更新于:2023-04-25
更新于:2023-04-25
概览
关联
7
项与 相关的药物靶点- |
作用机制- |
原研机构- |
在研适应症 |
非在研适应症- |
最高研发阶段 |
首次获批国家/地区 中国 |
首次获批日期2020-06-01 |
靶点- |
作用机制- |
原研机构- |
在研适应症 |
非在研适应症- |
最高研发阶段 |
首次获批国家/地区 中国 |
首次获批日期2020-05-31 |
靶点- |
作用机制- |
原研机构- |
在研适应症 |
非在研适应症- |
最高研发阶段 |
首次获批国家/地区 中国 |
首次获批日期2020-04-17 |
17
项与 相关的临床试验100 项与 相关的临床结果
登录后查看更多信息
0 项与 相关的专利(医药)
登录后查看更多信息
18
项与 相关的文献(医药)1996-08-31·Hecheng Xianwei Gongye
Manufacture of the fully crystallized fiber by one-step with heating tube
作者: Yao, Jun ; Gao, Yaguang ; Zhou, Yafang ; Yu, Yueli
The technol. innovation for ordinary high-speed spinning machine was carried out by using the heating tube. Fully crystallized filament of polyester was manufactured through the one-step method. The filament could be directly used in textile industry. By adjusting the temperature and the position of the heating tube, various filaments were obtained.
2006-08-31·Zhongguo Yaoke Daxue Xuebao
Preparation and evaluation in vivo and in vitro of melatonin controlled-release bilayer tablets
作者: Zhang, Tao ; Chen, Lan ; Zhou, Yuanda ; Yang, Yong ; Deng, Luhong
The melatonin controlled-release bilayer tablets were prepared and its pharmaceutical characteristics were studied. HPMC and stearic acid were used to prepare melatonin controlled- release bilayer tablets by solid dispersion method and double compression technol. In a randomized, crossover design, a single oral dose of melatonin tablets (3 mg) or melatonin controlled-release bilayer tablets (6 mg) was given to 6 Beagle dogs, and plasma concentration of melatonin was detected by HPLC. Pharmacokinetic parameters were estimated by 3P97 pharmacokinetic program. The peak levels (cmax) after administration of melatonin controlled-release bilayed tablets (6 mg) and melatonin tablets (3 mg) averaged (8.31 ± 5.11) ng/L and (11.27 ± 3.77) ng/mL at (1.00 ± 0.37) h and (0.50 ± 0.18) h. The mean elimination half lives (t1/2Ke) were (3.27 ± 0.89) h and (1.21 ± 0.52) h. AUC0-t were (38.03 ± 16.45) ng/mL h and (25.23 ± 7.71) ng/mL h, resp. The formulated melatonin controlled-release bilayer tablets exhibited both controlled-and rapid-release characteristics in vitro. The bioavailability of melatonin controlled-release bilayer tablets was lower than that of conventional release tablets. There existed no significant difference in tmax, but calculated MRT of controlled-release bilayer tablets was significantly greater than that of conventional release tablets. The in vitro release and in vivo absorption profiles were consistent with circadian rhythms in humans. There existed correlation between the in vitro release and in vivo absorption.
1995-06-08·Zhongguo Yaoxue Zazhi (Beijing)
Development of solid oral extended-release preparations
作者: Zeng, Dehui
A review with 43 references covering in vivo characteristics of solid oral extended-release preparations
87
项与 相关的新闻(医药)2023-04-23
·药通社
NMPA分别发布4月20日、21日药品批准证明文件送达信息,2批次共有118个受理号获批,其中6个为一致性评价受理号,分别为:西安海欣制药有限公司,醋氯芬酸片;仁和堂药业有限公司,马来酸氯苯那敏片;西南药业股份有限公司,异烟肼注射液;地奥集团成都药业股份有限公司,盐酸氯哌丁片;康芝药业股份有限公司,尼美舒利分散片;多多药业有限公司,氨酚曲马多片在20日发布的批件信息中,共有38个品种(51品规)获批上市,多个品种视同通过一致性评价,如:浙江仙琚制药的屈螺酮炔雌醇片(Ⅱ);扬子江药业的格列吡嗪控释片;鲁南贝特制药的奥美拉唑碳酸氢钠胶囊;迪沙药业的阿哌沙班片;上海安必生制药雷贝拉唑钠肠溶片;成都苑东生物的艾司奥美拉唑镁肠溶干混悬剂;烟台鲁银药业的盐酸帕洛诺司琼注射液;天地恒一制药的瑞舒伐他汀钙片等。 此外,诺诚健华的奥布替尼片获批上市,用于治疗复发/难治性边缘区淋巴瘤(MZL)患者。这是奥布替尼获批的第3项适应症,目前是中国首款且唯一获批治疗MZL适应症的BTK抑制剂。恒瑞医药的马来酸吡咯替尼片新适应症获批上市,与曲妥珠单抗和多西他赛联合,用于治疗表皮生长因子受体2(HER2)阳性、晚期阶段未接受过抗HER2治疗的复发或转移性乳腺癌患者。
2023-04-13
·赛柏蓝
4月13日,中国非处方药物协会发布《2022年度中国非处方药企业及产品榜》,榜单以企业全年以含税出厂价为准计算的非处方药和健康产品销售额为标准排序,此次统计调研首次纳入了健康产品(未申报数据的企业及产品不在公布之列)。 本次非处方药企业及产品榜共分为4部分,分别为生产企业排名、化学药排名、中成药排名以及大单品名单;健康产品相关榜单共3部分,分别为生产企业综合排名、分品类企业排名以及健康产品排名(完整榜单见文末附1-附7)。关注赛柏蓝,进入主页发消息界面对话框输入“OTC”并发送,获取pdf版完整榜单01150个化药、315个中成药登上OTC榜单涉179家药企 《2022年度中国非处方药生产企业综合统计排名》中共有100家企业上榜,其中Top10分别为华润三九、太极集团、扬子江、修正药业、强生、云南白药、李时珍医药、以岭、康恩贝、东阿阿胶。 完整排名见文末附1 对应《2022年度中国非处方药产品化学药综合统计排名》和《2022 年度中国非处方药产品中成药综合统计排名》来看,位居前列的企业往往也拥有更多上榜产品。 化药OTC产品榜单分为感冒类、止咳化痰类、抗过敏与抗眩晕类等十五个子类,每个子类下的上榜产品数量不一,共150个药品上榜,覆盖复方氨酚烷胺胶囊、氯雷他定片、小儿氨酚烷胺颗粒等114个大品种,涉及77家药企(完整排名见文末附2)。 OTC生产企业Top10中,强生中国个人健康护理(包含上海强生、西安杨森在内)在化药产品榜单中的上榜产品最多,达到14个;其次是太极集团西南药业股份有限公司,共11个产品上榜。 中成药OTC产品榜单分类更加具体,包括感冒咳嗽类、感冒抗病毒类、感冒暑湿类等二十四个子类,共315个药品上榜,覆盖黄连上清片、杞菊地黄丸、明目地黄丸等274个品种,涉及102家药企(完整排名见文末附3)。 OTC生产企业Top10中,太极集团(包括太极集团重庆桐君阁药厂、太极集团四川绵阳制药公司等8家企业)在中成药OTC产品榜单中的上榜产品最多,达到36个,占中成药OTC榜单总数的11%;其次是李时珍医药集团,23个产品上榜;华润三九共20个产品上榜,位于第三。 综合企业榜单与产品榜单来看,还有一些相对特殊的企业——上榜产品少,但企业排名高。 例如,东阿阿胶仅有一款阿胶/复方阿胶浆登上产品榜,但东阿阿胶却在企业榜单中“杀入”Top10,同时,该款产品也被纳入此次《2022年度中国非处方药黄金大单品名单》。东阿阿胶4月12日最新披露的2023年第一季度报显示,截至3月31日,其预估净利润在2.2亿元-2.45亿元之间,同比增长92%-114%。 《2022年度中国非处方药黄金大单品名单》包含18个药品,其中5个化药、13个中成药,共涉及17家企业。 02健康产品领域头部企业优势明显 在首次参与统计调研的健康产品及相关企业方面,上榜企业相对较少,共30家,Top5分别为上药药材、仁和、欧姆龙、康恩贝、北京同仁堂。 完整名单见文末附5 《2022 年度中国健康产品品类综合统计企业排名》按照保健食品、特医食品、健康食品、中药饮片、功能性日化用品、健康器具六个子类对企业进行了排名,其中,保健食品和健康食品领域上榜企业最多,均有15家,特医食品领域上榜企业最少,仅4家。 综合六个子类来看,共54家次企业上榜,去重后共涉及37家企业(完整名单见文末附6)。 在健康产品方面,《2022年度中国健康产品综合统计排名》同样是特医食品上榜产品数量最少,仅9款,除此之外的保健食品、健康食品、功能性日化用品、健康器具均有20款产品上榜(完整名单见文末附7)。 结合对应企业来看,可以发现头部企业在健康产品领域的优势较强,89款产品来自30家不同企业。例如,仁和(集团)发展有限公司共有10款产品上榜,占总数的11%;吉林修正健康股份有限公司也有7款产品上榜。 附1:附2:附3:附4:附5:附6:附7:END作者 | 陈芋微信号:DistillareChen不想错过赛柏蓝每日资讯,点击在看⬇️
2023-04-04
·赛柏蓝
2023年4月3日,在山东济南,国药太极、漱玉平民、山东广播电视台联合启动2023“太极劳动防暑节”。启动全倡导全社会参与公益,传递爱心,致敬最美劳动者。国药太极、漱玉平民高层领导,漱玉平民大区经理及各行业劳动者代表等400多人受邀参加本次启动仪式。山东漱玉平民大药房董事长李文杰出席活动太极股份常务副总经理袁永红太极股份营管中心常务副总经理韦平致辞太极股份营销管理中心常务副总经理韦平表示,国药太极公益活动已经持续二十多年,未来将继续开展“劳动最光荣 防暑要保障”的公益活动,关注、关爱上亿高温劳动者的将康,承担健康责任,弘扬社会正气。国药太极营管中心消化及代谢产品群副总经理苏中波在品牌建设方面,太极集团针对藿香正气口服液启动品牌升级工程——品类破圈增长。坚持“祛暑祛湿,来一瓶,太极藿香正气口服液”的品牌表达、坚持以满足客户为导向的产品发函规划,不断进行略升级,为品牌注入新能量。漱玉平民大药房营运中心总监宋晓娟“太极劳动防暑节”在高温劳动者关怀公益行动上,漱玉平民与太极集团保持着高度一致。漱玉平民将全力配合太极集团,积极开展“劳动防暑节”的公益捐赠活动。并对2023年全年公益活动、战略合作等细节进行了分解和部署,宋女士道:”希望今年共同打造的“劳动防暑节”能够成为漱玉平民与国药太极的标杆项目,履行企业的社会责任,关爱户外高温劳动者,助力他们赢战夏日。”启动会上,播放《致敬高温劳动者》公益宣传片,通过聚焦建筑工人、外卖小哥、电力工人等典型高温劳动者职业群体,他们“用汗水诠释敬业,用‘烤’验谱写奉献”,守护着城市的安全。向高温劳动者代表捐赠10000盒太极藿香正气液由环卫工人、外卖小哥、电焊工人等组成的劳动者代表受邀上台,太极股份营管中心副总经理王乐与漱玉平民大药房连锁股份有限公司总裁秦光霞向高温户外工作者代表一一捐赠10000盒太极藿香正气液,致敬他们的辛勤付出。军令如山、从令如流,掷地有声、言出必行。漱玉平民和太极集团将带着使命和责任,强强联合、通力合作,创造新的历史佳绩!国药太极常务副总经理袁永红、营管中心常务副总经理韦平、副总经理王乐、消化及代谢产品群副总经理苏中波、山东山海风行广告文化传媒有限公司总经理董经龙、漱玉平民大药房连锁股份有限公司董事长李文杰及全省漱玉平民大区经理等十五位嘉宾共同启动2023“太极劳动防暑节”,将本次活动推向高潮!漱玉平民大药房董事长李文杰作总结发言专注品牌品种,加强品牌合作。李董事长表示,2022年充满了机遇与挑战,我们取得了辉煌的成绩,特别感谢太极集团的大力支持,2023年,太极集团和漱玉平民再次携手并肩,共同努力,一定会创造无限可能,最后带领在场参会人员共同喊出口号:“祛暑祛湿,来一瓶,太极藿香正气液!”高温关怀,行胜于言。国药太极已连续二十余年开展公益活动,为数千万人民群众带去了夏日清凉。2023年,国药太极将把“关爱高温下的劳动者”大型公益活动提档升级,履行央企国企担当,践行社会责任,向更多高温作业的单位公益捐赠太极藿香正气口服液等防暑解暑物资,关爱更多高温下的劳动者。国药太极将秉承“呵护生命、关爱健康”的理念,2023年以“劳动最光荣,防暑要保障”为主题,聚焦高温下的劳动者,开展大型公益活动,也希望更多的爱心企业、爱心人士能参与其中!
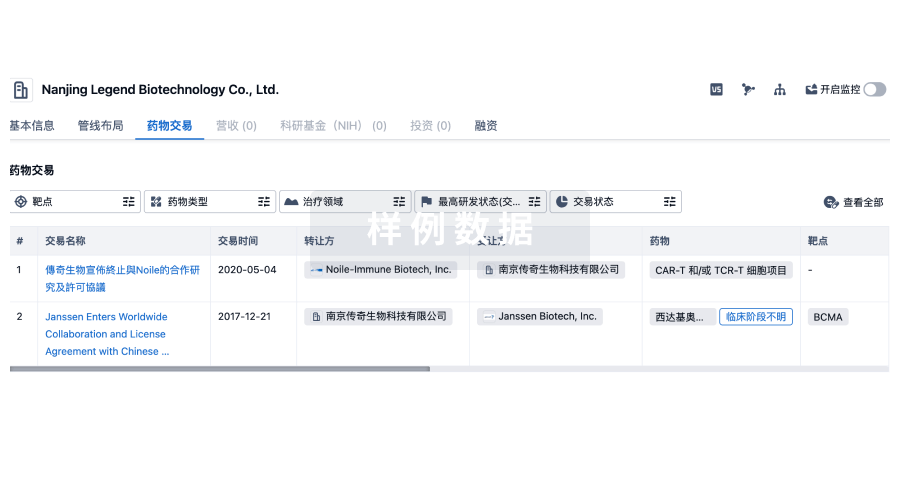
100 项与 相关的药物交易
登录后查看更多信息
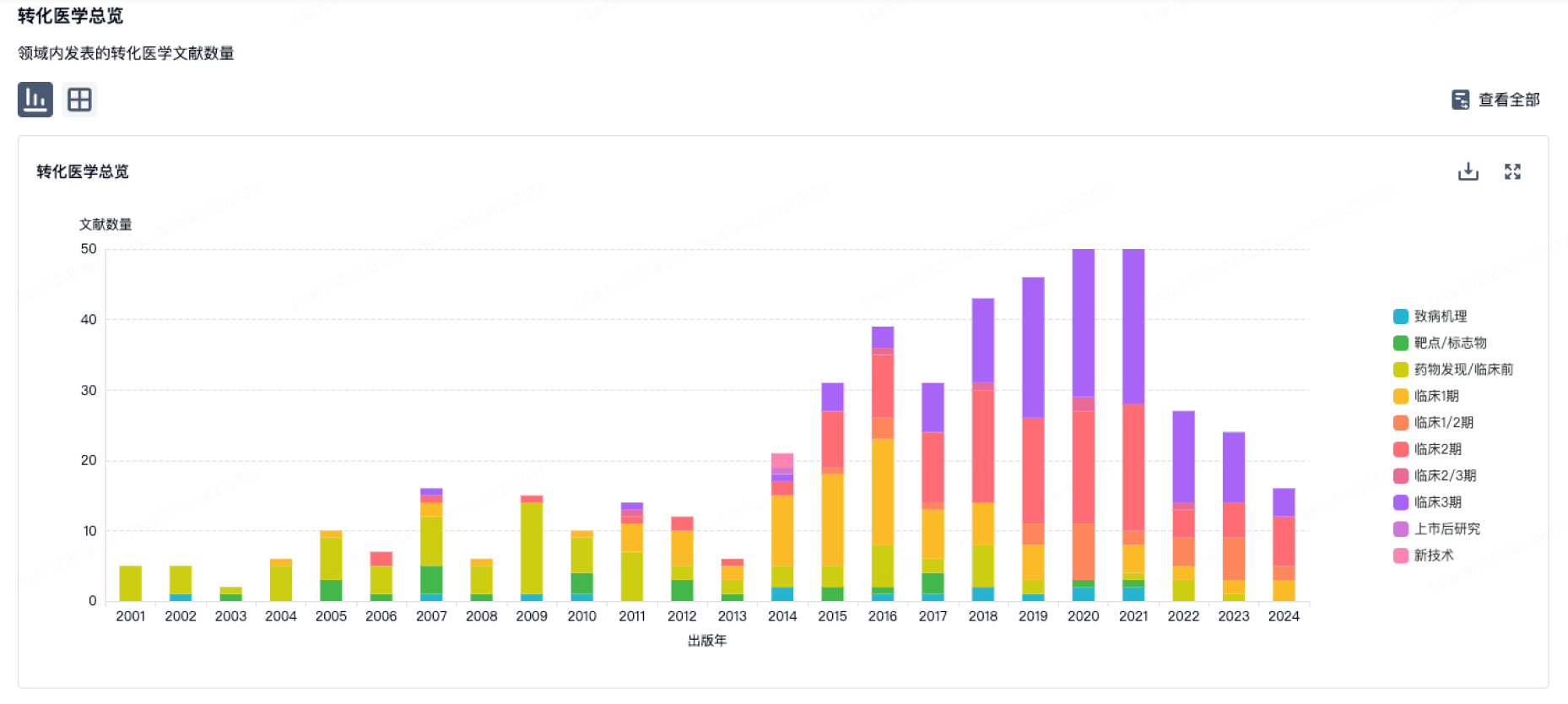
100 项与 相关的转化医学
登录后查看更多信息
组织架构
使用我们的机构树数据加速您的研究。
登录
或

管线布局
2024年09月16日管线快照
管线布局中药物为当前组织机构及其子机构作为药物机构进行统计,早期临床1期并入临床1期,临床1/2期并入临床2期,临床2/3期并入临床3期
批准上市
6
1
其他
登录后查看更多信息
药物交易
使用我们的药物交易数据加速您的研究。
登录
或
转化医学
使用我们的转化医学数据加速您的研究。
登录
或
营收
使用 Synapse 探索超过 36 万个组织的财务状况。
登录
或
科研基金(NIH)
访问超过 200 万项资助和基金信息,以提升您的研究之旅。
登录
或
投资
深入了解从初创企业到成熟企业的最新公司投资动态。
登录
或
融资
发掘融资趋势以验证和推进您的投资机会。
登录
或
标准版
¥16800
元/账号/年
新药情报库 | 省钱又好用!
立即使用
来和芽仔聊天吧
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用