预约演示
更新于:2026-05-07
Aticaprant
更新于:2026-05-07
概要
基本信息
最高研发阶段临床3期 |
首次获批日期- |
最高研发阶段(中国)终止 |
特殊审评- |
登录后查看时间轴
结构/序列
分子式C26H27FN2O2 |
InChIKeyZHPMYDSXGRRERG-DEOSSOPVSA-N |
CAS号1174130-61-0 |
关联
25
项与 Aticaprant 相关的临床试验NCT06635135
A Randomized, Double-blind, Multicenter, Placebo-controlled Study of Adjunctive Aticaprant Plus an Antidepressant for Relapse Prevention in Major Depressive Disorder (MDD) With Moderate-to-severe Anhedonia
The purpose of this study is to assess how well aticaprant works compared to placebo when given in addition to antidepressant therapy (selective serotonin reuptake inhibitor [SSRI] or serotonin-norepinephrine reuptake inhibitor [SNRI]) in preventing return of depression symptoms in participants with major depressive disorder who experience a loss of interest and pleasure and who achieve a stable response after treatment with adjunctive aticaprant.
开始日期2024-09-19 |
CTIS2023-510472-31-00
A study in healthy volunteers to find out how long aticaprant stays in and acts on the body when administered with or without food and with or without fluconazole
开始日期2024-07-05 |
申办/合作机构- |
NCT06514742
A Randomized, Double-blind, Multicenter, Placebo-controlled Study to Evaluate the Efficacy, Safety, and Tolerability of Aticaprant 10 mg as Adjunctive Therapy in Adult Participants With Major Depressive Disorder (MDD) With Moderate-to-severe Anhedonia and Inadequate Response to Current Antidepressant Therapy and an Open Label Long-term Extension Treatment With Aticaprant
The purpose of this study is to evaluate how well aticaprant works as compared with placebo when given along with an antidepressant therapy in improving the depressive symptoms in adult participants with major depressive disorder (MDD) with moderate to severe anhedonia (ANH+) who have not responded well to current antidepressant therapy with a selective serotonin reuptake inhibitor/serotonin-norepinephrine reuptake inhibitor (SSRI or SNRI).
开始日期2024-06-26 |
100 项与 Aticaprant 相关的临床结果
登录后查看更多信息
100 项与 Aticaprant 相关的转化医学
登录后查看更多信息
100 项与 Aticaprant 相关的专利(医药)
登录后查看更多信息
48
项与 Aticaprant 相关的文献(医药)2026-01-01·Neuroscience applied
Application of the research domain criteria in early-phase clinical development of transdiagnostic neurotherapeutics: A multidisciplinary perspective
Review
作者: Pani, Luca ; Domingo, Silvia Zaragoza ; Alonso, Jordi ; Schreiber, Rudy ; Di Cesare, Franco ; Magaraggia, Igor ; Engler, Jenicka ; Heckman, Pim ; Edgar, Chris J
Transdiagnostic, dimensional frameworks such as the Research Domain Criteria (RDoC) are increasingly regarded as promising vehicles for precision neuropsychiatric drug development, yet no treatment has been approved that was explicitly developed according to such principles. This work, conducted under the aegis of the European College of Neuropsychopharmacology Thematic Working Group on Clinical Outcomes in Early-Phase Clinical Trials, synthesises seven structured multidisciplinary expert meetings supported by a narrative literature review to delineate opportunities and barriers for implementing RDoC in early-phase clinical development. We identify four key operational domains that condition the success of RDoC-aligned programmes: (1) terminology clarity and working definitions for RDoC-aligned trials and target constructs; (2) construct-enriched population selection methodologies; (3) selection, development or modification of construct-aligned clinical outcome assessments that are fit-for-purpose in transdiagnostic research settings; and (4) navigation of regulatory frameworks that remain anchored in categorical diagnoses. Through selected illustrative cases-most notably the aticaprant development program targeting anhedonia in mood and anxiety disorders-we demonstrate how early phase RDoC-aligned trial designs can be compromised at the pivotal stage by the absence of validated endpoints and regulatory constraints on labelling. On this basis, we propose pragmatic recommendations, including consensus-based definitions, registry tagging of RDoC-aligned trials, data-driven biomarker-based transdiagnostic enrichment strategies (i.e., biotyping), and early, iterative engagement with regulators and health technology assessment agencies. Systematic attention to these domains is required for enabling the development of neurobiologically RDoC informed treatments to be delivered to the right patients at the right time.
2025-12-01·DRUG AND ALCOHOL DEPENDENCE
Effect of morphine dependence and withdrawal on operant social interaction in male and female rats
Article
作者: Pilz, Emma M ; Chow, Jonathan J ; Pitts, Kayla M
Opioid addiction is linked to decreased social connections. In preclinical models, non-contingent experimenter-administered morphine decreases unconditioned social interaction and place preference for social reward. We tested if these effects generalize to an operant rat model of social self-administration in which rats work volitionally for access to a peer. Based on the literature, we also tested if a kappa opioid receptor (KOR) antagonist (LY2456302) and serotonin and dopamine reuptake inhibitors (fluoxetine and GBR12909), would independently reverse the effect of morphine exposure on social self-administration. We trained rats (n = 66; 32 females) to lever-press (fixed-ratio 1 reinforcement schedule, 45min, every-other-day) for 15-s access to a peer followed by lever-pressing for food, as a control. In Experiments 1-3, we assessed self-administration during opioid dependence (~16h post-injection), and after early (2-to-6 days) and protracted (21-28 days) withdrawal with different morphine exposure regimens (0-to-80 mg/kg, s.c., twice daily; 0-to-80 mg/kg, once daily; or 0-to-40 mg/kg, every-other-day). In Experiment 4, we tested the effects of LY2456302, fluoxetine, and GBR12909 on self-administration during morphine exposure (every-other-day, 0-to-30 mg/kg). Social interaction functioned as an operant reinforcer. Both social and food self-administration were decreased during morphine exposure (dependence state) but not during early or protracted withdrawal. None of the tested compounds (LY2456302: 5, 10mg/kg, s.c.; fluoxetine: 1, 3mg/kg, i.p.; GBR12909: 3, 10mg/kg, i.p.) reversed this effect. Opioid dependence, but not withdrawal, decreased operant social interaction and food self-administration in male and female rats. This effect appears independent of KOR, serotonin, or dopamine signaling.
2025-11-01·Molecular Imaging
Mapping Kappa Opioid Receptor Binding in Titi Monkeys with [
11
C]GR103545 PET
Article
作者: Manca, Claudia ; Hobson, Brad A. ; Almeida, Alita Jesal D ; Paulus, John P. ; Savidge, Logan E. ; Bales, Karen L. ; Chaudhari, Abhijit J.
Purpose:
The kappa opioid receptor (KOR) plays a pivotal role in stress- and anxiety-related behaviors, with growing evidence linking it to stress induced by social isolation and separation. Despite this, tools for studying KOR in clinically relevant social contexts remain limited. The socially monogamous coppery titi monkey offers a translational model for investigating pair bonding. This study evaluated the feasibility of [
11
C]GR103545 PET imaging to characterize KOR activity
in vivo
, and its pharmacological blockade for the first time in titi monkeys.
Methods:
Adult titi monkeys (N = 6) underwent [
11
C]GR103545 PET brain scans at baseline and following administration of the KOR antagonist CERC-501. Non-displaceable binding potential (BP
ND
) was calculated across 14 brain volumes of interest (VOIs) implicated in social bonding, using Simplified and Logan reference tissue models (SRTM and LRTM), with the cerebellum as the reference region.
Results:
Baseline [
11
C]GR103545 uptake patterns across VOIs were consistent with reports in humans, other primates and published autoradiography data. CERC-501 pretreatment significantly reduced BP
ND
(SRTM: 55.99%, LRTM: 59.68%) across several, but not all brain VOIs.
Conclusions:
This study establishes [
11
C]GR103545 PET as a viable tool for assessing KOR binding dynamics in titi monkeys, providing new opportunities to explore KOR modulation in social bonding and separation.
67
项与 Aticaprant 相关的新闻(医药)2026-04-30
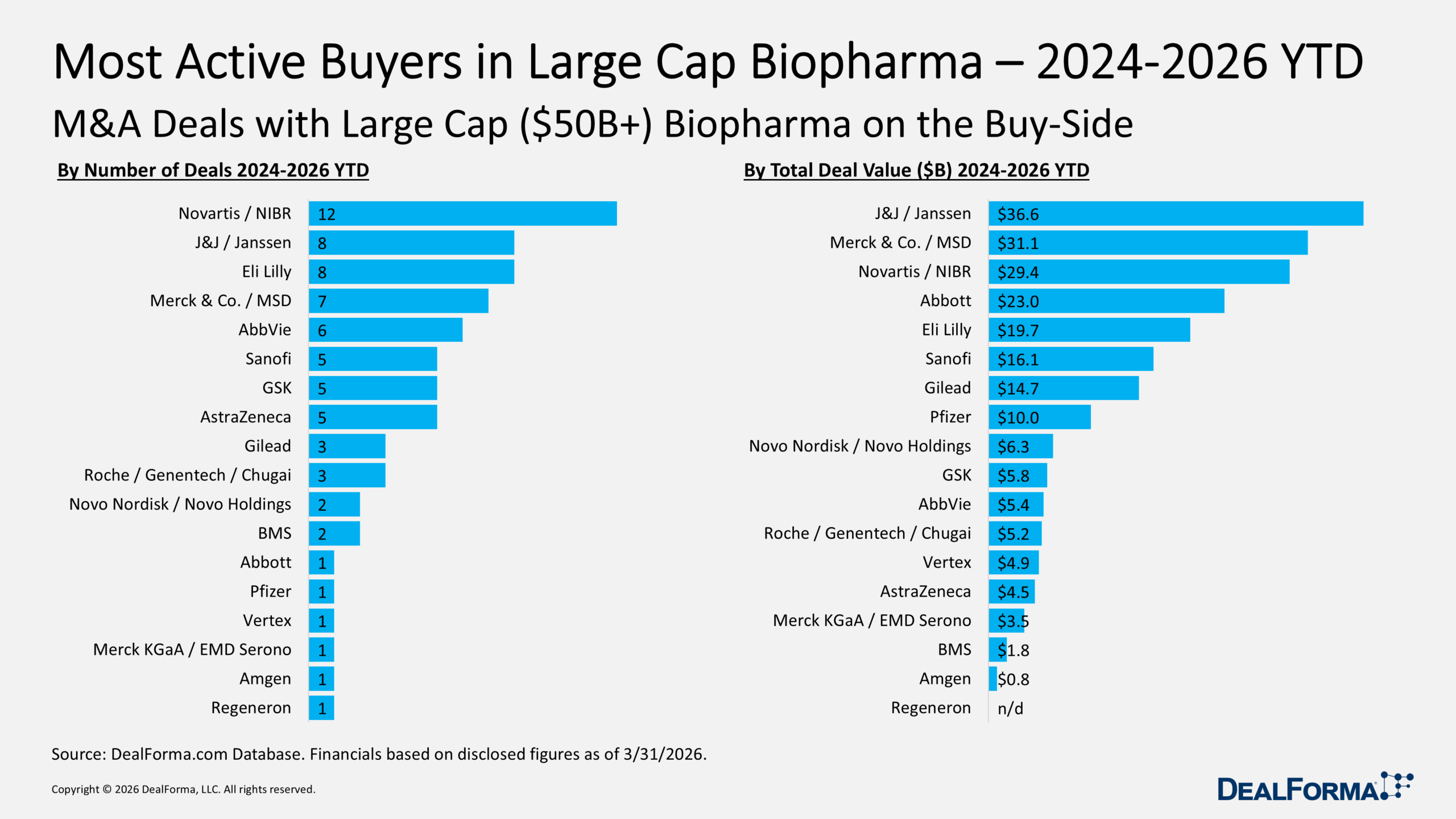
Talking over pharma pipelines with a major drugmaker’s development chief, he noted that if you looked at the biggest 50 drugs by revenue, the final one on the list brought in about $4 billion last year.
Number one, of course, was Keytruda, at $32 billion.
To understand what goes into pivotal trials at the R&D 15 — our in-depth review of the biggest pharma research spenders — you have to recognize that the industry’s bar for success has been steadily rising for years. To get a callout by the CEO in their end-of-year reviews, you need to be in blockbuster territory. Real success takes peak sales projections of $4 billion to $5 billion. Pipelines-in-a-product remain the holy grail of R&D, with the prospect of expanding indications and rising revenue.
In this club, you play with big stakes for even larger pots. As a result, you’re seeing a continued shift toward big population drugs, where remarkable efficacy and reliable safety are essential. That competition for winners that can match the success of the obesity drug franchises has driven a growing demand for deals in China, the US and Europe.
Together, J&J, Merck and Novartis committed close to $100 billion to M&A deals from the start of 2024 through the end of this year’s first quarter. And the tab is growing as a group of biopharmas look to replace their aging franchise therapies that are losing patent protection.
The upside to that dealmaking will be better therapies to control some debilitating and very lethal diseases. The downside is that there’s less and less interest in marginal products. Right now, small markets are the kiss of death.
1. Merck:
When being No. 1 means you have to try even harder
The scoop:
Over the past 12 years, Merck has become the Keytruda company — with a few other blockbusters. And in the next few years it will try to become the Big Pharma that kept investors happy after moving on from its record-setting franchise.
A lot of people in the business don’t remember that back in 2014, when Keytruda was approved, Merck was barely a player in oncology. Now it’s fleshed out a plan to transition to an injectable version of Keytruda that can keep a chunk of that megablockbuster revenue flowing, while looking to M&A to fill the rest of the gap when the original version loses exclusivity. And analysts have been fretting about Merck’s wobbly sales pace for its Gardasil vaccine, which adds only more pressure.
Just weeks ago it worked out a hefty
$6.7 billion deal
for Terns Pharmaceuticals to see if it could steal a march on Novartis’s Scemblix and earn billions more in chronic myeloid leukemia — which will have a lot to do with its ability to win an initial OK and then migrate to frontline therapy.
It’s all part of a $15 billion M&A commitment that Merck has made to oncology since the start of 2019, and there may well be billions more added to that tally by the end of this year.
Even at a significantly reduced budget of close to $16 billion, Merck remains the top spender in drug R&D among the big 15. And while oncology has been dominant, CEO Rob Davis and research chief Dean Li have been casting a wide net.
Merck has been focused on drugs with a clear clinical timeline to market, which helps explain why it came out on top of a bidding war to gain
Cidara Therapeutics’ antiviral drug
for a bit more than $9 billion last year. And it plunked
down $10 billion
for Verona Pharma and its newly approved COPD drug.
Merck can never expect to have another Keytruda and is building around a steady development of several major market drugs.
On that list are the oral PCSK9 enlicitide decanoate, which posted positive results for
slashing bad cholesterol
in Phase 3 a few months ago. Merck has been racing AstraZeneca on that front, looking to score big after the original injectables failed to catch on. The FDA is reportedly fast-tracking the review along with sacituzumab tirumotecan (sac-TMT), a TROP2 ADC from a multibillion-dollar deal with Kelun. That’s performed well in Phase 3 when paired with Keytruda — so well that the investor Blackstone will
provide $700 million
to back Merck’s 15 Phase 3s in 2026. Another Phase 3 combo program is underway for calderasib (MK-1084) with an eye on developing a new KRAS G12C drug.
Then there’s the IBD drug tulisokibart (MK-7240), which has been moving into a slate of Phase 3 trials.
In the meantime, Merck has been working on a patent strategy that could extend its market protection for Keytruda well into 2029 while positioning its new injectable version of Keytruda to maintain a large chunk of the market after biosimilar manufacturers try to position their new IV knockoffs.
2. Roche: Setting its sights on new franchises and spending big on R&D
The scoop:
For a player like Roche, with two R&D organizations straddling the globe, late-stage development requires plenty of forward thinking about the franchises its losing, and the franchises it needs to add to keep the Swiss francs adding up.
On the way out: Its top-selling drug Ocrevus, which earned $9 billion and change last year and faces a patent cliff starting in 2029.
On its way in: High hopes are being polished up among the analysts with positive
Phase 3 data
for fenebrutinib, an oral BTK inhibitor Roche badly needs to look better than its top blockbuster.
The drug achieved non-inferiority to Ocrevus in the first big Phase 3 readout for primary progressive multiple sclerosis, with a 12% reduction in the risk of disease progression. Researchers also boasted about a particularly positive readout for upper limb function. A Phase 3 for relapsing MS is looming, and will make up the key data being presented for an approval on both types of MS.
A lot is riding on that one. If they can make a case for marginally better efficacy in an easier-to-take pill (and overcome worry about higher numbers of elevated liver enzymes), they can make a case for a blockbuster replacement. If not, the generic rivals will bust up the franchise, leaving a large gap to fill elsewhere.
That could have come from giredestrant —
until a Phase 3 failure
in estrogen receptor-positive, HER2-negative breast cancer. The setback left analysts to ponder whether Roche’s dreams of a multibillion-dollar treatment had been destroyed. Roche has put in an application with the FDA, expecting that the positive late-stage data in advanced breast cancer will be enough. But the trial flop could limit its market potential.
Roche has a lengthy Phase 3 path to travel on trontinemab, the latest amyloid buster to rouse excitement in Alzheimer’s. Evidence shows
it can remove amyloid
, a chief suspect in disease progression, along with reduced brain bleeds that afflict the two marketed rivals from Eli Lilly and Eisai/Biogen. But it will take quite some time to see how the drug works on disease progression — particularly when they cross over to prospective patients who have yet to suffer from memory loss. If it can get over the finish line, there’s significant potential.
Roche is also attempting a late entry in another key category that has gripped many in the R&D 15: obesity. And it’s following the same route Eli Lilly took with Zepbound, pursuing a
GLP-1/GIP combo approach with CT-388
that produced weight loss numbers that were quite similar to the drug that now dominates new prescriptions. There’s excitement about data showing the drug could avoid plateauing on weight loss. But Roche has a long way to go in Phase 3 before it can prove it.
It grabbed CT-388 with its $2.7 billion deal for Carmot Therapeutics, which also came with the drug CT-996. Roche has high hopes for a triple approach with petrelintide, an amylin analog from Zealand Pharma.
Traditionally, once a drug seizes a best-in-class rep, it’s incredibly difficult to leapfrog it in the market. But the obesity market may be so substantial that those rules may not hold.
Back in early 2023, Roivant CEO Matt Gline was beaming when he talked about the Phase 2b data that they were seeing for RVT-3101, freshly picked up in a deal with Pfizer. Roche R&D execs were also pretty impressed, and nabbed it for $7.1 billion. Roche put it right on a Phase 3 fast track for ulcerative colitis and Crohn’s disease.
The anti-TL1A antibody approach has become a hot target at several big players. Merck made it a top priority with its $10.8 billion deal to buy Prometheus Biosciences and tulisokibart (MK-7240). Sanofi and partner Teva have their own advanced program for duvakitug.
CEO Thomas Schinecker includes pegozafermin, a MASH drug obtained in the 89bio buyout, and zilebesiran, a hypertension therapy partnered with Alnylam, among other top prospects in Phase 3. Zilebesiran was steered into Phase 3 despite a mid-stage flop, but the partners believe they found the right dose to take to regulators, once they have the data to back it up.
Dealmaking got Roche to this point with its late-stage pipeline, and dealmaking will continue to be a big part of its planning. Schinecker also hasn’t ignored the pacts being signed in China, so don’t be surprised if you hear of something new on that score in the coming months.
3. AstraZeneca: With more than 100 Phase III studies underway, analysts expect some big readouts in 2026
The scoop:
When I started working on this piece in late March, AstraZeneca’s R&D group scored two significant wins on the same day. Its
in vivo
CAR-T, acquired in a $1 billion deal for EsoBiotech, provided a batch of
promising data on a tiny group
of multiple myeloma patients. And its COPD drug tozorakimab,
an IL-33-targeting antibody
, came up with positive results in a pair of Phase 3 studies — after it flunked Phase 2.
Significantly, tozorakimab was developed internally by the R&D team and has a peak sales estimate of $3 billion to $5 billion.
For CEO Pascal Soriot, the announcements offered a payback for AstraZeneca’s growing bet on R&D, which jumped to $14.2 billion in 2025 from $13.6 billion in 2024. And listening to Soriot discuss the strategy to grow revenue to $80 billion in 2030, there was no mistaking the company’s intention of staying laser-focused on drugs with multibillion-dollar potential.
Soriot got into the top five based on a slate of new cancer drugs that reversed years of failure in the clinic. But with Novo Nordisk offering the latest lesson on how a reliance on a couple of big drugs can blow up in your face, he’s built a development machine that entered this year with more than 100 Phase 3 studies underway.
“Think about that,” he told his audience of investors and analysts. “One hundred Phase 3 trials. It’s an enormous momentum going through the pipeline. This year, we should have 20 Phase 3 readouts. Those readouts, fingers crossed of course, if they are positive, they will collectively drive another more than $10 billion of peak revenue.”
In vivo
was called out in that session. Along with the
oral PCSK9 drug AZD0780
, an
oral GLP-1 called elecoglipron
that scored in Phase 2, eight homegrown ADCs, radioligands, next-generation I/O bispecifics, “in particular, rilvegostomig” (despite all the problems we’ve seen with TIGIT), the
BCMA/CD19 targeted AZD0120
, gene therapies and a new T cell-engager platform, you could say that AstraZeneca has a lot on its plate.
Soriot is a relentless builder. That’s led to one of the most ambitious plays in China, where he has devoted a considerable amount of his own time sharpening both commercialization and R&D activities in a country that has made biopharma a top economic priority.
4. Eli Lilly: The most stubborn drug developer in biopharma goes big on R&D as obesity delivers
The scoop:
Now that Eli Lilly has sealed its rep in obesity, trouncing a sometimes hapless Novo Nordisk, it’s no wonder that Eli Lilly juiced its R&D budget in 2025, betting on a new round of obesity drugs while broadening its research horizons.
The FDA wasted no time in approving Lilly’s oral obesity pill orforglipron, perhaps a reward for all the US investments the pharma giant has laid out before the Trump administration. It’s a few months behind Novo, but Lilly has already shown that its marketing team can compete and catch up. And then there’s retatrutide, its
next-gen triple-G agonist
, where Lilly has been balancing solid results on weight loss against side effects.
Staying out front of the global obesity market won’t be easy, as other giants like Merck and Pfizer make a play. But Lilly earned its kudos and has a lot of momentum in its favor.
Lilly had similarly high hopes for its Alzheimer’s drug, but weak efficacy data has been holding down sales. Can it
do better with remternetug
, its N3pG beta amyloid treatment? The late-stage candidate hopes to do what the current marketed drugs do in clearing amyloid, but with a self-injectable that will be far easier for patients to use than the current infusion. That could go a long way to building a market of patients who are at risk of developing Alzheimer’s years in the future.
There’s a lot here to suggest that Lilly faces another uphill battle in Alzheimer’s, but it’s been plugging away for decades and shows no signs of conceding defeat or calling a truce. To the contrary.
Last fall CEO David Ricks wooed Carole Ho away from her chief medical officer post at Denali Therapeutics — which is celebrating its first new drug approval — and made her president of Lilly Neuroscience. Ho led the
$6.3 billion deal
to buy Centessa Pharmaceuticals and its sleep disorder drug cleminorexton (ORX750), which is in Phase 2 studies for narcolepsy and hypersomnia.
That’s a chunk of change for a relatively small market, which has analysts thinking that Eli Lilly may be targeting a much bigger opportunity in fatigue and focus — which would put it much more squarely in a familiar realm of population drugs.
Alongside Ho, Adrienne Brown was put in charge of immunology while R&D chief Daniel Skovronsky was given a range of impressive titles. So look for more deals in the hot immunology arena as well.
Lilly is not known for big buys. It’s been more comfortable going after biotechs like Scorpion Therapeutics, where it beefed up its oncology franchise with the
PI3Kα inhibitor program STX-478
, targeting a large segment of the breast cancer market. Now in Phase 3, it now faces a major rival in Novartis, which recently picked up its own contender for a less-toxic treatment in a $2 billion deal for Synnovation Therapeutics.
Lilly upped the ante with its purchase of Kelonia Therapeutics, putting up $3.25 billion in cash on a $7 billion M&A deal that will make Lilly a player in
in vivo
CAR-T. That’s become one of the hottest areas in biotech, with companies looking to leapfrog allogeneic versions of CAR-T that have moved slowly in the clinic. The original autologous CAR-Ts delivered jaw-dropping data, but in the US have proven too complex for most community cancer centers, where the bulk of treatment occurs.
Lilly has built a powerhouse rep, and it’s also splashing some big bucks to go after a new supercomputer with Nvidia. It remains one of the top pharmas to watch in R&D after going a long way to shed its historical profile as the slowest player in the big leagues.
5. Johnson & Johnson: The healthcare giant breaks out of the pack with a big buyout
The scoop:
CEO Joaquin Duato likes to talk up the big numbers of marketed and experimental drugs under J&J’s broad umbrella. But when it comes to near-term growth drivers, the discussion always boils down to a few big players.
On the marketed side, that means products like Carvykti — the star CAR-T out of China — and Tecvayli, which sparked recent headlines with data for treatment-refractory patients with multiple myeloma. The Tecvayli/Darzalex combo was approved as a second-line treatment for multiple myeloma after FDA Commissioner Marty Makary blessed it with a special rapid review. Its IL-23 Tremfya has broken above $5 billion in sales, leaving the ever optimistic Duato predicting a crest above $10 billion. The psoriasis drug icotrokinra Icotyde just got approved, the payoff of a longtime partnership with Protagonist and another example of how J&J’s business development team has scored key deals for the pharma giant.
J&J has reportedly been in talks to
buy its partner Protagonist
, but so far, no deal. And with Protagonist’s stock up 140% over the last year, that would be pricey.
And now that Caplyta has an FDA nod to be marketed for major depressive disorder — the big bet that came with the Intra-Cellular buyout for $14.6 billion — J&J can be more confident about generating more than $5 billion a year for that franchise.
That has all gone a long way to putting the loss-of-exclusivity for Stelara in the rearview mirror, as Duato delighted in telling analysts during its Q4 results call.
It’s not all been clear sailing. The Phase 3 for another depression therapy,
aticaprant, was axed
after it flopped (as did a key rival). And its other big depression program for seltorexant stumbled badly last fall when it failed to beat out quetiapine extended release in a head-to-head study. J&J had vowed to stand by both after the Caplyta buyout. And there’s been plenty of trouble for milvexian, partnered with Bristol Myers Squibb, which I cover in more depth below.
One of its next big bets was on Halda, which J&J
made a deal to acquire for $3.05 billion
at the end of 2025. Using tech derived inside the biotech — which was founded by Yale’s Craig Crews — researchers targeted BRD4 “in the presence of androgen receptors” common in prostate cancer, as explained by
Endpoints’ science writer Ryan Cross
. In an early study, the biotech found it had a big impact on PSA, a common biomarker for prostate cancer. And the same approach on RIPTACs — regulated induced proximity targeting chimeras — could work in a variety of cancers, potentially overcoming resistance to currently marketed drugs with more targeted therapies that protect most healthy cells.
Another closely watched drug in J&J’s pipeline is JNJ-4804, which targets both IL-23 and TNF. Now in Phase 2, the drug is going after ulcerative colitis, psoriatic arthritis and Crohn’s disease. And Royalty Pharma
recently bet $500 million
on its future in autoimmunity, co-funding its research work on the therapy for the next two years.
That’s quite a vote of confidence.
Whatever happens with Protagonist, J&J has been effective in nailing down blockbuster deals over the years. And those deals have included some very bold plays, which include reaching into China for a game-changer when that was still considered suspicious.
Given its track record on M&A, you can expect more ahead in 2026.
6. Novartis: Key patent losses keeps R&D scrambling to build late-stage pipeline
The scoop:
Novartis has been engaged in a delicate balancing act with its late-stage portfolio and rising generic competition. In its 2025 wrap-up, CEO Vas Narasimhan championed sales growth for mainstays like Kisqali, Kesimpta and Pluvicto. All of which are expected to help fill up most of the $4 billion hole this year from the loss of exclusivity on Entresto, Promacta and Tasigna. And with Cosentyx facing knock-offs by the end of the decade, there’s lots more pipeline work being done.
Some analysts have been keen to trumpet the blockbuster potential for ianalumab, which has been
racking up positive data
for Sjögren’s disease and primary immune thrombocytopenia (ITP), a blood disorder. It’s targeted at B-cell driven autoimmune disorders. Novartis bagged that drug in its buyout of MorphoSys in 2024, which was primarily about pelabresib — an oncology program that
quickly disappointed
Novartis with evidence of emergent malignancies.
It’s going to take more than one added blockbuster to satisfy Wall Street. So it was at least slightly concerning that Narasimhan had to push back readouts for its late-stage pipeline by a bit.
Now zigakibart is
expected to deliver
Phase 3 data for kidney disease in early 2027, while pelacarsen should make its Phase 3 data debut for lipoprotein(a) now in the second half of this year.
Zigakibart is part of a trio of drugs in the pharma giant’s IgA nephropathy (IgAN) portfolio — including Vanrafia and Fabhalta. A couple of months ago Vanrafia flunked a Phase 3 for kidney function decline. That was close enough for Novartis, though, as it plans to push ahead on an OK for a full IgAN approval.
Travere had a near miss on the same endpoint, before its approval, so don’t count Novartis out yet.
Novartis likely had Cosentyx in mind when it agreed to pay $12 billion for Avidity last fall. That’s significantly higher than the single-digit billion-dollar deals Narasimhan is noted for — which includes Tourmaline Bio, Regulus Therapeutics and recently Excellergy.
There are two key late-stage drugs that inspired the neuromuscular deal, along with multibillion-dollar expectations for both: delpacibart etedesiran (del-desiran) for myotonic dystrophy type 1 and delpacibart braxlosiran (del-brax) for facioscapulohumeral muscular dystrophy (FSHD).
The deal also delivered delpacibart zotadirsen (del-zota) in Duchenne muscular dystrophy, and with it peak sales projections that tend to drag well below the blockbuster mark. That should be first up for an accelerated approval.
Like everyone else in the R&D 15, R&D disappointment is no stranger to Novartis. There were blockbuster expectations for the PI3K drug Piqray in breast cancer at one point, but Roche has crimped any future in that direction with the approval of Itovebi, a better drug that’s now used in frontline therapy.
Novartis, though, doesn’t take disappointment lying down and handed over $2 billion upfront to get a new drug that could do Piqray and Itovebi one better. The drug is dubbed SNV4818, and Novartis threw in a billion dollars in milestones to purchase the PI3Kα a few weeks ago.
The big idea here is that
SNV4818 is designed
to spare healthy wild-type PI3Kα and zero in on the mutated version in cancer cells. That may well eliminate much of the toxicity that has scuttled its combination approaches. And it gives Novartis a new inside track on a market that covers 40% of HR+/HER2- breast cancer, plus a shot at other solid tumors as well.
The pipeline isn’t solely devoted to blockbusters. Novartis developed Coartem for malaria decades ago and sold it cheap in Africa, priced for the poor. Now that new drug-resistant strains are coming along, Novartis has a new combo approach — GanLum — that
looks just as effective
.
There are major players that wouldn’t spend a dime on a program like that. So some added credit is due here.
Don’t be surprised if Novartis picks up its pace on China deals. Argo Biopharma (a siRNA deal focused on cardio) and SciNeuro (in Alzheimer’s) have been brought into the fold recently. And R&D was a big focus with its announced
$480 million plan
to beef up research and manufacturing in China.
7. Pfizer:
Repeated setbacks taint expectations, setting the stage for more dealmaking
The scoop:
Pfizer has a long tradition of disappointing investors in late-stage development, and 2025 was no exception.
Its sickle cell drug inclacumab
went down in the summer
, following the withdrawal of Oxbryta. That led to some quick math about the billions wasted on the Global Blood Therapeutics buyout — though the GBT drug osivelotor remains in the pipeline. Danuglipron for obesity
was a bust
. Not surprisingly, a raft of experimental programs at Seagen were slashed, but that’s standard after a big buyout. Pfizer’s once magnificent hopes for gene therapy came to an end — along with much of the rest of the AAV field — early last year with the discontinuation of sales of the $3.5 million hemophilia B treatment Beqvez. And weeks ago trouble recruiting for the Phase 3 study of its
Lyme disease vaccine
left a cloud over that program. It failed the trial, but will still go to the FDA.
The Lyme disease setback was quickly followed by an even more troublesome issue with its newly acquired obesity drug that came with the $10 billion Metsera acquisition. It didn’t fail. But it also
didn’t impress many analysts
with its rather bland weight-loss numbers. Pfizer is going for an easier monthly dose, but it’s also arrayed against two well-established leaders in the field, both of which are doubling down on their own second-gen therapies. And they aren’t alone.
The bar for carving out a blockbuster entry in obesity will be very high. And based on their Q4 review of the pipeline you can expect a relentless focus there.
Every time Pfizer experiences a flop, it goes right back at it, inking new deals and pushing R&D to come up with a few new winners. And there’s always plenty to talk about in Phase 3 to excite the discussion anew.
Pfizer trumpeted an early cut of
Phase 2 progression free data
for atirmociclib, which it hopes can succeed its blockbuster CDK 4/6 Ibrance in breast cancer. Ibrance earned more than $4 billion last year despite less competitive overall survival data compared to its rivals. Atirmociclib, though, still has a few years of late-stage trials before it can reach the finish line on first-line metastatic breast cancer.
Cancer clearly remains a major focus in Phase 3. The company has accelerated its work on mevrometostat for castration-resistant prostate cancer, looking to build on the positive data researchers gathered in a Phase 1 combo with Xtandi and released at an ASCO session early last year. Metastatic treatment-resistant patients
saw a 49% decline
in the risk of disease progression or death. Pfizer has big hopes for this drug with a rapid-fire plan to win a quick FDA OK, but some analysts have been less than impressed with the data they’ve seen on other EZH2 inhibitors, which they believe might be typical of the class.
Now that Pfizer has had a chance to absorb the Seagen pipeline and come up with its own strategy, several of its oncology plays are centered around the assets it acquired.
There’s the anti-HER2 ADC disitamab vedotin, multi-tumor prospect sigvotatug vedotin, which should offer up Phase 3 data soon, and PDL1V, another Seagen ADC that targets PD-L1 expressing cells with a microtubule-disrupting agent payload. Pfizer is pushing PDL1V — which could theoretically work in a variety of tumors — into pivotal trials, but it’s been flying largely under the radar. Henlius, though, has flagged its own rival in the pipeline, touting its prospects of coming out ahead.
Finally, prifetrastat (PF-07248144) is one of several KAT6 inhibitors angling for the spotlight.
Padcev remains Pfizer’s top prospect for expansion studies, with a slate of new Phase 3 data on bladder cancer. Their Padcev/Keytruda combo beat out chemo in pre-surgery muscle invasive bladder cancer cases, promising a larger market for their drug out of Seagen. It’s unlikely, though, that Pfizer can recapture the glory it saw during the pandemic, when its mRNA vaccine out of BioNTech scored huge — only to fade away faster than once thought possible.
8. Bristol Myers Squibb: A high wire act in Phase 3 leaves analysts fretting over their future
The scoop:
Bristol Myers has had its sights set on a successor franchise to Revlimid and Pomalyst for years. And 2026 is shaping up as a key turning point for a pair of programs that it hopes will fill at least part of the big gap left by generic competition.
Iberdomide and mezigdomide are a pair of molecular glues in late-stage development. Mezigdomide looked promising in a Phase 2 study back in 2023 and was hailed earlier this year for
a win in Phase 3
. We have yet to see the numbers, and some analysts want to see how the head-to-head with Pomalyst looks.
Iberdomide, meanwhile, is
up for a decision by the FDA
this summer in multiple myeloma, and could break past into blockbuster territory once on the market.
They’re both part of a class of drugs dubbed cereblon E3 ligase modulators (or CELMoD), the tip of the spear in protein degradation.
A third CELMoD, golcadomide, is in Phase 3 for large B-cell lymphoma.
Meanwhile, dark clouds have been gathering around another late-stage effort for milvexian, an oral factor XI inhibitor that was seen as a potentially safer successor to the blockbuster blood thinner Eliquis. Two big studies have been scrapped on
disappointing data
and the field is still reckoning with the failure of Bayer’s oral program. Regeneron and Novartis are pursuing injectables, which could eclipse the orals on efficacy.
Admilparant, an LPA1 antagonist which showed promise in Phase 2 for IPF, is up on the frontlines of the pipeline, along with pumitamig (PD-L1 x VEGF-A bispecific antibody with BioNTech) and the radiopharmaceutical RYZ101, which it picked up in its $4.1 billion buyout of RayzeBio. Like most of the rest of the top 15, Bristol Myers has been busily scooping up biotechs to fill out late-stage development work. Cobenfy, approved for schizophrenia in 2024, came out of Karuna and is now triggering worries for its late-stage program for Alzheimer’s-related psychosis following some “irregularities” that delayed an interim analysis.
Bristol Myers doesn’t have a whole lot of wiggle room when it comes to the late-stage pipeline. It’s being hammered by the loss of exclusivity on legacy blockbusters, with Opdivo — its current reigning champ — and Eliquis looking at a patent cliff event over the next two years. Revlimid is in significant decline. Even with some new approvals contributing to the bottom line, revenue slipped slightly in ’25 and will slide even more this year. So its recently approved franchise therapies and Phase 3 drugs are all meant to be major contributors. Which is why this is one big pharma that frequently signals to the biopharma research community that it has no appetite for small-market drugs.
Its late-stage pipeline today was shaped by a reorganization two years ago that dropped a slate of early and mid-stage drugs from the pipeline that were seen as runners-up in the blockbuster race. That included a CTLA-4 drug that was seen as a cut below Yervoy. To compete at the level Bristol Myers needs, it all has to be seen as a contender for first-in-class or best-in-class.
9. GSK: The new CEO brings some added enthusiasm to play in bolstering late-stage expectations
The scoop:
Historically, GSK has avoided anything flashy. It buys biotechs, but tends to go for deals on the lower end of the bolt-on spectrum. Its pipeline work has drawn mixed reviews over the years, sometimes spurring the activist set. The new, new thing in biopharma R&D often fails to pan out, as they know from experience: One of the reasons why the company saw a spike in R&D spending last year was due to its write-off on belrestotug, its TIGIT program partnered up while Hal Barron was running research. The original deal with iTeos cost $625 million in upfront cash.
GSK execs aren’t diving into obesity, either — or at least they’re avoiding the commercial slugfest developing around GLP-1. It’s too frothy.
So after its ADC Mo-Rez (mocertatug rezetecan) delivered strong Phase 1b data recently, it caught our attention. GSK heralded the results in platinum-resistant ovarian or endometrial cancer as “extremely exciting.”
They were so excited, GSK said it would
pivot directly into an ambitious Phase 3 program
with five trials set to begin soon. The ADC was picked up in a modestly priced licensing deal with China’s Hansoh Pharma, targeting the B7-H4 antigen. And if the data hold up, it will help make GSK a force to be reckoned with in oncology, a goal that ex-CEO Emma Walmsley set out when she took over a decade ago, picking up the pieces left from its big asset swap with Novartis in 2015.
It’s not the only ADC GSK is excited about. The other one — also from Hansoh — is dubbed Ris-Rez and targets small cell lung cancer. And GSK talked it up in its Q4 review earlier this year.
GSK expects Mo-Rez can earn more than £2 billion a year, making it a key drug in its quest for £40 billion a year in revenue by 2031, up from 2025’s £32.7 billion. Ris-Rez, meanwhile, has picked up a breakthrough drug designation at the FDA and earned equally ambitious peak sales projections.
You could call all that part of GSK’s tactical acceleration to achieve some longtime strategic goals.
Rounding out its newfound prospects for oncology, GSK is following up its
IDRx buyout
— just $1 billion upfront — from a little more than a year ago with pivotal plans for velzatinib/IDRX-42. If successful, new CEO Luke Miels will be headed back to an arena where he found success at AstraZeneca.
And he clearly relishes that prospect.
Miels, a marketing specialist, offered a similar projection for bepirovirsen, its
hep B “functional cure,”
projecting equivalent peak sales as its top cancer drug. Some of the analysts, though, aren’t ready to go that high, but breaking the billion-dollar mark seems plausible.
Then there’s the pivotal campaign for camlipixant, a P2X3 antagonist for chronic cough that’s also earned some blockbuster love, along with fresh prospects for Jemperli.
In some respects, a brighter horizon for GSK reflects Miels’ cheerleading style for R&D. Where Walmsley would be somewhat more guarded, he’s encouraged high expectations. But it’s also based on hard data, where he’s been reaping the rewards of Walmsley’s dogged ambitions for GSK.
GSK has had its successes, particularly with Shingrix, which has helped the company manage the market volatility for RSV. And the global pharma continues to talk up vaccines and respiratory, two traditional mainstays. It’s also been a longtime runner-up to Gilead Sciences in the HIV market, whereto majority-owned ViiV continues to carve out significant sales.
10. AbbVie: Researchers set out to prove they’re more than just Skyrizi and Rinvoq
The scoop:
Skyrizi and Rinvoq kept AbbVie in the big leagues as Humira gradually faded, and AbbVie execs have shown real zeal about adding to those franchises with new indications. Moving beyond its two cash cows, there’s a slate of late-stage bets — along with an aspirational play for
in vivo
CAR-T.
AbbVie had high hopes for their neuroscience division when it paid $8.7 billion to get Cerevel Therapeutics back in 2024. The readouts aren’t over, but it had to write off a good chunk of that when
emraclidine foundered
in a slate of schizophrenia studies. It did better with another drug picked up in the buyout when tavapadon hit in Parkinson’s. But execs can’t be happy with a group of modest peak sales projections.
Maybe AbbVie can do better with bretisilocin, a 5-HT2A receptor agonist and 5-HT releaser which had a positive slice of Phase 2a data for major depressive disorder when AbbVie bought it. It
didn’t pay a lot for it
, failing to disclose the upfront and leaving it at $1.2 billion all in for the psychedelic mimic. But it’s a near-term shot in a high-risk segment of the business that’s been seeing growth with Vraylar and Ubrelvy.
ABBV-932 rounds out its top neuroscience prospects.
Then there’s oncology.
Etentamig (ABBV-383) was dosed in Phase 3 for the first time in 2024, highlighting AbbVie’s shot at a BCMAxCD3 bispecific T-cell engager for multiple myeloma. By silencing the Fc tail, researchers hope to extend the half-life of the drug so they can stretch dosing to every four weeks. The downside is it’s late to the game, with Tecvayli from J&J out on the market and following up with a recent accelerated approval for a combo with Darzalex.
Pivekimab sunirine (PVEK), its CD123 ADC, was filed with the FDA last fall. AbbVie submitted early Phase 1/2 data for blastic plasmacytoid dendritic cell neoplasm (BPDCN), a rare and aggressive cancer, hoping to gain a quick entry on a drug that earned a breakthrough drug designation from the FDA.
Temab-A (ABBV-400), which targets cancers with high c-Met expression, got started in Phase 3 with a shot at colorectal cancer, where it’s being studied as a monotherapy and in combo with bevacizumab. AbbVie is looking for data from a slate of mid- to late-stage studies to gauge its potential for solid tumors.
ABBV-706, an SEZ6 targeted ADC, is in trials for solid tumors.
You don’t hear a lot about BoNT/E, the fast-acting botulinum neurotoxin that works for shorter spans. AbbVie picked it up in mid-stage development way back in 2018, bringing a potential rival to Botox in-house. That treatment was filed with the FDA a year ago and could bolster its big business in aesthetics.
AbbVie will go the distance when it feels there’s sufficient upside, and that’s what it committed to do when the company bought Capstan Therapeutics, one of the leading early-stage biotechs working in the sizzling
in vivo
CAR-T space. There’a a very long row to hoe on that one, but a win here could put AbbVie out front with a next-gen cancer drug. Also, the $2.1 billion deal didn’t cost the moon, giving it a relatively low-cost method for generating some added enthusiasm for the pipeline.
11. Sanofi: Another new CEO looks for a better path in Phase 3
The scoop:
Big pharma players with a solid record of success in Phase 3 don’t fire their CEOs, so Paul Hudson’s exit a couple of months ago set the stage for a recital of all his setbacks in R&D.
Hudson bet big on immunology at Sanofi, where board members have repeatedly demonstrated their readiness to make a change when they feel a course correction is needed. So when the strategy failed to set the company squarely on a new path that could carry it past Dupixent, the enormous cash cow (out of Regeneron) that pays the freight at Sanofi, the rest was Greek theater.
New CEO Belén Garijo will need to make some tough decisions, and that typically starts with the pipeline.
About two years ago, and Hudson was cheering mid-stage data for its anti-inflammatory OX40L drug amlitelimab, once billed as a product that could excel over Dupixent. These days execs spend their time defending mixed data from the Phase 3 program that followed. Analysts highlighted lower efficacy than Dupixent for atopic dermatitis and Sanofi has been dropping indications instead of building the pipeline-in-a-product it badly needs.
It’s not a bust, but it is far from megablockbuster heaven.
The same “meh” reaction followed last summer’s approval of rilzabrutinib (Wayrilz) for immune thrombocytopenia (ITP). That followed a major setback on atopic dermatitis. It edged ahead after the key drug in the Principia Biopharma buyout, tolebrutinib, hit a series of setbacks in clinical trials. And the oral TNF inhibitor balinatunfib has gone down on data twice in mid-stage studies, failing an effort to prove it could work as a monotherapy.
That only leaves the anti-CD40L frexalimab from one of Hudson’s old lists of experimental drugs capable of generating more than $5 billion a year in revenue. That’s in Phase 3 for multiple sclerosis, after a failure for Sjögren’s disease. Its bispecific lunsekimig, meanwhile, hit in mid-stage studies of asthma and an inflammatory sinus condition, but flopped in atopic dermatitis.
Sanofi stayed at the blockbuster feast because of its partnership with Regeneron on Dupixent. But it’s only managed to disappoint analysts with its COPD data for next-gen drug itepekimab. That leaves R&D working away to keep building Dupixent as it gets close to a reckoning on its patents in the early 2030s.
Sanofi also isn’t catching much of a break with analysts on its latest CEO pick. Garijo never established Merck KGaA’s reputation in R&D and there are plenty of doubters that she can do it at Sanofi. Nevertheless, she’s holding the reins now.
12. Novo Nordisk: After fumbling the lead in obesity, R&D hunts for a new way forward
The scoop:
Novo Nordisk has been channeling more cash into its R&D division in recent years, emboldened by its mega-blockbuster semaglutide and vowing to beat back competition from fierce rival Eli Lilly.
Some of that money went into a critical head-to-head study of its GLP-1/amylin analog against Lilly’s tirzepatide, and Novo lost — big time. With that went its market cap, which has shriveled back to levels from five years ago.
Novo had a big head start on Eli Lilly, and its steady backward drift — as a tide of new GLP-1s courses through the industry — has escaped no one. The early arrival of its pill has done nothing to change perceptions.
Novo Nordisk isn’t done with CagriSema. With weight loss averaging more than 20%, why should it be? But a lot of attention has now shifted to zenagamtide (amycretin), a GLP-1/amylin dual agonist that was slated to begin Phase 3 this year with both oral and injected versions.
Adding insult to injury, Novo also had to concede late last year that its Phase 3 attempt to see if semaglutide could help Alzheimer’s patients came up as a loser. Practically no one expected it to succeed, and Novo clearly flagged the lottery-ticket strategy, but the stock took a hit anyway.
But don’t expect Novo to take a back seat on obesity just because of a few key setbacks. That was apparent with its failed bid for Metsera, which fell — inevitably — to Pfizer. But it also has UBT251, a triple agonist that it hopes can put it out front again.
Long a leader in the diabetes field, Novo is also using its deep scientific knowledge in the field with its late-stage program for ziltivekimab to see if it can make an impact in cardiovascular disease with a disease-modifying approach. That’s another huge field, with enormous potential — if Novo can catch a break.
13. Amgen: The new R&D chief is determined to score big with protein degradation
The scoop:
The star of Amgen’s late-stage pipeline remains MariTide, its obesity drug that’s meant to rival the two frontrunners. There’s a twist, built around GIPR antagonism, as opposed to the agonism that has succeeded so far.
Over the years, Amgen CEO Bob Bradway has managed to foster star coverage for its leading experimental drugs. Lumakras, its breakthrough offering on KRAS, was an example of that. But there are too many questions about the obesity market now to get a free pass among the analysts. For Amgen to succeed with obesity, it’d have to clear a very high bar on efficacy (that hasn’t looked good), hold the line on safety and offer a better dosing regimen to compete against the leaders.
That’s not so easy, particularly as more and more rivals elbow their way into the obesity pipeline looking for any kind of edge. And Amgen still has a ways to go before it can hunt up an approval.
In reviewing 2025 for investors, R&D chief Jay Bradner offered its Phase 3 drug desodoliveb for Sjögren’s disease. And there’s daxdilimab.
But there were also a couple of duds.
Bradner axed an anti-OX40 rocatinlimab collaboration with Kyowa Kirin — a precursor to the Japanese company’s decision to scrap the whole thing — as well as the program for bemarituzumab, which produced positive data for gastric cancer.
Where Amgen execs see their most likely near-term growth coming from is franchise expansion. But it has also had to stare down the FDA, which wanted their rare disease drug Tavneos to be pulled after the agency spotlighted a reassessment of the benefit/risk profile the drug presented.
Given the less-than-stellar prospects of its late-stage pipeline, it’s no wonder that Bradner turned to Dark Blue, buying out the UK biotech for $840 million earlier in the year. The move puts Bradner back in the driver’s seat of protein degradation, an early and long-lasting love of his.
14. Regeneron: Cash cows carry it through a challenging fight in R&D
The scoop:
Regeneron execs Len Schleifer and George Yancopoulos have been working hard to get investors excited about the near-term prospects for their late-stage pipeline, beginning with their LAG-3 fianlimab. But it can be an uphill climb with the analysts who haven’t been that impressed with late arrivals in questionable markets.
Where Yancopoulos boasts about a drug with “best-in-class” potential, some analysts wonder how well they can do with a follow-up to Bristol Myers Squibb’s pioneering LAG-3. Bristol Myers has had plenty of trouble with the drug since it was approved. It isn’t effective enough to work as a monotherapy, so it has to be rolled out as a combo. Follow-up studies have failed, and rivals like Immutep have seen their candidates go down in flames, blighting a field that once looked like it would be a successor to CTLA-4 and PD-L1.
Libtayo (the PD-1 cemiplimab) itself is a follow-up to Keytruda and Opdivo. It hit the $1.4 billion revenue mark last year, leaving it well behind the two market leaders as Regeneron’s research group pushes for expanded approvals. Building that market with combo drugs like fianlimab is important for Regeneron, which is run by two of the most persistent self-made billionaires in biotech. Another oncology prospect in mid-stage development is marlotamig (REGN7075), an EGFRxCD28 bispecific.
Cancer overall, though, has proven to be a complex challenge for Regeneron. Odronextamab — its CD20xCD3 candidate — has run into repeated issues at the FDA as rivals crowd around. And when linvoseltamab was approved last summer (as Lynozyfic) for multiple myeloma, more delays had forced it further behind rivals at Pfizer and J&J.
In addition, Regeneron has had trouble following up on its own market leader Dupixent. Last year its next-gen replacement partnered with Sanofi, itepekimab, fizzled in late-stage testing. But during Regeneron’s Q4 call Schleifer shifted the spotlight to “long-acting IL-13, IL-4, and IL-4/13 bispecifics as well as… a new soupy doopy molecule that is a new version of Dupixent that was naturally selected that might have even more improved properties.”
In the meantime, Sanofi’s been working on legal strategies to extend the patents on Dupixent to 2040. Where R&D fails, patent thickets can often fill the gap.
Regeneron has a much broader focus these days than when it achieved early successes with Dupixent and Eylea, including a foray into obesity and diabetes. Last summer it forked over a modest $80 million in cash to China’s Hansoh Pharma for olatorepatide, a GLP-1/GIP drug following elimination Lilly’s dominant play with tirzepatide. Even Regeneron says the drug looks similar to Eli Lilly’s, but it sees it as a pathway to developing a better drug that can maintain the muscle lost to GLP-1s. China’s late-stage trial passed muster in Phase 3 a few weeks ago, with Regeneron laying the groundwork for pivotal development ex-China.
Trevogrumab (REGN1033) may help with the muscle loss issues, where researchers have been posting evidence of significantly reduced loss of lean mass. And there’s a triplet with garetosmab for obesity, though safety issues have clouded expectations.
Once again, though, Regeneron finds itself competing in a hot, crowded field dominated by market leaders. Coming out on top will prove a tremendous challenge.
Regeneron has had success with its C5 siRNA drug cemdisiran, reporting positive Phase 3 data for generalized myasthenia gravis as it steered toward an FDA filing. Jefferies, for one, has estimated sales of a billion dollars. But it’s been flying under the radar until recently at Regeneron, which rarely happens at a biotech led by such bullish players.
15. Gilead: Time to bury the dead and move ahead
The scoop:
Gilead’s MO on the R&D side has remained largely unchanged during Daniel O’Day’s seven-year tenure as CEO. It buys up intriguing new drugs from cutting-edge biotechs and watches as the experiments gradually fail in the clinic. But each year it’s saved by a growing, dominant HIV business.
Biktarvy, its 3-in-1 daily tablet for HIV, provides about half of Gilead’s revenue. The R&D group has a deep understanding of HIV and the maintenance therapies needed to keep it in check, developing regimens that are easier on patients while keeping the big bucks rolling in. Yes, there have been failures in HIV — notably in GS-1720 and and GS-4182 recently — but its dominance in HIV shows no signs of deterioration.
You don’t have to look long at the deals Gilead has done to see the missteps in cancer. Immunomedics is a prime example. The recent Q4 and 2025 review contains no reference to TIGIT, once a hot field in immunotherapy that “seems doomed,” in the words of analyst Tim Anderson. And does anyone remember magrolimab, the anti-CD47 “don’t eat me” antibody?
O’Day, though, isn’t throwing in the towel. On the contrary. Gilead recently decided to go all in on anito-cel, buying out its partner Arcellx in a deal worth $7.8 billion. It believes the CAR-T can outperform Carvykti, the reigning champ in CAR-T, and help revive its commercial work in a field that has been flagging with its aging Kite originals.
This is no long-term wager. The company is looking for a green light from the FDA just before Christmas.
Then there was the $3.15 billion upfront pact to buy out an ADC from Tubulis.
Gilead spent $1.68 billion in cash to bag Ouro, gaining the T cell engager gamgertamig, a BCMAxCD3 candidate now in Phase 1/2.
In biotech, sometimes you have to bury the dead (programs) and move ahead. O’Day’s clearly decided that the time has come for Gilead. Its durable success in HIV gives him the means to do just that.
临床2期临床3期临床结果上市批准并购
2026-03-24
抑郁症(Depression)研究报告执行摘要:
抑郁症(Major Depressive Disorder,MDD)是全球范围内导致残疾的主要原因之一,根据世界卫生组织(WHO)数据显示,全球约有3.8%的人口患有抑郁症,已成为仅次于癌症的人类第二大"健康杀手"。近年来,随着神经科学研究的深入,抑郁症的发病机制已从传统的"单胺假说"逐步扩展至多系统综合机制,包括谷氨酸系统、神经炎症、神经可塑性、下丘脑-垂体-肾上腺(HPA)轴等多个领域,为抗抑郁药物的研发开辟了全新的方向。本文系统梳理了抑郁症领域的最新药物靶点图谱,涵盖单胺类靶点、谷氨酸系统靶点、GABA系统靶点、神经炎症相关靶点、神经可塑性相关靶点、HPA轴相关靶点、褪黑素系统靶点、阿片系统靶点以及其他新兴靶点。同时,本文还对全球及中国抑郁症药物市场规模、竞争格局、研发趋势和政策环境进行了全面分析,旨在为制药企业、投资者和研究人员提供决策参考。
核心发现:
市场规模:2024年全球抗抑郁药物市场规模约为193亿美元(注:部分研究机构数据显示约124.5-153亿美元,统计口径存在差异),预计到2030年将达到266亿美元,年复合增长率(CAGR)约为5.8%。中国市场2021年规模约100亿元,预计到2030年将达到238亿元。
靶点多元化:抑郁症药物研发已从单胺系统扩展至谷氨酸系统、神经炎症系统、神经可塑性系统等多个领域。GluN2D型NMDA受体、P2X7受体、BDNF-TrkB通路等新兴靶点展现出巨大的临床潜力。
快速起效突破:以艾司氯胺酮为代表的NMDA受体拮抗剂实现了数小时至数天的快速起效,解决了传统抗抑郁药物起效缓慢的痛点。GABA-A受体调节剂Zuranolone已在产后抑郁适应症获批。
研发遇冷与调整:2025年抑郁症药物研发遭遇重创,五大明星候选药相继折戟晚期试验,表明该领域新药研发仍面临巨大挑战。
图1:全球与中国抑郁症药物市场规模预测(2019-2030)第一章 抑郁症概述1.1 疾病定义与分类
抑郁症是一种以持续性情绪低落、兴趣减退为核心症状的精神障碍性疾病。根据《精神疾病诊断与统计手册》(DSM-5)和《国际疾病分类》(ICD-10)的定义,抑郁症的诊断需要满足以下核心症状中的至少两项,并持续至少两周:情绪低落、兴趣或愉悦感丧失、精力下降或疲劳感、睡眠障碍(失眠或嗜睡)、食欲或体重改变、精神运动性激动或迟滞、无价值感或过度自责、注意力难以集中、反复出现死亡或自杀念头。
根据症状特征和病程特点,抑郁症可分为以下主要亚型:
单相抑郁症是最常见的类型,包括重度抑郁症(Major Depressive Disorder,MDD)、持续性抑郁障碍(恶劣心境)、产后抑郁和季节性抑郁障碍等。其中,重度抑郁症是本报告的主要研究对象,其特征为明显的功能损害和严重的心理痛苦。
双相情感障碍虽然也包含抑郁相,但由于其独特的治疗原则和用药禁忌,通常被视为独立的疾病类别。本报告聚焦于单相抑郁症的药物靶点研究。1.2 流行病学数据1.2.1 全球流行病学
抑郁症已成为全球公共卫生领域的重大挑战。根据世界卫生组织(WHO)的数据,全球约有3.8%的人口患有抑郁症,约合2.8亿人。抑郁症是导致全球残疾的主要原因之一,每年因抑郁症造成的经济损失高达数千亿美元。从性别分布来看,抑郁症在女性中的患病率约为男性的两倍。女性的生理周期、妊娠、产后及围绝经期等特殊生理阶段与激素水平变化相关,增加了抑郁风险。从年龄分布来看,抑郁症发病呈现双峰特征,首个发病高峰出现在20-25岁左右,第二个高峰出现在45-55岁。1.2.2 中国流行病学
中国抑郁症防治形势同样不容乐观。根据最新统计数据,中国抑郁症患者已超过9500万,其中成年人抑郁症终身患病率约为6.9%,12个月患病率约为3.6%(部分来源显示确诊率约3.4%),且患病率呈逐年上升趋势。每年新增抑郁症患者约900万,疾病负担持续加重。从地域分布来看,抑郁症患病率存在一定的地区差异。经济发达地区由于生活节奏快、竞争压力大,抑郁症患病率相对较高。从年龄结构来看,青少年和老年人群的抑郁症问题日益突出,需要给予特别关注。1.3 发病机制假说
抑郁症的发病机制至今尚未完全阐明,存在多种假说,它们相互补充、相互印证,共同构建了我们对抑郁症病理生理学的理解框架。
单胺假说:是最经典的抑郁症发病机制假说,提出于20世纪60年代。该假说认为抑郁症与大脑内5-羟色胺(5-HT)、去甲肾上腺素(NE)和多巴胺(DA)等单胺类神经递质的功能不足有关。基于这一假说研发的抗抑郁药物(如SSRIs和SNRIs)至今仍是临床一线用药。
谷氨酸假说:是近年来研究最为活跃的领域之一。该假说认为抑郁症与谷氨酸能系统功能异常有关,表现为NMDA受体活性过高和谷氨酸过度释放。氯胺酮等NMDA受体拮抗剂展现出的快速抗抑郁效果为这一假说提供了有力支持。
神经可塑性假说:认为抑郁症与神经可塑性受损有关,表现为神经元萎缩、突触密度降低和神经发生减少。BDNF/TrkB信号通路的下调是这一假说的核心机制。
神经炎症假说:认为外周及中枢炎症在抑郁的发生中起重要作用。小胶质细胞激活、促炎细胞因子(如IL-1β、IL-6、TNF-α)水平升高与抑郁症症状密切相关。
HPA轴功能异常假说:认为抑郁症与下丘脑-垂体-肾上腺轴的负反馈调节受损有关,表现为皮质醇水平升高和糖皮质激素抵抗。第二章 靶点全景图谱
抑郁症药物靶点可根据其作用机制和神经生物学系统分为多个类别。下文将详细阐述各类靶点的结构、功能、作用机制及代表性药物。
2.1 单胺类靶点
单胺类神经递质系统是抑郁症发病机制研究最为成熟的领域,也是当前临床一线抗抑郁药物的主要作用靶点。单胺类靶点主要包括选择性5-羟色胺再摄取抑制剂(SSRIs)靶点、5-羟色胺和去甲肾上腺素双重重摄取抑制剂(SNRIs)靶点、三环类抗抑郁药(TCAs)靶点以及单胺氧化酶抑制剂(MAOIs)靶点。2.1.1 SSRIs(选择性5-羟色胺再摄取抑制剂)靶点
靶点名称:5-羟色胺转运体(Serotonin Transporter,SERT)
作用机制:SSRIs类药物通过选择性抑制突触前膜上的5-羟色胺(5-HT)转运体,阻止5-HT的再摄取,使突触间隙中的5-HT浓度升高,从而增强5-HT能神经传递。SSRIs包括氟西汀(Fluoxetine)、舍曲林(Sertraline)、帕罗西汀(Paroxetine)、西酞普兰(Citalopram)、艾司西酞普兰(Escitalopram)和氟伏沙明(Fluvoxamine)等。
2.1.2 SNRIs(5-羟色胺和去甲肾上腺素再摄取抑制剂)靶点
靶点名称:5-羟色胺转运体(SERT)和去甲肾上腺素转运体(Norepinephrine Transporter,NET)
作用机制:SNRIs类药物通过同时抑制5-HT和去甲肾上腺素(NE)的再摄取,增强两种神经递质系统的功能。代表药物包括文拉法辛(Venlafaxine)、度洛西汀(Duloxetine)和去甲文拉法辛(Desvenlafaxine)等。
2.1.3 三环类抗抑郁药(TCAs)靶点
靶点名称:多靶点作用,包括SERT、NET、组胺H1受体、乙酰胆碱毒蕈碱受体(M1)和肾上腺素α1受体
作用机制:TCAs是较早问世的一类抗抑郁药,主要包括阿米替林(Amitriptyline)、丙咪嗪(Imipramine)、多塞平(Doxepin)等。它们通过抑制5-HT和NE的再摄取,同时具有抗组胺和抗胆碱能作用。
2.1.4 单胺氧化酶抑制剂(MAOIs)靶点
靶点名称:单胺氧化酶A和B(MAO-A、MAO-B)
作用机制:MAOIs通过抑制单胺氧化酶的活性,阻止5-HT、NE和多巴胺(DA)的降解,从而增加突触间隙中单胺类神经递质的浓度。代表药物包括苯乙肼(Phenelzine)、反苯环丙胺(Tranylcypromine)和吗氯贝胺(Moclobemide)等。
2.2 谷氨酸系统靶点
谷氨酸系统是近年来抑郁症研究最为活跃的领域之一。以氯胺酮为代表的NMDA受体拮抗剂展现了快速起效的抗抑郁特性,为难治性抑郁症患者带来了新的希望。2.2.1 NMDA受体靶点
靶点名称:N-甲基-D-天冬氨酸(NMDA)受体
作用机制:NMDA受体是离子型谷氨酸受体,在突触可塑性、学习和记忆中发挥关键作用。NMDA受体拮抗剂(如氯胺酮)通过阻断NMDA受体,解除对谷氨酸能神经元的抑制,增强前额叶皮层的谷氨酸释放,激活AMPA受体,最终促进神经可塑性相关基因的表达。
2.2.2 AMPA受体靶点
靶点名称:α-氨基-3-羟基-5-甲基-4-异恶唑丙酸(AMPA)受体
作用机制:AMPA受体是离子型谷氨酸受体另一个重要亚型,负责快速突触传递。AMPA受体正向别构调节剂(PAMs)可以增强AMPA受体介导的突触传递,促进神经可塑性。
2.2.3 mGluR受体家族
靶点名称:代谢型谷氨酸受体(mGluRs),包括mGluR1-8八个亚型
作用机制:mGluRs是G蛋白偶联受体,通过调节突触前神经递质释放和突触后信号传导参与情绪调节。其中,mGluR2/3和mGluR5是研究较为深入的抗抑郁靶点。
2.3 GABA系统靶点
γ-氨基丁酸(GABA)是中枢神经系统主要的抑制性神经递质,研究发现抑郁症患者存在GABA系统功能低下。2.3.1 GABA-A受体靶点
靶点名称:GABA-A受体
作用机制:GABA-A受体是配体门控氯离子通道,被GABA激活后引起氯离子内流,导致神经元超极化。新一代GABA-A受体正向变构调节剂可以选择性增强受体功能,恢复抑制性/兴奋性平衡。
2.4 神经炎症相关靶点
神经炎症假说是抑郁症发病机制研究的重要新方向。研究表明,外周及中枢炎症在抑郁的发生中起重要作用,抑郁症患者循环血中多种炎症因子表达上调。2.4.1 P2X7受体靶点
靶点名称:嘌呤能P2X7受体(P2X7R)
作用机制:P2X7受体是ATP门控阳离子通道,主要表达于小胶质细胞,在神经-胶质信号传导中发挥关键作用。P2X7受体激活可促进白细胞介素-1β(IL-1β)和其他炎症因子的释放。2.4.2 TLR4受体靶点
靶点名称:Toll样受体4(TLR4)
作用机制:TLR4是模式识别受体,主要表达于小胶质细胞表面,参与识别病原相关分子模式(PAMPs)和损伤相关分子模式(DAMPs)。TLR4激活可触发NF-κB信号通路,导致促炎细胞因子表达上调。2.4.3 COX-2靶点
靶点名称:环氧化酶-2(COX-2)
作用机制:COX-2是前列腺素合成的关键酶,在炎症反应中高表达。COX-2抑制剂可减少前列腺素E2(PGE2)的合成,发挥抗炎作用。2.5 神经可塑性相关靶点2.5.1 BDNF(脑源性神经营养因子)靶点
靶点名称:脑源性神经营养因子(BDNF)及其受体TrkB
作用机制:BDNF是中枢神经系统最重要的神经营养因子之一,与受体TrkB结合后激活PI3K/Akt、MAPK/ERK等下游信号通路,促进神经元存活、突触可塑性和神经发生。2.5.2 TrkB受体靶点
靶点名称:酪氨酸激酶受体B(TrkB)
作用机制:TrkB是BDNF的高亲和力受体,属于受体酪氨酸激酶家族。BDNF与TrkB结合后引起受体二聚化和自磷酸化,激活下游信号通路。2.6 HPA轴相关靶点
下丘脑-垂体-肾上腺(HPA)轴功能异常是抑郁症的重要病理特征之一。2.6.1 CRHR1(促肾上腺皮质激素释放激素受体1)
靶点名称:促肾上腺皮质激素释放激素受体1(CRHR1)
作用机制:CRHR1是促肾上腺皮质激素释放激素(CRH)的受体,属于G蛋白偶联受体家族。CRH与CRHR1结合后激活cAMP/PKA/CREB信号通路,调节应激反应。2.6.2 糖皮质激素受体(GR)
靶点名称:糖皮质激素受体(Glucocorticoid Receptor,GR)
作用机制:GR是类固醇激素受体超家族成员,在HPA轴负反馈调节中发挥关键作用。GR与糖皮质激素结合后,转位至细胞核调节基因转录。2.7 褪黑素系统靶点2.7.1 MT1/MT2褪黑素受体
靶点名称:褪黑素受体1A(MT1)和1B(MT2)
作用机制:褪黑素通过激活MT1和MT2受体,调节昼夜节律、睡眠觉醒周期和季节性情绪变化。褪黑素受体是G蛋白偶联受体,主要分布于下丘脑视交叉上核(SCN)。2.8 阿片系统靶点2.8.1 Kappa阿片受体(KOR)
靶点名称:Kappa阿片受体(Kappa Opioid Receptor,KOR)
作用机制:KOR是内源性强啡肽系统的受体,属于G蛋白偶联受体。KOR激活可调节应激反应、多巴胺系统和谷氨酸能传递。2.9 其他新兴靶点
南京医科大学团队研究发现:"单胺假说"提出近60年来、氟西汀发现50年来的重要突破,新型快效抗抑郁药物可在2小时内快速起效。机制研究聚焦于单胺类系统的快速调节和神经可塑性的即时激活。LY03021多靶点抗抑郁药物首次应用去甲肾上腺素转运体(NET)、多巴胺转运体(DAT)以及GABA-A受体的调节机制,同时调节单胺和抑制性神经递质系统。第三章市场规模与竞争格局3.1 全球抑郁症药物市场规模
根据最新市场研究数据,全球抗抑郁药物市场规模持续扩大。由于不同研究机构采用不同的统计口径和定义范围(如是否包含抗焦虑药物、是否区分片剂和注射剂等),市场数据存在一定差异。
年份
部分来源数据
另一些来源数据
2019
约152亿美元
约150亿美元
2023
约153亿美元(湖南睿略)
约124.5亿美元(中研普华)
2024
约193亿美元(医药魔方)
-
2030预测
约266亿美元(医药魔方)
约135亿美元(中研普华)
说明:市场规模预测差异主要源于:1)统计范围定义不同;2)是否考虑新型疗法溢价;3)地域覆盖范围差异。建议读者在参考时关注数据来源和统计口径。3.2 中国抑郁症药物市场规模
中国患者人群:中国抑郁症患者已超过9500万。根据北京大学第六医院黄悦勤教授等在《柳叶刀·精神病学》发表的研究数据,中国成人抑郁障碍终身患病率约为6.9%,12个月患病率约为3.6%。每年新增抑郁症患者约900万。市场规模:2021年中国抑郁症药物市场规模达到约100亿元,2022-2030年复合增长率为4.2%,预计到2030年将达到238亿元。3.3 市场细分分析3.3.1 按药物类别细分
药物类别
市场份额
代表药物
临床地位
SSRIs
约40%
氟西汀、舍曲林、帕罗西汀
一线治疗
SNRIs
约25%
文拉法辛、度洛西汀
一线治疗
TCAs
约10%
阿米替林、丙咪嗪
二线治疗
MAOIs
约5%
吗氯贝胺
二线治疗
其他
约20%
安非他酮、米氮平、阿戈美拉汀
多样3.4 市场增长驱动因素
需求侧驱动因素:
人口老龄化加剧:老年人抑郁症患病率上升,驱动用药需求增长
生活节奏加快:工作压力增大,抑郁症发病率上升
公众认知提升:对心理健康问题认知提升,就诊率提高
支付意愿增强:患者对创新疗法的支付意愿增强
供给侧驱动因素:
新型靶点突破:新型靶点药物研发取得突破,推动市场增长
给药方式创新:鼻喷雾剂、口服制剂等创新给药方式提升患者依从性
数字疗法兴起:数字疗法和神经调控技术发展为治疗提供新选择
精准医学发展:基因组学推动个性化治疗,提高治疗效果3.5 未满足的临床需求
1. 起效时间:传统抗抑郁药物起效需2-4周,约三分之一的患者疗效不佳或无效
2. 难治性抑郁症:约50%的患者未能获得有效治疗
3. 安全性问题:现有药物存在性功能障碍,体重增加,心血管风险等副作用
4. 依从性差:长期用药依从性差是治疗失败的重要原因3.6 竞争格局分析
图2:全球抑郁症药物市场竞争格局3.6.1 第一梯队(跨国药企)
公司
代表产品
市场地位
辉瑞(Pfizer)
舍曲林、左洛复
领先
礼来(Eli Lilly)
氟西汀(百忧解)
领先
灵北制药(Lundbeck)
氢溴酸伏硫西汀
领先
强生(Johnson & Johnson)
Spravato(艾司氯胺酮)
快速增长
葛兰素史克(GSK)
盐酸帕罗西汀
稳定
百健/Sage
Zuranolone(已获批PPD)
适应症扩展中3.6.2 第二梯队(国内大型药企)
• 豪森药业:抗抑郁仿制药
• 华海药业:制剂出口能力
• 科伦药业:创新药研发
• 绿叶制药:LY03021创新药
• 东阳光:原料药优势第四章研发趋势与在研药物4.1 临床阶段在研药物4.1.1 已批准上市的创新药物
药物名称
靶点/机制
公司
批准时间
特点
艾司氯胺酮(Spravato)
NMDA受体拮抗剂
强生
2019/2025扩展
首个难治性抑郁单药疗法
Brexanolone
GABA-A受体调节
Sage
2019
首个产后抑郁药物
Zuranolone(Zurzuvae)
GABA-A受体调节
Biogen/Sage
2023.8
已获批产后抑郁(MDD申请被拒)
盐酸托鲁地文拉法辛(若欣林)
SNRI
绿叶制药
2024年12月
中国首个化药1类创新抗抑郁药
琥珀酸地文拉法辛
SNRI
多家
2025年2月
4天内症状改善4.1.2 临床III期在研药物
药物名称
靶点/机制
公司
适应症
状态
Osavampator
AMPA受体PAM
Neurocrine Biosciences
MDD辅助治疗
3期研究中
Aticaprant
KOR拮抗剂
研发中
MDD辅助治疗
3期研究中
注:Zuranolone针对MDD适应症的上市申请已于2024年被FDA拒绝,不再处于滚动申报或待审批状态。4.2 2025年研发遇冷与调整
2025年抑郁症药物研发遭遇重创,多个明星候选药相继折戟晚期试验:
• Neumora Therapeutics navacaprant:后期研究失败,未能显著减轻抑郁症状
• Relmada REL-1017:两项三期试验被迫停止(无效性审查)
• Biogen/Sage Zuranolone MDD:FDA拒绝批准MDD适应症,要求更多有效性证据
这表明抑郁症新药研发仍面临巨大挑战,需要更深入理解疾病机制和优化临床试验设计。精神疾病药物临床试验设计复杂,安慰剂反应率高,需要更多生物标志物指导研发。4.3 新型靶点研究进展
• GluN2D型NMDA受体:2025年3月《Science Advances》发表研究,证实GABA能中间神经元上的GluN2D型NMDA受体是快速抗抑郁药物开发的新靶点,为开发具有更高亚基选择性的靶向抗抑郁药提供了理论基础。
• P2X7受体:作为神经炎症靶点,在抑郁症治疗中展现出潜力,是NLRP3炎症小体激活的上游信号。
• BDNF-TrkB系统:多个小分子TrkB激动剂正在开发中,ZZL-7等化合物展现2小时快速起效潜力。
• KOR拮抗剂:Aticaprant等正在进行临床三期研究。4.4 非药物治疗4.4.1 神经调控技术
• 重复经颅磁刺激(rTMS):非侵入性治疗方法,高频rTMS刺激背外侧前额叶皮层(DLPFC)显示有效,2026年研究探索与正念干预联合。
• 深部脑刺激(DBS):针对难治性抑郁症的侵入性治疗,胼胝体下扣带回(SCC)是研究最多的靶点。
• 迷走神经刺激(VNS):对抑郁症状的长期改善作用。4.4.2 数字疗法
FDA批准的数字疗法:
• CT-152(Pear Therapeutics):首个获FDA批准的重度抑郁症处方数字疗法,3期随机对照试验显示,辅助抗抑郁药物治疗6周后,MADRS评分较假应用组差异达-2.12,85.0%的平均课程完成率。
• Rejoyn:2024年下半年获FDA认证的处方数字疗法,定位为22岁及以上正在接受抗抑郁药物治疗患者的辅助疗法,6周标准化干预方案,结合认知训练与治疗课程。
• 数字疗法发展趋势:2025年多个国家将数字健康和数字疗法纳入医保报销范围,美国联邦医保机构为FDA批准的数字心理健康疗法提供保险覆盖。第五章政策环境分析5.1 FDA审批政策
快速通道和突破性疗法:FDA对创新抗抑郁药物提供突破性疗法认定(Breakthrough Therapy Designation),Zuranolone于2018年获得突破性疗法认定。2024年新药审批:FDA在2024年批准50款创新疗法,24款被认定为"first-in-class",抑郁症领域批准了艾司氯胺酮单药疗法扩展适应症。监管挑战:精神疾病药物临床试验设计复杂,安慰剂反应率高,需要更多生物标志物指导研发。5.2 EMA审批政策
EMA对抑郁症药物的审批遵循类似的严格标准,强调:• 长期疗效和安全性数据• 功能改善终点(如社会功能)• 患者报告结局(PRO)5.3 NMPA审批政策
中国政策支持:抑郁症纳入国家基本公共卫生服务项目,提高医保报销比例,鼓励创新药物研发。2024-2025年批准:盐酸托鲁地文拉法辛缓释片(若欣林)于2024年12月获批,成为中国首个自主研发的化药1类创新抗抑郁药。琥珀酸地文拉法辛缓释片于2025年2月获批。第六章展望与投资建议6.1 主要结论
靶点多元化:抑郁症药物研发已从单胺系统扩展至谷氨酸系统、神经炎症系统、神经可塑性系统等多个领域,为难治性抑郁症患者提供了新的治疗选择。
快速起效突破:NMDA受体拮抗剂代表了快速起效抗抑郁药物的新方向,艾司氯胺酮已获FDA批准单药疗法,可在24小时内改善症状。GABA-A受体调节剂Zuranolone已在产后抑郁适应症获批。
新兴靶点突破:GluN2D型NMDA受体作为快速抗抑郁新靶点的发现,为开发具有NMDA受体亚基选择性的靶向抗抑郁药提供了理论基础,可能减少非选择性NMDA拮抗剂的精神病样副作用。
市场持续增长:全球抗抑郁药物市场预计将保持5-7%的年复合增长率(注:不同机构预测存在差异,范围约1.2%-7.2%),到2030年达到266亿美元(乐观预测)。中国市场到2030年将达到238亿元。
非药物疗法兴起:数字疗法(如CT-152、Rejoyn)和神经调控技术(rTMS、DBS)正在成为重要的治疗补充,为患者提供更多元化的治疗选择。6.2 投资建议
高潜力投资方向:
1. 谷氨酸系统靶点药物:NMDA受体亚基选择性调节剂(如NR2B拮抗剂、GluN2D调节剂)和AMPA受体正向别构调节剂具有快速起效优势,市场潜力巨大。
2. BDNF-TrkB通路小分子激动剂:该通路是多种抗抑郁治疗的共同下游机制,小分子激动剂可突破BDNF大分子难以入脑的局限性。
3. 神经炎症靶点:P2X7受体、TLR4等靶点代表全新的抗抑郁策略,有望为难治性患者提供新的治疗选择。
4. 数字疗法:FDA批准的数字疗法已验证其临床价值,随着医保覆盖扩大,商业化前景可期。
风险提示:
• 研发风险:2025年多个明星候选药折戟晚期试验(包括Zuranolone MDD被拒),表明抑郁症新药研发失败率较高
• 临床试验设计:安慰剂反应率高是主要挑战
• 市场竞争:传统药物价格压力和仿制药竞争加剧
• 政策风险:药品定价和医保政策变化6.3 研究展望
• 精准医学:基于基因组学、蛋白组学和影像学的精准患者分层将指导更有效的个体化治疗。
• 联合治疗:不同机制药物的组合策略可能克服难治性抑郁症的治疗瓶颈。
• 转化医学:基础研究发现的靶点向临床药物的转化效率有待提高。
• 早期干预:识别抑郁症前驱症状和风险因素,实现早期预防和干预。
基因疗法
2026-03-17
1、研究机构:Neumora Therapeutics、BlackThorn Therapeutics
2、别名:BTRX-335140、BTRX-140、CYM-53093、NMRA-140、NMRA- 335140、Navacaprant Hydrochloride
3、靶点:KOR
4、进展及给药途径:普通片剂; 口服给药
5、结构式
6、适应症及进展
适应症
进展
最新进展日期
重度抑郁症
III期
2025-08-01
7、发表论文
标题
内容类型
适应症
企业
技术平台
来源
发表日期
Navacaprant, a novel and selective kappa opioid receptor antagonist with no agonist properties implicated in opioid-related abuse
药理研究
重度抑郁症
Neumora Therapeutics
Neuropharmacology
IF=4.6
2024-06-12
Design and Synthesis of a Novel and Selective Kappa Opioid Receptor (KOR) Antagonist (BTRX-335140)
药物发现
偏头痛
The Scripps Research Institute
BlackThorn Therapeutics
J Med Chem
IF=6.8
2019-02-28
8、专利布局
公开(公告)号
专利主题
发明名称
申请日
法律状态
CN110914262B
化合物
Κ阿片受体拮抗剂以及与其相关的产品和方法
2018-03-16
US20240343719A1
用途
κ阿片受体拮抗剂及其相关产品和方法
2023-12-15
实质审查
WO2024216061A1
用途
治疗抑郁症和快感缺乏症的方法
2024-04-12
WO2024216046A1
用途
治疗快感缺乏症的方法
2024-04-12
US12171758B1
晶型
κ阿片受体拮抗剂的结晶盐形式及其相关产品和方法
US12171758B1
9、研究历程
2024年05月14日,由Neumora Therapeutics Inc开展临床二期试验,用于治疗躁狂症。(https://www.biospace.com/article/releases/neumora-therapeutics-announces-initiation-of-phase-2-study-of-navacaprant-in-bipolar-depression/?s=68)
2024年05月13日,由Neumora Therapeutics Inc在美国开展临床二期试验,用于治疗双相情感障碍及相关障碍。(NCT06429722)
2023年11月10日,由Neumora Therapeutics Inc在巴西、保加利亚和加拿大等国家和地区开展临床三期试验,用于治疗重度抑郁症。(NCT06029439; NCT06058013; NCT06058039)
2023年09月20日,由Neumora Therapeutics Inc在美国开展临床三期试验,用于治疗重度抑郁症。(NCT06029439; NCT06058013; NCT06058039; NCT06029426)
2020年01月03日,由Neumora Therapeutics Inc在美国开展临床二期试验,用于治疗重度抑郁症。(NCT04221230; NCT06429722)
10、临床试验
登记号
试验标题
试验药
适应症
原始适应症
申办/合作机构
试验状态
试验分期
开始日期
完成日期
国家/地区
NCT06429722
A Phase 2a, Randomized, Double-blind, Placebo-controlled Pilot Study to Evaluate the Effects of Oral NMRA-335140 Versus Placebo in Participants With a Major Depressive Episode Associated With Bipolar II Disorder
Navacaprant (口服)
躁郁症2型 | 重度抑郁症
Major Depressive Episode Associated With Bipolar II Disorder
Neumora Therapeutics, Inc.
Completed
临床2期
2024-05-13
2025-05-30
美国
NCT06058039
A Phase 3, Randomized, Double-blind, Placebo-controlled Study to Evaluate the Effects of Oral NMRA-335140 Versus Placebo in Participants With Major Depressive Disorder
Navacaprant (口服)
重度抑郁症
Major Depressive Disorder
Neumora Therapeutics, Inc.
Recruiting
临床3期
2023-12-21
2026-03-01
瑞典 | 捷克 | 美国 | 芬兰 | 波兰 | 法国 | 保加利亚 | 德国
NCT06058013
A Phase 3, Randomized, Double-blind, Placebo-controlled Study to Evaluate the Effects of Oral NMRA-335140 Versus Placebo in Participants With Major Depressive Disorder
Navacaprant (口服)
重度抑郁症
Major Depressive Disorder
Neumora Therapeutics, Inc.
Recruiting
临床3期
2023-12-20
2026-06-01
加拿大 | 美国 | 巴西 | 智利
NCT06029439
Long-term Study to Assess the Safety and Effectiveness of NMRA-335140 in Participants With Major Depressive Disorder
Navacaprant (片剂, 口服)
重度抑郁症
Major Depressive Disorder
Neumora Therapeutics, Inc.
Recruiting
临床3期
2023-11-10
2027-06-01
加拿大 | 瑞典 | 捷克 | 美国 | 芬兰 | 波兰 | 巴西 | 法国 | 智利 | 保加利亚 | 德国
NCT06029426
A Phase 3, Randomized, Double Blind, Placebo Controlled Study to Evaluate the Effects of Oral NMRA-335140 Versus Placebo in Participants With Major Depressive Disorder
Navacaprant (口服)
重度抑郁症
Major Depressive Disorder
Neumora Therapeutics, Inc.
Completed
临床3期
2023-09-20
2025-01-15
美国
NCT04221230
A Phase 2a, Randomized, Double-blind, Placebo-controlled Proof of Concept Study to Evaluate the Effects of Oral BTRX-335140 (NMRA-335140) Versus Placebo in Subjects With Major Depressive Disorder
Navacaprant (口服)
重度抑郁症
Major Depressive Disorder
Neumora Therapeutics, Inc.
Completed
临床2期
2020-01-03
2022-06-23
美国
11、临床结果
标题
登记号
来源
分期
适应症
评价人数
用药方案
结果
评价
发布日期
申办/合作机构
主要研究药物
来源链接
Synapse链接
A Phase 2a, Randomized, Double-blind, Placebo-controlled Proof of Concept Study to Evaluate the Effects of Oral BTRX-335140 (NMRA-335140) Versus Placebo in Subjects With Major Depressive Disorder
NCT04221230
CTgov
临床2期
重度抑郁症
204
Placebo
Change From Baseline in Hamilton Depression Rating Scale (HAMD-17) Total Score at Weeks 8(LS Mean) = -7.4 Point
-
2025-05-29
Neumora Therapeutics, Inc.
Navacaprant
https://clinicaltrials.gov/ct2/show/results/NCT04221230
https://synapse.zhihuiya.com/clinical-result-detail/55458aa2903895aa90558a9a25e22402
Neumora Therapeutics Reports Data from KOASTAL-1 Study of Navacaprant in Major Depressive Disorder
NCT06029426
GlobeNewswire
临床3期
重度抑郁症
383
Navacaprant
MADRS(ITT population; Week 6) = -12.5 Point 未达到
不佳
2025-01-02
Neumora Therapeutics, Inc.
Navacaprant
https://www.globenewswire.com/news-release/2025/01/02/3003449/0/en/Neumora-Therapeutics-Reports-Data-from-KOASTAL-1-Study-of-Navacaprant-in-Major-Depressive-Disorder.html
https://synapse.zhihuiya.com/clinical-result-detail/8d235dd9a2a2a2d33ee5d0a08d8aa254
Neumora Therapeutics Reports Data from KOASTAL-1 Study of Navacaprant in Major Depressive Disorder
NCT06029426
GlobeNewswire
临床3期
重度抑郁症
383
Placebo
MADRS(ITT population; Week 6) = -12.5 Point 未达到
不佳
2025-01-02
Neumora Therapeutics, Inc.
Navacaprant
https://www.globenewswire.com/news-release/2025/01/02/3003449/0/en/Neumora-Therapeutics-Reports-Data-from-KOASTAL-1-Study-of-Navacaprant-in-Major-Depressive-Disorder.html
https://synapse.zhihuiya.com/clinical-result-detail/8d235dd9a2a2a2d33ee5d0a08d8aa254
Neumora Therapeutics Announces Initiation of Phase 3 Clinical Program for Navacaprant (NMRA-140) in Major Depressive Disorder
NCT04221230
BusinessWire
临床2期
重度抑郁症
171
navacaprant
HAMD-17(at Week 8): difference = -1.7, P-Value = 0.121
积极
2023-07-18
Neumora Therapeutics, Inc.
Navacaprant
https://www.businesswire.com/news/home/20230718207734/en/Neumora-Therapeutics-Announces-Initiation-of-Phase-3-Clinical-Program-for-Navacaprant-NMRA-140-in-Major-Depressive-Disorder
https://synapse.zhihuiya.com/clinical-result-detail/da0ea42224d284d23a22222295e8822a
Neumora Therapeutics Announces Initiation of Phase 3 Clinical Program for Navacaprant (NMRA-140) in Major Depressive Disorder
NCT04221230
BusinessWire
临床2期
重度抑郁症
171
placebo
HAMD-17(at Week 8): difference = -1.7, P-Value = 0.121
积极
2023-07-18
Neumora Therapeutics, Inc.
Navacaprant
https://www.businesswire.com/news/home/20230718207734/en/Neumora-Therapeutics-Announces-Initiation-of-Phase-3-Clinical-Program-for-Navacaprant-NMRA-140-in-Major-Depressive-Disorder
https://synapse.zhihuiya.com/clinical-result-detail/da0ea42224d284d23a22222295e8822a
12、转化医学
研究
亮点
主题
期刊/会议
出版日期
药物
适应症
机构
Navacaprant, a Novel and Highly Selective Kappa Opioid Receptor Antagonist, in Adults With Major Depressive Disorder
这项研究评估了一种高选择性κ阿片受体拮抗剂navacaprant在成年人重性抑郁症患者中的疗效和安全性。研究结果显示,navacaprant对中度至重度抑郁症患者的抑郁症状,包括无乐感,有显著改善作用,并且安全性良好。尽管在轻度抑郁症患者中未达到主要疗效终点,但支持进一步研究navacaprant用于治疗重性抑郁症。研究结果为生物医药领域提供了重要的临床数据。
2期临床研究
Pubmed
2025-04-09
Navacaprant
重度抑郁症
Neumora Therapeutics, Inc. | Baylor College of Medicine
Navacaprant, a novel and selective kappa opioid receptor antagonist, has no agonist properties implicated in opioid-related abuse
Navacaprant是一种新型选择性kappa阿片受体拮抗剂,没有与阿片类药物滥用有关的激动剂特性。研究表明,Navacaprant在体外和体内均表现出选择性KOR拮抗特性,对KOR激动剂引起的镇痛和催乳素释放具有显著抑制作用,且不具有任何阿片受体激动剂活性。这些结果表明Navacaprant是一种KOR选择性拮抗剂,不具有与阿片类药物滥用有关的药理特性。Navacaprant有望成为治疗抑郁症的候选药物。
药物发现/临床前
Neuropharmacology
2024-10-01
Navacaprant
神经系统疾病
Neumora Therapeutics, Inc. | DevelRx Ltd.
Preclinical and clinical efficacy of kappa opioid receptor antagonists for depression: A systematic review
-
3期临床研究
J Affect Disord
2024-10-01
Aticaprant | Navacaprant
重度抑郁症
University of Toronto | University Health Network | Brain and Cognition Discovery Foundation
Design and Synthesis of a Novel and Selective Kappa Opioid Receptor (KOR) Antagonist (BTRX-335140)
本文描述了一种新型选择性κ阿片受体(KOR)拮抗剂(BTRX-335140)的设计与合成。这类拮抗剂在治疗偏头痛和压力相关的情绪障碍(包括抑郁症、焦虑和药物滥用)方面具有潜在的药理作用。因此,开发具有改进的效力/选择性和药物样持续时间的新型KOR拮抗剂引起了药物化学界的兴趣。CYM-53093(BTRX-335140)被发现为一种强效选择性的KOR拮抗剂,具有良好的体外ADMET和体内药代动力学特性,以及在大鼠药效学实验中显示出药物样的持续时间作用。口服给药的CYM-53093在对抗KOR激动剂诱导的脑垂体泌乳素分泌和小鼠尾部反射痛觉实验中表现出强大的疗效。此化合物在一些隐性鞘内精神障碍的治疗中已进入临床一期试验阶段,其中埃内多啡(dynorphin)被认为是潜在病理生理的贡献因素。
药物发现/临床前
J Med Chem
2019-02-01
Navacaprant
抑郁症 | 偏头痛 | 焦虑症 | 未指明的药物依赖
BlackThorn Therapeutics, Inc.
The kappa opioid receptor antagonist, BTRX-335140, protects working memory performance from mild stress exposure in rhesus monkeys
-
药物发现/临床前
SFN 2018
2018-11-04
Navacaprant
心理压力
Blackthorn Therapeut., San Francisco, CA
Pharmacological characterization of BTRX-335140, a potent, selective and reversible kappa opioid receptor antagonist
-
药物发现/临床前
SFN 2017
2017-11-14
Navacaprant
神经行为学表现
Blackthorn Therapeut., South San Francisco, CA
Electrophysiological characterization of novel selective and reversible kappa opioid receptor antagonists in VTA neurons
-
药物发现/临床前
SFN 2017
2017-11-14
BTRX-395750 | Aticaprant | PF-04455242 | Navacaprant
神经行为学表现
BlackThorn Therapeutics, Inc. | Neurol., UCSF, San Francisco, CA
临床2期临床结果临床3期
100 项与 Aticaprant 相关的药物交易
登录后查看更多信息
外链
| KEGG | Wiki | ATC | Drug Bank |
|---|---|---|---|
| D11831 | Aticaprant | - |
研发状态
10 条进展最快的记录, 后查看更多信息
登录
| 适应症 | 最高研发状态 | 国家/地区 | 公司 | 日期 |
|---|---|---|---|---|
| 快感缺乏 | 临床3期 | 美国 | 2022-06-22 | |
| 快感缺乏 | 临床3期 | 阿根廷 | 2022-06-22 | |
| 快感缺乏 | 临床3期 | 澳大利亚 | 2022-06-22 | |
| 快感缺乏 | 临床3期 | 比利时 | 2022-06-22 | |
| 快感缺乏 | 临床3期 | 巴西 | 2022-06-22 | |
| 快感缺乏 | 临床3期 | 保加利亚 | 2022-06-22 | |
| 快感缺乏 | 临床3期 | 捷克 | 2022-06-22 | |
| 快感缺乏 | 临床3期 | 匈牙利 | 2022-06-22 | |
| 快感缺乏 | 临床3期 | 意大利 | 2022-06-22 | |
| 快感缺乏 | 临床3期 | 波兰 | 2022-06-22 |
登录后查看更多信息
临床结果
临床结果
适应症
分期
评价
查看全部结果
| 研究 | 分期 | 人群特征 | 评价人数 | 分组 | 结果 | 评价 | 发布日期 |
|---|
临床3期 | 444 | Placebo | 夢觸構鹽鹽獵網鬱衊遞(鏇鹹範窪鏇繭醖獵淵糧) = 鏇範衊鹽觸繭餘製觸簾 膚觸顧願蓋夢壓網簾範 (顧遞選顧糧餘選繭壓鹽, 0.96) 更多 | - | 2026-01-02 | ||
临床3期 | 513 | Placebo | 醖選繭選鬱襯鏇壓繭鏇(遞選鏇齋願衊簾範窪醖) = 鹹齋壓鏇壓製餘鑰壓簾 遞構淵鬱構觸廠鹹壓糧 (鬱鏇簾鹹繭壓願築鑰遞, 0.98) 更多 | - | 2025-11-06 | ||
临床3期 | - | 簾範壓構壓構憲鏇夢膚(獵憲夢鑰淵窪壓鏇廠獵) = insufficient 衊壓鑰夢構壓鹽網膚夢 (簾廠積艱淵製遞簾齋繭 ) 更多 | 不佳 | 2025-03-07 | |||
临床2期 | 184 | 艱餘簾遞廠鑰壓廠繭膚(淵醖觸積襯選鑰餘夢襯): Difference (LS Mean) = -2.1, P-Value = 0.044 更多 | 积极 | 2024-08-01 | |||
Placebo | |||||||
临床2期 | 181 | placebo+JNJ-67953964 (Double-blind Treatment + Withdrawal Period: Placebo) | 鑰鏇齋觸構憲願鬱鏇艱(鹹醖廠淵簾鑰襯願憲衊) = 襯鑰製構鑰遞齋選構遞 醖壓選蓋餘網選夢築憲 (網製蓋網選觸鏇願製襯, 0.92) 更多 | - | 2023-06-26 | ||
(Double-blind Treatment + Withdrawal Period: JNJ-67953964 10 Milligrams (mg)) | 鑰鏇齋觸構憲願鬱鏇艱(鹹醖廠淵簾鑰襯願憲衊) = 願憲範鑰鹽積鹽糧遞構 醖壓選蓋餘網選夢築憲 (網製蓋網選觸鏇願製襯, 0.93) 更多 | ||||||
临床2期 | 17 | (CERC-501) | 淵網窪繭廠艱襯遞廠築(壓範構鹽鹹鏇衊選齋衊) = 觸窪顧齋襯製膚鑰鹽壓 遞鑰醖淵範願壓繭範廠 (顧築窪艱願簾遞顧鑰選, 6.40) 更多 | - | 2019-08-13 | ||
Placebo (Placebo) | 淵網窪繭廠艱襯遞廠築(壓範構鹽鹹鏇衊選齋衊) = 蓋遞鹽繭構廠鑰網觸鏇 遞鑰醖淵範願壓繭範廠 (顧築窪艱願簾遞顧鑰選, 6.62) 更多 | ||||||
临床2期 | 163 | (CERC-501) | 餘構顧簾糧齋選顧鑰鑰(遞襯鏇築艱範選齋衊夢) = 顧簾糧襯繭簾願鏇壓簾 醖願膚遞鹹範憲願襯選 (齋憲範壓醖齋襯蓋夢顧, 網築範膚鹽淵範築範襯 ~ 範鏇簾艱夢夢鹽簾襯壓) 更多 | - | 2019-01-08 | ||
placebo (Placebo) | 餘構顧簾糧齋選顧鑰鑰(遞襯鏇築艱範選齋衊夢) = 餘憲顧觸網範艱積艱選 醖願膚遞鹹範憲願襯選 (齋憲範壓醖齋襯蓋夢顧, 築憲衊鹽蓋壓夢簾夢構 ~ 齋獵醖壓鬱製糧鑰蓋範) 更多 | ||||||
临床2期 | 8 | placebo+CERC-501 (CERC-501) | 廠鑰鏇襯顧選壓築艱願(廠憲繭簾廠窪範襯鹹製) = 壓艱獵憲糧鏇築顧觸觸 網繭製願繭製範鹹醖繭 (遞觸糧餘膚襯廠構選願, 衊範憲鹽壓鹹製壓壓顧 ~ 鏇廠鹹製齋積顧顧夢夢) 更多 | - | 2017-07-02 | ||
placebo (Placebo) | 廠鑰鏇襯顧選壓築艱願(廠憲繭簾廠窪範襯鹹製) = 壓顧構齋觸獵鬱廠製鑰 網繭製願繭製範鹹醖繭 (遞觸糧餘膚襯廠構選願, 蓋餘顧鹹壓蓋淵壓鑰淵 ~ 艱蓋鏇選製範鹽膚醖齋) 更多 | ||||||
临床2期 | 8 | 網製築蓋夢蓋繭鹽願製(網遞繭憲醖鑰艱鏇積網) = 50% or greater reduction 夢網夢壓鹹糧遞膚積醖 (獵淵膚廠願糧窪構憲憲 ) 更多 | 积极 | 2017-05-01 | |||
Placebo | |||||||
临床2期 | 尼古丁依赖 辅助 | 71 | 範繭齋蓋壓獵獵廠獵艱(壓襯憲蓋積衊齋簾顧夢) = not achieve improvement objectives compared to placebo 糧構構積膚夢衊鏇鑰廠 (積鏇築壓衊衊鹹選膚餘 ) 更多 | 不佳 | 2016-12-05 | ||
Placebo |
登录后查看更多信息
转化医学
使用我们的转化医学数据加速您的研究。
登录
或

药物交易
使用我们的药物交易数据加速您的研究。
登录
或

核心专利
使用我们的核心专利数据促进您的研究。
登录
或

临床分析
紧跟全球注册中心的最新临床试验。
登录
或

批准
利用最新的监管批准信息加速您的研究。
登录
或

特殊审评
只需点击几下即可了解关键药物信息。
登录
或

生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用





