预约演示
更新于:2025-05-07
vaginal mucosa dry
阴道粘膜干燥
更新于:2025-05-07
基本信息
别名 DRYNESS VAGINAL、Dry;vagina、Dryness of vagina + [16] |
简介 An uncomfortable feeling of itching and burning in the vaginal opening resulting from inadequate vaginal lubrication. It is commonly seen during and after menopause, childbirth, or stressful conditions. It results in painful intercourse. |
关联
5
项与 阴道粘膜干燥 相关的药物靶点 |
作用机制 ERs调节剂 |
在研机构 |
非在研适应症 |
最高研发阶段批准上市 |
首次获批国家/地区 美国 |
首次获批日期2013-02-26 |
靶点 |
作用机制 ERs激动剂 |
最高研发阶段批准上市 |
首次获批国家/地区 日本 |
首次获批日期1960-11-18 |
靶点 |
作用机制 胶原蛋白调节剂 |
在研机构 |
原研机构 |
在研适应症 |
非在研适应症 |
最高研发阶段临床1期 |
首次获批国家/地区- |
首次获批日期1800-01-20 |
59
项与 阴道粘膜干燥 相关的临床试验NCT06900374
RF-Vaginale : Randomised Phase III Trial Evaluating the Efficacy and Safety of Vaginal Radiofrequency in the Management of Vulvovaginal Atrophy and Dryness in Patients Treated for Breast Cancer
This Phase III, randomized, balanced, parallel-group, multicenter trial aims to evaluate the effect of vaginal radiofrequency on symptoms of vaginal dryness at six months. The study compares vaginal radiofrequency treatment with non-hormonal hydration therapy in a population of patients undergoing adjuvant hormonal therapy with aromatase inhibitors, with or without LHRH agonists, for breast cancer.
Patients will be randomly assigned to one of two parallel treatment groups until the six-month assessment:
The first group will receive the reference treatment, which consists of local hydration using a hyaluronic acid-based treatment applied three times a week. Additionally, investigators may incorporate supportive measures at their discretion, such as physiotherapy or vaginal dilators.
The second group will receive the same reference treatment combined with vaginal radiofrequency therapy. This intervention consists of three sessions, each spaced 4 to 6 weeks apart.
Patients will be randomly assigned to one of two parallel treatment groups until the six-month assessment:
The first group will receive the reference treatment, which consists of local hydration using a hyaluronic acid-based treatment applied three times a week. Additionally, investigators may incorporate supportive measures at their discretion, such as physiotherapy or vaginal dilators.
The second group will receive the same reference treatment combined with vaginal radiofrequency therapy. This intervention consists of three sessions, each spaced 4 to 6 weeks apart.
开始日期2025-05-01 |
申办/合作机构  Centre Oscar Lambret Centre Oscar Lambret [+1] |
NCT06564883
Clinical Evaluation of Hydration and Acceptability of the Genital and Cutaneous Mucosa of a Vaginal Moisturizer in Vaginal Dryness in Menopausal Women
Hyaluronic Acid is indicated for the relief of vaginal dryness, itching, and irritation, or the reduction of vaginal lubrication with prolonged hydration effects for up to 72 hours. Hyaluronic Acid promotes vaginal moisture, providing symptom relief in various situations such as:
* Vaginismus and other sexual dysfunctions related to penetration
* Users of hormonal contraceptives with lubrication loss
* Postpartum and lactational period
* Post antibacterial or antifungal therapies
* During systemic oncological treatments (chemotherapy, radiotherapy, hormone therapy)
* Climacteric (urogenital atrophy)
* Post urogynecological surgeries (correction of urinary incontinence, genital dystopias, etc.)
* Post vulvovaginal abrasive, chemical, laser, or high-frequency therapies
* Post cosmetic and genital rejuvenation therapies
* Vaginismus and other sexual dysfunctions related to penetration
* Users of hormonal contraceptives with lubrication loss
* Postpartum and lactational period
* Post antibacterial or antifungal therapies
* During systemic oncological treatments (chemotherapy, radiotherapy, hormone therapy)
* Climacteric (urogenital atrophy)
* Post urogynecological surgeries (correction of urinary incontinence, genital dystopias, etc.)
* Post vulvovaginal abrasive, chemical, laser, or high-frequency therapies
* Post cosmetic and genital rejuvenation therapies
开始日期2025-03-01 |
NCT05379153
The Revitalize Trial: Reducing Vaginal Atrophy With Fractional CO2 Laser for Breast Cancer Survivors
This phase III trial tests whether vaginal fractional CO2 laser therapy works in treating vaginal dryness in breast cancer survivors. Vaginal dryness can be commonly experienced with normal aging, with menopause, or come about from medications used to treat breast cancer such as chemotherapy or endocrine therapies like tamoxifen or aromatase inhibitors. Vaginal fractional CO2 laser therapy may help reduce vaginal dryness.
开始日期2024-12-01 |
申办/合作机构 |
100 项与 阴道粘膜干燥 相关的临床结果
登录后查看更多信息
100 项与 阴道粘膜干燥 相关的转化医学
登录后查看更多信息
0 项与 阴道粘膜干燥 相关的专利(医药)
登录后查看更多信息
1,343
项与 阴道粘膜干燥 相关的文献(医药)2025-07-01·Nutrition
Navigating nutrition through the decades: Tailoring dietary strategies to women's life stages
Review
作者: Thakkar, Ayushi Atul ; Shelke, Smita Panditrao ; Mali, Dhanshri Revannath ; Harak, Shilpa Sudhakar
2025-05-01·Journal of Gynecology Obstetrics and Human Reproduction
Long-term efficacy of CO2 fractional laser in the treatment of genitourinary syndrome of menopause
Article
作者: Uzan, Pr Catherine ; Oueld Es Cheikh, Dr Eva ; Favier, Dr Amelia ; Canlorbe, Pr Geoffroy ; Morisot, Adeline ; Donnart, Dr Marion
2025-04-01·Supportive Care in Cancer
Genitourinary symptoms in women with breast cancer: frequency, severity and impact
Article
作者: Pearson, Antonia ; Lombard, Janine ; Hickey, Martha ; Chen, Jill ; Dhillon, Haryana M ; Campbell, Rachel ; Kiely, Belinda E
48
项与 阴道粘膜干燥 相关的新闻(医药)2025-03-06
THURSDAY, March 6, 2025 -- Hot flashes, night sweats and other symptoms of a difficult
menopause
could be early warning flags for dementia, a new study suggests.
Women who exhibit more menopause symptoms are more likely to develop early signs of dementia like decreased mental function and mild behavioral impairment, compared with women who have an easier transition, researchers reported in the journal
PLOS One
.
Results also showed that hormone replacement therapy did help ease the effects of menopause on behavioral impairment.
“It's fascinating that there is a link between the menopausal experience and subsequent changes in cognition and behavior,” senior investigator
Dr. Zahinoor Ismail
, a neuropsychiatrist at the University of Calgary in Canada, said in a news release. “I find that mind blowing.”
Women are known to have triple a man's risk of developing
Alzheimer’s disease
and related dementias, researchers said in background notes.
To examine this risk more closely, researchers analyzed data on nearly 900 women who’d gone through menopause.
The women reported the symptoms they suffered during menopause, and took tests measuring their brain function and behavior.
Symptoms of menopause could include irregular periods, hot flashes, chills, vaginal dryness, weight gain, slowed metabolism, night sweats, sleep problems, mood symptoms, inattention or forgetfulness and other unnamed symptoms, researchers said.
Women who reported more menopause symptoms tended to have more cognitive impairment later in life, researchers found.
They also had more behavioral problems associated with dementia, such as apathy, anxiety, irritability, lack of inhibition, loss of social graces or tact or inflexibility.
Researchers also asked women if they’d used hormone replacement therapy, which can ease the symptoms of menopause.
Those who had used hormone therapy had better behavioral impairment scores, but the therapy did not improve their cognitive scores, results show.
Some have speculated that the decline in estrogen levels caused by menopause might affect later brain health, researchers said.
Estradiol, a form of estrogen, is known to contribute to the development of synapses and neurons, “all of which decline in neurodegenerative diseases and dementia,” researchers wrote.
“In menopause, estradiol naturally declines,” the research team wrote. “Thus, the experience of menopausal symptoms may act as an indicator of how well females tolerate estradiol changes.”
However, they added that more research involving a larger group of women is needed to verify these findings, and to better understand the potential effects of menopause on brain health.
Whatever your topic of interest,
subscribe to our newsletters
to get the best of Drugs.com in your inbox.
2025-02-28
FRIDAY, Feb. 28, 2025 -- Many young women entering
menopause
suffer needlessly from symptoms related to the transition, a new study suggests.
More than half of women aged 30 to 35 have already developed moderate to severe symptoms of impending menopause, researchers found.
These include mood swings, delayed or absent periods, hot flashes, vaginal dryness, painful sex, heart palpitations and frequent urination.
Unfortunately, most wait decades before seeking treatment, under the mistaken belief that menopause symptoms that shouldn’t appear until their 50s, researchers said.
“We had a significant number of women who are typically thought to be too young for perimenopause tell us that they have high levels of perimenopause-related symptoms,” senior researcher
Liudmila Zhaunova
, director of science at Flo Health Inc., a London-based company that offers a pregnancy and ovulation tracking app.
Perimenopause is the transition period leading to menopause, researchers said.
For the study, researchers analyzed symptoms self-reported by more than 4,400 American women 30 and older who responded to a survey offered online and through the Flo app.
About 55% of women 30 to 35 reported problems that met the criteria for “moderate” or severe” menopause symptoms, researchers report in the journal
npj Women’s Health
.
That number increased to 64% among women 36 to 40, researchers noted.
But only 4% of women 30 to 35 and 7% of those 36 to 40 said they’d visited a medical professional to discuss menopause symptoms, results show.
“We also demonstrated that over a quarter of respondents in the youngest age group (30–35 years) had been told by a medical professional they were perimenopausal,” researchers wrote in their study.
Psychological symptoms like anxiety, depression and irritability took hold long before the physical symptoms of menopause set in, researchers said. Those symptoms peaked among women 41 to 45, then ebbed to their lowest in women 56 and older.
Physical problems like painful sex, bladder issues and vaginal dryness peaked in women 51 and older, and were lowest in women 30 to 35, researchers said.
Likewise, the “classic” symptoms of menopause like hot flashes peaked in women 51 to 55, and were least common among women 30 to 35.
Researchers said they hope their results will help fill an “alarming gap” in the understanding of perimenopause symptoms among younger women.
“This study is important because it plots a trajectory of perimenopausal symptoms that tells us what symptoms we can expect when and alerts us to the fact that women are experiencing perimenopausal symptoms earlier than we expected,” researcher
Dr. Jennifer Payne
, an expert in reproductive psychiatry at the University of Virginia School of Medicine, said in a news release.
Whatever your topic of interest,
subscribe to our newsletters
to get the best of Drugs.com in your inbox.
临床结果
2025-01-07
TUESDAY, Jan. 7, 2025 -- Women living in rural areas may experience a greater burden of psychological and somatic
menopause
symptoms, according to a study published in the January issue of
Menopause
.
Erin R. Dwyer, from the University of Washington School of Medicine in Seattle, and colleagues compared menopause experience in rural-, suburban-, and urban-residing women. The analysis included online survey responses from 1,531 participants.
The researchers found that rural residents had more prevalent symptoms in late menopause transition, including muscle aches and pains (urban, 49 percent; suburban, 49 percent; rural, 65 percent) and panic attacks (urban, 18 percent; suburban, 24 percent; rural, 30 percent). Rural residents also had more prevalent symptoms in postmenopause, including: mood swings (urban, 18 percent; suburban, 14 percent; rural, 23 percent), urinary incontinence (urban, 19 percent; suburban, 14 percent; rural, 23 percent), and
vaginal dryness
(urban, 22 percent; suburban, 29 percent; rural, 37 percent). Overall, vasomotor symptom prevalence was high (71 percent late menopause transition, 20 percent postmenopause), while current menopause hormone therapy use was low (11 percent late menopause transition, 11 percent postmenopause) and did not differ by residence.
"Overall low rates of menopause hormone therapy use suggest a need for education regarding hormone therapy, tailored to residential groups who rely on different resources on healthy aging," the authors write.
Two authors disclosed ties to the pharmaceutical industry.
分析
对领域进行一次全面的分析。
登录
或
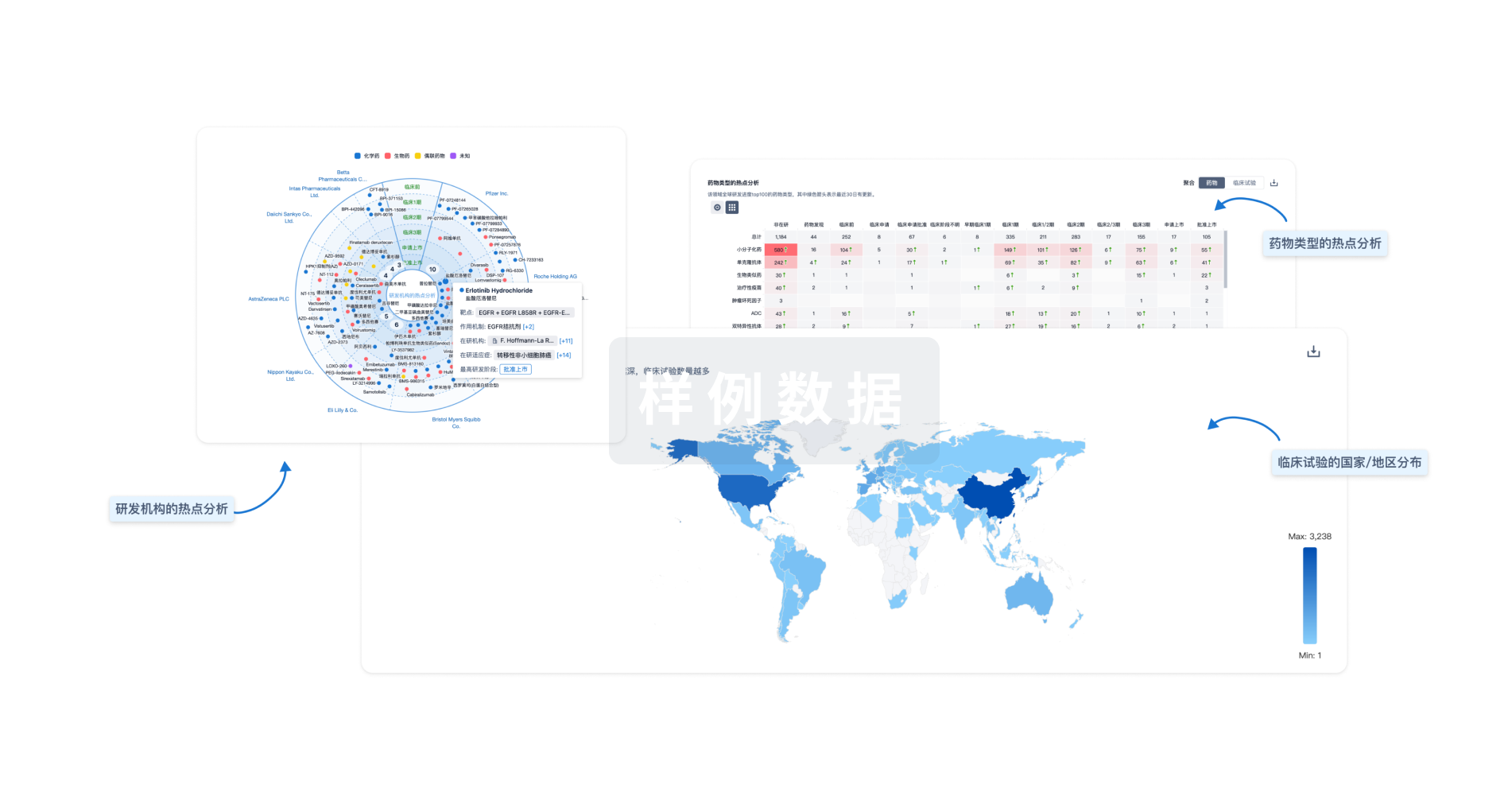
生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用





