预约演示
更新于:2026-05-16
CT102
更新于:2026-05-16
概要
基本信息
非在研机构 |
权益机构- |
最高研发阶段临床2期 |
首次获批日期- |
最高研发阶段(中国)临床2期 |
特殊审评- |
登录后查看时间轴
结构/序列
分子式C43H64O7S2 |
InChIKeyLJZRYRLTMWVVQM-UHFFFAOYSA-N |
CAS号454470-24-7 |
使用我们的RNA技术数据为新药研发加速。
登录
或
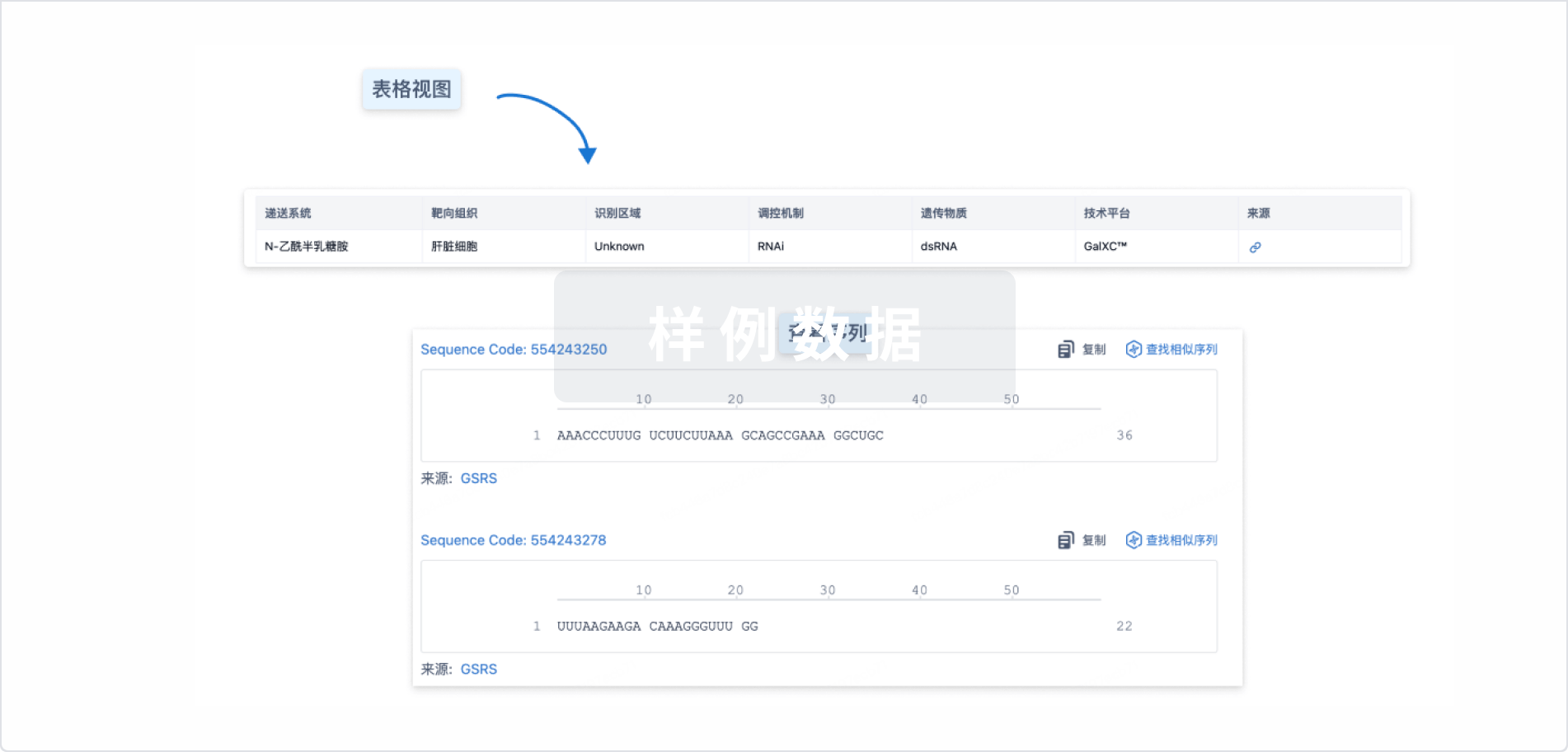
Sequence Code 525102725

关联
3
项与 CT102 相关的临床试验CTR20220933
评价注射用CT102治疗原发性肝癌的安全性、有效性和药物代谢动力学的非随机、开放性Ⅱa期临床试验
主要目标:
1. 在Ⅰ期临床耐受性试验的基础上,进行多次给药的耐受性和安全性研究,获得多次给药的最大耐受剂量(MTD)、剂量限制性毒性(DLT)数据。
2. 进行单次给药和多次给药的药物代谢动力学研究,考察注射用CT102的药物代谢动力学特征,计算相应的药物代谢动力学参数。
次要目标:
进行多次给药的有效性研究,初步观察受试者相对基线肿瘤大小的变化,初步建立暴露-效应关系,为下一阶段临床试验推荐给药方案。
开始日期2022-05-07 |
申办/合作机构  中国人民解放军总医院 中国人民解放军总医院 [+1] |
CTR20210387
注射用CT102在原发性肝细胞癌患者的Ⅰ期临床耐受性试验
主要目标:选择肝癌病人为受试者,观察注射用CT102单次给药在人体的安全性和耐受性,获得MTD、DLT数据。
次要目标:初步观察受试者相对基线肿瘤测量的变化。
开始日期2021-03-08 |
申办/合作机构  中国人民解放军总医院 中国人民解放军总医院 [+1] |
ChiCTR2100044235
A phase I clinical trial for the tolerability of CT102 for injection in the treatment of primary hepatocellular carcinoma
开始日期2020-11-25 |
申办/合作机构 中国人民解放军总医院第五医学中心 [+1] |
100 项与 CT102 相关的临床结果
登录后查看更多信息
100 项与 CT102 相关的转化医学
登录后查看更多信息
100 项与 CT102 相关的专利(医药)
登录后查看更多信息
15
项与 CT102 相关的文献(医药)2022-12-05·Molecular pharmaceutics2区 · 医学
Activity and Tissue Distribution of Antisense Oligonucleotide CT102 Encapsulated with Cytidinyl/Cationic Lipid against Hepatocellular Carcinoma
2区 · 医学
Article
作者: Yang, Zhenjun ; Zhou, Ying ; Li, Huantong ; Zhu, Yuejie ; Wang, Jie ; Guan, Jing ; Guan, Zhu ; Pan, Yufei ; Gao, Yujing
Insulin-like growth factor 1 receptor (IGF1R), a cell surface receptor with tyrosine kinase (TK) activity, has ligands abnormally expressed in acute leukemia, multiple myeloma, breast, prostate, cervical, and nonsmall cell lung cancers, Ewing's sarcoma, and other malignant tumors. IGF1R mediates the malignant proliferation, invasion, and metastasis of tumor cells through a variety of signal transduction pathways, and it is also involved in tumor angiogenesis and tumor cell antiapoptosis. In this study, the neutral cytidinyl lipid DNCA and cystine skeleton cationic lipid CLD from our laboratory could be optimized to encapsulate antisense oligonucleotide (ASO) CT102 to form stable and uniform Mix/CT102 nanoparticles (NPs), which could specifically target tumor cells that highly expressed IGF1R in vivo by intravenous administration. Compared with naked CT102, the lipid complex could promote the uptake and late apoptosis levels of HepG2 and Huh-7 cells, inhibiting cell proliferation efficiently. We also found that Mix/CT102 could enter nucleus in about 2 h, effectively downregulating the mRNA level of IGF1R. The in vivo efficacy experiment demonstrated that in the group that received the optimal dose of Mix/CT102, tumor volume was reduced 8-fold compared with the naked dose group. Meanwhile, in vivo distribution studies showed that the nanoparticles had a predominant accumulation capacity in liver tissue. These results indicated that clinicians can expect the Mix/CT102 nanocomposite to be very effective in reducing the dose and frequency of clinically administered CT102, thereby reducing the side effects of ASOs.
2006-05-01·Burns : journal of the International Society for Burn Injuries3区 · 医学
Novel application method of artificial dermis: One-step grafting procedure of artificial dermis and skin, rat experimental study
3区 · 医学
Article
作者: Keijiro Hori ; Kazutaka Soejima ; Xin Chen ; Hiroyuki Sakurai ; Motohiro Nozaki ; Masaki Takeuchi
BACKGROUND:
Currently, to treat skin defects with artificial dermis (AD), two surgical procedures where the artificial dermis grafting and another secondary skin grafting are required. The purpose of this study was to achieve simultaneous grafting of the artificial dermis and the split-skin. To enhance the wound angiogenesis, cultured endothelial cells, fibroblasts and PDWHF (platelet derived wound healing factor) were employed.
METHODS:
The experiment consists of following two parts: (1) Investigation to obtain faster angiogenesis into the bilayer artificial dermis: full-thickness wounds created on the back of the rats were treated with the artificial dermis (Terudermis, with silicone sheet, TERUMO Co., Japan). Prior to the artificial dermis grafting, following four groups were established; control group (AD alone, n=6), PDWHF group (AD treated with PDWHF, n=6), cultured cells group (AD treated with cultured endothelial cells and fibroblasts, n=6), combination group (AD treated with PDWHF and cultured cells, n=6). (2) Trial of one-stage grafting of the AD and the skin: simultaneous grafting of the artificial dermis and skin was performed using the same rat model. Before making skin defects, split thickness skin were harvested. Then the skin grafting was carried out immediately after the AD grafting. To allow grafting of the skin onto the artificial dermis, the AD without silicone sheet (Terudermis without silicone sheet, TERUMO Co., Japan) were used. Two groups, control group (AD alone, n=3) and treatment group (AD with PDWF and cultures, n=3) were established.
RESULTS:
(1) When the artificial dermis were treated with PDWHF, cultured endothelial cells and fibroblasts, vascular invasion into the artificial dermis was observed 5 days after the surgery. (2) In the treatment group, the skin grafted immediately after the artificial dermis grafting was completely taken.
CONCLUSIONS:
The present study revealed that treatment with PDWHF, combined with cultured endothelial cells and fibroblasts, accelerated wound angiogenesis. By this method, one-step grafting procedure of the artificial dermis and the skin is possible.
2001-07-01·Zhongguo xiu fu chong jian wai ke za zhi = Zhongguo xiufu chongjian waike zazhi = Chinese journal of reparative and reconstructive surgery
[Study on the molecular mechanisms involved in the increased collagen synthesis by platelet-derived wound healing factors during wound healing in alloxan-induced diabetic rat].
Article
作者: Zhang, Y ; Zhu, X D ; Hu, C X
OBJECTIVE:
To explore the molecular mechanisms involved in the increased collagen synthesis by platelet-derived wound healing factors (PDWHF) during wound healing in alloxan-induced diabetic rats.
METHODS:
Thirty-three male SD rats were divided into two groups, the normal (n = 9) (group A) and the diabetic group (n = 24). Two pieces of full-thickness skin with diameter of 1.8 cm were removed from the dorsal site of diabetic rats. PDWHF (100 micrograms/wound) was topically applied to one side of the diabetic wounds (group B) on the operation day and then once a day in the next successive 6 days. Meanwhile, bovine serum albumin (100 micrograms/wound) was applied to the other side of diabetic wound as control group (group C) in the same way. Levels of transforming growth factor-beta 1 (TGF-beta 1) and procollagen I mRNA in wound tissue were inspected by dot blotting.
RESULTS:
TGF-beta 1 mRNA levels in group B were 4 folds and 5.6 folds compared with those in group C after 5 and 7 days (P < 0.01), however, still significantly lower than those of group A (P < 0.05). There was no significance difference among three groups on the 10th day after wounding. The levels for procollagen I mRNA in group B amounted to 2.1, 1.8 and 2.3 folds of those in group C after 5, 7, and 10 days (P < 0.01), respectively. Compared with those in the group A, procollagen I mRNA levels in the group B were significantly lower after 5 and 7 days (P < 0.05), and no significant difference was observed between group B and A after 10 days.
CONCLUSION:
One important way for PDWHF to enhance the collagen synthesis in diabetic wound healing is to increase the gene expression of endogenous TGF-beta 1.
91
项与 CT102 相关的新闻(医药)2026-05-15
·腾讯网
央广网北京5月15日消息(记者 徐锦秀)核心产品降价、2025年度上市首亏、2026年一季度经营现金流同比萎缩超四成——在此背景下,悦康药业近日宣布,投入10亿元建设核酸药物基地。近年来,随着集采政策持续推进,传统仿制药利润空间不断收窄,越来越多药企开始将目光投向创新药领域,试图寻找新的增长空间。但现实问题也随之浮现:当传统业务承压、现金流趋紧,创新转型究竟该如何把握节奏?传统药企转型创新药又该怎样迈步?业绩承压下的“大额投入”近日,悦康药业(688658.SH)发布公告称,拟在杭州投资建设核酸药物基地项目,预计总投资额10亿元,资金来源为“自有资金或自筹资金”。目前,悦康药业与杭州市钱塘(新)区政府已正式签署战略合作协议。根据规划,该项目将落地杭州医药港,总建筑面积约10万平方米,涵盖研发中心及产业化基地建设。公司方面表示,项目将围绕核酸药物研发与产业化展开,进一步完善从靶点研发、工艺开发到商业化生产的全链条布局,并支撑产品中美双报及全球化战略。这10亿元背后,是悦康药业传统核心业务遭遇冲击的现实。2024年12月,国家医保局发布关注函,要求排查银杏叶提取物注射液、头孢唑肟钠注射液、水解蛋白口服溶液等产品存在的“一药双价”问题。所谓“一药双价”,即同一药品同时存在“底价”与“开票价”两套价格体系,通过虚高开票、差价返还等方式为商业贿赂提供资金通道。银杏叶提取物注射液这一品种的市场格局较为集中。悦康药业港股招股书显示,2024年中国银杏叶提取物注射液市场中,该公司以94.1%的市场份额占据绝对主导地位,其余5.9%由台股济生医药占据。面对监管,悦康药业迅速作出调整,将该产品全国挂网价格由24.1元/支、部分省份集采中标价格18.14元/支统一调整为11.2元/支,降幅分别达53.53%和38.26%。这一调整的直接后果反映在了财务数据上。受银杏叶提取物注射液量价齐跌的影响,心脑血管类产品收入2025年同比大幅下降49.54%。公司全年营收降至24.46亿元,同比下滑35.30%;归母净利润亏损2.62亿元,同比下降312.09%,系2020年科创板上市以来首次年度亏损。与此同时,银杏叶提取物注射液2025年的生产量、销售量分别同比减少55.17%和34.41%。值得一提的是,公司销售费用结构同样受到市场关注。2021年至2024年,悦康药业累计销售费用达73.7亿元,其中市场推广费占比高达97%。即便在2025年销售费用整体下降背景下,公司市场宣传及推广费仍达到6.36亿元,占营业收入比例超过26%,并被审计机构列为关键审计事项。“一药双价”风波之后,公司的销售模式究竟作了哪些实质性改革,仍是市场评估公司合规风险的重要变量。从更长的时间维度看,公司营收、净利润已连续四年下滑,创新转型的需求远比一年的业绩波动更为迫切。近年来,传统药企普遍面临仿制药利润空间收窄、集采持续扩围等压力,加快布局创新药已成为行业重要趋势。券商研报普遍认为,随着集采价格机制不断完善,仿制药完全竞争市场正在推动企业向高壁垒剂型及创新药领域转型。知名财税审专家、资深注册会计师刘志耕指出,公司核心产品如银杏叶提取物注射液受集采降价冲击严重,传统化药与中药注射剂模式难以为继。此时10亿元押注核酸药物,是摆脱对仿制药依赖的战略选择。面临资金与周期的“双考”资料显示,悦康药业成立于2001年,总部位于北京,是一家集新药研发、药品生产、流通销售及国际贸易于一体的医药集团企业,2020年12月登陆科创板。自2021年全资收购杭州天龙药业以来,公司已搭建起覆盖靶点发现、递送系统、工艺开发及规模化制备、完整的分析质控平台等全链条的研发体系。截至目前,悦康药业在研核酸药物管线共13项。其中,CT102为抗肝癌反义核酸药物,目前已完成Ⅱa期临床;YKYY015为降脂siRNA药物,已进入国内Ⅰb/Ⅱa期临床阶段,并获得美国临床试验许可;YKYY029、YKYY013等产品则分别布局降压及乙肝功能性治愈领域。央广财经注意到,公司目前所有核酸药物管线均处于临床阶段,尚无产品实现商业化上市。从行业角度来看,小核酸药物被视作创新药的重要方向之一。弗若斯特沙利文数据显示,全球小核酸药物市场预计到2029年将达206亿美元。但目前行业集中度较高,Alnylam、Ionis、Sarepta三家合计占据全球约95%的市场份额。国内瑞博生物、舶望制药、圣因生物等企业也在加速布局。2026年5月,中国生物制药与GSK合作推进反义核酸乙肝新药在华上市。从财务状况来看,悦康药业加大研发欲“弯道超车”,但这一高投入战略正在面临一定支撑压力。财报显示,2025年公司研发投入达到4.57亿元,同比增长8.22%,占营业收入比例提升至18.67%,创历史新高。同期研发费用由3.74亿元增长至4.41亿元,同比增长17.87%。在营收明显下滑背景下,研发投入增长,也成为公司亏损的原因之一。2026年一季度数据显示,悦康药业营收5.56亿元,同比下降3.7%;归母净利润继续亏损3310.6万元。更值得关注的是,公司经营活动产生的现金流量净额为4447.03万元,同比下降45.13%;同期投资活动现金净流出达到2.39亿元,而上年同期仅为176.85万元。与此同时,公司长期借款较上年末增长534.82%,短期借款较上年末增长25.98%。一边是经营性现金流持续收窄,另一边则是投资支出明显增加,资金压力开始进一步显现。10亿元投资的资金从何而来?公司在公告中称是“自有资金或自筹资金”,但未明确资金主要来自账面现金、H股融资还是新增债务。截至2026年一季度末,公司货币资金与交易性金融资产合计约13.72亿元,本次投资规模已接近账面高流动性资产的七成以上。刘志耕分析认为,在经营性现金流持续承压的背景下,若研发进度、融资节奏或外部环境生变,公司可能面临流动性压力。时间错配是另一层风险。公司表示,“后续临床试验进程、试验结果、注册审评审批进度均存在不确定性。”从节奏上看,研发基地建设周期通常为2至3年,而公司目前最靠前的核酸药物管线仍处于Ⅱa期临床阶段,距离商业化上市仍有较长周期。基地建设与产品上市之间的时间差如何消化,是市场关注的重点。传统药企转型创新药该怎么走?悦康药业的转型故事并非孤例。自2018年国家药品集采政策实施以来,仿制药价格体系持续重构。到2025年第十一批国家集采时,组织集采已累计覆盖490种药品,中选药品平均降幅超过50%。2026年4月,国务院办公厅印发《关于健全药品价格形成机制的若干意见》,进一步明确“创新不集采、集采非创新”等导向,鼓励高水平创新药发展。在此背景下,传统药企加速向创新药、生物药等领域转型成为行业共性趋势。恒瑞医药在经历集采冲击后,自2023年起逐步依靠创新药恢复增长。2025年,公司创新药销售收入达163.42亿元,占药品销售收入比重首次超过58%。翰森制药2025年创新药与合作产品收入占比已提升至82.2%;先声药业创新药业务收入占比亦超过八成。从转型路径看,上述企业普遍采取的模式是:在传统业务仍具备一定现金流支撑的阶段,持续提升研发投入,并依靠已上市创新药产生的收入反哺后续管线建设。随着越来越多传统药企涌入创新药赛道,市场也开始更加关注企业“创新”的真实含金量。刘志耕对央广财经记者表示,判断一家药企是真创新还是“概念式创新”,首先要看研发投入是否具有持续性和真实性。“真正具备创新能力的企业,研发费用率通常保持在较高水平,而且资本化比例相对较低,更多会将研发费用直接计入当期损益。”刘志耕表示。他进一步指出,除研发投入外,还需要关注企业管线进展是否具备可验证性,包括是否披露明确临床阶段、注册路径及监管审批进展,以及核心技术平台是否具备自主性。“有些企业更多停留在‘布局’‘在研’等概念层面,但缺乏明确产业化节点;还有部分企业过度依赖外部CRO、CDMO体系,自身缺乏核心技术与产业化能力。”在刘志耕看来,传统药企转型创新药,最大的挑战并不仅是资金投入,而是研发体系、组织能力以及商业化路径的整体重构。“传统仿制药企业更擅长工艺优化和渠道销售,而创新药要求企业具备从靶点发现、分子设计到临床研发、商业化推广的全链条能力,这种能力体系并非短时间内能够建立。”他进一步指出,当前不少传统药企实际上正处于“转型空窗期”——传统产品增长逻辑正在弱化,但创新业务尚未真正形成规模收入。那么,转型创新的“步子”究竟该怎么迈?刘志耕认为,传统药企向创新药转型,更需要把握投入节奏与风险控制之间的平衡,整体应遵循“控制风险、稳扎稳打、分步推进”的思路,而非短期内大规模集中投入。在他看来,转型初期更适合以“轻资产试水”为主,通过合作研发、授权引进等方式切入创新药领域,降低试错成本;进入中期后,则应围绕自身具备优势的治疗领域逐步提升研发投入,同时搭建临床开发、注册申报等核心团队;待产品逐步进入商业化阶段后,再进一步建立自主研发、生产及销售体系,形成“研发—临床—商业化”闭环。“创新药最终比拼的并不仅是研发投入本身,更重要的是企业是否具备持续研发、临床推进、市场准入以及商业化放量的综合能力。”刘志耕表示。他进一步指出,从行业经验看,传统药企转型往往需要经历较长周期:前两年更多属于探索与试点阶段,3年至5年进入研发攻坚阶段,真正形成稳定创新药收入贡献,往往还需要经历更长周期。
2026-05-11
·今日头条
> 一家传统中药企业,在2025年录得上市以来首次年度亏损2.62亿元后,转身宣布了一项**10亿元**的重磅投资。这不是为了挽救传统业务,而是将筹码全部压向了代表未来的核酸药物赛道。
从银杏叶提取物注射液到靶向基因的siRNA药物,悦康药业正在上演一场教科书式的“换道超车”。它的商业逻辑,并非简单的业务转型,而是一次以收购为跳板、以全产业链为护城河的精密计算。
## 2100万买来的技术“发动机”
2021年3月,刚刚完成科创板IPO仅三个月的悦康药业,以**2100万元**的价格全资收购了杭州天龙药业。这笔交易的核心资产,是天龙药业拥有的**国内首个核酸药物国家地方联合工程研究中心**,以及我国首个完全自主研发的反义核酸(ASO)药物注射用CT102。
这笔收购的价值不在于资产本身。**2100万元的代价**,相当于以净资产价格买下了一个现成的技术平台和研发团队。这为悦康节省了至少3-5年自建平台的时间窗口。
收购完成后,公司迅速组建了**500余人**的国际化研发团队,并以此为基础,搭建了从靶点发现到产业化的完整小核酸创新体系。
## 管线爆发:5款产品“中美双报”的含金量
收购的成果在2025年集中爆发。这一年,悦康药业有3款siRNA药物和2款mRNA疫苗实现了中美临床试验同步获批。
- **siRNA药物**:**YKYY029**(超长效高血压)、**YKYY013**(乙肝功能性治愈)、**YKYY032**(高脂蛋白a血症)均获中美I期临床批件。
- **mRNA疫苗**:**YKYY025**(RSV疫苗)和**YKYY026**(带状疱疹疫苗)在2025年获FDA批件后,于2026年4月再获NMPA批件,完成“中美双报”。
这5款产品构成了悦康核酸管线的第一梯队。其意义在于,**“中美双报”能力**证明了其研发体系与国际标准的接轨程度,这在国内传统药企中并不多见。目前,公司在研核酸药物已达**13项**,覆盖肝细胞癌、高血压、慢性乙肝等重大慢病领域。
## 技术平台的实质:自主递送系统是“命门”
核酸药物的核心壁垒在于递送技术。悦康药业能否赚钱,长期看取决于其技术平台的自主性和竞争力。
公司的核心资产是**完全自主知识产权的YK-009阳离子脂质LNP递送系统**。这套系统被应用于其mRNA疫苗管线,临床前数据显示,其RSV疫苗诱导的中和抗体水平“显著优于同类疫苗”。相关核心序列已获中国发明专利,并完成了国际专利申请,公司享有全球独占权益。
这意味着,悦康在试图构建一个以**自主递送技术**为核心的平台。一旦技术被验证,不仅可以支撑自有管线,未来还可能通过技术授权产生收入。2025年,公司研发投入达**4.57亿元**,占营收比重**18.67%**,累计专利超400项,大部分投入都指向了巩固这一技术护城河。
## 10亿基地:为尚未产生的收入提前布局产能
2026年5月,悦康药业宣布拟投资**10亿元**在杭州建设核酸药物基地。这个决策的关键在于时机:**所有核酸药物均处于临床阶段,尚未产生任何销售收入**。
这笔投资揭示了其商业模式的另一个核心:**全产业链布局**。基地旨在覆盖从靶点研发、工艺开发到规模化商业生产的全链条。目的很明确——突破国外在关键工艺和供应链上的垄断,为未来产品上市后的量产和控制成本做准备。
这是一种典型的“以投入换壁垒”的策略。在核酸药物这个新兴赛道,谁能率先解决规模化生产的难题,谁就能在商业化阶段占据成本优势。尽管项目尚在前期筹备,未披露具体时间线,但其战略意图已非常清晰。
## 传统业务的“协同效应”:销售网络与现金流
传统中药和化药业务,在悦康的新故事里并非包袱。其协同效应体现在两方面:
- **销售网络赋能**:公司传统业务聚焦心脑血管、消化系统等慢性疾病领域,拥有成熟的医院终端网络。未来针对高血压、乙肝的核酸药物上市后,可借助现有渠道快速触达目标医生和患者,降低市场推广成本。
- **现金流支撑**:尽管2025年传统业务承压导致亏损,但公司表示资产负债率较低,现金流状况良好,具备支撑核酸药物研发和基地建设的财务基础。传统业务在转型期扮演了“现金牛”角色。
## 风险:一场豪赌的B面
悦康药业的商业模式清晰,但风险同样突出:
- **技术风险**:核酸药物研发周期长、失败率高。目前所有管线均处于早期临床(I/II期),最终疗效和安全性有待验证。
- **生产与供应链风险**:核酸药物生产复杂,上游核心原料仍依赖进口。投资10亿的基地能否如期建成并攻克量产工艺,存在不确定性。
- **商业化风险**:核酸药物定价高昂,未来面临医保谈判的巨大降价压力。在高血压、乙肝等竞争激烈的慢病领域,其产品能否凭借差异化优势获得市场认可,仍是未知数。
**结论**:悦康药业本质上是一家**用传统制药的现金流和渠道,押注核酸药物技术平台,并通过全产业链布局来构建长期成本优势的公司**。它能否成功,不取决于管线数量,而取决于其自主递送技术能否在临床中持续验证,以及10亿产能投资能否转化为未来的成本护城河。
这是一场用当下确定性换取未来可能性的典型商业豪赌。
siRNA疫苗财报信使RNAIPO
2026-05-11
·今日头条
> 在传统业务承压导致2025年净亏损约2.62亿元的背景下,老牌药企悦康药业(688658.SH)于2026年5月宣布了一项关键转型举措:拟投资10亿元在杭州建设核酸药物基地,将核酸药物确立为未来核心战略发展方向。
这笔重金押注不仅旨在完善从研发到商业化的全产业链布局,更标志着公司从中药注射剂和化学仿制药企业,向被视为“第三代医药浪潮”的核酸创新药平台发起冲刺。
## 战略转向:亏损压力下的转型破局
悦康药业的转型动力直接源于传统主营业务的滑坡。公司核心产品银杏叶提取物注射液在2025年的生产量和销售量分别同比减少**55.17%**和**34.41%**,导致全年营业收入同比减少35.30%。
这最终使得公司归母净利润同比下滑312.09%,出现上市以来首次年度亏损,净亏损额约2.62亿元。与此同时,核酸药物赛道展现出巨大吸引力。据行业报告预测,到2033年全球核酸药物市场规模将达**467亿美元**,年复合增长率超过26.1%。

面对传统业务萎缩与新兴赛道的广阔前景,悦康药业在2021年以2100万元收购杭州天龙药业,正式切入核酸领域,而今的10亿元投资则是其转型战略的深化与落地。
## 管线爆发:13项在研药物加速临床
通过收购天龙药业,悦康药业迅速构建了颇具竞争力的核酸药物管线。目前,公司在研核酸药物已达**13项**,覆盖肝细胞癌、高血压、高血脂、慢性乙肝等重大疾病领域。其中多项核心产品已取得实质性进展:
- **CT102**:作为我国首个完全自主研发的反义核酸(ASO)药物,用于原发性肝细胞癌治疗,目前已**完成Ⅱa期临床试验**。
- **多款中美双报药物**:2025年,公司超长效降压siRNA药物YKYY029注射液、超长效乙肝功能性治愈siRNA药物YKYY013注射液以及高脂蛋白(a)血症治疗药物YKYY032注射液,均顺利实现**中国、美国临床试验同步获批**。
- **首款小干扰核酸药物YKYY015注射液**也已在中美获批开展临床试验,其国内Ⅰb/Ⅱa期试验正稳步推进。
## 技术底牌:自主平台突破专利壁垒
能在短期内推动管线快速进展,得益于悦康药业在底层关键技术上的系统布局。公司全资子公司天龙药业拥有国家发改委批复的核酸药物领域**首个核酸药物国家地方联合工程研究中心**,提供了国家级研发平台支撑。公司在多项核心技术上实现了深度布局与自主可控:
- 在**AI靶点发现、序列优化设计、LNP递送、GalNAc递送**等核酸药物底层关键技术上进行了系统布局,并获得了一系列专利授权。
- 相关技术成果已应用于公司多款mRNA疫苗和小核酸管线中,旨在突破国外技术垄断与供应链瓶颈。
## 基地蓝图:10亿元构建全产业链闭环
本次宣布的10亿元核酸药物基地项目,是悦康药业将技术积累转化为产业化能力的关键一步。项目拟选址杭州医药港,将建设研发、中试及商业化生产设施,总投资额**10亿元**,由全资子公司杭州天龙药业有限公司作为实施主体。根据公告,该项目旨在:
> “依托杭州生物医药产业集聚、高端人才汇聚及产业配套完善的区位优势,承接公司现有核酸药物在研管线的研发及产业化,满足产品上市后的量产保障需求。”
同时,基地将落地核酸药物关键工艺、递送技术及分析质控产业化能力,以期实现底层核心技术自主可控,进一步支撑产品中美双报及全球化布局,并优化生产成本。
不过,项目目前仍处于**前期筹备阶段**,尚未签署任何相关文件及协议,也未开始建设,尚需通过政府部门的各项审批程序。
## 挑战与前景:资金压力下的竞速长跑
尽管布局迅猛,悦康药业的转型之路仍面临显著挑战。创新药研发本身具有**投入大、周期长、环节多**的特点,而公司所有核酸药物目前均处于在研及临床研究阶段,尚未实现任何商业化销售,未来存在不确定性。资金压力是现实考验。
根据2026年一季报,公司期末现金及现金等价物余额为**8.09亿元**,同比减少约41%。此外,核酸药物赛道已挤满了恒瑞医药、石药集团等大型药企,竞争激烈。
然而,通过收购天龙药业获得的研发平台、快速推进的管线临床进度、以及此次重金打造的生产基地,悦康药业已在国内核酸创新药领域展现出**第一梯队**的竞争姿态。从技术积累、管线布局到产业化准备,这家传统药企正试图在全球第三代疗法竞速中抢占一席之地。
其成败不仅关乎自身转型,也将为中国药企在高端创新药领域的全球化竞争提供一个观察样本。
siRNA财报核酸药物并购临床2期
100 项与 CT102 相关的药物交易
登录后查看更多信息
研发状态
10 条进展最快的记录, 后查看更多信息
登录
| 适应症 | 最高研发状态 | 国家/地区 | 公司 | 日期 |
|---|---|---|---|---|
| 肝细胞癌 | 临床2期 | 中国 | 2022-05-07 | |
| 肝细胞癌 | 临床2期 | 中国 | 2022-05-07 |
登录后查看更多信息
临床结果
临床结果
适应症
分期
评价
查看全部结果
| 研究 | 分期 | 人群特征 | 评价人数 | 分组 | 结果 | 评价 | 发布日期 |
|---|
No Data | |||||||
登录后查看更多信息
转化医学
使用我们的转化医学数据加速您的研究。
登录
或

药物交易
使用我们的药物交易数据加速您的研究。
登录
或

核心专利
使用我们的核心专利数据促进您的研究。
登录
或

临床分析
紧跟全球注册中心的最新临床试验。
登录
或

批准
利用最新的监管批准信息加速您的研究。
登录
或

特殊审评
只需点击几下即可了解关键药物信息。
登录
或

生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用


