预约演示
更新于:2025-05-07
c-met mutant solid tumour
c-met阳性实体瘤
更新于:2025-05-07
基本信息
别名- |
简介- |
关联
7
项与 c-met阳性实体瘤 相关的药物靶点 |
作用机制 c-Met抑制剂 |
原研机构 |
在研适应症 |
最高研发阶段批准上市 |
首次获批国家/地区 中国 |
首次获批日期2023-11-14 |
作用机制 微管蛋白抑制剂 [+1] |
在研机构 |
原研机构 |
非在研适应症- |
最高研发阶段临床2期 |
首次获批国家/地区- |
首次获批日期1800-01-20 |
靶点 |
作用机制 c-Met抑制剂 |
原研机构 |
非在研适应症- |
最高研发阶段临床1/2期 |
首次获批国家/地区- |
首次获批日期1800-01-20 |
8
项与 c-met阳性实体瘤 相关的临床试验NCT06669117
A First-In-Human (FIH) Phase I/II Open-label, Multicentre, Dose Escalation and Expansion Trial of VERT-002 in Patients With Locally Advanced or Metastatic Solid Tumors Including Non-small Cell Lung Cancer (NSCLC) Harboring Mesenchymal-Epithelial Transition (MET) Alterations
The goal of this clinical trial is to investigate the safety, the activity of VERT-002, and the optimal safe dose to be used, in participants with solid tumors including non-small cell lung cancer.
开始日期2024-10-22 |
CTR20202395
评价注射用RC108治疗c-Met阳性晚期恶性实体瘤患者的安全性、药代动力学和有效性的Ⅰ/Ⅱa期临床研究
主要目的:确定注射用RC108的安全性和耐受性;
次要目的:初步评价注射用RC108的药代动力学特点;初步评价注射用RC108的免疫原性;初步评价注射用RC108的疗效;生物标志物评价。
开始日期2021-03-10 |
申办/合作机构 |
NCT04617314
A Phase I Study to Evaluate the Safety, Pharmacokinetics, and Effect of RC108-ADC for Injection in Subjects with C-Met Positive Advanced Malignant Solid Tumors
This study will evaluate the safety, pharmacokinetics, and effect of RC108-ADC for injeciton in subjects with c-Met positive advanced malignant solid tumors.
开始日期2021-03-10 |
申办/合作机构 |
100 项与 c-met阳性实体瘤 相关的临床结果
登录后查看更多信息
100 项与 c-met阳性实体瘤 相关的转化医学
登录后查看更多信息
0 项与 c-met阳性实体瘤 相关的专利(医药)
登录后查看更多信息
9
项与 c-met阳性实体瘤 相关的文献(医药)2023-10-01·Investigational new drugs
A novel bivalent anti-c-MET/PD-1 bispecific antibody exhibits potent cytotoxicity against c-MET/PD-L1-positive colorectal cancer.
Article
作者: Yao, Y ; Ji, P ; Wang, X ; Gu, C ; Li, D ; Wu, J ; Shang, A ; Liu, W ; Quan, W ; Sun, Z
2023-03-01·British Journal of Clinical Pharmacology
Effect of capmatinib on the pharmacokinetics of substrates of CYP3A (midazolam) and CYP1A2 (caffeine) in patients with MET‐dysregulated solid tumours
Article
作者: Rahmanzadeh, Gholamreza ; Chen, Xinhui ; You, Benoit ; Pognan, Nathalie ; Cui, Xiaoming ; Isambert, Nicolas ; Mau‐Sorensen, Morten ; Giovannini, Monica ; Kapoor, Shruti ; López‐López, Rafael ; Quinlan, Michelle
2020-12-01·Lung Cancer
MET-mutant cancer and immune checkpoint inhibitors: A large database analysis
Letter
作者: Wang, Linlin ; Li, Xuanzong ; Wang, Ruozheng
分析
对领域进行一次全面的分析。
登录
或
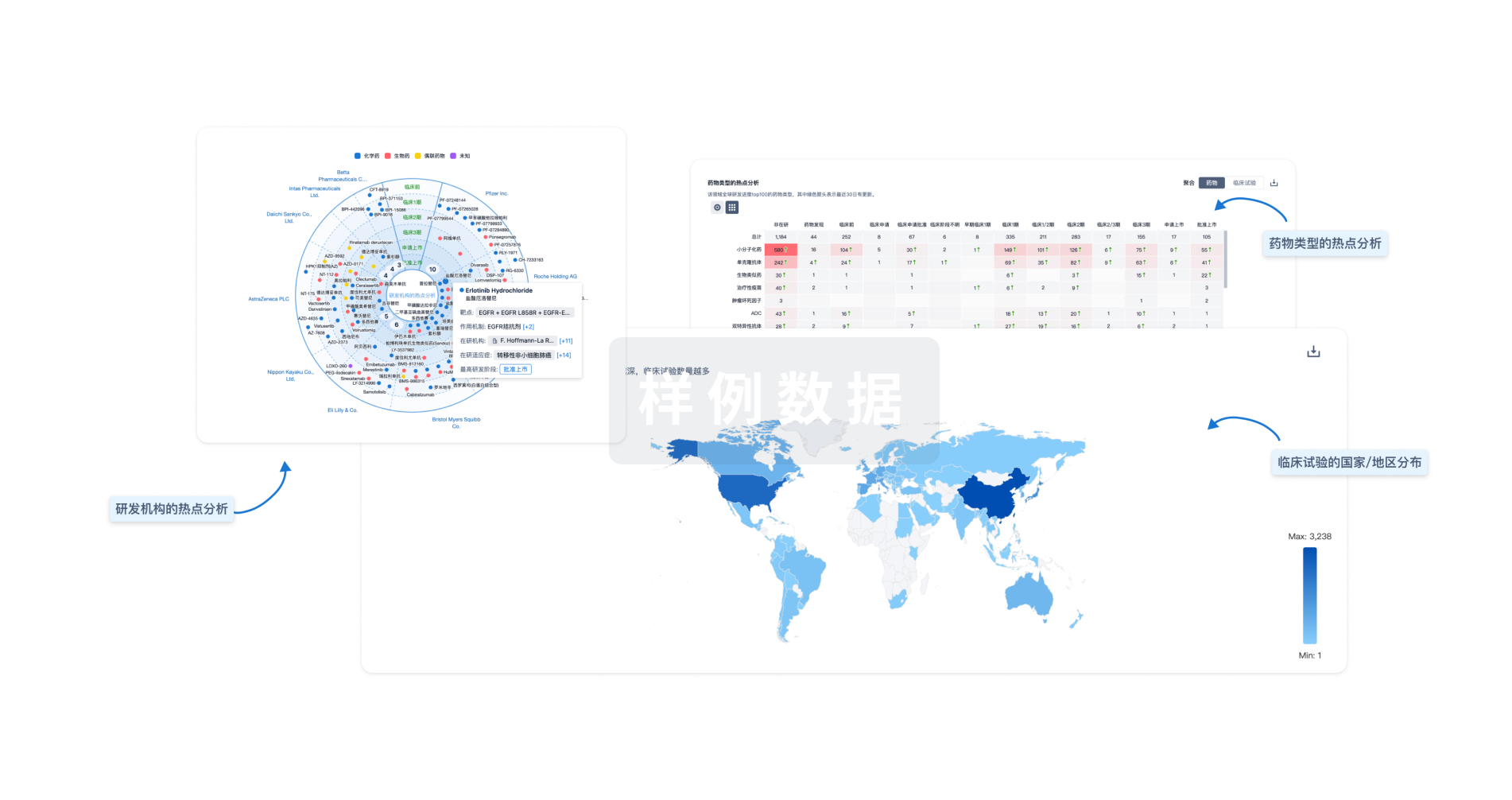
生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用



