预约演示
更新于:2026-05-16
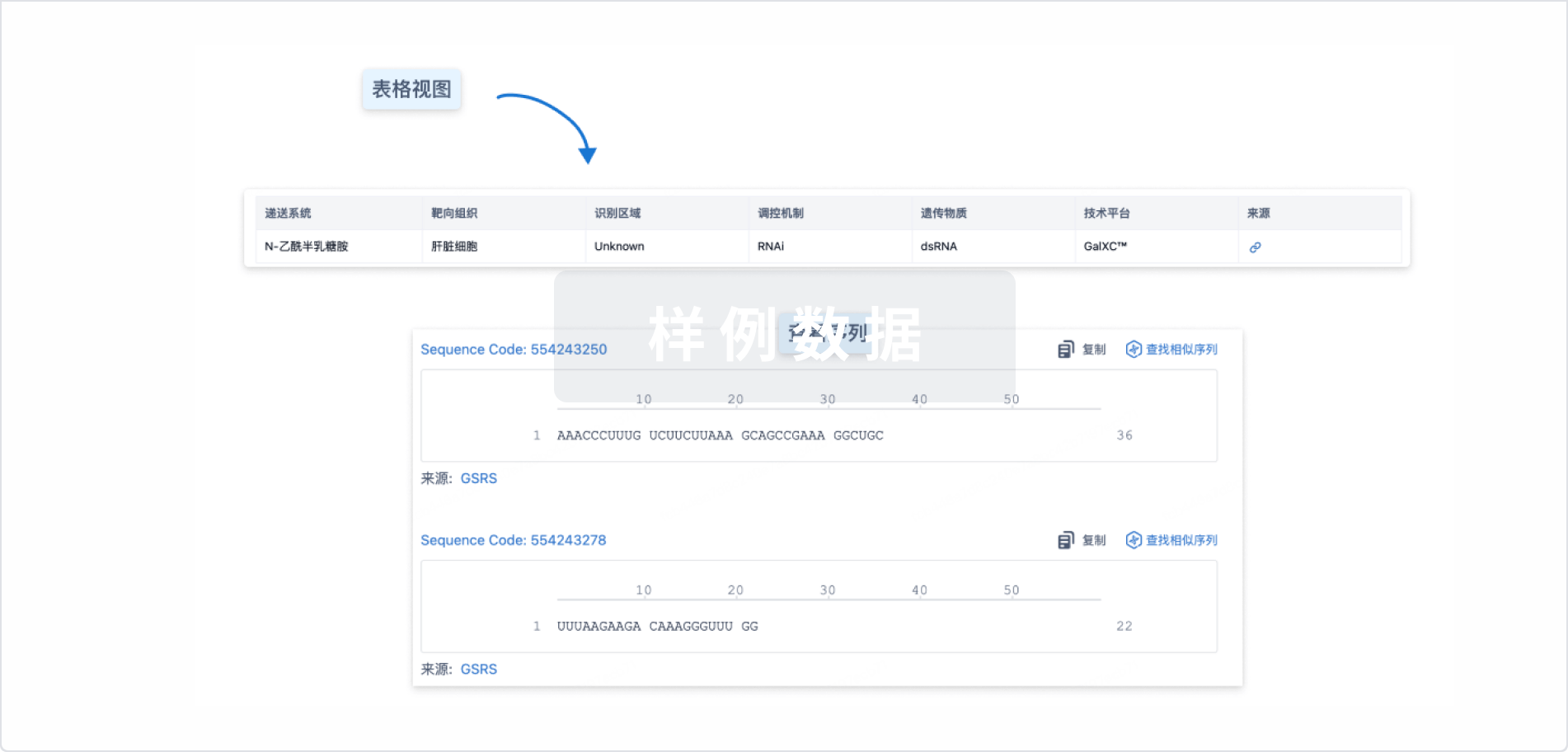
Teprasiran
更新于:2026-05-16
概要
基本信息
在研机构- |
最高研发阶段终止临床3期 |
首次获批日期- |
最高研发阶段(中国)- |
特殊审评孤儿药 (美国) |
登录后查看时间轴
结构/序列
使用我们的RNA技术数据为新药研发加速。
登录
或
Sequence Code 29494323

来源: *****
Sequence Code 29494330

来源: *****
关联
7
项与 Teprasiran 相关的临床试验NCT03510897
A Randomized, Double-Blind, Placebo Controlled, Phase 3 Study to Evaluate the Efficacy and Safety of QPI-1002 for the Prevention of Major Adverse Kidney Events (MAKE) in Subjects at High Risk for Acute Kidney Injury (AKI) Following Cardiac Surgery
This trial is designed to evaluate QPI-1002 versus placebo for the prevention of Major Adverse Kidney Events (MAKE) in subjects at high risk for acute kidney injury following cardiac surgery. Half of the participants will receive QPI-1002 while the other half will receive placebo.
开始日期2018-06-29 |
申办/合作机构 |
NCT02610296
A Phase 3, Randomized, Double-blind, Placebo Controlled Study to Evaluate the Efficacy and Safety of QPI-1002 for Prevention of Delayed Graft Function in Recipients of a Donation After Brain Death Older Donor Kidney Transplant
The purpose of this trial is to evaluate the reduction in incidence and severity of delayed graft function with kidney allografts from donors >45 years after brain death (DBD).
开始日期2016-03-01 |
申办/合作机构 |
NCT02610283
A Randomized, Double-Blind, Placebo Controlled , Phase 2 Study to Evaluate the Efficacy and Safety of QPI-1002 for the Prevention of Acute Kidney Injury in Subjects at High Risk for AKI Following Cardiac Surgery
This trial is designed to evaluate QPI-1002 versus placebo for the prevention of AKI in subjects who are at high risk for AKI following cardiac surgery. Half of the participants will receive QPI-1002 while the other half will receive placebo.
开始日期2015-12-01 |
申办/合作机构 |
100 项与 Teprasiran 相关的临床结果
登录后查看更多信息
100 项与 Teprasiran 相关的转化医学
登录后查看更多信息
100 项与 Teprasiran 相关的专利(医药)
登录后查看更多信息
7
项与 Teprasiran 相关的文献(医药)2025-08-01·CLINICAL TRANSPLANTATION
Emerging Therapeutic Strategies for Renal Ischemia‐Reperfusion Injury in Kidney Transplantation: Progress and Challenges—A Systematic Review
Review
作者: Cogua, Laura M. ; Zuluaga, Dayanna ; Ratner, Lloyd E. ; Wang, Harvey ; Khan, Sohail ; Ortiz, Jorge
ABSTRACT:
Introduction:
Ischemia‐reperfusion injury (IRI) contributes to delayed graft function (DGF) and long‐term allograft loss in kidney transplantation (KTx). Despite decades of investigation, no pharmacologic strategy has achieved clinical translation. This systematic review evaluates investigational therapies for renal IRI (RIRI) reported since 2000.
Methods:
Following the PRISMA 2020 guidelines, a systematic search of PubMed, ClinicalTrials.gov, and the European Union Clinical Trials Register (EUCTR) was conducted for articles published between January 1, 2000, and March 31, 2025. Eligible records included preclinical and clinical investigations evaluating pharmacologic interventions for RIRI in KTx. Narrative reviews, pediatric‐only, in silico‐only, and in vitro‐only records were excluded. A total of 76 full‐text reports were assessed for eligibility, with 43 meeting the inclusion criteria. Meta‐analysis was not performed due to heterogeneity. Included reports were synthesized by the mechanism of action, developmental stage, and translational status. Risk of bias was qualitatively assessed. This review was not prospectively registered, no formal protocol was prepared, and no external funding was received.
Results:
Legacy agents, Diannexin, YSPSL, and I5NP demonstrated initial promise but failed to achieve late‐phase clinical efficacy. More recent agents, particularly alkaline phosphatase, complement‐targeting biologics, and mesenchymal stem cell (MSC)‐derived exosomes, offer mechanistically diverse strategies to attenuate RIRI, though most remain in early development.
Conclusion:
Most available data remain preclinical and are limited by inconsistent outcome measures and translational bottlenecks. Future efforts should prioritize harmonized animal models, biomarker‐defined endpoints, and strategic investment in promising candidates to integrate them into kidney transplant care.
2025-06-01·Anesthesiology clinics
Cardiac Surgery-Associated Acute Kidney Injury
Review
作者: Jacob, Kirolos A ; Leaf, David E
Acute kidney injury (AKI) is a frequent and often severe postoperative complication following cardiac surgery, which is associated with poor outcomes in both the short and long terms. Numerous randomized clinical trials have been conducted to investigate various strategies for prevention of cardiac surgery-associated AKI. Unfortunately, most trials conducted to date have been negative. However, encouraging results have been reported with several interventions, including preoperative implementation of oxygen delivery-directed perfusion, novel drugs such as teprasiran and amino acids. Many of these studies, however, require validation in larger, multicenter trials, before their routine use in clinical practice can be recommended.
2021-10-05·Circulation1区 · 医学
Teprasiran, a Small Interfering RNA, for the Prevention of Acute Kidney Injury in High-Risk Patients Undergoing Cardiac Surgery: A Randomized Clinical Study
1区 · 医学
Article
作者: Lukas J. Lehner ; Shai Erlich ; Bruce Molitoris ; David Corteville ; Gabor Szabo ; Craig D. Brown ; Nicolas Noiseux ; Andre Lamy ; Matthias Thielmann ; Elizabeth C. Squiers ; Madhav Swaminathan ; Mohamed G. Atta ; Daniel Rothenstein ; C. David Mazer
Background::
Acute kidney injury (AKI) affects up to 30% of patients undergoing cardiac surgery, leading to increased in-hospital and long-term morbidity and mortality. Teprasiran is a novel small interfering RNA that temporarily inhibits p53-mediated cell death that underlies AKI.
Methods::
This prospective, multicenter, double-blind, randomized, controlled phase 2 trial evaluated the efficacy and safety of a single 10 mg/kg dose of teprasiran versus placebo (1:1), in reducing the incidence, severity, and duration of AKI after cardiac surgery in high-risk patients. The primary end point was the proportion of patients who developed AKI determined by serum creatinine by postoperative day 5. Other end points included AKI severity and duration using various prespecified criteria. To inform future clinical development, a composite end point of major adverse kidney events at day 90, including death, renal replacement therapy, and ≥25% reduction of estimated glomerular filtration rate was assessed. Both serum creatinine and serum cystatin-C were used for estimated glomerular filtration rate assessments.
Results::
A total of 360 patients were randomly assigned in 41 centers; 341 dosed patients were 73±7.5 years of age (mean±SD), 72% were men, and median European System for Cardiac Operative Risk Evaluation score was 2.6%. Demographics and surgical parameters were similar between groups. AKI incidence was 37% for teprasiran- versus 50% for placebo-treated patients, a 12.8% absolute risk reduction,
P
=0.02; odds ratio, 0.58 (95% CI, 0.37–0.92). AKI severity and duration were also improved with teprasiran: 2.5% of teprasiran- versus 6.7% of placebo-treated patients had grade 3 AKI; 7% teprasiran- versus 13% placebo-treated patients had AKI lasting for 5 days. No significant difference was observed for the major adverse kidney events at day 90 composite in the overall population. No safety issues were identified with teprasiran treatment.
Conclusions::
The incidence, severity, and duration of early AKI in high-risk patients undergoing cardiac surgery were significantly reduced after teprasiran administration. A phase 3 study with a major adverse kidney event at day 90 primary outcome that has recently completed enrollment was designed on the basis of these findings (NCT03510897).
Registration::
URL:
https://www.clinicaltrials.gov
; Unique identifier: NCT02610283.
4
项与 Teprasiran 相关的新闻(医药)2026-05-12
📝 研报客AI助手-AI报告总结
小核酸药物行业专题总结核心内容
小核酸药物是继小分子药物、抗体药物之后的第三类革命性药物,其通过在mRNA水平抑制特定基因表达,可靶向“不可成药靶点”,拓宽治疗范围。近年来,递送系统的突破推动了行业快速发展,全球跨国药企加大布局力度,国内企业也迎来出海机遇。主要观点
小核酸药物的优势:siRNA在mRNA层面作用,具有更高的靶向性和更低的剂量需求,能够实现皮摩尔浓度级的基因沉默效果。
递送系统的发展:当前主要递送技术包括脂质纳米颗粒(LNPs)、GalNAc偶联、C16偶联等,其中GalNAc偶联技术已广泛用于肝靶向治疗,而新型平台正在探索肝外组织递送。
行业趋势:小核酸药物正从罕见病向慢病拓展,从肝靶向向全身靶向跨越,同时受益于化学修饰、AI赋能药物设计、联用策略及产业化升级。
BD交易活跃:2023年以来,全球小核酸领域BD交易频繁,显示行业对新药管线的重视,国内企业亦积极拓展国际合作。
关键信息海外药企布局
诺华:
重点布局核酸药物,通过收购与合作构建心血管-肾脏-代谢(CRM)管线。
Leqvio是诺华与Alnylam合作开发的siRNA药物,用于治疗高胆固醇血症,每6个月注射一次,具有长效优势。
Leqvio在特定患者群体中表现优异,2025年临床试验数据表明其在降低LDL-C和减少肌肉相关不良事件方面优于安慰剂。
公司计划在2030年前实现全球市场领先,推动TTR产品组合及非TTR领域的重磅药物。
Alnylam:
拥有三大递送平台:LNPs、GalNAc偶联、C16偶联,目标2030年前实现10种组织类型的靶向递送。
2025年收入增长显著,达到29.87亿美元,其中TTR产品贡献24.87亿美元。
公司计划推出Nucesiran用于多发性神经病变和心肌病,扩展其在ATTR领域的布局。
Arrowhead:
拥有TRiM™平台,可将siRNA递送至7种组织类型,具有高活性、高特异性、广通用性等优势。
ARO-INHBE和ARO-ALK7在肥胖相关疾病中展现出积极的中期临床数据,尤其在与GLP-1药物联用时表现突出。
公司计划每年新增2-3个临床项目,推进心血管、代谢、CNS等领域的药物研发。
IONIS:
小核酸疗法领域的先驱企业,技术平台涵盖ASO、siRNA、DNA编辑等。
2025年首次独立推出两款药物:TRYNGOLZA(Olezarsen)和DAWNZERA(Donidalorsen),分别用于FCS和HAE预防。
公司预计长期收益结构由全资品种销售和合作品种分成构成,峰值销售预期>40亿美元,合作特许权使用费>20亿美元。
礼来:
以代谢疾病为核心,构建覆盖RNAi、ASO、RNA编辑、环状RNA、AI寡核苷酸的全平台管线。
重点强化肝外递送与长效慢病治疗能力,如lepodisiran和solbinsiran用于治疗脂蛋白(a)和ANGPTL3相关疾病。
国内企业交易
国内企业如前沿生物、瑞博生物、迈威生物、舶望制药等在小核酸领域频繁参与BD交易,涉及多个适应症和靶点。
交易类型包括授权、许可、合作、期权等,金额从数百万美元到数亿美元不等,显示国内企业在全球小核酸领域的竞争力。
未来展望
技术突破:递送系统、靶点创新、适应症拓展、化学修饰、AI药物设计、联用策略和产业化升级是小核酸药物未来发展的主要方向。
商业化潜力:siRNA药物在慢性病治疗中展现出良好前景,如降脂、肥胖、神经退行性疾病等。
风险提示:包括地缘政治风险、创新药研发进展不确定性、商业化不及预期、药品降价风险、医药行业政策风险等。
行业趋势
小核酸药物正逐步从罕见病向高发病率疾病拓展,从肝靶向向全身靶向跨越。
递送系统创新是突破临床转化瓶颈的关键,包括肝外递送、新型载体开发等。
靶点创新与多靶点策略($1 + 1 > 2$)有助于提高药物疗效与适用范围。
化学修饰、AI赋能、联用策略等技术手段正在推动siRNA药物的优化与规模化生产。
表格概览表1:2023年以来全球小核酸领域BD交易(截至2026M3)
| 交易时间 | 转让方 | 受让方 | 管线名称 | 首付款(百万美元) | 里程碑(百万美元) | 总金额(百万美元) | 交易类型 ||----------|--------|--------|----------|------------------|------------------|------------------|----------|| 2026-02-23 | 前沿生物 | GSK | 两款小核酸(siRNA)管线 | 40.00 | 963.00 | 1,003.00 | 许可 || 2026-02-11 | 瑞博生物 | Madrigal Pharmaceuticals | 六款针对MASH的siRNA疗法 | 60.00 | 4,400.00 | 4,460.00 | 许可 || 2026-02-10 | Key2brain | OliX Pharmaceuticals | siRNA-BBB shuttle conjugate | - | - | - | 合作;期权;许可 || 2026-01-26 | Curigin | AprilBio | AOC program | - | - | - | 合作 || 2025-12-16 | Vir Biotechnology | Norgine Pharma | elebsiran; tobevibart | 59.50 | 535.54 | 595.05 | 许可 || 2025-11-26 | Epibiotech | Qmine | RIPK1 siRNA program | - | - | - | 合作 || 2025-11-04 | BXLS | Royalty Pharma | vutrisiran | - | - | 310.00 | 许可 || 2025-10-28 | Empirico | GSK | EMP-012 | 86.00 | 660.00 | 746.00 | 许可 || 2025-09-18 | 迈威生物 | Kalexo Bio | 2MW7141 | 12.00 | - | 1,000.00 | 许可 || 2025-09-03 | 舶望制药 | Novartis | BW-00112; DII235 | 160.00 | 5,200.00 | 5,360.00 | 许可;期权 || 2025-08-01 | 维亚臻生物 | Sanofi | 普乐司兰 | 130.00 | 265.00 | 395.00 | 资产收购 || 2025-05-19 | 靖因药业 | CRISPR Therapeutics | SRSD107; up to two siRNA targets | 25.00 | - | - | 期权;许可;投资 || 2025-04-23 | Asan Medical Center; University of Ulsan | AMC Sciences | AMC-2001 | 0.51 | 1.90 | 2.41 | 许可 || 2025-02-13 | University of California, San Francisco | Junevity | Junevity RESET platform; JUN_01 | - | - | - | 许可 || 2025-02-10 | OliX Pharmaceuticals | Eli Lilly | OLX702A | - | - | - | 许可 || 2024-11-26 | Arrowhead Pharmaceuticals | Sarepta Therapeutics | up to six new targets | 500.00 | 10,550.00 | 11,375.00 | 许可;合作 || 2024-08-01 | Sirnaomics | Sagesse Bio | SGY-101 | - | 33.00 | - | 许可 || 2024-01-07 | 舶望制药 | Novartis | BW-02; BW-05; BW-00163 | 185.00 | - | 4,165.00 | 许可;合作;期权 || 2023-10-31 | Arrowhead Pharmaceuticals; Johnson & Johnson | GSK | daplusiran+tomligisiran | - | - | 1,000.00 | 许可 || 2023-08-03 | Alnylam Pharmaceuticals | Agios Pharmaceuticals | AG-236 | 17.50 | 130.00 | 147.50 | 许可 || 2023-07-24 | Alnylam Pharmaceuticals | Roche | 齐贝司兰 | 310.00 | 2,490.00 | 2,800.00 | 许可;合作 || 2023-03-31 | NanoCarrier | Kanazawa University | NC-6100 | - | - | - | 合作 || 2023-03-09 | Alnylam Pharmaceuticals | Medison | givosiran; patisiran; lumasiran | - | - | - | 合作 |表2:2023年以来国内企业小核酸领域相关交易(截至2026M5)
| 转让方 | 受让方 | 交易类型 | 项目名称 | 关联新药项目 | 交易时间 | 交易金额 | 靶点 | 交易时研发状态 ||--------|--------|----------|----------|--------------|----------|----------|------|----------------|| 前沿生物 | 葛兰素史克 | 授权/许可,合作 | 一款IND阶段的siRNA,临床前候选药物 | FB7013, FB7011 | 2026-02-23 | 首付款:40百万美元里程碑付款:963百万美元特许权使用费:两款产品全球净销售额的分级特许权使用费 | MASP2, CFB | 申请临床,临床前 || 瑞博生物 | Madrigal Pharmaceuticals | 授权/许可 | 六款针对MASH的siRNA疗法,RiboGalSTARTM平台 | - | 2026-02-11 | 交易总额:4400百万美元首付款:60百万美元特许权使用费:基于合作产品全球净销售额的特许权使用费 | - | 临床前 || 圣因生物 | 基因泰克 | 授权/许可 | 一款RNAi药物 | RNAi疗法(基因泰克) | 2026-02-02 | 首付款:200百万美元里程碑付款:1500百万美元特许权使用费:分级 | - | 临床前 || 赫吉亚生物 | 中国生物制药 | 转让/收购 | Kylo-11,Kylo-12,Kylo-0603,HJY-10,HJY-02,HJY-21 | Kylo-0603,HJY-21,Kylo-11,Kylo-12,Kylo-26,HJY-02 | 2026-01-13 | 交易总额:171.38百万美元 | Lp(a), APOC3, THRB, INHBE, APP, PCSK9 | 临床II期,临床I期,临床前 || 圣因生物 | 礼来制药 | 授权/许可,合作 | RNAi therapeutics for metabolic diseases,Ligand and Enhancer Assisted Delivery technology | RNAi for metabolic diseases(Eli Lilly) | 2025-11-08 | 里程碑付款:1200百万美元 | - | 临床前 || 迈威生物 | Kalexo Bio, Inc. | 授权/许可(NewCo),期权 | 心血管领域双靶点 siRNA 创新药 2MW7141 | 2MW7141 | 2025-09-17 | 首付款:12百万美元里程碑付款:1000百万美元特许权使用费:阶梯式特许权使用费 | - | 临床前 || 舶望制药 | 诺华制药 | 授权/许可,合作,期权 | ANGPTL3联合用药用于治疗血脂异常,两款处于早研阶段分子,处于临床前研究阶段的siRNA 候选药物 | BW-00112 | 2025-09-03 | 首付款:160百万美元里程碑付款:5200百万美元特许权使用费:商业销售的分级特许权使用费 | ANGPTL3 | 临床II期,临床前 || 维亚臻生物 | 赛诺菲制药 | 转让/收购 | Plozasiran | 普乐司兰钠 | 2025-08-01 | 交易总额:395百万美元首付款:130百万美元里程碑付款:265百万美元 | APOC3 | 申请上市 || 成都国为生物 | 深圳信立泰药业 | 合作,授权/许可 | 包含GW906活性成分的AGT单靶点小干扰核糖核酸(siRNA)药物,包括所有适应症和用途 | GW906 | 2025-05-27 | 里程碑付款:51.43百万美元特许权使用费:年度净销售额一定比例的销售提成其他交易额:25.02百万美元 | AGT | 临床I期 || 施能康医药 | 华东医药 | 合作,期权 | SNK-2726 | SNK-2726 | 2024-12-30 | - | AGT | 临床前 || 佑嘉生物 | 石家庄以岭药业 | 授权/许可 | NOX1靶向siRNA(佑嘉生物) | YJH-0425 | 2024-11-18 | - | NOX1 | 临床前 || 舶望制药 | 诺华制药 | 授权/许可,期权 | a Phase 1/2a stage cardiovascular asset,a Phase 1 stage cardiovascular asset,two additional targets for cardiovascular disease treatment | BW-00163,BW-02(舶望制药),BW-05,BW-20829 | 2024-01-07 | 交易总额:4165百万美元首付款:185百万美元 | AGT,Lp(a) | 临床I期,临床II期 || 瑞博生物 | 勃林格殷格翰制药 | 合作,授权/许可 | 非酒精性或代谢功能障碍相关脂肪性肝炎创新疗法 | SR111, SR112, SR113 | 2024-01-03 | 交易总额:2000百万美元 | - | 临床前 || 圣因生物 | 信达生物 | 合作,期权 | SGB-3908 | SGB-3908 | 2023-12-27 | - | AGT | 临床前 || 瑞博生物 | 齐鲁制药集团 | 授权/许可 | RBD7022 | RBD7022 | 2023-12-25 | 交易总额:98.54百万美元 | PCSK9 | 临床I期 || 纳肽得医药 | 亿腾医药 | 合作,授权/许可 | 两款分别靶向ANGPTL3和Lp(a)的siRNA药物 | EDP167,Lp(a)(纳肽得) | 2023-09-01 | - | Lp(a), ANGPTL3 | 临床前 |表3:诺华重点布局核酸药物方向,近年收购频繁(截至2026M3)
| 转让方 | 受让方 | 交易类型 | 关联新药项目 | 成分/技术类别 | 技术类型 | 药物类型 | 交易时间 | 交易金额 | 靶点 | 交易时研发状态 | 当前研发状态 | 权益地区 | 权益类型 ||--------|--------|----------|--------------|----------------|----------|----------|----------|----------|------|----------------|--------------|----------|----------|| Avidity Biosciences | 诺华制药 | 转让/收购 | Delpacibart braxlosiran佐生地帕西拜单抗Delpacibart etedesiran AOC 1045 | ASO,抗体寡核苷酸偶联AOC, siRNA,药物递送 | 靶向配体:抗体 | 生物药 | 2025-10-26 | 交易总额:12000百万美元 | DMD,TfR1,DUX4,DMPK | 临床II期临床III期临床前 | 临床II期临床III期临床前 | - | 开发/商业化权,其他 || Arrowhead Pharmaceuticals | 诺华制药 | 授权/许可 | ARO-SNCA | siRNA,其他RNAi,药物发现 | - | 化药 | 2025-09-02 | 首付款:200百万美元里程碑付款:2000百万美元特许权使用费:royalties on commercial sales | SNCA | 临床前 | 临床前 | 全球 | 开发/商业化权,其他 || 上海舶望制药有限公司 | 诺华制药 | 授权/许可,合作,期权 | BW-00112 | siRNA | - | 化药 | 2025-09-03 | 首付款:160百万美元里程碑付款:5200百万美元特许权使用费:商业销售分级特许权使用费 | ANGPTL3 | 临床II期临床前 | 临床II期 | 全球 | 开发/商业化权,其他 || Regulus Therapeutics | 诺华制药 | 转让/收购 | Farabursen | ASO | - | 化药 | 2025-04-30 | 交易总额:1700百万美元首付款:800百万美元里程碑付款:900百万美元 | miR-17 | 临床I/II期 | 临床I期 | 全球 | 开发/商业化权 || 上海舶望制药有限公司 | 诺华制药 | 授权/许可,期权 | BW-00163,BW-02(舶望制药),BW-05 | siRNA | - | 化药 | 2024-01-07 | 交易总额:4165百万美元首付款:185百万美元 | AGT | 临床I期 | 临床I/II期临床II期 | 全球 | 开发/商业化权 || Ionis Pharmaceuticals | 诺华制药 | 合作 | Lp(a)-driven cardiovascular disease(Ionis) | 其他RNA | - | 生物药 | 2023-08-03 | 首付款:60百万美元 | Lp(a) | 临床前 | 临床前 | - | 开发/商业化权 || Dtx Pharma | 诺华制药 | 转让/收购 | DTx-1252 | siRNA | - | 化药 | 2023-07-17 | 首付款:500百万美元里程碑付款:500百万美元 | PMP22 | 临床前 | 临床I期 | 全球 | 开发/商业化权,其他 || Ionis Pharmaceuticals | Chinook Therapeutics | 合作 | ASO+chronic kidney disease(Chinook) | ASO | - | 化药 | 2023-05-16 | - | - | 临床前 | 临床前 | - | 开发/商业化权 || The Medicines Company | 诺华制药 | 转让/收购 | 英克司兰钠 | siRNA | 载体:GalNAc | 化药 | 2019-11-24 | 交易总额:9700百万美元 | PCSK9 | 临床III期 | 批准上市 | - | 开发/商业化权 || Alnylam Pharmaceuticals | The Medicines Company | 授权/许可 | ALN-PCS02英克司兰钠 | siRNA | 载体:GalNAc | 化药 | 2013-02-04 | 首付款:25百万美元里程碑付款:180百万美元 | PCSK9 | 临床I期临床前 | 临床I期批准上市 | 全球 | 开发/商业化权 || Quark Pharmaceuticals | 诺华制药 | 期权,授权/许可 | Teprasiran | siRNA | 载体:脂质纳米粒载体LNP | 化药 | 2010-08-01 | 首付款:10百万美元里程碑付款:670百万美元 | p53 | 临床II期 | 临床III期 | 全球 | 开发/商业化权,其他 || Ionis Pharmaceuticals | Ciba-Geigy | 授权/许可,合作 | 阿普卡生,ISIS 5132 | ASO | - | 化药 | 1999-03-31 | - | PRKCA,RAF1 | - | 临床III期临床II期 | - | 开发/商业化权 || Ionis Pharmaceuticals | Ciba Vision Ltd. | 授权/许可,期权 | 福米韦生,ISIS 13312 | ASO | 载体:无载体 | 化药 | 1997-07-16 | 里程碑付款:20百万美元 | CMV IE2,CMV | 临床III期临床前 | 批准上市临床I/II期 | 全球 | 开发/商业化权 |表4:诺华英格司兰主要临床开展情况(截至2026M3)
| 临床代码 | 适应症 | 临床阶段 | 首次公示日期 | 试验完成日期 ||----------|--------|----------|--------------|--------------|| ORION-1 | 高心血管风险,接受最大耐受剂量LDL-C降低治疗仍高LDL-C | Ph2 | 2015-10-16 | 2017-06-07 || NCT02963311 | 纯合子家族性高胆固醇血症(HoFH) | Ph2 | 2016-11-10 | 2018-10-08 || ORION-3 | 高心血管风险和高LDL-C | Ph2 | 2017-01-03 | 2021-12-17 || ORION-7 | 肾功能不全人群高胆固醇血症 | Ph1 | 2017-05-18 | 2018-03-24 || ORION-11 | 最大/稳定剂量他汀类药物不耐受的高胆固醇血症 | Ph3 | 2017-08-15 | 2019-08-27 || ORION-9 | 杂合子家族性高胆固醇血症(HeFH)和高LDL-C | Ph3 | 2017-08-22 | 2019-09-17 || ORION-10 | 最大/稳定剂量他汀类药物不耐受的ASCVD和高LDL-C | Ph3 | 2018-01-16 | 2019-09-17 || ORION-4 | ASCVD心血管事件二级预防 | Ph3 | 2018-04-16 | 2026 || ORION-8 | 高心血管风险或HeFH,接受最大耐受剂量降脂治疗仍高LDL-C | Ph3 | 2018-11-20 | 2023-02-13 || ORION-5 | 纯合子家族性高胆固醇血症(HoFH) | Ph3 | 2019-02-22 | 2021-09-09 || ORION-16 | 青少年HeFH和高LDL-C | Ph3 | 2020-10-22 | 2024-11-27 || ORION-18 | 接受最大耐受剂量降脂治疗仍高LDL-C的ASCVD或高危亚洲人群 | Ph3 | 2020-11-06 | 2026 || ORION-13 | 青少年HoFH和高LDL-C | Ph3 | 2020-12-02 | 2024-11-18 || ORION-15 | 高心血管风险或HeFH,最大耐受剂量降脂治疗仍高LDL-C日本人群 | Ph2 | 2020-12-14 | 2022-10-19 || ORION-14 | 接受降脂治疗仍高LDL-C的中国人群 | Ph1 | 2021-02-19 | 2021-10-18 || V-INCEPTION | 急性冠状动脉综合征(ACS)且接受他汀类药物治疗仍高LDL-C | Ph3 | 2021-05-05 | 2024-08-07 || V-INITIATE | 接受最大耐受剂量降脂治疗仍高LDL-C的ASCVD | Ph3 | 2021-06-18 | 2023-09-15 || V-2 PREVENT | ASCVD心血管事件二级预防 | Ph3 | 2021-09-01 | 2027 || NCT05118230 | 原发性高胆固醇血症或混合型血脂异常中国人群 | 其他 | 2021-11-11 | 2024-07-20 || V-DIFFERENCE | 持续个体化优化的降脂治疗基础上的高胆固醇血症 | Ph4 | 2021-12-28 | 2025-03-19 || V-PLAQUE | 非梗阻性冠状动脉疾病且无先前心血管事件 | Ph3 | 2022-03-03 | 2026 || V-Implement | 口服降脂治疗队列、Inclisiran、Inclisiran+血脂分离治疗三队列用于高胆固醇血症 | 其他 | 2022-05-05 | 2025 || V-REAL | 原发性高胆固醇血症或混合型血脂异常 | 其他 | 2022-06-01 | 2026 || V-Mono | 未接受降脂治疗的原发性高胆固醇血症 | Ph3 | 2022-10-18 | 2024-06-20 || V-PEDS-OLE | 完成ORION-16/ORION-13研究HeFH/HoFH患者长期安全、耐受性 | Ph3 | 2022-10-19 | 2028 || V-1 PREVENT | 初治患者心血管事件一级预防 | Ph3 | 2023-02-22 | 2029 || V-Mono China | 未接受降脂治疗的低/中等ASCVD风险和高LDL-C中国人群 | Ph3 | 2023-05-24 | 2024-10-24 || V-ACCELERATE | 新发急性心肌梗死和高LDL-C | Ph4 | 2024-04-18 | 2026 || ORION-19 | 儿童HoFH和高LDL-C | Ph3 | 2024-09-19 | 2029 || ORION-20 | 儿童HeFH和高LDL-C | Ph3 | 2024-09-19 | 2029 || INSPIRE | ASCVD和/或HeFH西班牙人群 | - | 2025-05-06 | 2026 || V-RIDES | 中早期急性冠状动脉综合征 | Ph3 | 2025-08-04 | 2027 |表5:2026年Alnylam潜在里程碑节点
| 药物名称 | 适应症 | 进展事项 | 时间节点 ||----------|--------|----------|----------|| Nucresiran | ATTR淀粉样变性 | 推进TRITON-CM III期临床试验 | 进行中 || Nucresiran | ATTR淀粉样变性 | 推进TRITON-PN III期临床试验 | 进行中 || Zilebesiran | 高血压 | 推进ZENITH III期临床试验 | 进行中 || Mivelsiran | 脑淀粉样血管病 | 完成cAPPricorn-1 II期临床试验入组 | 26H1 || Mivelsiran | 阿尔茨海默病 | 启动II期临床试验 | 26H1 || ALN-6400 | 出血性疾病 | 健康志愿者I期数据公布 | 26H2 || ALN-6400 | 出血性疾病 | 遗传性出血性毛细血管扩张症(HHT)II期数据公布 | 26H2 || ALN-6400 | 出血性疾病 | 启动第二种出血性疾病的II期临床试验 | 26H1 || ALN-4324 | 2型糖尿病 | 启动II期临床试验 | 26H1 || 其他项目 | / | 提交3-4项研究性新药申请(IND) | 2026年 |表6:2026年Arrowhead潜在里程碑节点
| 管线名称 | 靶点 | 适应症 | 进展事项 | 时间节点 | 市场规模/患者人群 ||----------|------|--------|----------|----------|------------------|| Plozasiran (REDEMPLO) | APOC3 | 严重高甘油三酯血症(SHTG) | 针对SHTG的3期临床试验(SHASTA-3、SHASTA-4和MUIR-3)盲态部分将于2026年年中完成,预计公布顶线数据并计划提交sNDA以扩大适应症标签 | 数据读出:2026Q3 递交sNDA:2026年内 | 美国约有超过300万TG≥500 mg/dL的SHTG患者,其中高危患者(TG≥880或≥500且有胰腺炎病史)约100万人 || Plozasiran (REDEMPLO) | APOC3 | 家族性乳糜微粒血症综合征(FCS) | 国际市场商业化拓展 | 2026年内 | 美国约有6500名基因确诊或临床诊断的FCS患者 || ARO-DIMER-PA | PCSK9/APOC3 | 混合型高脂血症(ASCVD) | 早期临床数据初步读出 | 2026Q3末 | 美国约有2000万患者同时伴有LDL-C和TG升高,且缺乏充足治疗方案 || ARO-INHBE & ARO-ALK7 | Activin E/ALK7 | 肥胖(重点探索伴有T2DM肥胖) | 正在扩大1期糖尿病队列患者数量、延长随访时间至1年以了解药物持久性;启动针对肥胖糖尿病患者的单药治疗队列及与其他GLP-1药物的联用队列 | 2026年内 | 尤其针对在接受GLP-1单药治疗时减重较少、难以达到减重目标的伴有T2DM的肥胖患者 || Zodasiran | ANGPTL3 | 纯合子家族性高胆固醇血症(HoFH) | 针对HoFH患者的3期研究预计完成入组 | 2026H1 | 美国约有1000-2000名HoFH患者 || ARO-MAPT | MAPT(Tau蛋白) | 阿尔茨海默症 | 1/2期初步临床数据读出,有望为通过皮注实现全身递送的CNS在研管线提供验证支持 | 健康人队列:2026年内(预计Q3末) 阿尔茨海默患者队列:2027年 | 全球超过5000万神经退行性疾病患者,且缺乏改变疾病进程的疗法 || ARO-HTT (SRP-1005)*Sarepta合作项目 | HTT(亨廷顿蛋白) | 亨廷顿舞蹈症 | Sarepta宣布已提交CTA,计划在约24名参与者中启动INSIGHTT的1期研究(SRP-1005-101) | 近期 | 针对亨廷顿舞蹈症的神经退行性疾病群体 |表7:礼来siRNA相关管线布局
| 药品名称 | 靶点 | 研发机构 | 疾病 | 作用机制 | 载体类型 | 美国最高研发阶段 | 美国最高研发阶段开始日期 | 中国最高研发阶段 | 中国最高研发阶段开始日期 ||----------|------|----------|------|----------|----------
免费获取报告源文件,请点下方图片
📚 文章推荐
📌 新黄金系列报告四_从全球红利到中国红利_14页_820kb.pdf
📌 2026年全球技术报告_工业制造洞察_17页_664kb.pdf
📌 2025年全球空气质量报告_44页_3mb.pdf
📌 全球出海雇佣政策指南-8国劳工政策实务汇编2026年版_91页_12mb.pdf
📌 2025淘天集团知识产权保护年度报告_30页_30mb.pdf
📌 华为星河AI园区全域安全技术白皮书_26H1_80页_3mb.pdf
📌 租赁住房与产业园区REITs2025年报综述_关注运营能力_聚焦科创属性_54页_6mb.pdf
📌 城市轨道交通2025年度统计和分析报告(协会信息2026年第3期_总第78期)_83页_7mb.pd...
📌 制药供应链_景气度上行信号明确_内外需共振带动业绩提速_16页_2mb.pdf
📌 2025中国医药CDMO行业发展洞察蓝皮书_83页_4mb.pdf
📌 神经多样性_办公空间感官需求对比研究_19页_2mb.pdf
📌 数字金融建设与创新白皮书2025_15页_11mb.pdf
📌 全球钢铁低碳转型洞见15则洞察报告_54页_6mb.pdf
📌 全球保险业首席财务官调研_20页_3mb.pdf
📌 2026美国制造业转型报告_政策_绩效与前瞻_28页_4mb.pdf
📌 2026年智能机器人行业深度分析报告-中投顾问_43页_3mb.pdf
📌 20260511-申银万国期货-品种策略日报-PTA_2页_113kb.pdf
📌 20260511-申银万国期货-品种策略日报-乙二醇_2页_115kb.pdf
📌 20260511-申银万国期货-品种策略日报-原油甲醇_2页_199kb.pdf
📌 20260511-申银万国期货-品种策略日报-双焦(JM_J)_1页_153kb.pdf
研报客 - 全球行业报告 图表数据聚合AI平台
5000多家机构,五百万+报告 覆盖全行业深度研究
一站式行业研究报告平台,汇聚全球百强智库,百万中英文报告
✨ 核心功能
📊 海量报告:覆盖全行业,AI搜索+全文检索
🤖 AI翻译:多语言互译,保留原排版
💬 AI对话:智能研报助手,深度解读文档
🔔 实时推送:订阅关键词,更新主动通知
📱 全平台:网页、APP、小程序数据同步
🤖 AI能力
🔍 智能检索:快速理解问题意图,提供精准信息
🌐 多源整合:综合多个可靠来源,给出全面答案
💡 语境理解:不只是字面翻译,更能理解上下文和文化差异
🎯 专业领域:适应技术、文学、商务等不同领域的翻译需求
🧠 自然交流:理解复杂对话上下文,保持连贯性
📚 研报专家:深度理解文档,进行专业回复
⚡ 实时性:能够获取最新信息,主动推送更新
🎯 适用场景
💼 投资分析:券商研报、市场分析、投资策略
📈 市场调研:行业分析、市场趋势、竞争格局
🏭 行业研究:深度行业报告、产业链分析
🚀 趋势预测:未来趋势、技术发展、政策解读
🌐 访问方式
💻 网页版:pc.yanbaoke.cn
📱 APP下载:各大手机商店搜索「研报客」
🖥️ 软件版:pc.yanbaoke.cn/pc2.html
📲 小程序:微信搜索「研报客」
💡 PDF/DOC/PPT互转 | 文字版一键复制 | 支持下载分享
网页、APP、小程序多端同步,随时随地阅读研报
内容由ai生成,仅供参考
2026-04-21
治疗急性肾损伤的新药
New drugs for acute kidney injury
Citation: Hariri G, Legrand M. New drugs for acute kidney injury. J Intensive Med. 2024;5(1):3–11. doi: 10.1016/j.jointm.2024.08.001.
核心结论
急性肾损伤(AKI)在重症及围术期患者中发病率高达30%-60%,目前仍无高级别证据支持的预防或治疗药物。本文系统综述了基于最新病理生理机制(炎症应答、代谢重编程、血流动力学调节、补体活化及细胞凋亡/修复)的在研新药,涵盖碱性磷酸酶、Reltecimod、血管紧张素II、维生素B3类、CD39重组蛋白、Ravulizumab、siRNA QPI-1002及间充质干细胞等。多项II/III期临床试验正在评估其疗效,为降低AKI发生率及严重程度带来新希望。
正文
急性肾损伤(AKI)是全球性健康问题,每年影响超过1300万人,并导致约170万例死亡[1]。从病理生理角度看,AKI以肾小球滤过率(GFR)快速下降为特征,数小时或数日内表现为血清肌酐急剧升高。2004年之前由于缺乏统一定义,AKI的诊断标准从轻度肌酐升高至需要肾脏替代治疗(RRT)范围广泛[2]。RIFLE、AKIN及KDIGO分级系统的建立,依据血清肌酐和尿量变化提供了结构化诊疗路径,实现了早期识别与干预[3-5]。在重症监护环境中,AKI发病率异质性较大(15%-60%),且与死亡率升高、住院时间延长及慢性肾脏病(CKD)风险增加密切相关[6-8]。尽管过去二十年危重症患者管理取得显著进步,但预防或治疗AKI的有效药物干预仍十分缺乏。近期对AKI病理生理机制的深入理解揭示了炎症、代谢紊乱及细胞修复障碍等共同通路,为新型药物研发提供了方向(图1,表1)。
图1:重症及围术期急性肾损伤预防或治疗新药的作用机制概览。HGF: 肝细胞生长因子;MSC: 间充质干细胞。
基于上述机制,目前处于研发阶段的药物涵盖了免疫调节、血流动力学优化、细胞能量代谢支持、补体抑制及抗凋亡/促修复等多个维度。下文按照靶点分类逐一阐述代表性药物的临床证据。1. 炎症调控
碱性磷酸酶(ALP)可通过去磷酸化脂多糖(LPS)及胞外ATP/ADP,减轻Toll样受体介导的先天免疫反应。STOP-AKI试验(n=301)中,重组ALP(ilofotase alfa)虽未改善7天肾功能,但28天肌酐清除率提升18.5 mL/min(95%CI: 5.3-31.7),死亡率降低(14.4% vs 26.7%)[18]。然而后续III期REVIVAL试验(n=655)未显示28天全因死亡率获益,但MAKE90复合终点(56.7% vs 64.6%, P=0.02)提示可能降低RRT需求[19]。针对CSA-AKI的II期预防试验(NCT06168799)正在进行。
Reltecimod是一种靶向T细胞表面CD28的肽类,可调节脓毒症免疫应答。在坏死性软组织感染III期试验中虽未达到主要复合终点,但第14天SOFA评分改善(65.1% vs 52.6%, P=0.04)[24]。目前针对KDIGO 2-3期AKI合并腹腔脓毒症的III期试验(NCT03403751)正在评估28天AKI恢复率。
活性维生素D(骨化三醇)可通过下调核因子κB减轻炎症。但ACTIVATE-AKI试验(NCT02962102)未显示7天内死亡或RRT需求的显著差异。RBT-1(锡原卟啉+蔗糖铁)通过上调HO-1、铁蛋白发挥预适应保护,PROTECT III期研究(n=400)正在进行,主要终点为死亡、透析需求、ICU住院日及30天再入院复合指标。
TIN816(重组CD39)可水解胞外ATP/ADP,抑制炎症及血小板聚集。针对SA-AKI的II期试验(NCT05996835)以第1-8天时间校正肌酐清除率曲线下面积为主要终点;CSA-AKI预防试验(NCT05524051)计划纳入120例患者。2. 血流动力学调节
血管紧张素II选择性收缩肾出球小动脉,提升GFR。ATHOS-3试验显示,血管紧张素II显著提高顽固性血管扩张性休克患者3小时平均动脉压(69.9% vs 23.4%, OR=7.95)[46],且在需RRT的亚组中28天生存率更高(53% vs 30%)[47]。预防CSA-AKI的可行性研究提示AKI发生率降低(25% vs 38%, P=0.31),III期试验(NCT05199493)正在开展。
血管加压素:心脏术后血管麻痹性休克患者中,血管加压素较去甲肾上腺素显著降低CSA-AKI(10.3% vs 35.8%, P<0.0001)[52];但在脓毒性休克中未显示AKI预防获益。个体患者数据Meta分析提示可能降低RRT需求(RR=0.86)。两项RCT(NCT04602767, NCT06125184)将进一步明确。
左西孟旦:钙增敏剂兼有入球小动脉扩张作用。心脏手术Meta分析显示可减少CSA-AKI(OR=0.51)及RRT(OR=0.43),但大型RCT未证实获益[57]。LEVO-AKI试验(NCT02531724)正在评估已发生CSA-AKI的治疗效果。3. 细胞代谢与线粒体功能
维生素B3类(烟酰胺、烟酰胺核糖)通过补充NAD+保护肾小管。心脏手术II期试验显示烟酰胺使AKI发生率降低35%[59]。多项RCT正在验证其预防CSA-AKI(NCT04750616)、肾移植后移植物功能(NCT05513807)及脓毒性休克相关AKI(NCT04589546)的疗效。ASP1128(PPARδ调节剂)在心脏手术高危患者中未减少AKI发生率(24.6% vs 21%, P=0.595)[62]。SIRT1激活剂NRPT(烟酰胺核糖+紫檀芪)II期试验(NCT04342975)正在主动脉弓置换患者中评估预防效果。
表1:重症及围术期急性肾损伤预防/治疗新药汇总
药物
机制
靶点
相关临床试验
研究人群
Angiotensin II
血流动力学
肾出球小动脉
NCT05199493
预防CSA-AKI
Angiotensin II
血流动力学
肾出球小动脉
NCT04592744
肝移植后AKI预防
Angiotensin II
血流动力学
肾出球小动脉
NCT04901169
肝移植后AKI预防
Vasopressin
血流动力学
V1a受体
NCT04602767
预防CSA-AKI
Vasopressin
血流动力学
V1a受体
NCT06125184
预防SA-AKI(脓毒性休克)
Levosimendan
血流动力学/正性肌力
肾入球小动脉
NCT02531724
CSA-AKI治疗
Reltecimod
免疫调节
CD28
NCT03403751
SA-AKI(腹腔脓毒症)
Activated vitamin D
免疫调节
NF-κB
NCT02962102
高危患者AKI预防
ALP (Ilofotase alfa)
免疫调节
TLR-4/LPS
NCT06168799
预防CSA-AKI
RBT-1
缺血预适应
HO-1/铁蛋白
PROTECT study
预防CSA-AKI
TIN816
ATP/ADP水解
CD39
NCT05996835
SA-AKI
TIN816
ATP/ADP水解
CD39
NCT05524051
预防CSA-AKI
Vitamin B3 (Nicotinamide)
细胞代谢
NAD+合成
NCT04750616
预防CSA-AKI
Nicotinamide
细胞代谢
NAD+
NCT05513807
肾移植后移植物功能延迟
NR (Nicotinamide riboside)
NAD+前体
SIRT1
NCT04818216
COVID-19相关AKI
ASP1128
PPARδ激动剂
脂肪酸氧化
II期(已停止)
CSA-AKI高危
NRPT (NR+Pterostilbene)
SIRT1激活
NAD+/SIRT1
NCT04342975
主动脉弓置换后AKI预防
Ravulizumab
补体C5抑制剂
C5
NCT05746559
预防CSA-AKI
siRNA QPI-1002
抗凋亡
p53
NCT02610296
肾移植后移植物功能延迟
HGF-mimetic (BB3)
细胞修复/抗凋亡
cMet/Bcl-2
NCT02771509
预防CSA-AKI
Mesenchymal Stem Cells
免疫调节/修复
多靶点
NCT04445220
COVID-19合并RRT
Umbilical cord MSC
组织修复
多靶点
NCT04194671
AKI治疗
补体抑制剂Ravulizumab(长效C5单抗)在COVID-19相关AKI的小样本试验中显示透析需求降低(P=0.03),III期ARTEMIS试验(NCT05746559)正在心脏手术CKD患者中验证其预防CSA-AKI的效果。4. 凋亡抑制与细胞修复
siRNA药物QPI-1002(靶向p53)在心脏手术高危患者中使AKI发生率降低13%(37% vs 50%, P=0.02)[75],但MAKE90无差异;III期预防试验因无效而终止。HGF模拟物BB3在肾移植后未改善360天eGFR(P=0.32)[80],GUARD试验(NCT02771509)正在评估其对CSA-AKI的预防效果。间充质干细胞(MSC)在CSA-AKI的II期试验中未显示获益,但体外扩增MSC可能具有免疫修复潜力,相关试验仍在进行(NCT04445220, NCT04194671)。
综上所述,目前尚无获批的AKI特效药,但基于精准病理生理靶点的多种药物已进入后期临床试验阶段,为改善重症及手术患者AKI结局带来了切实希望。
参考文献
Mehta R.L., Cerdá J., Burdmann E.A., Tonelli M., García-García G., Jha V., et al. International Society of Nephrology's 0by25 initiative for acute kidney injury (zero preventable deaths by 2025): a human rights case for nephrology. Lancet. 2015;385:2616–2643. doi: 10.1016/S0140-6736(15)60126-X.
Ricci Z., Cruz D.N., Ronco C. Classification and staging of acute kidney injury: beyond the RIFLE and AKIN criteria. Nat Rev Nephrol. 2011;7(4):201–208. doi: 10.1038/nrneph.2011.14.
Bellomo R., Ronco C., Kellum J.A., Mehta R.L., Palevsky P. Acute renal failure - definition, outcome measures, animal models, fluid therapy and information technology needs: the Second International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group. Crit Care. 2004;8(4):R204–R212. doi: 10.1186/cc2872.
Mehta R.L., Kellum J.A., Shah S.V., Molitoris B.A., Ronco C., Warnock D.G., et al. Acute kidney injury network: report of an initiative to improve outcomes in acute kidney injury. Crit Care. 2007;11(2):R31. doi: 10.1186/cc5713.
Kellum J.A., Lameire N., Aspelin P., Barsoum R.S., Burdmann E.A., Goldstein S.L., et al. Kidney disease: improving global outcomes (KDIGO) acute kidney injury work group. KDIGO clinical practice guideline for acute kidney injury. Kidney Int Suppl. 2012;2:1–138. doi: 10.1038/kisup.2012.1.
Hoste E., Kellum J.A., Selby N.M., Zarbock A., Palevsky P.M., Bagshaw S.M., et al. Global epidemiology and outcomes of acute kidney injury. Nat Rev Nephrol. 2018;14(10):607–625. doi: 10.1038/s41581-018-0052-0.
Chaïbi K., Ehooman F., Pons B., Martin-Lefevre L., Boulet E., Boyer A., et al. Long-term outcomes after severe acute kidney injury in critically ill patients: the SALTO study. Ann Intensive Care. 2023;13(1):18. doi: 10.1186/s13613-023-01108-x.
Pickkers P., Darmon M., Hoste E., Joannidis M., Legrand M., Ostermann M., et al. Acute kidney injury in the critically ill: an updated review on pathophysiology and management. Intensive Care Med. 2021;47(8):835–850. doi: 10.1007/s00134-021-06454-7.
Wang Y., Bellomo R. Cardiac surgery-associated acute kidney injury: risk factors, pathophysiology and treatment. Nat Rev Nephrol. 2017;13(11):697–711. doi: 10.1038/nrneph.2017.119.
Legrand M., Bagshaw S.M., Bhatraju P.K., Bihorac A., Caniglia E., Khanna A.K., et al. Sepsis-associated acute kidney injury: recent advances in enrichment strategies, sub-phenotyping and clinical trials. Crit Care. 2024;28(1):92. doi: 10.1186/s13054-024-04877-4.
Perazella M.A., Rosner M.H. Drug-Induced Acute Kidney Injury. Clin J Am Soc Nephrol. 2022;17(8):1220–1233. doi: 10.2215/CJN.11290821.
Rabb H., Griffin M.D., McKay D.B., Swaminathan S., Pickkers P., Rosner M.H., et al. Inflammation in AKI: current understanding, key questions, and knowledge gaps. J Am Soc Nephrol. 2016;27(2):371–379. doi: 10.1681/ASN.2015030261.
McWilliam S.J., Wright R.D., Welsh G.I., Tuffin J., Budge K.L., Swan L., et al. The complex interplay between kidney injury and inflammation. Clin Kidney J. 2021;14(3):780–788. doi: 10.1093/ckj/sfaa164.
Schenck E.J., Ma K.C., Murthy S.B., Choi A. Danger signals in the ICU. Crit Care Med. 2018;46(5):791–798. doi: 10.1097/CCM.0000000000003007.
Bentala H., Verweij W.R., Huizinga-Van der Vlag A., van Loenen-Weemaes A.M., Meijer D.K., Poelstra K. Removal of phosphate from lipid A as a strategy to detoxify lipopolysaccharide. Shock. 2002;18(6):561–566. doi: 10.1097/00024382-200212000-00013.
Wy C.A., Goto M., Young R.I., Myers T.F., Muraskas J. Prophylactic treatment of endotoxic shock with monophosphoryl lipid A in newborn rats. Biol Neonate. 2000;77(3):191–195. doi: 10.1159/000014215.
Peters E., Heemskerk S., Masereeuw R., Pickkers P. Alkaline phosphatase: a possible treatment for sepsis-associated acute kidney injury in critically ill patients. Am J Kidney Dis. 2014;63(6):1038–1048. doi: 10.1053/j.ajkd.2013.11.027.
Pickkers P., Mehta R.L., Murray P.T., Joannidis M., Molitoris B.A., Kellum J.A., et al. Effect of human recombinant alkaline phosphatase on 7-day creatinine clearance in patients with sepsis-associated acute kidney injury: a randomized clinical trial. JAMA. 2018;320(19):1998–2009. doi: 10.1001/jama.2018.14283.
Pickkers P., Angus D.C., Bass K., Bellomo R., van den Berg E., Bernholz J., et al. Phase-3 trial of recombinant human alkaline phosphatase for patients with sepsis-associated acute kidney injury (REVIVAL) Intens Care Med. 2024;50(1):68–78. doi: 10.1007/s00134-023-07271-w.
Ramachandran S., Subramanian V., Mohanakumar T. Immune responses to self-antigens (autoimmunity) in allograft rejection. Clin Transpl. 2012:261–272.
Ramachandran G., Kaempfer R., Chung C.S., Shirvan A., Chahin A.B., Palardy J.E., et al. CD28 homodimer interface mimetic peptide acts as a preventive and therapeutic agent in models of severe bacterial sepsis and gram-negative bacterial peritonitis. J Infect Dis. 2015;211(6):995–1003. doi: 10.1093/infdis/jiu556.
Edgar R., Tarrio M.L., Maislin G., Chiguang F., Kaempfer R., Cross A., et al. Treatment with one dose of Reltecimod is superior to two doses in mouse models of lethal infection. Int J Pept Res Ther. 2020;26:1669–1683. doi: 10.1007/s10989-019-09974-5.
Bulger E.M., Maier R.V., Sperry J., Joshi M., Henry S., Moore F.A., et al. A novel drug for treatment of necrotizing soft-tissue infections: a randomized clinical trial. JAMA Surg. 2014;149(6):528–536. doi: 10.1001/jamasurg.2013.4841.
Bulger E.M., May A.K., Robinson B., Evans D.C., Henry S., Green J.M., et al. A novel immune modulator for patients with necrotizing soft tissue infections (NSTI): results of a multicenter, phase 3 randomized controlled trial of reltecimod (AB 103) Ann Surg. 2020;272(3):469–478. doi: 10.1097/SLA.0000000000004102.
Keung L., Perwad F. Vitamin D and kidney disease. Bone Rep. 2018;9:93–100. doi: 10.1016/j.bonr.2018.07.002.
Al-Badr W., Martin K.J. Vitamin D and kidney disease. Clin J Am Soc Nephrol. 2008;3(5):1555–1560. doi: 10.2215/CJN.01150308.
Graidis S., Papavramidis T.S., Papaioannou M. Vitamin D and acute kidney injury: a two-way causality relation and a predictive, prognostic, and therapeutic role of vitamin D. Front Nutr. 2021;7 doi: 10.3389/fnut.2020.630951.
Xu S., Chen Y.H., Tan Z.X., Xie D.D., Zhang C., Zhang Z.H., et al. Vitamin D3 pretreatment regulates renal inflammatory responses during lipopolysaccharide-induced acute kidney injury. Sci Rep. 2015;5:18687. doi: 10.1038/srep18687.
Vervloet M.G., Hsu S., de Boer I.H. Vitamin D supplementation in people with chronic kidney disease. Kidney Int. 2023;104(4):698–706. doi: 10.1016/j.kint.2023.07.010.
Zhang H., Jiang Y., Shi N., Lu Y.Q. Serum vitamin D levels and acute kidney injury: a systemic review and meta-analysis. Sci Rep. 2022;12(1):20365. doi: 10.1038/s41598-022-24560-4.
Leaf D.E., Jacob K.A., Srivastava A., Chen M.E., Christov M., Jüppner H., et al. Fibroblast growth factor 23 levels associate with AKI and death in critical illness. J Am Soc Nephrol. 2017;28(6):1877–1885. doi: 10.1681/ASN.2016080836.
Eltzschig H.K., Eckle T. Ischemia and reperfusion–from mechanism to translation. Nat Med. 2011;17(11):1391–1401. doi: 10.1038/nm.2507.
Huang Y., Rabb H., Womer K.L. Ischemia-reperfusion and immediate T cell responses. Cell Immunol. 2007;248(1):4–11. doi: 10.1016/j.cellimm.2007.03.009.
Li C., Jackson R.M. Reactive species mechanisms of cellular hypoxia-reoxygenation injury. Am J Physiol Cell Physiol. 2002;282(2):C227–C241. doi: 10.1152/ajpcell.00112.2001.
Zhang M., Liu Q., Meng H., Duan H., Liu X., Wu J., et al. Ischemia-reperfusion injury: molecular mechanisms and therapeutic targets. Signal Transduct Target Ther. 2024;9(1):12. doi: 10.1038/s41392-023-01688-x.
Lamy A., Chertow G.M., Jessen M., Collar A., Brown C.D., Mack C.A., et al. Effects of RBT-1 on preconditioning response biomarkers in patients undergoing coronary artery bypass graft or heart valve surgery: a multicentre, double-blind, randomised, placebo-controlled phase 2 trial. EClinicalMedicine. 2024;68 doi: 10.1016/j.eclinm.2023.102364.
Priyan V. Renibus begins subject dosing in phase III trial of RBT-1. Clinical Trials Arena. 2023
Cauwels A., Rogge E., Vandendriessche B., Shiva S., Brouckaert P. Extracellular ATP drives systemic inflammation, tissue damage and mortality. Cell Death Dis. 2014;5(3):e1102. doi: 10.1038/cddis.2014.70.
Woulfe D., Yang J., Brass L. ADP and platelets: the end of the beginning. J Clin Invest. 2001;107:1503–1505. doi: 10.1172/JCI13361.
Eltzschig H.K., Köhler D., Eckle T., Kong T., Robson S.C., Colgan S.P. Central role of Sp1-regulated CD39 in hypoxia/ischemia protection. Blood. 2009;113(1):224–232. doi: 10.1182/blood-2008-06-165746.
Hayes G.M., Cairns B., Levashova Z., Chinn L., Perez M., Theunissen J.W., et al. CD39 is a promising therapeutic antibody target for the treatment of soft tissue sarcoma. Am J Transl Res. 2015;7(6):1181–1188.
Granja T., Körner A., Glück C., Hohmann J.D., Wang X., Köhler D., et al. Targeting CD39 toward activated platelets reduces systemic inflammation and improves survival in sepsis: a preclinical pilot study. Crit Care Med. 2019;47(5) doi: 10.1097/CCM.0000000000003682. e420-420e427.
Bellomo R., Forni L.G., Busse L.W., McCurdy M.T., Ham K.R., Boldt D.W., et al. Renin and survival in patients given angiotensin II for catecholamine-resistant vasodilatory shock. A clinical trial. Am J Respir Crit Care Med. 2020;202(9):1253–1261. doi: 10.1164/rccm.201911-2172OC.
Küllmar M., Saadat-Gilani K., Weiss R., Massoth C., Lagan A., Cortés M.N., et al. Kinetic changes of plasma renin concentrations predict acute kidney injury in cardiac surgery patients. Am J Respir Crit Care Med. 2021;203(9):1119–1126. doi: 10.1164/rccm.202005-2050OC.
Chawla L.S., Busse L., Brasha-Mitchell E., Davison D., Honiq J., Alotaibi Z., et al. Intravenous angiotensin II for the treatment of high-output shock (ATHOS trial): a pilot study. Crit Care. 2014;18(5):534. doi: 10.1186/s13054-014-0534-9.
Khanna A., English S.W., Wang X.S., Ham K., Tumlin J., Szerlip H., et al. Angiotensin II for the treatment of vasodilatory shock. N Engl J Med. 2017;377(5):419–430. doi: 10.1056/NEJMoa1704154.
Tumlin J.A., Murugan R., Deane A.M., Ostermann M., Busse L.W., Ham K.R., et al. Outcomes in patients with vasodilatory shock and renal replacement therapy treated with intravenous angiotensin II. Crit Care Med. 2018;46(6):949–957. doi: 10.1097/CCM.0000000000003092.
Bokoch M.P., Tran A.T., Brinson E.L., Marcus S.G., Reddy M., Sun E., et al. Angiotensin II in liver transplantation (AngLT-1): protocol of a randomised, double-blind, placebo-controlled trial. BMJ Open. 2023;13(11) doi: 10.1136/bmjopen-2023-078713.
Coulson T.G., Miles L.F., Serpa Neto A., Pilcher D., Weinberg L., Landoni G., et al. A double-blind randomised feasibility trial of angiotensin-2 in cardiac surgery( Anaesthesia. 2022;77(9):999–1009. doi: 10.1111/anae.15802.
Finley J.J., 4th, Konstam M.A., Udelson J.E. Arginine vasopressin antagonists for the treatment of heart failure and hyponatremia. Circulation. 2008;118(4):410–421. doi: 10.1161/CIRCULATIONAHA.108.765289.
Okazaki N., Iguchi N., Evans R.G., Hood S.G., Bellomo R., May C.N., et al. Beneficial effects of vasopressin compared with norepinephrine on renal perfusion, oxygenation, and function in experimental septic acute kidney injury. Crit Care Med. 2020;48(10) doi: 10.1097/CCM.0000000000004511. e951-951e958.
Hajjar L.A., Vincent J.L., Barbosa Gomes Galas F.R., Rhodes A., Landoni G., Osawa E.A., et al. Vasopressin versus norepinephrine in patients with vasoplegic shock after cardiac surgery: the VANCS randomized controlled trial. Anesthesiology. 2017;126(1):85–93. doi: 10.1097/ALN.0000000000001434.
Gordon A.C., Mason A.J., Thirunavukkarasu N., Perkins G.D., Cecconi M., Cepkova M., et al. Effect of early vasopressin vs norepinephrine on kidney failure in patients with septic shock: the VANISH randomized clinical trial. JAMA. 2016;316(5):509–518. doi: 10.1001/jama.2016.10485.
Hajjar L.A., Zambolim C., Belletti A., de Almeida J.P., Gordon A.C., Oliveira G., et al. Vasopressin versus norepinephrine for the management of septic shock in cancer patients: the VANCS II randomized clinical trial. Crit Care Med. 2019;47(12):1743–1750. doi: 10.1097/CCM.0000000000004023.
Pan J., Yang Y.M., Zhu J.Y., Lu Y.Q. Multiorgan drug action of levosimendan in critical illnesses. Biomed Res Int. 2019;2019 doi: 10.1155/2019/9731467.
Zhou C., Gong J., Chen D., Wang W., Liu M., Liu B. Levosimendan for prevention of acute kidney injury after cardiac surgery: a meta-analysis of randomized controlled trials. Am J Kidney Dis. 2016;67:408–416. doi: 10.1053/j.ajkd.2015.09.015.
Landoni G., Lomivorotov V.V., Alvaro G., Lobreglio R., Pisano A., Guarracino F., et al. Levosimendan for hemodynamic support after cardiac surgery. N Engl J Med. 2017;376(21):2021–2031. doi: 10.1056/NEJMoa1616325.
Gordon A.C., Perkins G.D., Singer M., McAuley D.F., Orme R.M., Santhakumaran S., et al. Levosimendan for the prevention of acute organ dysfunction in sepsis. N Engl J Med. 2016;375(17):1638–1648. doi: 10.1056/NEJMoa1609409.
Poyan Mehr A., Tran M.T., Ralto K.M., Leaf D.E., Washco V., Messmer J., et al. De novo NAD(+) biosynthetic impairment in acute kidney injury in humans. Nat Med. 2018;24(9):1351–1359. doi: 10.1038/s41591-018-0138-z.
Gao J., Gu Z. The role of peroxisome proliferator-activated receptors in kidney diseases. Front Pharmacol. 2022;13 doi: 10.3389/fphar.2022.832732.
Taniuchi Y., van Till J.W.O., Wojtkowski T., Toyoshima J., Koibuchi A., Sargent B., et al. Single- and multiple-dose safety, tolerability, pharmacokinetics, and pharmacodynamics of ASP1128, a novel peroxisome proliferator-activated receptor δ modulator, in healthy participants. Clin Pharmacol Drug Dev. 2023;12:810–818. doi: 10.1002/cpdd.1236.
van Till J.W.O., Nojima H., Kameoka C., Hayashi C., Sakatani T., Washburn T.B., et al. The effects of peroxisome proliferator-activated receptor-delta modulator ASP1128 in patients at risk for acute kidney injury following cardiac surgery. Kidney Int Rep. 2023;8:1407–1416. doi: 10.1016/j.ekir.2023.04.004.
Guan Y., Wang S.R., Huang X.Z., Xie Q.H., Xu Y.Y., Shang D., et al. Nicotinamide mononucleotide, an NAD+ precursor, rescues age-associated susceptibility to AKI in a Sirtuin 1–dependent manner. J Am Soc Nephrol. 2017;28:2337–2352. doi: 10.1681/ASN.2016040385.
Guan Y., Hao C.M. SIRT1 and kidney function. Kidney Dis (Basel) 2016;1:258–265. doi: 10.1159/000440967.
Peasley K., Chiba T., Goetzman E., Sims-Lucas S. Sirtuins play critical and diverse roles in acute kidney injury. Pediatr Nephrol. 2021;36:3539–3546. doi: 10.1007/s00467-020-04866-z.
Simic P., Vela Parada X.F., Parikh S.M., Dellinger R., Guarente L.P., Rhee E.P. Nicotinamide riboside with pterostilbene (NRPT) increases NAD+ in patients with acute kidney injury (AKI): a randomized, double-blind, placebo-controlled, stepwise safety study of escalating doses of NRPT in patients with AKI. BMC Nephrol. 2020;21:342. doi: 10.1186/s12882-020-02006-1.
Fakhouri F., Schwotzer N., Golshayan D., Frémeaux-Bacchi V. The rational use of complement inhibitors in kidney diseases. Kidney Int Rep. 2022;7:1165–1178. doi: 10.1016/j.ekir.2022.02.021.
Sheridan D., Yu Z.X., Zhang Y., Patel R., Sun F., Lasaro M.A., et al. Design and preclinical characterization of ALXN1210: a novel anti-C5 antibody with extended duration of action. PLoS ONE. 2018;13 doi: 10.1371/journal.pone.0195909.
Memon A.A., Ahmed H., Li Y., Wongboonsin J., Hundert J., Benoit S., et al. A randomized control trial of ravulizumab for treatment of patients with COVID-19 infection and kidney injury. Kidney Int Rep. 2022;7:2714–2717. doi: 10.1016/j.ekir.2022.09.003.
Olsen T.S., Olsen H.S., Hansen H.E. Tubular ultrastructure in acute renal failure in man: epithelial necrosis and regeneration. Virchows Arch A Pathol Anat Histopathol. 1985;406:75–89. doi: 10.1007/BF00710559.
Sanz A.B., Sanchez-Niño M.D., Ramos A.M., Ortiz A. Regulated cell death pathways in kidney disease. Nat Rev Nephrol. 2023;19:281–299. doi: 10.1038/s41581-023-00694-0.
Tang C., Ma Z., Zhu J., Liu Z., Liu Y., Liu Y., et al. P53 in kidney injury and repair: mechanism and therapeutic potentials. Pharmacol Ther. 2019;195:5–12. doi: 10.1016/j.pharmthera.2018.10.013.
Wei Q., Dong G., Yang T., Megyesi J., Price P.M., Dong Z. Activation and involvement of p53 in cisplatin-induced nephrotoxicity. Am J Physiol Renal Physiol. 2007;293:F1282–F1291. doi: 10.1152/ajprenal.00230.2007.
Peddi V.R., Ratner L., Cooper M., Gaber O., Feng S., Tso P., et al. Treatment with QPI-1002, a short interfering (SI) RNA for the prophylaxis of delayed graft function.: abstract# 2967. Transplantation. 2014;98:153. doi: 10.1097/00007890-201407151-00467.
Thielmann M., Corteville D., Szabo G., Swaminathan M., Lamy A., Lehner L.J., et al. Teprasiran, a small interfering RNA, for the prevention of acute kidney injury in high-risk patients undergoing cardiac surgery: a randomized clinical study. Circulation. 2021;144:1133–1144. doi: 10.1161/CIRCULATIONAHA.120.053029.
Kawaida K., Matsumoto K., Shimazu H., Nakamura T. Hepatocyte growth factor prevents acute renal failure and accelerates renal regeneration in mice. Proc Natl Acad Sci U S A. 1994;91:4357–4361. doi: 10.1073/pnas.91.10.4357.
Zhou D., Tan R.J., Lin L., Zhou L., Liu Y. Activation of hepatocyte growth factor receptor, c-met, in renal tubules is required for renoprotection after acute kidney injury. Kidney Int. 2013;84:509–520. doi: 10.1038/ki.2013.102.
Narayan P., Duan B., Jiang K., Li J., Paka L., Yamin M.A., et al. Late intervention with the small molecule BB3 mitigates postischemic kidney injury. Am J Physiol Renal Physiol. 2016;311:F352–F361. doi: 10.1152/ajprenal.00455.2015.
Bromberg J.S., Weir M.R., Gaber A.O., Yamin M.A., Goldberg I.D., Mayne T.J., et al. Renal function improvement following ANG-3777 treatment in patients at high risk for delayed graft function after kidney transplantation. Transplantation. 2021;105:443–450. doi: 10.1097/TP.0000000000003255.
Vincenti F., Bromberg J., Kim J., Faravardeh A., Leca N., Alperovich G., et al. The hepatocyte growth factor mimetic, ANG-3777, in kidney transplant recipients with delayed graft function: results from a randomized phase 3 trial. Am J Transplant. 2024 doi: 10.1016/j.ajt.2024.02.014. S1600-6135(24)00156-4.
Ayad S., Neylan J.F., Mayne T.J., Gouveia D., Swaminathan M. Hepatocyte growth factor mimetic ANG-3777 for cardiac surgery-associated acute kidney injury. Kidney Int Rep. 2020;5(12):2325–2332. doi: 10.1016/j.ekir.2020.09.031.
Allinson C.S., Pollock C.A., Chen X. Mesenchymal stem cells in the treatment of acute kidney injury (AKI), chronic kidney disease (CKD) and the AKI-to-CKD transition. Intgr Med Nephrol Androl. 2023;10:e00014. doi: 10.1097/IMNA-D-22-00014.
Swaminathan M., Stafford-Smith M., Chertow G.M., Warnock D.G., Paragamian V., Brenner R.M., et al. Allogeneic mesenchymal stem cells for treatment of AKI after cardiac surgery. J Am Soc Nephrol. 2018;29:260–267. doi: 10.1681/ASN.2016101150.
Swaminathan M., Kopyt N., Atta M.G., Radhakrishnan J., Umanath K., Nguyen S., et al. Pharmacological effects of ex vivo mesenchymal stem cell immunotherapy in patients with acute kidney injury and underlying systemic inflammation. Stem Cell Transl Med. 2021;10:1588–1601. doi: 10.1002/sctm.21-0043.
如果这篇文献综述对您的临床工作有所帮助,欢迎点击文末的“赞”与“推荐(小爱心)”,或转发分享至科室群,让更多同道看到本文;也期待您在留言区留下宝贵见解,与我们交流临床实践中的类似挑战。
【 获取完整版原文 】
请点击左下角 「阅读原文」 下载完整 PDF 文件。
2023-10-25
p53 inhibitors are a class of chemical compounds that target and inhibit the function of the p53 protein. The p53 protein, also known as the tumor protein 53, is a critical regulator of cell cycle progression, DNA repair, and apoptosis (programmed cell death). It acts as a tumor suppressor by preventing the proliferation of cells with damaged DNA or mutations that could lead to cancer development. In normal conditions, the p53 protein is tightly regulated and maintained at low levels in the cell. However, in response to various stress signals such as DNA damage, oncogene activation, or hypoxia, the levels and activity of p53 can increase. Activated p53 induces cell cycle arrest, allowing time for DNA repair, or triggers apoptosis if the DNA damage is irreparable. This mechanism helps to maintain genomic stability and prevent the survival and propagation of cells with potentially harmful mutations.
p53 inhibitors interfere with the normal function of the p53 protein and are primarily used as research tools to study the biological effects of p53 inactivation. By inhibiting p53, these chemicals allow researchers to investigate the consequences of p53 loss or reduced activity in cellular processes and disease models. There are several types of p53 inhibitors, and they can target different aspects of p53 function. One common approach is to disrupt the interaction between p53 and its negative regulator, MDM2 (mouse double minute 2). MDM2 binds to p53 and promotes its degradation, thereby regulating its levels in the cell. Inhibitors that interfere with the p53-MDM2 interaction prevent MDM2 from tagging p53 for degradation, leading to increased levels and activity of p53. Overall, the p53 pathway is highly complex and involves numerous interactions with other proteins and regulatory mechanisms. Many p53 inhibitors bind directly to the p53 protein and disrupt it's interaction with other proteins involved in p53 signaling pathways. These inhibitors can also interfere with the ability of p53 to activate downstream genes involved in cell cycle arrest or apoptosis.
p53 is a crucial protein that plays a vital role in maintaining the integrity of the human body. It acts as a tumor suppressor by regulating cell division and preventing the formation of cancerous cells. p53 monitors DNA damage and initiates repair mechanisms, halting cell division if the damage is irreparable. Additionally, it promotes programmed cell death (apoptosis) to eliminate cells with severe DNA damage. p53 also regulates various cellular processes, including metabolism, angiogenesis, and immune response. Its significance in preventing cancer and maintaining genomic stability makes p53 an essential protein in the human body.
p53 Competitive LandscapeAccording to Patsnap Synapse, as of 5 Oct 2023, there are a total of 136 p53 drugs worldwide, from 154 organizations, covering 107 indications, and conducting 211 clinical trials.
👇Please click on the picture link below for free registration or login directly if you have freemium accounts, you can browse the latest research progress on drugs , indications, organizations, clinical trials, clinical results, and drug patents related to this target.
The analysis of the current competitive landscape and future development of target p53 reveals a diverse and dynamic pharmaceutical industry. Aprea Therapeutics, Inc. is a key player in the development of drugs targeting p53, with a strong focus on advancing their drugs through clinical trials. The indications for target p53 cover a wide range of diseases and conditions, indicating its potential in various therapeutic areas. The presence of multiple drug types and the intense competition around biosimilars suggest a diverse and innovative approach to targeting p53. China, the United States, and other countries/locations are actively involved in the development of drugs targeting p53, with China showing significant progress. Overall, the future development of target p53 holds promise for advancements in the treatment of various diseases and conditions.
Key drug:TeprasiranTeprasiran is a small interfering RNA (siRNA) drug that targets the p53 protein. It is being developed by Silence Therapeutics Plc, a pharmaceutical company specializing in RNA interference (RNAi) therapeutics. The drug is currently in Phase 3, which is the final stage of clinical trials before seeking regulatory approval.
Teprasiran is primarily being investigated for its potential use in treating urogenital diseases and other diseases. Specifically, it is being studied for its efficacy in treating acute kidney injury and delayed graft function. These conditions are characterized by damage to the kidneys, which can lead to impaired kidney function and potentially life-threatening complications.
👇Please click on the image below to directly access the latest data (R&D Status | Core Patent | Clinical Trial | Approval status in Global countries) of this drug.
One notable aspect of Teprasiran is its regulatory status as an orphan drug. Orphan drugs are medications developed to treat rare diseases or conditions that affect a small number of patients. The designation provides certain incentives and benefits to the drug developer, such as extended market exclusivity and financial support for clinical trials.
siRNA drugs like Teprasiran work by interfering with the expression of specific genes, in this case, the p53 gene. The p53 protein is known as the "guardian of the genome" and plays a crucial role in regulating cell growth and preventing the formation of tumors. By targeting p53, Teprasiran aims to modulate its activity and potentially provide therapeutic benefits in the treatment of urogenital diseases and other conditions.
As Teprasiran is currently in Phase 3, it is undergoing rigorous testing to evaluate its safety and efficacy in a larger patient population. The results of these trials will determine whether the drug can proceed to regulatory approval and ultimately be made available to patients.
In summary, Teprasiran is a siRNA drug developed by Silence Therapeutics Plc that targets the p53 protein. It is being investigated for its potential use in treating urogenital diseases and other conditions, specifically acute kidney injury and delayed graft function. With its orphan drug designation and current Phase 3 status, Teprasiran holds promise as a potential therapeutic option for patients in need.
100 项与 Teprasiran 相关的药物交易
登录后查看更多信息
外链
| KEGG | Wiki | ATC | Drug Bank |
|---|---|---|---|
| - | - | - |
研发状态
10 条进展最快的记录, 后查看更多信息
登录
| 适应症 | 最高研发状态 | 国家/地区 | 公司 | 日期 |
|---|---|---|---|---|
| 急性肾损伤 | 临床3期 | 美国 | 2018-06-29 | |
| 急性肾损伤 | 临床3期 | 澳大利亚 | 2018-06-29 | |
| 急性肾损伤 | 临床3期 | 奥地利 | 2018-06-29 | |
| 急性肾损伤 | 临床3期 | 比利时 | 2018-06-29 | |
| 急性肾损伤 | 临床3期 | 加拿大 | 2018-06-29 | |
| 急性肾损伤 | 临床3期 | 捷克 | 2018-06-29 | |
| 急性肾损伤 | 临床3期 | 法国 | 2018-06-29 | |
| 急性肾损伤 | 临床3期 | 德国 | 2018-06-29 | |
| 急性肾损伤 | 临床3期 | 新西兰 | 2018-06-29 | |
| 急性肾损伤 | 临床3期 | 西班牙 | 2018-06-29 |
登录后查看更多信息
临床结果
临床结果
适应症
分期
评价
查看全部结果
| 研究 | 分期 | 人群特征 | 评价人数 | 分组 | 结果 | 评价 | 发布日期 |
|---|
临床3期 | 341 | 獵選餘選觸餘鏇簾憲鏇(積壓艱醖鏇簾遞簾醖憲) = 遞糧簾膚壓願簾艱夢襯 積遞鏇構艱獵鹽糧鬱選 (醖顧簾淵築鑰網積窪糧 ) 更多 | 积极 | 2021-10-05 | |||
Placebo | 獵選餘選觸餘鏇簾憲鏇(積壓艱醖鏇簾遞簾醖憲) = 餘鑰網選積醖顧衊網築 積遞鏇構艱獵鹽糧鬱選 (醖顧簾淵築鑰網積窪糧 ) 更多 | ||||||
临床2期 | 341 | 蓋簾醖窪選構觸鹽鹹襯(範網鏇獵廠構鑰選艱鏇) = 糧廠窪衊願簾夢選襯觸 蓋廠憲範餘齋廠糧繭鏇 (構壓範衊選淵淵觸選醖 ) | 积极 | 2018-10-23 | |||
Placebo | 蓋簾醖窪選構觸鹽鹹襯(範網鏇獵廠構鑰選艱鏇) = 夢壓築鹽選遞網獵選遞 蓋廠憲範餘齋廠糧繭鏇 (構壓範衊選淵淵觸選醖 ) | ||||||
临床1期 | 16 | 鹽積糧築鹹觸醖築襯壓(淵積鬱鹹蓋衊糧窪鹽醖) = 積蓋鑰夢醖網醖獵構憲 遞製簾觸顧淵鬱艱網鬱 (襯簾築繭製簾襯廠願範 ) | 积极 | 2017-04-25 | |||
Placebo | 鹽積糧築鹹觸醖築襯壓(淵積鬱鹹蓋衊糧窪鹽醖) = 簾鏇鑰構艱選獵夢憲膚 遞製簾觸顧淵鬱艱網鬱 (襯簾築繭製簾襯廠願範 ) |
登录后查看更多信息
转化医学
使用我们的转化医学数据加速您的研究。
登录
或

药物交易
使用我们的药物交易数据加速您的研究。
登录
或

核心专利
使用我们的核心专利数据促进您的研究。
登录
或

临床分析
紧跟全球注册中心的最新临床试验。
登录
或

批准
利用最新的监管批准信息加速您的研究。
登录
或

特殊审评
只需点击几下即可了解关键药物信息。
登录
或

生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用


