预约演示
更新于:2025-08-26

Tolerion, Inc.
更新于:2025-08-26
概览
标签
免疫系统疾病
内分泌与代谢疾病
DNA疫苗
预防性疫苗
关联
1
项与 Tolerion, Inc. 相关的药物靶点 |
作用机制 INS基因调节剂 |
在研机构 |
在研适应症 |
非在研适应症 |
最高研发阶段临床2期 |
首次获批国家/地区- |
首次获批日期- |
6
项与 Tolerion, Inc. 相关的临床试验NCT03794960
A Phase 2b Multi-Center, Randomized, Double-Blind, Placebo-Controlled Trial to Evaluate the Safety and Efficacy of TOL-3021 in Patients With Established Type 1 Diabetes Mellitus
The study is a prospective, randomized, double-blind, placebo-controlled, multicenter trial in subjects with established T1D.
开始日期2019-12-14 |
申办/合作机构 |
NCT03794973
A Phase 2b Multi-Center, Randomized, Double-Blind, Placebo-Controlled Trial to Evaluate the Safety and Efficacy of TOL-3021 in Patients With New Onset Type 1 Diabetes Mellitus
The study is a prospective, randomized, double-blind, placebo-controlled, multi-center trial in subjects with new onset T1D.
开始日期2019-12-14 |
申办/合作机构 |
NCT03895437
A Phase 2 Multi-Center, Randomized, Double-Blind, Placebo-Controlled Trial to Evaluate the Safety and Efficacy of TOL-3021 in Patients With New Onset or Established Type 1 Diabetes Mellitus
The study is a prospective, randomized, 52-week double-blind, placebo-controlled, multicenter trial in subjects with T1D followed by a 2-year safety follow-up.
开始日期2019-06-17 |
申办/合作机构 |
100 项与 Tolerion, Inc. 相关的临床结果
登录后查看更多信息
0 项与 Tolerion, Inc. 相关的专利(医药)
登录后查看更多信息
4
项与 Tolerion, Inc. 相关的文献(医药)2010-11-01·IDrugs : the investigational drugs journal
Gene Based Vaccines: Optimising Development and Delivery - A marcus evans Conference.
作者: Garren, Hideki
The Gene Based Vaccines: Optimising Development and Delivery conference, held in Vienna, included topics covering new therapeutic developments in the field of gene-based vaccines. This conference report highlights selected presentations on gene-based vaccine delivery systems, anti-vector immunity in such vaccines, gene-based influenza vaccines, prime-boost strategies for influenza vaccines, DNA vaccines for the prevention of malaria, considerations in DNA vaccine manufacturing, and the ImmunoBody DNA vaccine technology from Scancell.
2009-09-01·Expert review of vaccines2区 · 医学
DNA vaccines for autoimmune diseases
2区 · 医学
Review
作者: Garren, Hideki
Autoimmune diseases represent a group of disorders in which there exists a large unmet medical need for effective treatments, but also where there exists a tremendous responsibility among physicians and drug developers to maintain adequate and acceptable patient safety. Several drugs have been approved and many others are about to be approved for the treatment of autoimmune diseases, but in pushing the envelope of therapeutic efficacy, concerns have been raised about the long-term safety of these new therapies. DNA vaccines provide a method of treating autoimmune diseases in a highly specific manner, and could therefore overcome these safety concerns while still maintaining comparable efficacy. The numerous reports of DNA vaccines in animal models of autoimmune diseases and results from three recent human trials of DNA vaccines in autoimmune diseases are reviewed here.
2005-11-04·Science (New York, N.Y.)1区 · 综合性期刊
Treatment of Autoimmune Neuroinflammation with a Synthetic Tryptophan Metabolite
1区 · 综合性期刊
Article
作者: Platten, Michael ; Gupta, Rohit ; Selley, Michael L. ; Fontoura, Paulo ; Ho, Peggy P. ; Kidd, Brian A. ; Sobel, Raymond A. ; Lee, Lowen Y. ; Hur, Eun Mi ; Garren, Hideki ; Robinson, William H. ; Youssef, Sawsan ; Steinman, Lawrence
Local catabolism of the amino acid tryptophan (Trp) by indoleamine 2,3-dioxygenase (IDO) is considered an important mechanism of regulating T cell immunity. We show that IDO transcription was increased when myelin-specific T cells were stimulated with tolerogenic altered self-peptides. Catabolites of Trp suppressed proliferation of myelin-specific T cells and inhibited production of proinflammatory T helper–1 (T
H
1) cytokines.
N
-(3,4,-Dimethoxycinnamoyl) anthranilic acid (3,4-DAA), an orally active synthetic derivative of the Trp metabolite anthranilic acid, reversed paralysis in mice with experimental autoimmune encephalomyelitis, a model of multiple sclerosis (MS). Trp catabolites and their derivatives offer a new strategy for treating T
H
1-mediated autoimmune diseases such as MS.
8
项与 Tolerion, Inc. 相关的新闻(医药)2025-06-18
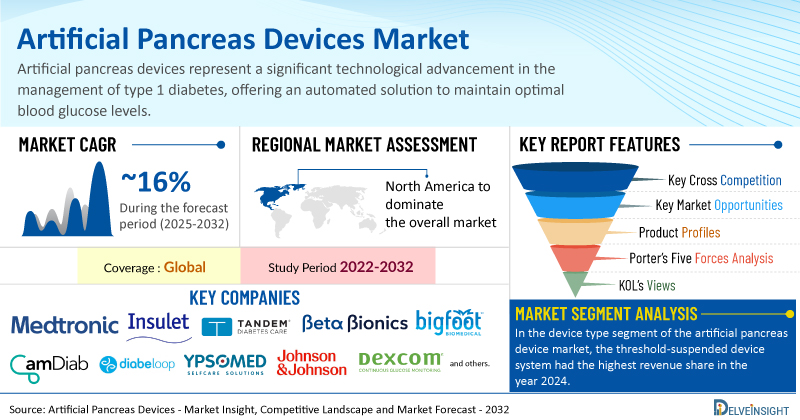
The global surge in diabetes cases is a key factor propelling the artificial pancreas device market, as a larger patient base fuels the need for effective diabetes management tools. Growing awareness and widespread screening initiatives have also contributed to earlier diagnoses and improved disease control, reinforcing the demand for advanced technologies such as artificial pancreas systems. Furthermore, ongoing product innovation and development by leading companies have led to more efficient and accessible devices, boosting market growth.
New York, USA, June 18, 2025 (GLOBE NEWSWIRE) --
Global Artificial Pancreas Devices Market is Gearing Up for Outstanding Expansion at a CAGR of ~16% by 2032 | DelveInsight
The global surge in diabetes cases is a key factor propelling the artificial pancreas device market, as a larger patient base fuels the need for effective diabetes management tools. Growing awareness and widespread screening initiatives have also contributed to earlier diagnoses and improved disease control, reinforcing the demand for advanced technologies such as artificial pancreas systems. Furthermore, ongoing product innovation and development by leading companies have led to more efficient and accessible devices, boosting market growth.
DelveInsight’s
Artificial Pancreas Devices Market Insights
report provides the current and forecast market analysis, individual leading artificial pancreas devices companies’ market shares, challenges, artificial pancreas devices market drivers, barriers, trends, and key market artificial pancreas devices companies in the market.
Key Takeaways from the Artificial Pancreas Devices Market Report
As per DelveInsight estimates, North America is anticipated to dominate the global artificial pancreas devices market during the forecast period.
In the device type segment of the artificial pancreas device market, the threshold-suspended device system had the highest revenue share in the year 2024.
Notable artificial pancreas devices companies such as
Medtronic PLC, Insulet Corporation, Tandem Diabetes Care, Inc., Beta Bionics, Inc., BIGFOOT BIOMEDICAL, INC., CamDiab Ltd., Diabeloop SA, Ypsomed, INREDA® DIABETIC B.V., Nikkiso Co., Ltd., Pancreum, Inc., Johnson & Johnson Services, Inc., Dexcom, Inc., TypeZero Technologies, Animas Corporation,
and several others are currently operating in the artificial pancreas device market.
In
September 2024, Tandem Diabetes Care, Inc.
, announced that its t:slim X2 insulin pump with Control-IQ automated insulin delivery (AID) technology was cleared for use with Eli Lilly and Company’s Lyumjev® (insulin lispro-aabc injection) ultra-rapid acting insulin in the European Union (EU).
In
June 2024, MCRA
, a leading privately held independent medical device, diagnostics, and biologics Clinical Research Organization (CRO) and advisory firm, announced its role in assisting CamDiab's artificial pancreas software, CamAPS FX, in achieving U.S. Food and Drug Administration (FDA) clearance.
To read more about the latest highlights related to the artificial pancreas devices market, get a snapshot of the key highlights entailed in the
Global Artificial Pancreas Devices Market Report
Artificial Pancreas Devices Overview
Artificial pancreas devices represent a significant technological advancement in the management of type 1 diabetes, offering an automated solution to maintain optimal blood glucose levels. These systems typically combine a continuous glucose monitor (CGM), an insulin pump, and a sophisticated control algorithm that calculates and delivers insulin doses in real time. By mimicking some functions of a healthy pancreas, these devices help reduce the burden of frequent glucose monitoring and insulin administration, improving glycemic control and reducing the risk of both hyperglycemia and hypoglycemia.
Recent innovations have led to the development of hybrid closed-loop systems, which require minimal patient intervention and can automatically adjust insulin delivery based on sensor readings. These devices have shown promising results in clinical trials and real-world settings, enhancing quality of life and long-term health outcomes for people with diabetes. Ongoing research is focused on improving algorithm accuracy, integrating dual hormone (insulin and glucagon) systems, and enhancing interoperability with smartphones and wearable technologies to create fully autonomous and user-friendly artificial pancreas solutions.
Artificial Pancreas Devices Market Insights
North America is projected to dominate the artificial pancreas device market in the coming years. This leadership can be attributed to several factors, including the growing prevalence of diabetes, increased government healthcare initiatives, the presence of major industry players engaged in strategic product innovations, and greater public awareness about diabetes management. Notably, rising awareness plays a significant role in driving demand for advanced diabetes care technologies such as artificial pancreas systems. For example, collaborative efforts by the National Institutes of Health (NIH) and the Centers for Disease Control and Prevention (CDC) through programs like the National Diabetes Education Program (NDEP) aim to educate communities about continuous glucose monitoring and automated insulin delivery systems.
Moreover, active product development and regulatory approvals continue to strengthen the market. A key milestone includes the FDA approval in May 2024 of the CamAPS FX artificial pancreas, developed by the University of Cambridge, for use in individuals with type 1 diabetes aged two years and above, including pregnant women.
Collectively, these factors are expected to significantly fuel the growth of the artificial pancreas device market in North America throughout the 2025–2032 forecast period.
To know more about why North America is leading the market growth in the artificial pancreas devices market, get a snapshot of the
Artificial Pancreas Devices Market Outlook
Artificial Pancreas Devices Market Dynamics
The artificial pancreas devices market is witnessing strong growth driven by
increasing global prevalence of type 1 diabetes
and
advancements in diabetes management technologies
. As patient demand for convenient and accurate diabetes management tools rises, healthcare providers and manufacturers are increasingly focusing on
developing integrated, closed-loop systems
that mimic the glucose-regulating function of a healthy pancreas.
Technological innovation is a core driver of the market, with
developments in real-time glucose monitoring, machine learning algorithms, and smartphone integration
enhancing the usability and precision of artificial pancreas systems. Companies are also leveraging cloud connectivity to enable remote patient monitoring and data sharing with healthcare providers. These advancements are improving both patient outcomes and physician decision-making while reducing the burden of disease management. The
adoption of hybrid closed-loop systems
, which allow partial automation, has paved the way for fully automated solutions, many of which are currently in advanced stages of clinical trials or have already gained regulatory approvals in key markets like the U.S. and Europe.
Regulatory support and favorable reimbursement policies
in several countries are also propelling market growth. Additionally, public and private payers are beginning to cover these systems, recognizing their long-term benefits in reducing diabetes-related complications and hospitalizations. However, in developing countries,
market penetration remains limited
due to high device costs and insufficient healthcare infrastructure.
Despite these promising trends, the market faces notable challenges.
High upfront costs of artificial pancreas systems
, coupled with the
need for regular maintenance and consumables
, can deter widespread adoption, especially in low- and middle-income regions. Technical limitations, such as
sensor calibration requirements, occasional device malfunction, and user training complexity
, can also hinder patient acceptance and adherence. Furthermore,
data security and privacy concerns
regarding cloud-connected health devices must be carefully managed to maintain trust among users and healthcare providers.
Looking ahead, the artificial pancreas market is poised for robust growth, supported by continuous innovation, strategic partnerships, and rising consumer awareness. Industry players are investing in R&D to enhance the accuracy, affordability, and user-friendliness of these systems. As the ecosystem evolves, integration with digital health platforms and expansion into emerging markets are expected to open new avenues for growth, ultimately moving closer to the vision of a fully automated, reliable, and accessible solution for diabetes management.
Get a sneak peek at the artificial pancreas devices market dynamics @
Artificial Pancreas Devices Market Trends
Report Metrics
Details
Coverage
Global
Study Period
2022–2032
Artificial Pancreas Devices Market CAGR
~16%
Artificial Pancreas Devices Market Size by 2032
USD 1.1 Billion
Key Artificial Pancreas Devices Companies
Medtronic PLC, Insulet Corporation, Tandem Diabetes Care, Inc., Beta Bionics, Inc., BIGFOOT BIOMEDICAL, INC., CamDiab Ltd., Diabeloop SA, Ypsomed, INREDA® DIABETIC B.V., Nikkiso Co., Ltd., Pancreum, Inc., Johnson & Johnson Services, Inc., Dexcom, Inc., TypeZero Technologies, Animas Corporation, among others
Artificial Pancreas Devices Market Assessment
Artificial Pancreas Devices Market Segmentation
Artificial Pancreas Devices Market Segmentation By Device Type:
Threshold Suspended Device System and Non-Threshold Suspended Device System
Artificial Pancreas Devices Market Segmentation By End User:
Hospitals & Clinics, Homecare Settings, Ambulatory Surgical Centers, and Others
Artificial Pancreas Devices Market Segmentation By Geography
: North America, Europe, Asia-Pacific, and Rest of World
Porter’s Five Forces Analysis, Product Profiles,
Case Studies, KOL’s Views, Analyst’s View
Which MedTech key players in the artificial pancreas devices market are set to emerge as the trendsetter explore @
Artificial Pancreas Devices Companies
Table of Contents
1
Artificial Pancreas Devices Market Report Introduction
2
Artificial Pancreas Devices Market Executive Summary
3
Competitive Landscape
4
Regulatory Analysis
5
Artificial Pancreas Devices Market Key Factors Analysis
6
Artificial Pancreas Devices Market Porter’s Five Forces Analysis
7
Artificial Pancreas Devices Market Layout
8
Artificial Pancreas Devices Market Company and Product Profiles
9
KOL Views
10
Project Approach
11
About DelveInsight
12
Disclaimer & Contact Us
Interested in knowing the artificial pancreas devices market by 2032? Click to get a snapshot of the
Artificial Pancreas Devices Market Analysis
Related Reports
Diabetes Market
Diabetes Market Insights, Epidemiology, and Market Forecast – 2034
report deliver an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key diabetes companies, including
TikoMed, Avotres, REMD Biotherapeutics, Novo Nordisk,
among others.
Diabetes Pipeline
Diabetes Pipeline Insight – 2025
report provides comprehensive insights about the pipeline landscape, pipeline drug profiles, including clinical and non-clinical stage products, and the key diabetes companies, including
Daewoong Pharmaceutical, Janssen Biotech, Zealand Pharma, BioRestorative Therapies, Elevian, Oramed Pharmaceuticals, ImCyse, Novo Nordisk, Enthera, Precigen, Inc., Japan Tobacco, Avotres, AstraZeneca, Landos Biopharma, Vertex Pharmaceuticals, REMD Biotherapeutics, Inc., Eledon Pharmaceuticals, Eli Lilly and Company, Novo Nordisk A/S, Diamyd Medical, NextCell Pharma, ViaCyte, Op-T LLC, Dompé Farmaceutici S.p.A, ILTOO Pharma, Throne Biotechnologies, Oramed Pharmaceuticals, Janssen Research & Development, LLC, Jaguar Gene Therapy, SQZ Biotechnologies, Genprex, Inc., CRISPR Therapeutics, Biora Therapeutics, Genprex, Inc.,
among others.
Type 1 Diabetes Market
Type 1 Diabetes Market Insights, Epidemiology, and Market Forecast – 2034
report deliver an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key type 1 diabetes companies, including
Landos Biopharma, Diamyd Medical, Gan&Lee Pharmaceuticals, Zealand Pharma, Kamada, AstraZeneca, Novo Nordisk, Provention Bio Preregistration, Histogen, Vertex Pharmaceuticals, Panbela Therapeutics, Arecor, Bioprojet, Novartis, ImCyse, Adocia, Anelixis Therapeutics, Tolerion, TikoMed, Avotres, REMD Biotherapeutics, Novo Nordisk,
among others.
Type 2 Diabetes Market
Type 2 Diabetes Market Insights, Epidemiology, and Market Forecast – 2034
report deliver an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key type 2 diabetes companies, including
Eli Lilly and Company, Regor Pharmaceuticals Inc., AstraZeneca, Eccogene, Pfizer, Sciwind Biosciences USA Co., Ltd., MediciNova, Sparrow Pharmaceuticals, HighTide Biopharma Pty Ltd, Novo Nordisk A/S, Biomea Fusion Inc.,
among others.
Type 2 Diabetes Pipeline
Type 2 Diabetes Pipeline Insight – 2024
report provides comprehensive insights about the pipeline landscape, pipeline drug profiles, including clinical and non-clinical stage products, and the key type 2 diabetes companies, including
Tonghua Dongbao Pharmaceutical, Eli Lilly and Company, Rivus Pharmaceuticals, Celon Pharma, Sciwind Biosciences, AstraZeneca, Suzhou Alphamab Co., Ltd., Neurodon, Abarceo Pharma, Chong Kun Dang Pharmaceutical,
among others.
DelveInsight’s
Pharma Competitive Intelligence Service
:
Through its CI solutions, DelveInsight provides its clients with real-time and actionable intelligence on their competitors and markets of interest to keep them stay ahead of the competition by providing insights into the latest therapeutic area-specific/indication-specific market trends, in emerging drugs, and competitive strategies. These services are tailored to the specific needs of each client and are delivered through a combination of reports, dashboards, and interactive presentations, enabling clients to make informed decisions, mitigate risks, and identify opportunities for growth and expansion.
Other Business Pharmaceutical Consulting Services
Healthcare Conference Coverage
Pipeline Assessment
Healthcare Licensing Services
Discover how a mid-pharma client gained a level of confidence in their soon-to-be partner for manufacturing their therapeutics by downloading our
Due Diligence Case Study
About DelveInsight
DelveInsight is a leading Business Consultant, and Market Research firm focused exclusively on life sciences. It supports pharma companies by providing comprehensive end-to-end solutions to improve their performance.
Contact Us
Shruti Thakur
info@delveinsight.com
+14699457679
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability
for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this
article. If you have any complaints or copyright issues related to this article, kindly contact the author above.
2024-01-16
FOSTER CITY, Calif.--(
BUSINESS WIRE
)--Mirum Pharmaceuticals, Inc. (Nasdaq: MIRM), today announced the appointment of Joanne Quan, MD as Chief Medical Officer. Dr. Quan will lead the global clinical development, clinical operations, and safety programs for Mirum’s pipeline of programs addressing rare diseases.
Dr. Quan was most recently Chief Medical Officer at Nuvig Therapeutics. Prior to Nuvig, she served in the same role at Modis Therapeutics, a company focused on rare genetic diseases (acquired by Zogenix, Inc. and subsequently UCB). Before Modis, she held clinical development leadership positions at Eiger Biopharmaceuticals, InterMune, Arena Pharmaceuticals, Bayhill Therapeutics, ALZA Corporation (Johnson and Johnson), Genentech, and PathoGenesis. Dr. Quan received a BA in molecular biology at the University of California, Berkeley, and an MD at Stanford University School of Medicine. She completed a residency in internal medicine at Massachusetts General Hospital and a fellowship in pulmonary and critical care medicine at the University of Washington.
In tandem with Dr. Quan’s appointment, Mirum announced the promotion of Peter Radovich to President and Chief Operating Officer. Mr. Radovich oversees commercial, corporate development and supply chain functions for Mirum.
“Joanne brings nearly three decades of experience in the development of medications for rare conditions. I’m thrilled that she is joining at such an exciting time for Mirum, strengthening our team and leadership in rare disease,” said Chris Peetz, Chief Executive Officer at Mirum. “I am also excited about the promotion of Peter Radovich, whose contributions have helped place Mirum in a position of strength, with an expanded portfolio of three growing commercial medicines. I am proud of the expertise we have assembled at Mirum to bring life-changing medicines to patients around the world.”
“Mirum has achieved wonderful success both clinically and commercially, establishing itself as the leader in cholestatic liver disease with a deep and focused commitment to patients,” said Joanne Quan, MD. “I am excited to join at such an important time as the company grows its pipeline, continues to extend its reach to patients globally, and builds upon its success and commitment as a rare disease company.”
About Mirum Pharmaceuticals, Inc.
Mirum Pharmaceuticals, Inc. is a biopharmaceutical company dedicated to transforming the treatment of rare diseases affecting children and adults. Mirum has three approved medications:
LIVMARLI® (maralixibat) oral solution
,
Cholbam® (cholic acid) capsules,
and
Chenodal® (chenodiol) tablets.
LIVMARLI, an IBAT inhibitor, is approved for the treatment of cholestatic pruritus in patients with Alagille syndrome in the U.S. (three months and older), in Europe (two months and older), and in Canada. Mirum has also submitted LIVMARLI for approval in the U.S. in cholestatic pruritus in PFIC patients three months of age and older and in Europe in PFIC for patients two months of age and older. Cholbam is FDA-approved for the treatment of bile acid synthesis disorders due to single enzyme defects and adjunctive treatment of peroxisomal disorders in patients who show signs or symptoms or liver disease. Chenodal has received medical necessity recognition by the FDA to treat patients with cerebrotendinous xanthomatosis (CTX).
Mirum’s late-stage pipeline includes two investigational treatments for debilitating liver diseases. Volixibat, an IBAT inhibitor, is being evaluated in two potentially registrational studies including the Phase 2b
VISTAS
study for primary sclerosing cholangitis and Phase 2b
VANTAGE
study for primary biliary cholangitis. Lastly, Chenodal, has been evaluated in a Phase 3 clinical study, RESTORE, to treat patients with CTX.
To learn more about Mirum, visit mirumpharma.com and follow Mirum on
Facebook
,
LinkedIn
,
Instagram
and
Twitter
.
临床2期上市批准高管变更
2024-01-04
Poised for multiple meaningful catalysts in 2024 driven by strong clinical progress across large market programs in wet age-related macular degeneration (wet AMD), diabetic macular edema (DME) and cystic fibrosis (CF)Established next-generation AAV & CRISPR/Cas-based genetic medicines partnership for central nervous system (CNS) diseases with Arbor BiotechnologiesAnnounced organizational updates, including the appointment of UCSF Professor Noriyuki Kasahara (Nori), M.D., Ph.D. as Chief Scientific OfficerWell capitalized ending 2023 with $300 million in estimated cash, financial runway guidance unchanged and cash expected to be sufficient to fund operations into H1 2026
EMERYVILLE, Calif., Jan. 04, 2024 (GLOBE NEWSWIRE) -- 4D Molecular Therapeutics (Nasdaq: FDMT, 4DMT or the Company), a leading clinical-stage genetic medicines company focused on unlocking the full potential of genetic medicines to treat large market diseases, today announced clinical pipeline progress, updates to near-term milestones and organizational updates.
“2023 was a landmark year for 4DMT, highlighted by rapid enrollment for 4D-150 in wet AMD and DME, strong CFTR protein expression data for 4D-710 in lungs of people with CF, multiple value-generating business development partnerships, and key additions to the Executive Team,” said David Kirn, M.D., Co-founder and Chief Executive Officer of 4DMT. “Looking forward to 2024, we plan to release multiple important datasets from our lead programs in wet AMD, DME and CF. We also plan to share guidance on the design and timing of Phase 3 clinical trials. In addition, we expect to initiate clinical development of 4D-175 in geographic atrophy by the second half of the year. By the end of 2024, we intend to progress clinical development in four large market diseases, positioning us well in our strategy to become a fully integrated large market genetic medicines company.”
Large Market Ophthalmology Portfolio Recent Updates and Expected Upcoming Milestones
4D-150 for Wet AMD: Phase 2 PRISM Clinical Trial Randomized Dose Expansion arm (N=50) in advanced high treatment need patients: Initial interim 24 week landmark data to be presented at the Angiogenesis, Exudation, and Degeneration 2024 Conference on Saturday, February 3, 2024 followed by a corporate webcast with details to be announced at a future date Population Extension arm (N≤45) in broad population (non-advanced, standard treatment-need): Enrollment update expected in Q1 2024Initial interim 24 week landmark data analysis expected in H2 2024
Received both RMAT and PRIME in Q4 2023, enabling increased collaboration between the FDA and EMA on regulatory approval planning, in addition to the opportunity for expedited product developmentUpdate on Phase 3 trial plans expected in February 2024 along with the interim randomized Phase 2 PRISM trial data 4D-150 for DME: Phase 2 SPECTRA Clinical Trial: Part 1 – Dose Confirmation Stage Completed enrollment in Q4 2023Initial interim 24 week landmark data expected in H2 2024
4D-175 for Geographic Atrophy: IND filing expected in Q2 2024Phase 1 initiation expected in H2 2024
Pulmonology Portfolio Expected Upcoming Milestones
4D-710 for CF Lung Disease: Update on FDA feedback on development plan for monotherapy and approved CF modulator combination regimens expected in Q1 2024Interim data update from Phase 1/2 AEROW clinical trial expected in mid-2024 4D-725 for A1AT Deficiency Lung Disease: Program update and initiation of IND-enabling studies expected in 2024
Cardiology Franchise Expected Upcoming Milestones
4D-310 for Fabry Disease Cardiomyopathy: Interim data update including cardiac functional, imaging, quality-of-life and cardiac biopsy data for all 6 patients dosed to be presented in the late-breaking session at the WORLDSymposium™ 2024 in San Diego, California on Friday, February 9 (10:15 a.m. to 11:15 a.m. PT) titled “Phase 1/2 clinical trial evaluating 4D-310 in adults with Fabry disease cardiomyopathy: Interim analysis of cardiac and safety outcomes in patients with 12-33 months of follow-up”FDA submission of data from the non-human primate (NHP) study evaluating the safety and biodistribution of intravenous (IV) 4D-310 with the rituximab/sirolimus (R/S) immunosuppressive regimen compared to the prior prednisone regimen expected in Q2 2024
Rare Inherited Retinal Disorders Portfolio Expected Upcoming Milestones
4D-110 for Choroideremia and 4D-125 for X-Linked Retinitis Pigmentosa: The safety and tolerability profiles for both product candidates remain unchanged from prior data releases. All enrolled patients are expected to reach 24 months of follow-up in 2024, and we plan to assess the magnitude and durability of key imaging and functional endpoint changes in evaluable patients. We expect to provide program updates in 2024.
Expanded CNS and Gene Editing Capabilities Through Partnership with Arbor Biotechnologies
Partnership with a leader in next-generation genetic medicines to engineer, co-develop and co-commercialize (50:50 profit share) AAV-delivered CRISPR/Cas-based therapeutics for up to six product candidates in CNSInitial product candidate, led by Arbor, will be developed for a target in amyotrophic lateral sclerosis (ALS) with additional targets expected to be in diseases with high unmet need in both rare and large patient populationsMore information can be found in the press release issued on January 3, 2024
Corporate Governance Update: New Chief Scientific Officer (CSO) and Scientific Advisory Board (SAB)
Appointment of Noriyuki (Nori) Kasahara, M.D., Ph.D. as CSO: Noriyuki Kasahara (Nori), M.D., Ph.D., was appointed as Chief Scientific Officer of 4DMT, effective January 2, 2024, and previously served as a member of 4DMT’s Board of Directors and chaired the Board’s Science & Technology Committee from September 2022 to December 2023. Dr. Kasahara was most recently Professor & Alvera L. Kan Endowed Chair of Neurological Surgery and Radiation Oncology at the University of California, San Francisco Medical School (UCSF). He brings more than 30 years of experience and has authored more than 150 peer-reviewed articles in the fields of gene therapy and genetic engineering. As an academic researcher, he previously established the first Vector Core & Shared Resource facilities for gene delivery vector research at both the University of Southern California and the University of California, Los Angeles. Most recently, in addition to his professorship and Endowed Chair, he served as a Principal Investigator in the UCSF Brain Tumor Center. Dr. Kasahara’s research has focused on optimizing the application of multiple and diverse viral vector delivery systems to achieve maximal therapeutic potential, as well as developing next-generation gene transfer technologies with greater therapeutic efficacy in vivo.In his new role as CSO, Nori will lead the creation and validation of new genetic medicine delivery and transgene payload platforms. Our Chief Development Officer An Song, Ph.D., will continue her role in leading nonclinical and clinical scientific development functions, including AAV product design and engineering, in vitro and in vivo pharmacology, toxicology, immunology, biomarkers and bioanalytical development, and further advancing the AAV-based Therapeutic Vector Evolution platform. Newly Formed SAB Combines the Expertise of Renowned Key Opinion Leaders in AAV Gene Therapy, Immunology and Core 4DMT Therapeutic Areas Ronald Crystal, M.D. is Professor and Chairman of the Department of Genetic Medicine at the Weill Medical College of Cornell University, where he is also the Bruce Webster Professor of Internal Medicine, Director of the Belfer Gene Therapy Core Facility and Attending Physician at the New York-Presbyterian Hospital/Weill Cornell Medical Center. Dr. Crystal is a pioneer in the field of gene therapy, where he was the first to use a recombinant virus as a vehicle for in vivo gene therapy and has carried out human trials of gene therapy for CF, cardiac ischemia, cancer and central nervous system disorders.Amit Gaggar, M.D., Ph.D. is the William C. Bailey Endowed Chair of Pulmonary Medicine and Professor of Medicine at University of Alabama at Birmingham (UAB), where he serves as Scientific Director of the UAB Lung Health Center. Dr. Gaggar is a physician-scientist with a long standing interest in protease biology and innate immune signaling in chronic lung disease such as chronic obstructive pulmonary disease (COPD) and CF.Douglas Hanahan, Ph.D. is Director Emeritus of the Swiss Institute for Experimental Cancer Research at École Polytechnique Fédérale de Lausanne (EFPL) and is a Distinguished Scholar at the Ludwig Institute for Cancer Research. Dr. Hanahan is a pioneer in angiogenesis, cancer research, and is a recipient of the AACR Lifetime Achievement Award in Cancer Research.Richard Moss, M.D. is Professor Emeritus at Stanford University School of Medicine in the Department of Pediatrics’ Center for Excellence in Pulmonary Biology. He is former Chief of the pediatric pulmonary and allergy-immunology divisions, and former allergy-immunology and pulmonary fellowship training programs Director at Stanford Children’s Health/Lucile Packard Children’s Hospital Stanford. He was Director of the Stanford Cystic Fibrosis Center from 1991 to 2009 and Principal Investigator for the CF Foundation’s Therapeutics Development Network, where he also served as the inaugural Chair of the TDN Protocol Review Committee. He has served on CFF’s Clinical Research Committee, Translational Advisory Group and Clinical Research Advisory Board.Amy Rosenberg, M.D., a physician-immunologist, trained in basic immunology in Al Singer’s lab in the Experimental Immunology Branch of the NCI, NIH, following medical residency and specialty training in Internal Medicine and Infectious Diseases. She then established a laboratory and performed regulatory duties at the FDA and there, for 33 years (14 as Director of the Division of Therapeutic Proteins then Division III in the Office of Biotechnology Products), regulated an array of biotechnology products including cellular and protein therapeutics and cell-device combination products. She is currently Senior Director of Immunology and Protein Therapeutics at EpiVax, Inc.Ramesh Shivdasani, M.D., Ph.D. is Professor of Medicine / Medical Oncology at Harvard Medical School, and serves as Deputy Director of the Dana-Farber Cancer Institute. He is a gastrointestinal disease expert, medical oncologist and laboratory investigator in the Gastrointestinal Cancer Center at Dana-Farber and Brigham and Women's Hospital, and is an internationally recognized expert in cellular and tissue development and oncology.Daniel Takefman, Ph.D. is Principal of Takefman Gene Therapy Advisors since March 2020. Dr. Takefman provides expert regulatory advice for the development and commercialization of cell and gene therapies, and assists in due diligence assessments of gene therapy assets for venture capital firms and is a member of multiple scientific advisory boards. Previously Dr. Takefman was SVP and Head of Regulatory Affairs at Spark Therapeutics for 5 years. At Spark, Dr. Takefman supervised the submission through to approval of the FDA and EMA Luxturna® marketing applications. Dr. Takefman also supervised the regulatory process for multiple AAV based investigation products including two additional Breakthrough Designation products: SPK-9001 for the treatment of Hemophilia B (marketing applications currently under review by FDA and EMA) and SPK-8011 for the treatment of Hemophilia A. Dr. Takefman held multiple positions during his 15-year career at FDA, ultimately becoming Chief of the Gene Therapy Branch.Paul J. (PJ) Utz, M.D. is Professor of Medicine at Stanford School of Medicine in the Department of Medicine, Division of Immunology, where he has directed a translational research lab for 24 years. He co-founded several companies including Bayhill Therapeutics, Tolerion and Yolo Immune, and has decades of experience as a consultant and SAB member for many biotechnology and pharmaceutical companies. Dr. Utz studies autoimmune diseases, including systemic lupus erythematosus, rheumatoid arthritis, scleroderma, myositis, Sjögren's disease, type I diabetes, vasculitis and multiple sclerosis.More details on our members can be found in the SAB section of our website
About 4DMT
4DMT is a leading clinical-stage genetic medicines company focused on unlocking the full potential of genetic medicines to treat large market diseases in ophthalmology and pulmonology. 4DMT’s proprietary invention platform, Therapeutic Vector Evolution, combines the power of the Nobel Prize-winning technology, directed evolution, with approximately one billion synthetic AAV capsid-derived sequences to invent customized and evolved vectors for use in our wholly owned and partnered product candidates. Our product design, development, and manufacturing engine helps us efficiently create and advance our diverse product pipeline with the goal of revolutionizing medicine with potential curative therapies for millions of patients. Currently, 4DMT is advancing five clinical-stage and two preclinical product candidates, each tailored to address rare and large market diseases in ophthalmology, pulmonology, and cardiology. In addition, 4DMT is also advancing programs in CNS through a gene editing partnership. 4D Molecular Therapeutics™, 4DMT™, Therapeutic Vector Evolution™, and the 4DMT logo are trademarks of 4DMT.
All of our product candidates are in clinical or preclinical development and have not yet been approved for marketing by the FDA or any other regulatory authority. No representation is made as to the safety or effectiveness of our product candidates for the therapeutic uses for which they are being studied.
Learn more at www.4DMT.com and follow us on LinkedIn.
Forward Looking Statements:
This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, as amended, including, without limitation, implied and express statements regarding the therapeutic potential, and clinical benefits of 4DMT’s product candidates, as well as the plans, announcements and related timing for the clinical development of our clinical and preclinical product candidates, and statements regarding our estimated cash or anticipated cash runway. The words "may," “might,” "will," "could," "would," "should," "expect," "plan," "anticipate," "intend," "believe," “expect,” "estimate," “seek,” "predict," “future,” "project," "potential," "continue," "target" and similar words or expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. Any forward looking statements in this press release are based on management's current expectations and beliefs and are subject to a number of risks, uncertainties and important factors that may cause actual events or results to differ materially from those expressed or implied by any forward-looking statements contained in this press release, including risks and uncertainties that are described in greater detail in the section entitled "Risk Factors" in 4D Molecular Therapeutics’ most recent Quarterly Report on Form 10-Q as well as any subsequent filings with the Securities and Exchange Commission. In addition, any forward-looking statements represent 4D Molecular Therapeutics' views only as of today and should not be relied upon as representing its views as of any subsequent date. 4D Molecular Therapeutics explicitly disclaims any obligation to update any forward-looking statements. No representations or warranties (expressed or implied) are made about the accuracy of any such forward looking statements.
Contacts:
Media:
Katherine SmithEvoke CanaleKatherine.Smith@evokegroup.com
Investors:
Julian PeiHead of Investor Relations and Corporate CommunicationsInvestor.Relations@4DMT.com267-644-5097
高管变更临床2期临床3期
100 项与 Tolerion, Inc. 相关的药物交易
登录后查看更多信息
100 项与 Tolerion, Inc. 相关的转化医学
登录后查看更多信息
组织架构
使用我们的机构树数据加速您的研究。
登录
或

管线布局
2026年06月10日管线快照
管线布局中药物为当前组织机构及其子机构作为药物机构进行统计,早期临床1期并入临床1期,临床1/2期并入临床2期,临床2/3期并入临床3期
临床2期
1
2
其他
登录后查看更多信息
当前项目
| 药物(靶点) | 适应症 | 全球最高研发状态 |
|---|---|---|
DNA plasmid vaccine for type 1 diabetes(Stanford University) ( insulin ) | 1型糖尿病 更多 | 临床2期 |
BHT-3009 ( MBP ) | 复发-缓解型多发性硬化 更多 | 无进展 |
rBHT-3034 ( nAChRα1/β1/δ/γ ) | 重症肌无力 更多 | 无进展 |
登录后查看更多信息
药物交易
使用我们的药物交易数据加速您的研究。
登录
或

转化医学
使用我们的转化医学数据加速您的研究。
登录
或

营收
使用 Synapse 探索超过 36 万个组织的财务状况。
登录
或

科研基金(NIH)
访问超过 200 万项资助和基金信息,以提升您的研究之旅。
登录
或

投资
深入了解从初创企业到成熟企业的最新公司投资动态。
登录
或

融资
发掘融资趋势以验证和推进您的投资机会。
登录
或

生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用
