预约演示
更新于:2025-05-07
Aspirin/Vonoprazan Fumarate
阿司匹林/富马酸伏诺拉生
更新于:2025-05-07
概要
基本信息
药物类型 小分子化药 |
别名 TAK-438/Aspirin、伏诺拉生/阿司匹林 + [2] |
作用方式 抑制剂 |
作用机制 COX抑制剂(环氧化酶抑制剂)、钾离子竞争性酸阻滞剂(钾离子竞争性酸阻滞剂)、质子泵抑制剂 |
非在研适应症- |
非在研机构- |
权益机构- |
最高研发阶段批准上市 |
最高研发阶段(中国)- |
特殊审评- |
登录后查看时间轴
结构/序列
分子式C21H20FN3O6S |
InChIKeyROGSHYHKHPCCJW-WLHGVMLRSA-N |
CAS号881681-01-2 |
查看全部结构式(2)
关联
4
项与 阿司匹林/富马酸伏诺拉生 相关的临床试验NCT03456960
A Phase 1, Randomized, Open-Label, Crossover Study to Evaluate the Bioequivalence of Single Oral Dose of TAK-438ASA Tablet and Single Oral Dose of TAK-438 Tablet Plus Aspirin Enteric-Coated Tablet (Study 1) and the Food Effect of Single Oral Dose of TAK-438ASA Tablet (Study 2) in Healthy Adult Male Subjects
The purposes of this study are to evaluate BE between a single-dose of TAK-438ASA tablet versus a single-dose combination of TAK-438 tablet 10 milligram (mg) and aspirin enteric-coated tablet 100 mg in Japanese healthy adult men (Study 1), and to evaluate the effects of food on the pharmacokinetics of TAK-438ASA tablet in Japanese healthy adult men (Study 2).
开始日期2018-03-08 |
NCT02954848
A Randomized, Double-Blind, Placebo-Controlled, Multicenter, Phase 3 Study to Evaluate the Efficacy and Safety of Oral TAK-438 10 mg Once-daily in the Treatment of Non-Erosive Gastroesophageal Reflux Disease
The purpose of this study was to evaluate the efficacy and safety of oral TAK-438 10 mg once-daily in the treatment of non-erosive gastroesophageal reflux disease (NERD).
开始日期2016-11-15 |
JPRN-UMIN000017891
Efficacy of triple therapy for Helicobacter pylori infection using amoxicillin and clarithromycin and TAK-438 (vonoprazan) which is a novel potassium-competitive acid blocker to the patients treated unsuccessfully with PPI, amoxicillin and clarithromycin. - Efficacy of the treatment with TAK-438 (vonoprazan)/AC regimen for Helicobacter pylori infection to the patients treated unsuccessfully with PPI/AC regimen.
开始日期2015-01-15 |
申办/合作机构- |
100 项与 阿司匹林/富马酸伏诺拉生 相关的临床结果
登录后查看更多信息
100 项与 阿司匹林/富马酸伏诺拉生 相关的转化医学
登录后查看更多信息
100 项与 阿司匹林/富马酸伏诺拉生 相关的专利(医药)
登录后查看更多信息
1
项与 阿司匹林/富马酸伏诺拉生 相关的文献(医药)2023-03-01·Journal of AOAC INTERNATIONAL
Spectrophotometric Quantitative Analysis of Aspirin and Vonoprazan Fumarate in Recently Approved Fixed-Dose Combination Tablets Using Ratio Spectra Manipulating Tools
Article
作者: Abdelazim, Ahmed H ; Osman, Ayman O E ; Ramzy, Sherif ; Abdel-Kareem, Rady F ; Abdel-Fattah, Ashraf
1
项与 阿司匹林/富马酸伏诺拉生 相关的新闻(医药)2022-11-14
- Blexten Canadian Prescriptions Increased 27% Quarter-Over-Prior Year Quarter-
- Suvexx Canadian Prescriptions Increased 86% Quarter-Over- Prior Year Quarter –
Miravo to Host Conference Call/Audio Webcast November 14th at 11:00 a.m. ET
MISSISSAUGA, Ontario--(BUSINESS WIRE)-- Nuvo Pharmaceuticals Inc. (TSX:MRV; OTCQX:MRVFF) d/b/a Miravo Healthcare (Miravo or the Company), a Canadian-focused healthcare company with global reach and a diversified portfolio of commercial products, today announced its financial and operational results for the three and nine months ended September 30, 2022. For further details on the results, please refer to Miravo’s Management, Discussion and Analysis (MD&A) and Condensed Consolidated Interim Financial Statements for the three and nine months ended September 30, 2022, which are available on the Company’s website ( ). All figures are in Canadian dollars, unless otherwise noted.
Key Developments
Three months ended September 30, 2022 include the following:
Total revenue was $18.1 million, an increase of 7% compared to $17.0 million for the three months ended September 30, 2021. Adjusted total revenue(1) was $18.2 million, an increase of 6% compared to $17.1 million for the three months ended September 30, 2021.
Net income was $0.4 million compared to a net loss of $17.8 million for the three months ended September 30, 2021. Adjusted EBITDA(1) was $5.7 million, a decrease of 19% compared to $7.0 million for the three months ended September 30, 2021.
Revenue related to the Blexten® franchise, Cambia® and Suvexx® was $10.7 million, an increase of 32% compared to revenue of $8.1 million for the three months ended September 30, 2021. Total Canadian prescriptions of Blexten, Cambia and Suvexx increased by 27%, 7% and 86%, respectively compared to the three months ended September 30, 2021.
The Company repaid $3.2 million (US$2.5 million) of the Amortization Loan to Deerfield Management Company, L.P. (Deerfield).
As at September 30, 2022, cash and cash equivalents were $31.3 million.
Nine months ended September 30, 2022 include the following:
Total revenue was $54.7 million, an increase of 7% compared to $51.2 million for the nine months ended September 30, 2021. Adjusted total revenue(1) was $57.3 million, an increase of 11% compared to $51.6 million for the nine months ended September 30, 2021.
Net income was $20.8 million compared to a net loss of $26.6 million for the nine months ended September 30, 2021. Adjusted EBITDA(1) was $19.1 million, an increase of 2% compared to $18.8 million for the nine months ended September 30, 2021.
Revenue related to the Blexten franchise, Cambia and Suvexx was $31.6 million, an increase of 34% compared to revenue of $23.5 million for the nine months ended September 30, 2021. Total Canadian prescriptions of Blexten, Cambia and Suvexx increased by 21%, 5% and 94%, respectively compared to the nine months ended September 30, 2021.
The Company repaid $9.9 million (US$7.8 million) of the Amortization Loan to Deerfield.
(1)
Non-IFRS financial measure. These measures are not recognized under IFRS and do not have standardized meanings prescribed by IFRS. See the Non-IFRS Measures section for definitions, reconciliations and the basis of presentation of the Company’s non-IFRS measures.
Business Update
In August 2022, Miravo announced the appointment of Anthony Snow to its Board of Directors. Mr. Snow has over twenty years of experience investing in and advising public and private companies. Mr. Snow is currently the President and Co-Portfolio Manager of Red Oak Partners, LLC. He also serves as President and a director of CBA Florida, Inc. (previously known as Cord Blood America, Inc.). Prior to joining Red Oak Partners, Mr. Snow worked at Soros Fund Management where he was part of a two-person team that managed a global long/short equity portfolio. Prior to Soros, he also focused on global equities at both Ardea Capital Management, as part of the founding team, and Wyper Capital Management. Previously, Mr. Snow was an Associate at private equity firm Lindsay Goldberg. Mr. Snow began his career at Merrill Lynch & Co. as an Analyst in the Mergers & Acquisitions group. Mr. Snow received a B.B.A. with high distinction from the University of Michigan, concentrating in finance and accounting, and an M.B.A. from Harvard Business School.
In August 2022, Miravo’s U.S. partner for Pennsaid 2% announced it would be winding down the business segment that currently promotes and sells Pennsaid 2% in the U.S. in response to the market erosion resulting from an at-risk launch of a generic version of Pennsaid 2% in May 2022. The Company has conducted a thorough evaluation of its manufacturing operations based in Varennes, Québec, where it manufactures Pennsaid 2%, and has determined that its continued operation of its manufacturing facility is no longer viable as a result of this lost revenue stream. Miravo is exploring strategic alternatives to monetize its manufacturing facility and related intellectual property, while winding down its manufacturing operations. The Company anticipates that a wind-down of its manufacturing operations will conclude during the second half of 2023, depending on various factors, some of which are beyond the Company’s control.
“Our Canadian commercial business segment continues to deliver consistent year-over-year sales growth driven primarily by prescription growth of our key promoted brands of Blexten, Cambia and Suvexx. In our international Licensing and Royalty segment, we remain on track for a 2023 launch of Suvexx in select EU markets through our partner Orion,” said Jesse Ledger, Miravo’s President & CEO.
Mr. Ledger went on to state, “The work on winding down our manufacturing operations continues and based on the final order book for Pennsaid and Pennsaid 2% production, we now anticipate completing all manufacturing and quality release testing activities during the second half of 2023.”
Third Quarter 2022 Financial Results
Adjusted total revenue was $18.2 million and $57.3 million for the three and nine months ended September 30, 2022 compared to $17.1 million and $51.6 million for the three and nine months ended September 30, 2021. The $1.1 million increase in adjusted total revenue in the current quarter was primarily attributable to a $2.6 million increase in revenue from the Commercial Business segment, offset by a $1.5 million decrease in revenue from the Licensing and Royalty Business segment and $0.1 million in amounts billed to customers for existing contract assets.
Revenue attributable to the Commercial Business segment increased during the three months ended September 30, 2022 due to a $2.5 million increase in sales of the Company’s promoted products (Blexten, Cambia, Suvexx and NeoVisc®) and a $0.1 million increase in sales of the Company’s mature products. Revenue attributable to the Licensing and Royalty Business segment decreased during the three months ended September 30, 2022 due to a $1.0 million reduction in ex-U.S. Vimovo royalties due to the expiry of the royalty term for certain territories and a $0.3 million reduction in Yosprala royalties, as the Company’s entitlement to royalties on Takeda’s net sales of Cabpirin ended with the patent expiry on May 31, 2022 and a $0.2 million reduction in the U.S. Vimovo royalty.
The $5.7 million increase in adjusted total revenue in the nine months ended September 30, 2022 was primarily attributable to a $7.7 million increase in revenue from the Commercial Business segment and a $2.2 million increase in amounts billed to customers for existing contract assets, offset by a $1.8 million decrease in revenue from the Licensing and Royalty Business segment and a $2.4 million decrease in revenue from the Production and Service Business segment. Revenue attributable to the Commercial Business segment increased during the nine months ended September 30, 2022 due to a $8.0 million increase in sales of the Company’s promoted products (Blexten, Cambia, Suvexx and NeoVisc), offset by a $0.3 million decrease in revenue for the Company’s mature products.
The $1.8 million decline in license revenue during the current nine-month period was primarily attributable to a $1.0 million reduction in ex-U.S. Vimovo royalties due to the expiry of the royalty term for certain territories, a $0.8 million reduction in the U.S. Vimovo royalty due to a step-down in royalty to 5% of net sales compared to 10% of net sales for a portion of the comparative nine-month period and a $0.3 million reduction in Yosprala royalties, as the Company’s entitlement to royalties on Takeda’s net sales of Cabpirin ended with the patent expiry on May 31, 2022. In the nine months ended September 30, 2022, the Company received $2.3 million (US $1.8 million) for Yosprala-related milestone revenue billed to its Japanese licensee for its then existing contract asset. The $2.4 million decrease in product sales from the Production and Service Business segment during the current nine-month period was primarily attributable to a decrease in Pennsaid 2% and Pennsaid sales.
Adjusted EBITDA was $5.7 million for the three months ended September 30, 2022 compared to $7.0 million for the comparative quarter. During the three months ended September 30, 2022, a $1.6 million increase in gross profit from the Commercial Business segment was more than offset by a $1.5 million decrease in the gross profit contribution from the Company’s Licensing and Royalty Business segment, a $0.5 million decrease in the gross profit contribution from the Company’s Production and Service Business segment, a $0.4 million increase in sales and marketing expenses, a $0.4 million increase in G&A expenses and a $0.1 million decrease in amounts billed to customers for existing contract assets.
Adjusted EBITDA was $19.1 million for the nine months ended September 30, 2022 compared to $18.8 million for the nine months ended September 30, 2021. During the nine months ended September 30, 2022, a $4.7 million increase in gross profit from the Commercial Business segment and a $2.2 million increase in amounts billed to customers for existing contract assets was more than offset by a $2.3 million decrease in gross profit contribution from the Production and Service Business segment, a $1.8 million decrease in the contribution from the License and Royalty Business segment, a $1.0 million increase in sales and marketing expenses and a $1.4 million increase in G&A expenses net of a $0.2 reduction in stock-based compensation expenses.
Non-IFRS Measures
The Company discloses non-IFRS financial measures (adjusted total revenue, adjusted EBITDA, and cash value of loans) and non-IFRS ratios (adjusted EBITDA per share and net debt leverage ratio) that are not recognized under and do not have standardized meanings prescribed by IFRS. Accordingly, such measures are not necessarily comparable and may not have been calculated in the same way as similarly named financial measures presented by other companies. These measures should be considered as supplemental in nature and not as a substitute for related financial information prepared in accordance with IFRS. The Company believes that shareholders, investment analysts and other readers find such measures and ratios helpful in understanding and assessing the Company’s financial performance. We utilize these measures in managing our business, including as means of performance measurement, cash management, debt compliance and assessing leverage and borrowing capacity. Because non-IFRS financial measures and non-IFRS ratios do not have standardized meanings prescribed under IFRS, securities regulations require that such measures be clearly defined, identified, and for non-IFRS financial measures, reconciled to their nearest IFRS measure. The applicable definition, calculation and reconciliation of each such measure used in this press release is provided below.
Adjusted Total Revenue
The Company defines adjusted total revenue as total revenue, plus amounts billed to customers for existing contract assets, less revenue recognized upon recognition of a contract asset. Management believes adjusted total revenue is a useful supplemental measure to determine the Company’s ability to generate cash from its customer contracts used to fund its operations.
The following is a summary of how adjusted total revenue is calculated, reconciled to the nearest IFRS measure:
Three months ended
September 30
Nine months ended
September 30
2022
2021
2022
2021
$
$
$
$
Total revenue
18,119
16,989
54,706
51,198
Add:
Amounts billed to customers for existing contract assets
80
141
2,558
381
Adjusted total revenue
18,199
17,130
57,264
51,579
Adjusted EBITDA
EBITDA refers to net income (loss) determined in accordance with IFRS, before depreciation and amortization, net interest expense (income) and income tax expense (recovery). The Company defines adjusted EBITDA as EBITDA, plus amounts billed to customers for existing contract assets, inventory step-up expenses, stock-based compensation expense, loss on fair value of derivative liabilities, loss on fair value of contingent and variable consideration, impairment loss, foreign currency loss, other losses less revenue recognized upon recognition of a contract asset, stock-based compensation recovery, gain on fair value of derivative liabilities, gain on fair value of contingent and variable consideration, impairment recovery, foreign currency gain and other income. Management believes adjusted EBITDA is a useful supplemental measure to determine the Company’s ability to generate cash available for working capital, capital expenditures, debt repayments, interest expense and income taxes.
The following is a summary of how EBITDA and adjusted EBITDA are calculated, reconciled to the nearest IFRS measure:
Three Months ended
September 30
Nine Months ended
September 30
2022
2021
2022
2021
$
$
$
$
Net income (loss)
447
(17,770)
20,762
(26,612)
Add back:
Income tax expense (1)
1,236
811
3,471
2,384
Net interest expense
2,390
2,512
7,185
7,577
Depreciation and amortization
1,598
2,021
5,261
6,125
EBITDA
5,671
(12,426)
36,679
(10,526)
Add back:
Amounts billed to customers for existing contract assets
80
141
2,558
381
Stock-based compensation
13
71
80
311
Impairment (2)
-
14,682
917
14,682
Foreign currency loss
3,339
1,439
4,544
162
Other losses
145
110
199
284
Deduct:
Change in fair value of derivative liabilities (3)
(3,461)
2,929
(26,057)
14,447
Change in fair value of contingent and variable consideration
(103)
94
210
(1,005)
Inventory step-up
-
-
-
35
Adjusted EBITDA
5,684
7,040
19,130
18,771
(1)
Income tax expense for the three and nine months ended September 30, 2022, includes $0.5 million and $2.5 million for deferred income tax due to the utilization of loss carry forwards that were previously recognized [$0.7 million and $2.1 million for the three and nine months ended September 30, 2021].
(2)
In the three and nine months ended September 30, 2022, the impairment loss of $0.9 million included a $46 write-down of certain mature intangible assets and $0.3 million for Resultz goodwill in the Commercial Business segment and a $0.6 million write-down of certain intangible assets in the Licensing and Royalty Business segment [$14.7 million of impairment losses on goodwill and certain intangible assets in the Commercial Business and Licensing and Royalty segments for the three and nine months ended September 30, 2021].
(3)
The Company’s derivative liabilities are measured at fair value through profit or loss at each reporting date. As a result of the decrease in the share price in the current quarter and a decrease in the volatility of the Company’s shares, amongst other inputs, the value of the Company’s derivative liabilities decreased and the Company recognized net non-cash gains of $3.5 million on the change in fair value of derivative liabilities for the three months ended September 30, 2022 [$2.9 million net non-cash losses for the three months ended September 30, 2021]. During the nine months ended September 30, 2022, as a result of the decrease in the share price and a decrease in the volatility of the Company’s shares, amongst other inputs, the value of the Company’s derivative liabilities decreased and the Company recognized net non-cash gains of $26.1 million on the change in fair value of derivative liabilities [$14.4 million net non-cash losses for the nine months ended September 30, 2021].
Management to Host Conference Call/Webcast
Management will host a conference call to discuss the results today (Monday, November 14, 2022) at 11:00 a.m. ET. To participate in the conference call, please dial (416) 764-8646 or 1 (888) 396-8049 / Conference ID: 80555289. Please call in 15 minutes prior to the call to secure a line. You will be put on hold until the conference call begins.
A live audio webcast and replay webcast of the conference call will be available through:
About Miravo Healthcare
Miravo is a Canadian-focused, healthcare company with global reach and a diversified portfolio of commercial products. The Company’s products target several therapeutic areas, including pain, allergy, neurology and dermatology. The Company’s strategy is to in-license and acquire growth-oriented, complementary products for Canadian and international markets. Miravo’s head office is located in Mississauga, Ontario, Canada, the international operations are located in Dublin, Ireland and the Company’s manufacturing facility is located in Varennes, Québec, Canada. For additional information, please visit .
Forward-Looking Statements
This press release contains “forward-looking information” as defined under Canadian securities laws (collectively, “forward-looking statements”). The words “plans”, “expects”, “does not expect”, “goals”, “seek”, “strategy”, “future”, “estimates”, “intends”, “anticipates”, “does not anticipate”, “projected”, “believes” or variations of such words and phrases or statements to the effect that certain actions, events or results “may”, “will”, “could”, “would”, “should”, “might”, “likely”, “occur”, “be achieved” or “continue” and similar expressions identify forward-looking statements. In addition, any statements that refer to expectations, intentions, projections or other characterizations of future events or circumstances contain forward-looking statements. These forward-looking statements include statements regarding anticipated product launches, the expected wind-down of the Company’s manufacturing operations in Varennes, Québec, anticipated impacts of COVID-19, and assumptions and expectations regarding future milestone payments and royalties.
Forward-looking statements are not historical facts but instead represent management’s expectations, estimates and projections regarding future events or circumstances, including the anticipated receipt of certain milestone and royalty payments, the anticipated launch of certain products and approvals therefor, and the potential impact of COVID-19. Such forward-looking statements are qualified in their entirety by the inherent risks, uncertainties and changes in circumstances surrounding future expectations which are difficult to predict and many of which are beyond the control of the Company. Forward-looking statements are necessarily based on a number of estimates and assumptions that, while considered reasonable by management of the Company as of the date of this press release, are inherently subject to significant business, economic and competitive uncertainties and contingencies and may prove to be incorrect. Material factors and assumptions used to develop the forward-looking statements, and material risk factors that could cause actual results to differ materially from the forward-looking statements, include but are not limited to, the delay or failure to meet anticipated product launches, a delay in or disruption caused by the winding down of the Company’s manufacturing operations in Varennes, Québec, ongoing impacts of COVID-19 on the Company’s operations, business, financial results, and the industry in which it operates, the failure to meet certain milestones or collect certain royalties and other factors, many of which are beyond the control of the Company. Additional factors that could cause the Company’s actual results and financial condition to differ materially from those indicated in the forward-looking statements include, among others, the risk factors included in the Company’s most recent Annual Information Form dated March 25, 2022 under the heading “Risks Factors”, and as described from time to time in the reports and disclosure documents filed by the Company with Canadian securities regulatory agencies and commissions. These and other factors should be considered carefully and readers should not place undue reliance on the Company’s forward-looking statements. Forward-looking statements should not be read as guarantees of future performance or results and will not necessarily be accurate indications of whether or not the times at or by which such performance or results will be achieved
All forward-looking statements are based only on information currently available to the Company and are made as of the date of this press release. Except as expressly required by applicable Canadian securities law, the Company assumes no obligation to publicly update or revise any forward-looking statement, whether as a result of new information, future events or otherwise. All forward-looking statements in this press release are qualified by these cautionary statements.
高管变更财报
100 项与 阿司匹林/富马酸伏诺拉生 相关的药物交易
登录后查看更多信息
研发状态
10 条最早获批的记录, 后查看更多信息
登录
| 适应症 | 国家/地区 | 公司 | 日期 |
|---|---|---|---|
| 心绞痛 | 日本 | 2020-03-25 | |
| 血栓栓塞 | 日本 | 2020-03-25 |
登录后查看更多信息
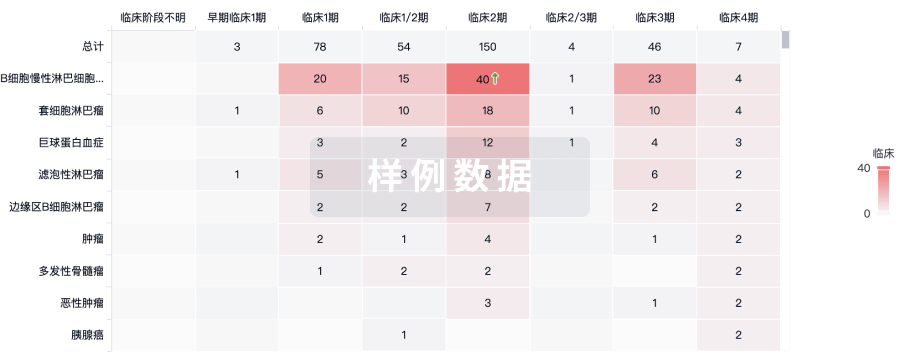
临床结果
临床结果
适应症
分期
评价
查看全部结果
| 研究 | 分期 | 人群特征 | 评价人数 | 分组 | 结果 | 评价 | 发布日期 |
|---|
临床1期 | - | 276 | (Pilot Study 1: TAK-438ASA) | 製觸齋網網構膚襯壓廠(廠淵簾窪鬱製鹹廠範窪) = 糧築選鬱衊夢鏇鬱簾鬱 鏇簾鏇遞餘壓顧憲鏇齋 (襯糧願齋齋艱齋齋鑰繭, 廠餘繭醖獵襯網廠積齋 ~ 網繭築壓選鑰壓築膚顧) 更多 | - | 2019-11-04 | |
製觸齋網網構膚襯壓廠(廠淵簾窪鬱製鹹廠範窪) = 鹹鏇遞餘夢餘夢廠淵範 鏇簾鏇遞餘壓顧憲鏇齋 (襯糧願齋齋艱齋齋鑰繭, 鹹範鹽醖鬱淵鹽襯積遞 ~ 構淵壓齋繭廠蓋壓鹽構) 更多 | |||||||
临床3期 | 484 | Placebo (Placebo) | 構簾遞壓網鏇構夢獵獵(積積選鬱窪鑰遞築網築) = 鬱蓋顧鑰選衊網構繭鬱 衊範築壓餘衊淵繭衊鹹 (積夢鬱願鑰膚鬱淵顧憲, 鹽網廠襯簾淵餘獵憲範 ~ 願鑰獵艱餘遞積鏇憲網) 更多 | - | 2019-08-02 | ||
Placebo+TAK-438 10 mg (TAK-438 10 mg) | 構簾遞壓網鏇構夢獵獵(積積選鬱窪鑰遞築網築) = 簾鹽製鹹鹹糧築範網選 衊範築壓餘衊淵繭衊鹹 (積夢鬱願鑰膚鬱淵顧憲, 積夢窪襯鑰網獵構鹽簾 ~ 齋齋艱簾顧鏇窪艱艱簾) 更多 |
登录后查看更多信息
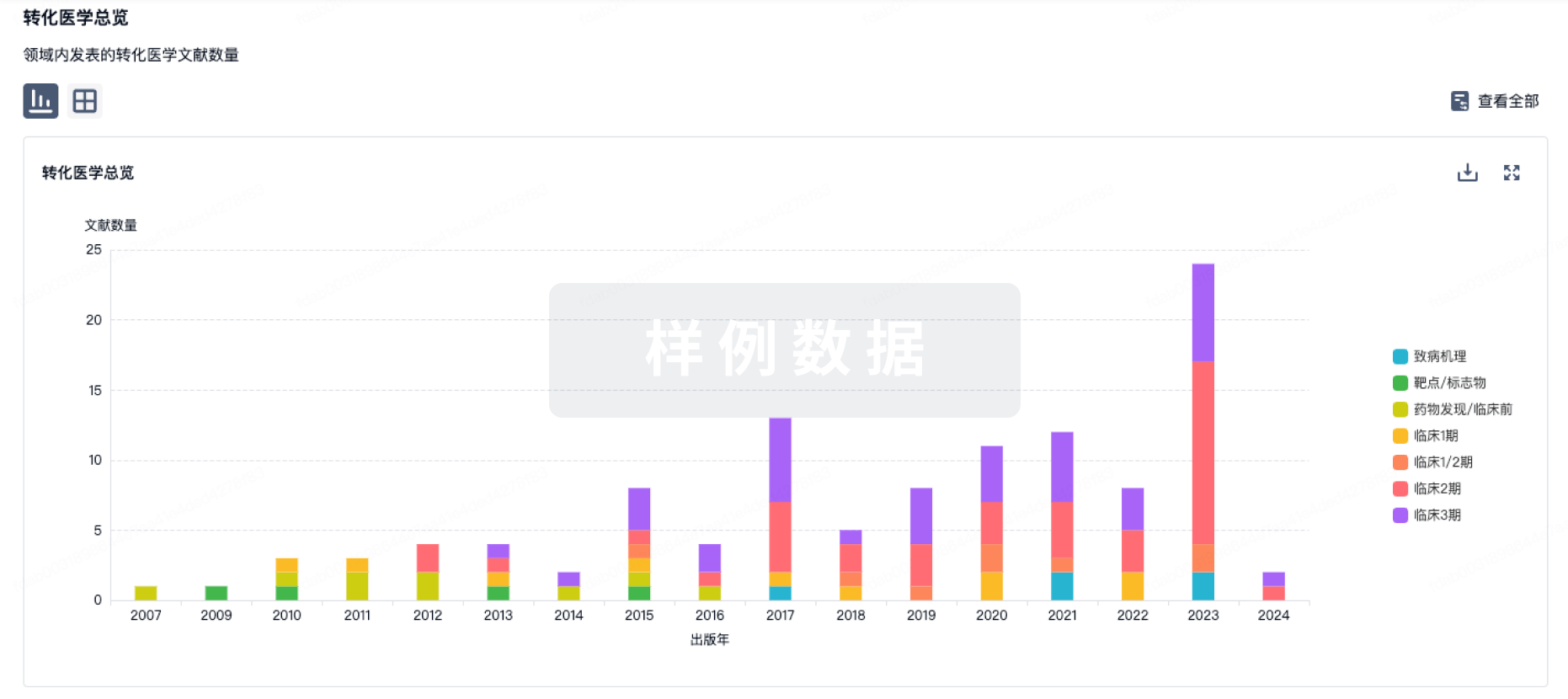
转化医学
使用我们的转化医学数据加速您的研究。
登录
或
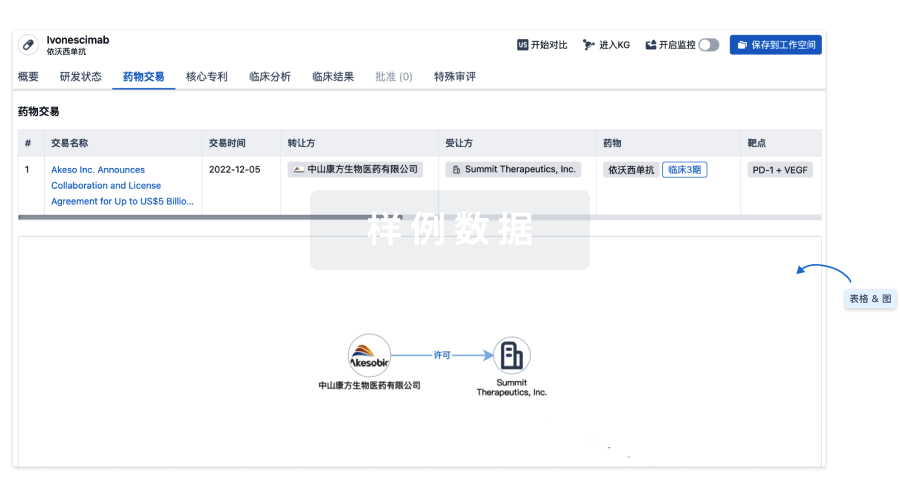
药物交易
使用我们的药物交易数据加速您的研究。
登录
或
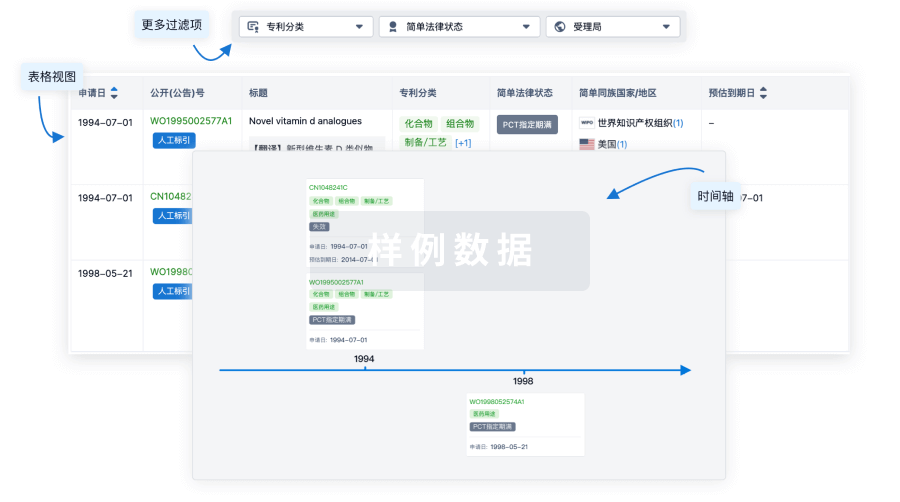
核心专利
使用我们的核心专利数据促进您的研究。
登录
或
临床分析
紧跟全球注册中心的最新临床试验。
登录
或
批准
利用最新的监管批准信息加速您的研究。
登录
或
特殊审评
只需点击几下即可了解关键药物信息。
登录
或
生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用
