更新于:2024-05-01
Yichang Humanwell Pharmaceutical Co. Ltd.
更新于:2024-05-01
概览
关联
100 项与 Yichang Humanwell Pharmaceutical Co. Ltd. 相关的临床结果
登录后查看更多信息
0 项与 Yichang Humanwell Pharmaceutical Co. Ltd. 相关的专利(医药)
登录后查看更多信息
10
项与 Yichang Humanwell Pharmaceutical Co. Ltd. 相关的文献(医药)2020·Zhongguo Yiyao Gongye Zazhi
Improved Synthetic Process of Rilpivirine Hydrochloride
作者: Guo Jianfeng ; Chen Yi ; Li Li'e ; Tian Luanyuan ; Wang Miao
The synthetic process of the antiviral drug rilpivirine hydrochloride(1) was improved. Et formyl acetate(2) reacted with S-methylisothioure(3) to give 2-(methylthio)-4-hydroxyl-pyrimidine(4). Compound 4 reacted with 4-aminobenzonitrile(5) in ethylene glycol di-Me ether in the presence of methanesulfonic acid to give 4-[(4-hydroxypyrimidin-2-yl)amino]benzonitrile(6), and the reaction temperature decreased from 200 °C to 110-120 °C. Compound 6 reacted with HBr/CH3 COOH to obtain 4-[(4-bromopyrimidin-2-yl)amino]benzonitrile(7). 4-Bromo-2,6-dimethylaniline(8) reacted with acrylonitrile(9) in the presence of palladium acetate and tris(Omethylphenyl)phosphine instead of 10% Pd/C to give(E)-3-(4-amino-3,5-dimethylphenyl)acrylonitrile(10), by which the cis-isomer was reduced from 20% to 0.60%. Meanwhile, compound 10 was purified by recrystallization instead of column chromatog. in the work-up, so that the yield of this step was increased from 48% to 87%. Compound 7 was condensed with 10 under the action of 1,1,3,3-tetramethylguanidine(TMG) as acid binding agent, and the title compound in a purity of 99.92% was obtained via a salt formation with oxalic acid, and the content of cis-isomer was reduced to 0.08%. The improved process was simple and convenient with a total yield of 52%(based on 2).
2020·Zhongguo Xinyao Zazhi
Synthesis of morphine-6-β-D-glucuronic acid glycosides
作者: Guo Jian-feng ; Wang Miao ; Tian Luan-yuan ; Li Li-e ; Chen Yi ; Li Yu-qiong
Objective: To study the Synthesis process of morphine-6-β-D-glucuronic acid glycosides. Methods: Using morphine sulfate as raw material, 3-benzoyl morphine was prepared by esterification reaction. Using 1, 2, 3, 4-four-O-acetyl-β-D-glucuronic acid Me ester as raw material, the intermediate 1-three fluorine acetyl groups and 2,3,4-three-O-acetyl-β-D-glucuronic acid Me ester was prepared by ester exchange reaction. The intermediate 4 was coupled with intermediate 6, followed by glycosidic reaction to give 3-benzoyl acetyl glycoside(7), and then morphine-6-β-D-glucuronic acid glycosides was prepared by hydrolysis reaction. Results: The structure of M6 G was confirmed by ∼1H-NMR, ∼(13)C-NMR, high resolution MS and X-ray diffraction, with an overall yield of 40.3%. The purity was 99.8% determined by HPLC.Conclusion: This synthetic process has advantages of easy operation, good quality, high yield of total product, high yield and stable quality of glycosidic reaction, and is suitable for industrial production
2015-08-10·Zhongguo Yiyao Gongye Zazhi
Synthesis of salidroside
作者: Guo, Jianfeng ; Fu, Yigang ; Wang, Menghua ; Lu, Jinliang ; Zhang, Guolong
Salidroside was synthesized from glucose via iso-Bu acylation and trifluoroacetylation to give 1-trifluoroacetyl-2,3,4,6-O-tetraisobutyrylglucose (5) which was subjected to glycosylation with 4-(2-hydroxyethyl) Ph benzoate to obtain 1-[2-[4-(benzoyloxy) phenethoxy] ethyl]-2,3,4,6-O-tetraisobutyryl-β-D-glucopyranoside (7), followed by deprotection under basic conditions with an overall yield of 55% and purity of 99.5%. Compounds 5 and 7 had not been reported in the literature.
11
项与 Yichang Humanwell Pharmaceutical Co. Ltd. 相关的新闻(医药)2022-07-18
·生物探索
导语:科兴新冠口服药获批;加拿大现新冠后遗症“安乐死”;白云山头孢拉定通过仿制药一致性评价……生物探索与您一同关注“药”闻,探索生物科技的价值!01科兴新冠口服药获批7月17日,科兴制药全资子公司深圳科兴收到国家药品监督管理局签发的《药物临床试验批准通知书》,国家药品监督管理局批准同意深圳科兴与深圳安泰维生物医药有限公司合作开发的新冠小分子口服药SHEN26胶囊进行临床试验。据披露,SHEN26是一款新型冠状病毒聚合酶(RdRp)抑制剂,可以通过抑制病毒核酸合成达到抗病毒效果。临床前研究显示,SHEN26在体内外都表现出显著的抗SARSCoV-2作用,对新冠病毒及其变异株的抑制活性显著,对原始株和已知重要变异株均表现出抗病毒活性,同时药物机制明确,口服生物利用度好,在动物重复给药上安全性好,无致突变风险,无脱靶效应。02加拿大现新冠后遗症“安乐死”近日,加拿大一名50多岁女子Tracey Thompson因长期新冠后遗症申请“安乐死”。据悉,Thompson之前是一名厨师,但她现在连站着喝水都有困难。新冠康复后,Thompson视力变差,日落前后变得视线模糊;难以消化食物;嗅觉和味觉都发生了变化;呼吸困难等等。由于无法重回工作岗位,Thompson已经长达26个月没有收入,预计再过5个月就将耗尽积蓄。由于“长新冠”没有明确的定义,Thompson一时难以申请安大略省的残疾生活补助。长期无法工作,没有收入来源,同时也缺乏政府支援,这让Thompson深陷绝望之中,因此,她决定申请合法安乐死。Thompson表示:“据我所知,我符合标准。因为我病得很重,而且没有治愈方法,不必等到绝症才能申请安乐死……”03白云山头孢拉定通过仿制药一致性评价近日,A股公司白云山(600332.SH)发布:分公司广州白云山医药集团股份有限公司白云山制药总厂(白云山制药总厂)收到国家药品监督管理局签发的《药品补充申请批准通知书》(通知书编号:2022B02676、2022B02677),头孢拉定胶囊(0.125g、0.25g)已通过仿制药质量和疗效一致性评价。白云山制药总厂的头孢拉定胶囊于1992年1月在国内正式上市,并于2020年12月 24日向国家药品监督管理局递交一致性评价申请,于2021年1月21日获得受理。头孢拉定为第一代头孢菌素类抗生素,适用于敏感菌所致的急性咽炎、扁桃体炎、中耳炎、支气管炎和肺炎等呼吸道感染、泌尿生殖道感染及皮肤软组织感染等。04真实生物阿兹夫有望成为我国首个口服抗新冠药物近日,河南真实生物科技有限公司宣布,近日已正式向国家药品监督管理局提交阿兹夫定治疗新型冠状病毒适应症的上市申请并获得受理。获批开展Ⅲ期临床试验两年多的阿兹夫定,有望成为我国首个具有完全自主知识产权的口服抗新冠药物。据了解,阿兹夫定是全球首个双靶点抗艾滋病创新药,已在中国、美国等多个国家申请专利并获授权。作为一款抗病毒小分子口服药,阿兹夫定具有广谱抑制RNA病毒复制的作用,而新冠病毒同属RNA作为遗传物质的病毒,因此该药对新冠病毒有抑制作用。05华人福医药盐酸氢吗啡酮缓释片申报生产获受理近日,人福医药(600079)公告,控股子公司宜昌人福近日收到国家药品监督管理局核准签发的盐酸氢吗啡酮缓释片申报生产的《受理通知书》。盐酸氢吗啡酮是一种阿片类药物,属于吗啡衍生物,主要用于镇痛,目前国内仅有宜昌人福同品种注射液获批上市,暂无口服剂型批准上市销售,2021年,宜昌人福盐酸氢吗啡酮注射液销售额约为4亿元。截至目前,宜昌人福该项目累计投入约4500万元。整理|文竞择排版|文竞择
End
往期精选
围观新冠疫苗皮肤不良反应:80.2%出现在女性身上!包括荨麻疹、带状疱疹……
热文
一份外卖吃下去,万亿颗微塑料入体,你知道对身体有多大影响吗?
热文
晚饭吃太晚,疾病找上门!你把握住这一最佳晚餐时间了吗?热文
两针13300元真的能预防新冠?全球唯一一款新冠预防药已在海南开放使用!
热文
Al机器人又添用武之地!可预测胚胎质量,提高试管婴儿成功率
点击“阅读原文”,了解更多~
一致性评价仿制药创新药小分子药物疫苗
2022-07-17
市场和政策始终是动态的,但国家的方向是明确的:让老百姓用上质优价廉的药品,是一个不会走回头路的方向。集采始终是企业向前发展不可逾越的一个阶段性政策,但同时对于创新、对于市场销售来说又是一个很好的机会。当5年、10年之后再回头看,企业不会遗憾地说,当时有这样的一个机会,但是没有把握住。7月14号,第七批集采的拟中标结果公布,295家企业的488个产品参与投标,217家企业的327个产品获得拟中选资格,48%的平均降幅看似温和又“暗藏杀机”:奥美拉唑以28进10、入选者降幅均超过90%获封“最卷品种”;抗癌药仑伐替尼降幅高达97%,先声药业以最低价3.2元中标,抗癌药的价格赶不上一支雪糕······有人唏嘘,有人震惊。但这次集采也带给了行业和企业更多的思考:集采已经进行了七次,未来的方向是什么?在这次集采中,企业采用的报价策略是什么?规则有哪些变化?集采之后的企业又该如何应对接下来的发展?Q:E药经理人A:风云药谈创始人 张廷杰Q:当开标的结果出来之后,有没有哪些品种的结果是出乎你的意料的?你怎么看最终的报价结果?A:在国采之前,我(风云药谈)对36个产品做过报价预测,相对结果来看,90%左右的价格都在预期之内。也有几个品种的降幅确实是非常大。以竞争最激烈的奥美拉唑为例,最终是28进10的格局,从结果上来看也是非常“惨烈”,而惨烈的结果是中标价格已经非常接近产品的成本。如果核算原料成本的话,按照每支40毫克的规格,每支原料药的成本大概不到一毛钱,但是从结果来看,奥美拉唑注射剂的第一顺位中标价还不到7毛钱,再加上西林瓶和胶塞等原辅包材、运费、配送费,实际上这个价格已经是极限接近成本了。另外一个品种奥司他韦,目前它的原料还是不便宜的,在国采前又有几家产品通过一致性评价,在开标之前我就预测,如果广东东阳光投一个很低的价格,就可能拿市场最高的份额,而原有宜昌人福的品牌奥司他韦可以销售高价,从市场策略上看是非常成功的。从结果上来看,第一顺位1.13元的价格也是非常接近成本的报价,说明企业也确实是这种低价的策略,同时也证明了了风云药谈的逻辑和东阳光的报价策略不谋而合。还有丙酚替诺福韦这个品种,从第一顺位的0.36元到第五顺位的0.56元,价格都是比较低的,这个产品的限价在18元左右,如果按照原研的销售价格体系来算,原研可能会在30元左右,那就相当于降幅达到了99%。虽然这次的平均降幅在49%左右,表面上看是并没有太大的降幅,但因为此次限价偏低,有些品种的竞争还是比较激烈的,大家又都是比较“拼”的状态,所以最终有些品种虽然价格“打”的比较狠,在开标之前,都是整体有预期的。Q:这次集采跟前几批集采的话,有哪些比较大的不同?A:从政策端大概有九个方面的不同。一是纳入的条件发生了一定的变化。国采第2至5批的政策已经比较成熟,当时都是3家入围,第七批的话是要综合满足4家。实际上在这次集采没有正式发布之前,也有相关的信息说会有数量上的提高。这次集采也给了我们一个信号,今后的集采在纳入的数量级上会逐渐有所提升,包括稳定在4家或者提高到5家6家都是有可能的;二是时间节点发生了变化。从政策角度,之前六批集采的规定,都是在报价之前产品通过一致性评价就可以,但是现在的要求是要在报价之前的5天拿到批文。所以在7月7号这天前后,NMPA网站上也公布了很多产品获批的情况,包括奥曲肽注射剂在7号的零时卡点获批了三家。这对于企业来说,竞争格局可能会发生很多变化,企业就需要重新测算降幅、评定预期。三是开标地点的变化。因为有疫情,从上海改到了南京,但大家还是非常理解的。四是选区上的变化。以前是只有一个主供地区,并没有备供的策略,增加了备选选区的策略后,整体时间就拉长了。此前基本上是12号报价截止,13号就会公布最终结果,但是到现在为止,国采官网的选区还没有公布,说明选区的增多也增加了非常多的工作量。五是限价的变化。相较于二到五批集采,整体的限价是偏低的。举个例子,国产第五批是有60个左右的产品,取得原研的价格是有27个,而国产第七批取得原研的价格只有4个,这个限价给的还是偏低。六是差比价要求的变化。此次集采差比价关系参照现有规则,根据剂型、规格(装量差异按照含量差比价计算)、包装数量计算,不考虑包装、剂型的差异,这次对于部分依靠此类剂型优势抢市场的企业来说,价格压力更大。七是“品间熔断”规则缩小价差。原有确定拟中选的1.8倍的熔断机制、大于等于50%的降幅以及单位可比价小于等于0.1元的三个条件之一的基础上,还需满足同品种中非最高顺位/同品种最低顺位的“差比值”,进一步淘汰价差较大的6个品种最高价格。这也就导致企业在报价的时候会更加的谨慎。八是持有人和委托生产关系的变化,也是第七批集采的一大亮点。这次对关联企业批件转让、委托生产进行了约束,还要求中选企业履约期间持续拥有中选药品的国内有效注册批件。不再只是企业负责人为同一人或存在直接控股、管理关系的情形,增加了企业之间存在该品种部分有效注册批件转让的情形。因为这种情况没有中标的企业也有,所以相当于这次集采在极大的程度上挡住了这些人“钻漏洞”的情况。九是采购量设置更加细化。比如—部分抗生素产品带量比例较普通药品带量比例降低10%;特殊级抗生素药品进一步降低10%,较普通药品带量比例降低20%等。Q:备选地区的设置是怎样规定的?A:在此前几批的集采中,部分产品出现了断供或者供应紧张的情况。在这样的情况下,出台了备选的政策。这是国采首次设置备选的政策,但是在地方的带量采购中已经有过类似的方案,比如广东和河南的集采联盟,就有了针对国采和省采产品的备选方案。增加备选一方面是保证国采产品中标之后的充分供应,另一方面对于企业非中标区域的价格也是有了保证。但每个企业选择备选选区的逻辑是不一样的,在联采办的第七批的主选和备供地区公示之后,大家可以去看一下。Q:在已经有产品竞争格局的预期之下,企业是如何采取报价策略的?A:报价策略也分为几种情况,如果是一个大进位的情况,比如二甲双胍、奥帕瑞西布等竞争比较激烈的品种,没什么特别的技巧,拼的就是企业原料药生产情况、供应情况和购买成本,在生产上是否有达到极限成本的优势。所以大进位的品种通用的报价策略,基本上就是要接近产品的极限成本。对于有原研参与的品种,国产品种一般会围绕50%到65%的降价区间进行报价。从大体的逻辑上,竞争的厂家越多,通常降价幅度就会越大,这是一个逻辑上的正相关。但是统计数据来看,可能也不会完全按照这个逻辑来,因为大进位品种已经充分经过了市场的杀价。企业在制定降幅的时候,第一会根据同组产品限价,第二根据成本,第三也会根据同组企业的投标风格。Q:仑伐替尼最低报出了3.2元的价格,平均拟中标价格也在18元左右,有人说,“卖抗癌药不如卖钟薛高”,您如何看待这个结果和这种看法?A:在国采报价之前,我对仑伐替尼这个产品进行过价格预测,当时预测的是,大家会围绕着8~10元左右的价格进行角逐,进入到第二阶段的限价也不会超过20。从结果上来说,大致上是在这个范围内。先声的这个报价,可能是属于个例。因为这类产品的价格一般给的限价还是比较高的,在集采中也很少会出现同组的所有厂家报价降幅都超过90%。一般来讲,报价跨度最低和最高中标价的降幅基本上是能拉开10~15个百分点,从这个角度来说,这个品种的报价有一些特殊,这种情况不能评价先声的报价或者代表集采的报价,只能算一个个例。从行业的角度来说,中国的医药行业正在通过集采走过一个降价提质、企业集中度提高的过程,政策的实行也在倒逼企业去做创新,让老百姓用上更好的药品。Q:相较于前几次集采,这次涉及到的原研品种和参与的跨国药企的数量相对有所提升,但最终只有4个产品中标,出现这些情况的原因是什么?A:其实从大样本数据来讲,外企的报价从第二批到第七批并没有发生什么变化。每一批集采都会参与、产品降幅也比较大、参与的数量也很可观,只是最终落标了而已。Q:对于有多品种进入集采的企业尤其是传统企业来说,在销售队伍的建设方面会不会承受非常大的压力?A:这个问题实际上是所有企业都在考虑和正在面对的问题。我们做一种假设,所有的企业不去做创新、不去开拓渠道,只在原有的这些产品上去做一致性评价的努力,之后纳入集采。老的产品集采完了之后,却没有新的产品跟上,在这样的情况下,销售队伍解散的逻辑是成立的。但是企业都是在不断向前发展的,以齐鲁为例,虽然每一批集采的产品有十几个,但是每年获批的产品比这个数量更多。像贝伐珠单抗、齐鲁用了两三年的时间就做到了差不多30亿的规模。风云药谈有计算过这个数据,目前,齐鲁所有集采产品从公立医院样本的放大上,也达不到30亿元的规模。对于像齐鲁这样的企业来说,他不只有参加集采这些品种,同时有新产品上市的放量,就能弥补集采造成的影响。Q:对于集采,大家认为是一个“以价换量”,扩张市场的原则,但是像恩替卡韦这个品种,在第一批集采落地当年之后,又逐渐的回到了原来的格局,原研在公立医院的占有率依然稳居第一,为什么会出现这种情况?A:从数据上来看,原研的产品尤其是口服制剂,在集采之后出现了销量反弹的情况,但注射剂产品虽然有个别品种销量增长,但是总体上是受到了影响的。一方面是看产品。在同类型中比较新的产品,比如卡格列净、利伐沙班等产品,从全球的角度来说也是比较新的产品,在国内上市的时间比较短,在医生端、患者端教育还没有太充分,在进入集采之后增长情况就会受阻。另一方面,如果目前医保支付标准或者挂网价格还是实行双轨制的话,可能原研在终端的销售确实会抢夺阵地。这种情况从目前来看还比较普遍。可能是医疗机构的人员觉得原研和仿制药之前还是存在质量差距。国内企业可以更多的关注一下同类产品中原研的销售情况,包括推广策略等,对于国内企业还是很有借鉴意义的。Q:联盟集采、地方集采可能相对国采来说,规则会更复杂,这会不会给企业带来一些“困扰”?A:集采政策一直在完善。地方的规则如果从统一的角度来讲确实很乱,但是从另一个层面来说,统一的模式会始终只对某一类企业、某一类产品有利,对于产品多样性的企业来说,也有失公允。在公平和公正之间,地方集采的多种模式的探索值得去考量。登记邮箱信息订阅E药经理人信息服务扫描二维码
仿制药带量采购
2022-07-16
·医药观澜
▎药明康德内容团队编辑根据中国国家药监局药品审评中心(CDE)官网数据,近期多款1类创新药临床试验申请通过“默示许可”,包括抗CD20和CD3双抗、Axl和Mer双抑制剂、钇[90Y]炭微球注射液、PI3Kγ抑制剂、靶向蛋白降解剂等等,针对适应症包括恶性肿瘤、疼痛、难治性抑郁症、骨关节炎等。本文,我们将挑选部分1类新药作介绍,供读者参阅(排名不分先后)。君实生物:注射用JS203作用机制:抗CD20和CD3双抗适应症:复发难治B细胞非霍奇金淋巴瘤CD20属于B淋巴细胞限制性分化抗原,是B细胞淋巴瘤的治疗靶点之一。CD3是T细胞表面的重要标志,通过CD3介导T细胞特异性攻击肿瘤细胞,是T细胞导向的双特异性抗体的主要作用机制。根据君实生物公告,JS203为该公司自主研发的重组人源化抗CD20和CD3双特异性抗体,由抗CD20段和抗CD3段组成,它通过联结并活化T细胞(结合CD3)和淋巴瘤细胞(结合CD20),从而有效促进T细胞杀伤淋巴瘤细胞。临床前体内药效试验显示该药具有较为显著的抑瘤效果。本次,JS203获批临床的适应症为:复发难治B细胞非霍奇金淋巴瘤。纽瑞特医疗:NRT6003注射液作用机制:放射性微球适应症:转移性肝癌根据纽瑞特医疗新闻稿,NRT6003注射液为其自主研发的钇[90Y]炭微球注射液。这是一款选择性内照射治疗(SIRT)用放射性微球,是以炭微球为载体负载高活度的放射性钇[90Y]而得。具体治疗过程中,通过经导管血管造影术(DSA)介入手术将钇[90Y]炭微球经肝动脉灌注至肿瘤供血动脉,钇[90Y]炭微球随血流分布并阻滞于肿瘤组织微小动脉和毛细血管中,钇[90Y]发射纯β射线,最终通过选择性内照射治疗杀死肿瘤细胞达到治疗目的。据悉,该产品平均能量为0.9367MeV,最大组织穿透距离为11mm,平均组织穿透距离为2.5mm。此外,为最大程度降低医护人员在给药过程中受到的放射性辐照剂量和避免放射性物质泄露,纽瑞特医疗还设计了配套的给药管路系统,可实现远程、高效、安全的给药路径。本次,该产品获批临床的适应症为转移性肝癌。图片来源:123RF瀚晖制药:HS248片作用机制:PI3Kγ抑制剂适应症:晚期实体瘤PI3Kγ是磷脂酰肌醇-3-激酶(PI3K)的一种亚型。研究发现,PI3K信号通路的过度活跃与肿瘤的进展、肿瘤微血管密度的增加、癌细胞的趋化性和侵袭性增强有显著的相关性,因此PI3K信号通路被研究人员视为开发癌症治疗药物的关键靶点之一。根据海正药业公告,其全资子公司瀚晖制药申报的HS248是一种高选择性的PI3Kγ抑制剂,通过促进巨噬细胞向抗肿瘤的表型极化,增强肿瘤微环境中CD8+T细胞的活化来抑制肿瘤生长和转移。此外,通过抑制PI3Kγ,HS248可以克服免疫检查点抑制剂的耐药性,与PD-1等免疫检查点抑制剂进行联用,有望为PD-1抑制剂耐药性患者带来更多获益。本次该药在中国获批临床,拟用于治疗晚期实体瘤(尿路上皮癌、三阴性乳腺癌、黑色素瘤等)。人福医药:RFUS-144注射液作用机制:阿片受体激动剂适应症:急慢性疼痛、瘙痒症根据人福医药公告,其控股子公司宜昌人福药业申报的RFUS-144注射液是一种选择性阿片受体激动剂,临床拟用于治疗疼痛和瘙痒。该药近期在中国获批多项临床默示许可,针对适应症为急慢性疼痛、瘙痒症。疼痛是临床最常见的就诊原因之一,阿片类药物是临床常用的镇痛药物之一。此外,根据文献报道,瘙痒或痒是一种激发欲望去搔抓的感觉,多种疾病都会导致瘙痒的发生。研究证实内源性或外源性内啡肽参与瘙痒的发生,应用阿片μ受体拮抗剂与阿片κ受体激动剂可有效抑制瘙痒。索元生物:liafensine片作用机制:抗抑郁新药适应症:难治性抑郁症DB104(liafensine)是一款潜在“first-in-class”新药,可特异性地抑制在抑郁症中起重要作用的5-羟色胺、去甲肾上腺素和多巴胺的再摄取。该药最初由百时美施贵宝(BMS)/AMRI公司开发,索元生物于2017年获得了该在研新药在全球的研发、生产及销售权利。据悉,索元生物获得liafensine 后,通过其独特的生物标志物发现平台,发现了一个全新的与DB104降低抑郁症主要临床指标MADRS评分高度相关的生物标志物,命名为DGM4 (Denovo Genomic Marker 4),它可特异性地预测TRD患者接受DB104治疗后的疗效。2022年1月,DB104获美国FDA批准开展国际2b期临床试验,用于治疗难治性抑郁症(TRD)。根据索元生物早先新闻稿介绍,该临床试验是索元生物继DB102淋巴瘤国际3期、脑胶质瘤临床3期后,开展的第三个国际临床试验,也将是公司在中枢神经系统(CNS)领域开展的首个生物标志物指导下的临床试验。本次该药在中国获批临床,拟定适应症为难治性抑郁症。图片来源:123RF诺诚健华:ICP-490片作用机制:靶向蛋白降解剂适应症:多发性骨髓瘤、非霍奇金淋巴瘤等血液肿瘤据诺诚健华新闻稿介绍,ICP-490片是其自主研发的新型靶向蛋白降解剂,它通过与CRL4CRBN-E3泛素连接酶复合物的底物受体CRBN特异性结合,可诱导淋巴转录因子Ikaros和Aiolos的泛素化降解,从而诱导细胞凋亡,抑制肿瘤细胞的生存及增殖,发挥直接抗肿瘤作用。此外,ICP-490还可刺激T细胞活化,释放白介素IL-2,增强效应T细胞的功能,发挥免疫调节效应。本次该药获批在多发性骨髓瘤、非霍奇金淋巴瘤等血液肿瘤中开展临床试验。博生吉医药:TAA06注射液作用机制:B7-H3靶向CAR-T细胞注射液适应症:复发/难治性神经母细胞瘤B7-H3(又称CD276)属于B7免疫共刺激和共抑制家族,在正常人体组织中表达水平较低,但在人恶性肿瘤中异常高表达,这使得B7-H3成为治疗多种肿瘤的潜力靶点。根据博生吉医药新闻稿,TAA06注射液是该公司自主研发的一款B7-H3靶向嵌合抗原受体(CAR)-T细胞注射液。针对实体肿瘤复杂的免疫微环境,博生吉医药在TAA06注射液的靶点选择、CAR结构优化、工艺开发、以及临床方案上进行了系统性的创新和优化。在前期动物模型中,该药展现出单次静脉给药就完全清除全部肿瘤的显著疗效。本次,TAA06获批临床的适应症为复发/难治性神经母细胞瘤。康禾生物:注射用K11作用机制:重组人成纤维细胞生长因子适应症:骨关节炎、退行性软骨磨损等根据康禾生物新闻稿,注射用K11是该公司自主研发的重组人成纤维细胞生长因子(FGF)。它通过激活FGF受体,促进软骨细胞的增殖,实现对软骨层修复的作用,达到逆转病情的效果。本次K11获批临床的适应症为:促进软骨再生,可适用于骨关节炎、退行性软骨磨损等病变的治疗。康禾生物表示将于近期启动该药的1期临床试验。除了上述1类新药,还有其他创新药也已经在近期获批临床,如辉瑞(Pfizer)公司的Axl和Mer双抑制剂PF-07265807和潜在“first-in-class”的抗真菌疗法fosmanogepix(注射液和片剂剂型)、甘李药业每周注射一次的超长效胰岛素周制剂GZR4、新码生物注射用重组人源化抗CD70单抗-AS269偶联物ARX305等等,本文不再一一列举。希望这些在研疗法后续临床开发顺利进行,早日为患者造福。参考资料:[1]中国国家药监局药品审评中心(CDE)官网.Retrieved June 15,2022, From https://www.cde.org.cn/main/xxgk/listpage/4b5255eb0a84820cef4ca3e8b6bbe20c[2]各公司官网及公开资料本文来自药明康德内容团队,欢迎个人转发至朋友圈,谢绝媒体或机构未经授权以任何形式转载至其他平台。转载授权请在「医药观澜」微信公众号留言联系我们。其他合作需求,请联系wuxi_media@wuxiapptec.com。免责声明:药明康德内容团队专注介绍全球生物医药健康研究进展。本文仅作信息交流之目的,文中观点不代表药明康德立场,亦不代表药明康德支持或反对文中观点。本文也不是治疗方案推荐。如需获得治疗方案指导,请前往正规医院就诊。
细胞疗法创新药放射疗法First in Class蛋白降解靶向嵌合体抗体免疫疗法合作
100 项与 Yichang Humanwell Pharmaceutical Co. Ltd. 相关的药物交易
登录后查看更多信息
100 项与 Yichang Humanwell Pharmaceutical Co. Ltd. 相关的转化医学
登录后查看更多信息
管线布局
2024年05月04日管线快照
无数据报导
登录后保持更新
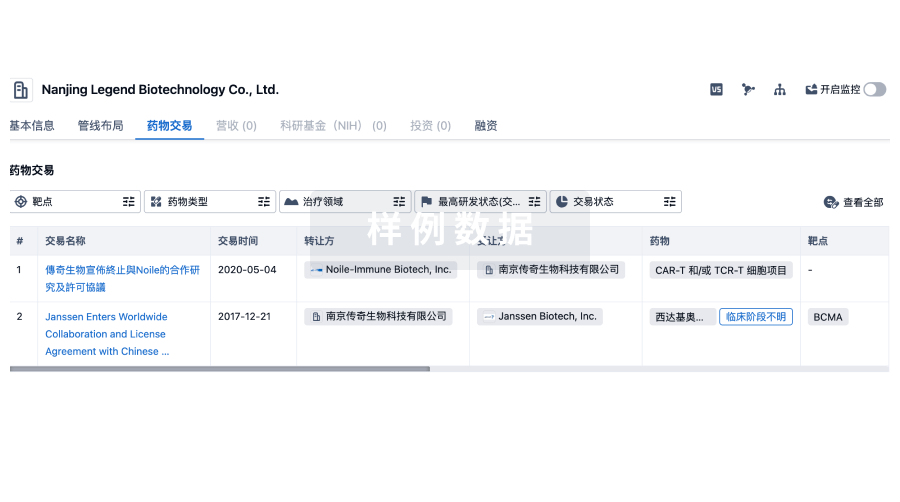
药物交易
使用我们的药物交易数据加速您的研究。
登录
或
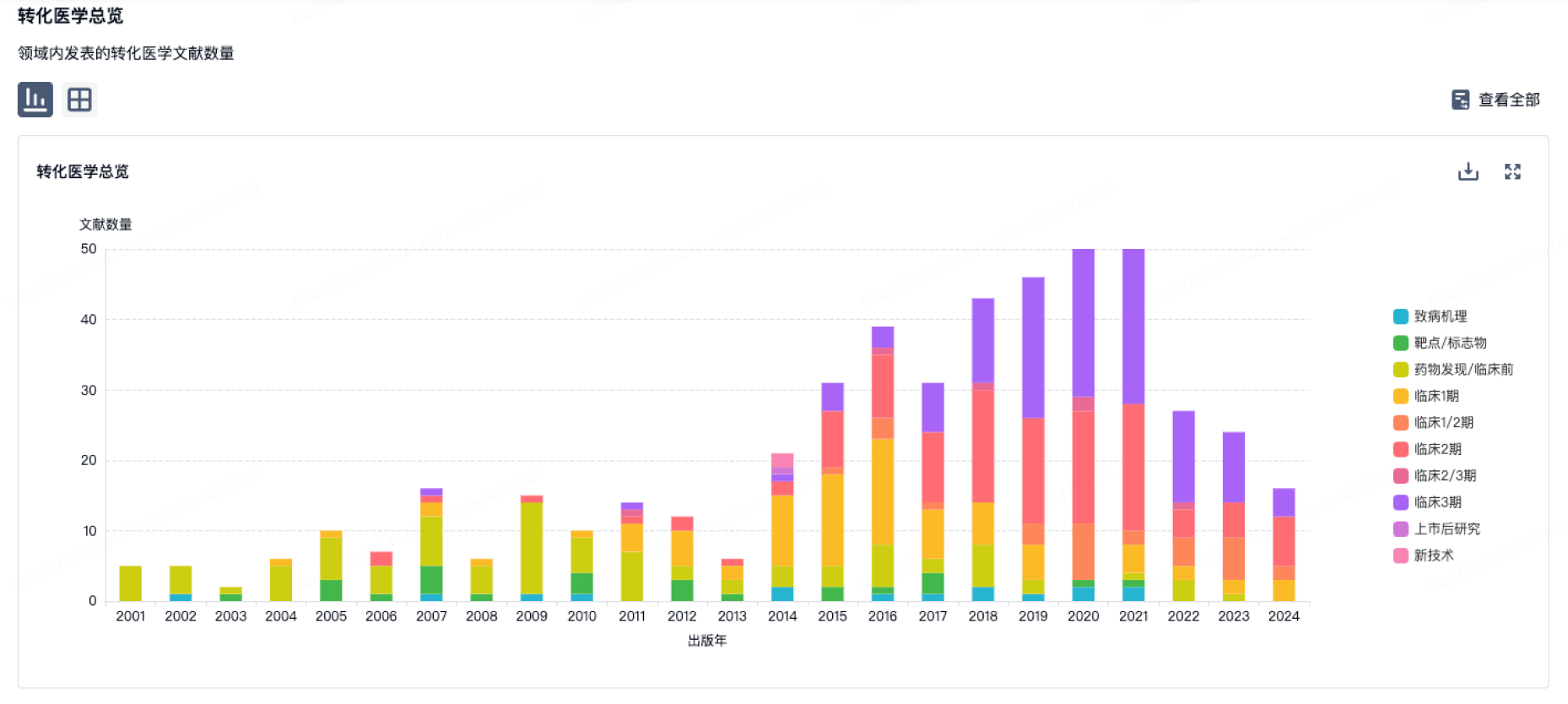
转化医学
使用我们的转化医学数据加速您的研究。
登录
或
营收
使用 Synapse 探索超过 36 万个组织的财务状况。
登录
或
科研基金(NIH)
访问超过 200 万项资助和基金信息,以提升您的研究之旅。
登录
或
投资
深入了解从初创企业到成熟企业的最新公司投资动态。
登录
或
融资
发掘融资趋势以验证和推进您的投资机会。
登录
或
标准版
¥16800
元/账号/年
新药情报库 | 省钱又好用!
立即使用
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用