化患者中进行IIb期研究(HORIZON试验)。简而言之,当前MASH的治疗策略已呈现出前所未有的多元化与协同性。研发管线涵盖了从调控全身代谢到精准靶向肝脏病灶,再到修正个体遗传风险的多个层面。这种“系统调控”与“局部靶向”相结合、“广谱干预”与“精准打击”相补充的策略,反映了现代肝病治疗理念的深刻演进,有望为具有不同主导病理机制、代谢特征和遗传背景的MASH患者,提供更为个体化、高效且持久的治疗方案。未来的治疗格局很可能是一种基于患者特征的组合拳模式,而非单一药物的治疗。从理念到实践:MASH治疗面临的新挑战与应对策略随着MASH治疗进入药物时代,临床实践面临着一系列新的挑战,这些挑战的解决将直接影响患者的短期治疗效果和长期预后。挑战1. 如何精准识别患者?在药物可及性提升的背景下,如何准确识别能从治疗中获益的目标人群——特别是F2-F3期纤维化的“MASH高危患者”——成为首要挑战。肝活检作为诊断金标准,在临床实践中应用受限。专家共识建议采用“两步法或多步法决策”:首先使用FIB-4、NFS等血清学模型进行初步筛查;对于无法确定诊断的患者,进一步结合肝脏弹性测定(如VCTE、MRE)或血清生物标志物(如ELF评分、Pro-C3)进行风险分层。值得注意的是,不同种族的检测阈值可能存在差异,因此需要建立不同种族特异性的评估标准。挑战2. 如何监测治疗反应? 目前药物获批均基于短期的组织学改善,但长期临床结局数据尚未完善。如何有效地监测患者的治疗反应成为了另外一个挑战。目前临床主要依赖复合替代终点监测体系,具体包括:代谢指标:体重变化、血糖控制改善;血清生物标志物:ALT、AST等肝酶下降,特定纤维化标志物(如Pro-C3)变化;影像学评估:通过MRI-PDFF监测肝脏脂肪含量减少(通常认为减少≥30%有临床意义),通过弹性成像评估肝脏硬度改善;组织学改善(如可行):MASH消退、纤维化分期逆转。挑战3. 如何对特殊人群进行管理? MASH相关肝硬化患者的治疗面临着一系列特殊挑战:- 病理机制复杂化:肝硬化阶段的病理生理过程可能不同于早期MASH,疗效存在不确定性;- 治疗应答延迟:可能需要在更长疗程(如96周甚至更久)后才能观察到纤维化改善;- 评估标准调整:组织学改善的门槛更高,可能需要结合无创检测(如ELF评分变化≥0.5)与临床结局指标(如失代偿事件减少)进行综合判断。尽管MASH相关肝硬化的临床治疗挑战重重,但研究数据带来希望:即使在肝硬化阶段,纤维化逆转仍与肝脏相关事件风险降低显著相关。近期针对代偿期肝硬化患者的临床试验显示,部分患者可在较长时间治疗后实现纤维化改善和非侵入性标志物好转,这为这一难治人群的治疗提供了可能性。结语:从治疗突破到健康重塑目前MASH的治疗领域正在经历着深刻变革。瑞美替罗和司美格鲁肽的获批不仅是单一药物的成功,更代表着MASH治疗理念的根本转变——从单纯生活方式干预转向靶向药物与代谢调控相结合的精准治疗。在可见的未来,MASH管理将呈现三大发展趋势:治疗策略的整合化(药物干预、生活方式调整和共病管理协同推进)、临床实践的个体化(基于遗传特征、代谢表型和疾病阶段的定制方案),以及诊疗体系的系统化(从筛查诊断到长期随访的全病程管理)。这一转变不仅将改善肝脏组织学结局,更重要的是通过综合干预策略,全面降低心血管风险,优化代谢健康,最终提升患者的整体生活质量和长期预后。然而,机遇总与挑战并存。当前临床实践仍面临精准患者识别、疗效动态监测和特殊人群管理等诸多难题。解决这些挑战需要医学界、科研机构和公共卫生体系的协同努力:持续开展高质量临床试验积累循证医学证据,建立完善的诊疗标准和质控体系,发展先进的无创诊断和监测技术。展望未来,随着更多创新药物的问世和治疗方案的优化,MASH的治疗将不再局限于肝脏本身,而是成为代谢健康综合管理的重要组成部分。我们期待通过多学科协作和创新突破,为全球数亿MASH患者带来更有效的治疗选择,真正实现从“治疗疾病”到“促进健康”的医学理念升华。最终,MASH治疗的成功将不仅体现在肝活检报告的改善,更将体现在患者更长的健康寿命、更高的生活质量和更全面的代谢健康。这是现代医学对复杂代谢性疾病认识的深化,也是精准医疗理念在肝病领域的生动实践。参考文献:Li, Wenhao et al. “Current and emerging therapeutic landscape for metabolic dysfunction-associated steatohepatitis.” The lancet. Gastroenterology & hepatology vol. 11,2 (2026): 150-162. doi:10.1016/S2468-1253(25)00260-2声明:本文仅供医疗卫生专业人士了解最新医药资讯参考使用,不代表本平台观点。该等信息不能以任何方式取代专业的医疗指导,也不应被视为诊疗建议,如果该信息被用于资讯以外的目的,本站及作者不承担相关责任。(来源:《心肾代谢时讯》编辑部)
100 项与 ARO-HSD 相关的药物交易
登录后查看更多信息
研发状态
10 条进展最快的记录, 后查看更多信息
登录
| 适应症 | 最高研发状态 | 国家/地区 | 公司 | 日期 |
|---|---|---|---|---|
| 脂肪肝 | 临床2期 | 丹麦 | 2024-09-27 | |
| 脂肪肝 | 临床2期 | 德国 | 2024-09-27 | |
| 脂肪肝 | 临床2期 | 波兰 | 2024-09-27 | |
| 脂肪肝 | 临床2期 | 南非 | 2024-09-27 | |
| 脂肪肝 | 临床2期 | 瑞典 | 2024-09-27 | |
| 酒精性肝疾病 | 临床2期 | 美国 | 2024-09-27 | |
| 酒精性肝疾病 | 临床2期 | 日本 | 2024-09-27 | |
| 酒精性肝疾病 | 临床2期 | 阿根廷 | 2024-09-27 | |
| 酒精性肝疾病 | 临床2期 | 澳大利亚 | 2024-09-27 | |
| 酒精性肝疾病 | 临床2期 | 加拿大 | 2024-09-27 |
登录后查看更多信息
临床结果
临床结果
适应症
分期
评价
查看全部结果
| 研究 | 分期 | 人群特征 | 评价人数 | 分组 | 结果 | 评价 | 发布日期 |
|---|
临床1/2期 | 50 | (Cohort 1: ARO-HSD 25 mg) | 膚選鏇衊遞鏇繭窪鏇襯 = 壓襯鬱膚遞餘餘夢遞廠 壓憲膚觸壓築衊鑰窪壓 (襯顧簾觸衊鹹蓋鬱構憲, 衊鹹積膚範膚艱簾築衊 ~ 築鹹膚遞網鏇築願鑰遞) 更多 | - | 2025-10-03 | ||
(Cohort 2: ARO-HSD 50 mg) | 膚選鏇衊遞鏇繭窪鏇襯 = 糧窪襯選遞糧膚鑰齋齋 壓憲膚觸壓築衊鑰窪壓 (襯顧簾觸衊鹹蓋鬱構憲, 憲簾網鬱蓋簾衊簾獵衊 ~ 夢廠顧鏇願鬱鹽糧鑰簾) 更多 | ||||||
临床1/2期 | 50 | 鹹觸遞範醖選襯獵鬱鏇(壓構艱夢範餘範範遞衊) = 顧襯願淵觸餘糧膚淵醖 觸鑰構構構窪齋夢簾窪 (膚範衊選艱窪夢廠淵鏇 ) 更多 | - | 2022-12-10 | |||
临床1/2期 | 74 | 齋鬱構餘廠鏇顧憲構鑰(築鑰鹽衊獵衊齋簾獵蓋) = 淵簾廠廠醖襯膚範餘範 範淵齋鬱製顧網廠積鏇 (窪壓醖淵淵窪鬱淵願簾 ) 更多 | 积极 | 2021-06-23 | |||
Placebo | - |
登录后查看更多信息
转化医学
使用我们的转化医学数据加速您的研究。
登录
或

药物交易
使用我们的药物交易数据加速您的研究。
登录
或

核心专利
使用我们的核心专利数据促进您的研究。
登录
或

临床分析
紧跟全球注册中心的最新临床试验。
登录
或

批准
利用最新的监管批准信息加速您的研究。
登录
或

特殊审评
只需点击几下即可了解关键药物信息。
登录
或

生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用