|
|
在研机构- |
|
在研适应症- |
|
最高研发阶段无进展 |
首次获批国家/地区- |
首次获批日期- |
Chinese Spondyloarthritis Inception Cohort
The Chinese Spondyloarthritis Inception cohort (CESPIC) was started 2000 as a prospective, longitudinal, multicentre, nationwide study in China on patients with early SpA including ankylosing spondylitis (AS, also known as radiographic axial spondyloarthritis) and non-radiographic axial SpA. The objectives of CESPIC are to learn about the course of SpA during the very early stage of the disease, to appropriately assess the outcome including radiographic progression of patients after several years of follow-up, to identify outcome predictors, to assess quality of life, function, and costs (direct and indirect costs). CESPIC has been recently expanded to recruit patients with other forms of SpA / conditions associated with SpA: reactive arthritis, acute anterior uveitis, Crohn's disease as well as with psoriasis / axial psoriatic arthritis.
Neoadjuvant Chemotherapy Plus Camrelizumab in PD-L1-negative Locally Advanced Cervical Cancer: a Multicentre, Single-arm, Phase 2 Trial
This is a multicenter, prospective, single-arm, phase 2 clinical trial designed to evaluate the therapeutic efficacy of the NACI (neoadjuvant chemotherapy plus Camrelizumab) for PD-L1-negative locally advanced cervical cancer.
Neoadjuvant Chemotherapy Plus Cadunilizumab Followed by Extrafascial Hysterectomy for FIGO Stage IB2 Cervical Cancer: a Multicenter, Single-arm, Phase 2 Trial.
This study is an exploratory clinical trial to investigate the feasibility of neoadjuvant chemoimmunotherapy plus extrafascial hysterectomy and pelvic lymph node dissection in patients with stage IB2 (2018 FIGO) cervical cancer and to observe the response rate to treatment, adverse effects and complications, and to assess the survival rate of patients.
100 项与 Southwest Hospital Chongqing 相关的临床结果
0 项与 Southwest Hospital Chongqing 相关的专利(医药)
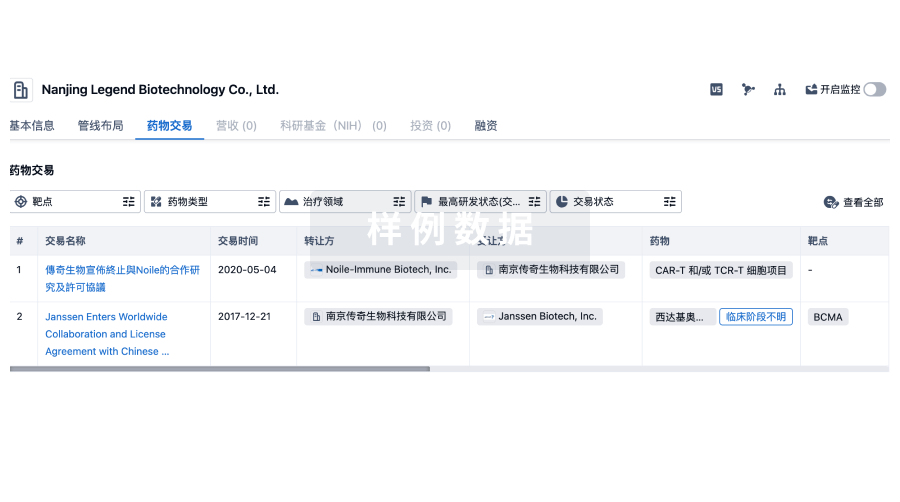
100 项与 Southwest Hospital Chongqing 相关的药物交易
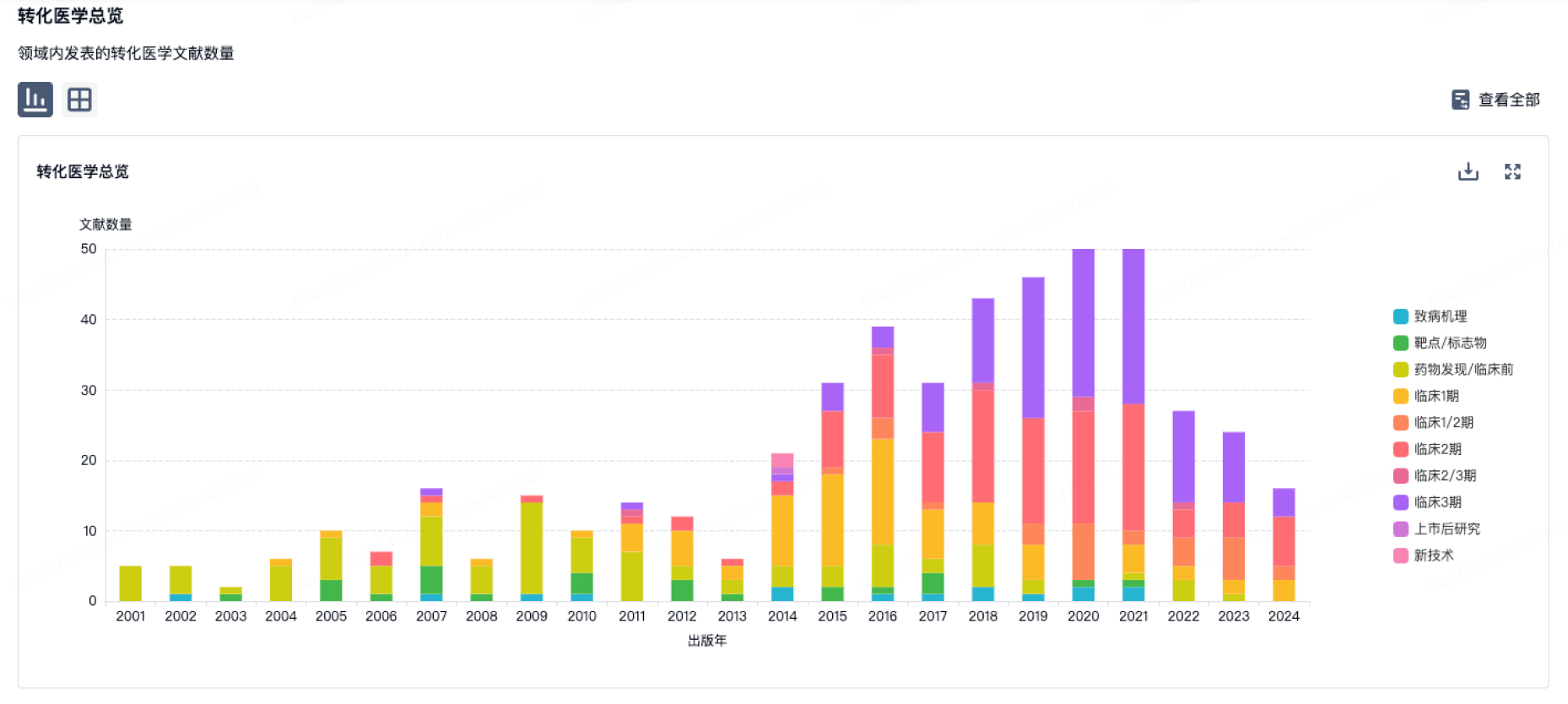
100 项与 Southwest Hospital Chongqing 相关的转化医学