预约演示
更新于:2025-05-07
Vitamin a Deficiency
维生素a缺乏症
更新于:2025-05-07
基本信息
别名 Deficiencies, Vitamin A、Deficiency of vitamin A、Deficiency, Vitamin A + [38] |
简介 A nutritional condition produced by a deficiency of VITAMIN A in the diet, characterized by NIGHT BLINDNESS and other ocular manifestations such as dryness of the conjunctiva and later of the cornea (XEROPHTHALMIA). Vitamin A deficiency is a very common problem worldwide, particularly in developing countries as a consequence of famine or shortages of vitamin A-rich foods. In the United States it is found among the urban poor, the elderly, alcoholics, and patients with malabsorption. (From Cecil Textbook of Medicine, 19th ed, p1179) |
关联
6
项与 维生素a缺乏症 相关的药物靶点 |
作用机制 RARs激动剂 |
原研机构 |
非在研适应症- |
最高研发阶段批准上市 |
首次获批国家/地区 日本 |
首次获批日期1976-08-10 |
作用机制 RARs激动剂 [+1] |
原研机构 |
非在研适应症- |
最高研发阶段批准上市 |
首次获批国家/地区 美国 |
首次获批日期1949-05-18 |
99
项与 维生素a缺乏症 相关的临床试验NCT06450925
A Randomized Double Blinded Trial of Vitamin A Supplementation in Allogeneic Stem Cell Transplantation.
The investigators hypothesize that single oral high dose supplementation with vitamin A will reduce the incidence of moderate-severe chronic graft-versus-host disease (GVHD) compared with placebo.
开始日期2025-03-01 |
NCT06759493
Validation and Simplification of the Retinol Isotope Dilution Technique in Burkinabe Schoolchildren
Two cross-sectional, repeated-pass, community-based studies will be carried out during the school year. The first study will be conducted from November to December. This period corresponds to the end of the rainy season and the end of the mango season, which is one of the main sources of vitamin A in the Orodara region.
The second survey will be carried out 5 to 6 months (April-May of the following year) after data collection for the first study, corresponding to the mango season. The same schoolchildren recruited during the first phase of data collection (5 to 6 months earlier) will be the participants in the second study. The interest of this study is to use nuclear techniques to implement a strategy for the precise and accurate determination of Vitamin A status in order to reinforce existing Vitamin A interventions and policies.
The second survey will be carried out 5 to 6 months (April-May of the following year) after data collection for the first study, corresponding to the mango season. The same schoolchildren recruited during the first phase of data collection (5 to 6 months earlier) will be the participants in the second study. The interest of this study is to use nuclear techniques to implement a strategy for the precise and accurate determination of Vitamin A status in order to reinforce existing Vitamin A interventions and policies.
开始日期2024-11-21 |
NCT06372366
Relationship Between Vitamins Deficiency and Caries Experience Among a Group of Egyptian Children: A Cross-Sectional Study
Dental caries is a worldwide condition characterized by localized destruction of dental hard tissue by acidic by-products from bacterial fermentation of dietary carbohydrates . Dental caries is considered to be the single most common chronic childhood disease, and its prevalence is thought to have increased recently.
开始日期2024-07-15 |
申办/合作机构 |
100 项与 维生素a缺乏症 相关的临床结果
登录后查看更多信息
100 项与 维生素a缺乏症 相关的转化医学
登录后查看更多信息
0 项与 维生素a缺乏症 相关的专利(医药)
登录后查看更多信息
4,850
项与 维生素a缺乏症 相关的文献(医药)2025-12-01·Molecular Biology Reports
A detailed comparative in silico and functional analysis of ccd1 gene in maize gives new insights of its expression and functions
Article
作者: Mandal, Pranab Kumar ; Poudel, Ankur ; Gaikwad, Kishor ; Phogat, Sachin ; Banu, Nuzat ; Roy, Jeet ; Muthusamy, Vignesh ; Chanda, Sagnik ; Hossain, Firoz ; Madhavan, Jayanthi
2025-06-01·International Journal of Paleopathology
Archeometric detection of mercury: A paleopharmacological case study of skeletal remains of a child with vitamin deficiencies (Rouen, France, late 18–19th centuries)
Article
作者: Thomann, Aminte ; Lefrais, Yannick ; Colombo, Antony ; Dutour, Olivier ; Zinn, Alexandra
2025-05-01·Veterinary Medicine and Science
Stillbirth Caused by Vitamin A Deficiency in Captive African Lion Cubs (Panthera leo)
Article
作者: Khordadmehr, Monireh ; Ghorani, Mohammadreza ; Nofouzi, Katayoon ; Sadri, Hassan ; Jigari‐Asl, Farinaz ; Gharehaghajlou, Yashar
50
项与 维生素a缺乏症 相关的新闻(医药)2025-04-30
- Encouraging start to first independent launch with TRYNGOLZATM -
- On track for second independent launch with donidalorsen PDUFA August 21, 2025 -
- Increasing 2025 financial guidance by more than 20% -
CARLSBAD, Calif. --(BUSINESS WIRE)--Apr. 30, 2025-- Ionis Pharmaceuticals, Inc. (Nasdaq: IONS) (the “Company”) today reported financial results for the first quarter ended March 31, 2025 .
“With an encouraging start to the TRYNGOLZA launch for familial chylomicronemia syndrome, the first of four independent launches expected over the next two years, Ionis’ new chapter as a fully integrated, commercial-stage biotechnology company is well underway,” said Brett P. Monia , Ph.D., chief executive officer, Ionis. “We look forward to continued momentum this year, including our second independent launch for donidalorsen in hereditary angioedema and Phase 3 results for olezarsen for severe hypertriglyceridemia in the third quarter. We also continue to advance our next wave of wholly owned neurology medicines, including ION582 for Angelman syndrome, which is on track to start Phase 3 development shortly. Our advancing pipeline of transformational medicines, together with strong commercial and financial execution, position Ionis to deliver increasing value for all stakeholders.”
First Quarter 2025 Summary Financial Results(1):
Three months ended March 31 ,
2025
2024
(amounts in millions)
Total revenue
$
132
$
119
Operating expenses
$
278
$
269
Operating expenses on a non-GAAP basis
$
249
$
238
Loss from operations
$
(146
)
$
(150
)
Loss from operations on a non-GAAP basis
$
(117
)
$
(119
)
(1)
Reconciliation of GAAP to non-GAAP basis contained later in this release.
Recent Financial Highlights
Revenue increased 10% in the first quarter of 2025 compared to the same period last year, driven by higher commercial revenue including new TRYNGOLZA product revenue following approval in late December and higher SPINRAZA and WAINUA royalty revenue Operating expenses increased slightly in the first quarter of 2025 driven by commercialization efforts for TRYNGOLZA, donidalorsen and WAINUA Increased 2025 financial guidance reflects recent successful licensing transactions:
Full Year 2025 Guidance
Previous Guidance
New Guidance
Revenue
> $600 million
$725-750 million
Operating loss on a non-GAAP basis
< $495 million
< $375 million
Cash, cash equivalents and short-term investments
~$1.7 billion
~$1.9 billion
Recent Highlights - Marketed Medicines
TRYNGOLZATM (olezarsen), the first-ever treatment for adults living with familial chylomicronemia syndrome (FCS) as an adjunct to diet, generated net product sales of over $6 million in its first full quarter following approval in the U.S. on December 19, 2024 Commercialization rights in countries outside of the U.S. , Canada and China licensed to Sobi EU approval decision anticipated in H2:2025 WAINUATM (eplontersen) (WAINZUA in EU) for the treatment of adults with polyneuropathy of hereditary transthyretin-mediated amyloidosis (ATTRv-PN) achieved commercial and regulatory milestones: Generated sales of $39 million resulting in royalty revenue of $9 million in the first quarter of 2025 Launch underway in numerous countries, including the EU following approval by the European Commission (EC); additional global submissions in progress to expand WAINUA access SPINRAZA® (nusinersen) for the treatment of spinal muscular atrophy (SMA) generated global sales of $424 million resulting in royalty revenue of $48 million in the first quarter of 2025 Higher dose nusinersen under review for marketing approval in U.S. (PDUFA date of September 22, 2025 ) and EU
Commercialization rights in countries outside of the U.S. , Canada and China licensed to Sobi EU approval decision anticipated in H2:2025
Generated sales of $39 million resulting in royalty revenue of $9 million in the first quarter of 2025 Launch underway in numerous countries, including the EU following approval by the European Commission (EC); additional global submissions in progress to expand WAINUA access
Higher dose nusinersen under review for marketing approval in U.S. (PDUFA date of September 22, 2025 ) and EU
Recent Highlights - Late-Stage Wholly Owned Pipeline
Olezarsen on track for topline Phase 3 data from supportive ESSENCE study in Q2:2025 and pivotal CORE and CORE2 studies in patients with severe hypertriglyceridemia (sHTG) in Q3:2025, positioning olezarsen to potentially treat this second, more prevalent patient population with high unmet need Published Phase 3 study design and baseline characteristics for CORE, CORE2 and ESSENCE studies in the American Heart Journal , highlighting the potential to generate robust and meaningful data Donidalorsen on track for launch this year as the first RNA-targeted prophylactic treatment for people with hereditary angioedema (HAE), assuming approval: Under review for marketing approval in U.S. (PDUFA date of August 21, 2025 ) and EU Presented positive Phase 2 open label extension (OLE) study data demonstrating an overall sustained mean reduction in HAE attack rates of 96% in patients treated up to 196 weeks with every four weeks or every eight weeks dosing Published positive patient-reported outcomes (PROs) from Phase 3 OASIS-HAE study in Allergy showing donidalorsen significantly improved quality of life (QoL) and other PROs compared to placebo ION582 on track to initiate Phase 3 development in Angelman syndrome (AS) in Q2:2025
Published Phase 3 study design and baseline characteristics for CORE, CORE2 and ESSENCE studies in the American Heart Journal , highlighting the potential to generate robust and meaningful data
Under review for marketing approval in U.S. (PDUFA date of August 21, 2025 ) and EU Presented positive Phase 2 open label extension (OLE) study data demonstrating an overall sustained mean reduction in HAE attack rates of 96% in patients treated up to 196 weeks with every four weeks or every eight weeks dosing Published positive patient-reported outcomes (PROs) from Phase 3 OASIS-HAE study in Allergy showing donidalorsen significantly improved quality of life (QoL) and other PROs compared to placebo
Recent Highlights - Partnered Pipeline
Pelacarsen Phase 3 Lp(a) HORIZON study design and baseline characteristics published in the American Heart Journal highlighting the potential to generate robust and meaningful data Licensed sapablursen global development and commercialization rights to Ono Pharmaceutical Co. generating $280 million in an upfront payment, with the potential to earn up to $660 million in additional payments plus royalties in the mid-teen percentage range on annual net sales IONIS-MAPTRx (BIIB080) received FDA Fast Track designation for the treatment of Alzheimer’s disease
First Quarter 2025 Financial Results
“We are increasing our 2025 financial guidance including raising revenue guidance by more than 20 percent due to our strong first quarter results and recent successful licensing transactions. We are also substantially improving our operating loss and cash guidance and now expect to end the year with approximately $1.9 billion in cash. Our strong financial position and commitment to drive operating leverage position Ionis to advance our strategic priorities and successfully navigate the dynamic macroeconomic environment,” said Elizabeth L. Hougen , chief financial officer, Ionis. “Our first quarter results reflected encouraging early performance in the TRYNGOLZA launch, adding product revenue to our P&L for the first time. Moving forward, the three additional independent launches anticipated over the next couple of years position Ionis to deliver substantial and growing product revenue. This product revenue coupled with anticipated increasing royalty revenue from multiple partner launches and disciplined investment, position Ionis to achieve sustained positive cash flow.”
Revenue
Ionis’ revenue was comprised of the following:
Three months ended March 31 ,
2025
2024
Revenue
(amounts in millions)
Commercial revenue:
Product sales, net:
TRYNGOLZA sales, net
$
6
$
-
Total product sales, net
6
-
Royalty revenue:
SPINRAZA royalties
48
38
WAINUA royalties
9
1
Other royalties
7
10
Total royalty revenue
64
49
Other commercial revenue:
TEGSEDI and WAYLIVRA revenue, net
6
9
Other revenue
-
1
Total other commercial revenue
6
10
Total commercial revenue
76
59
Research and development revenue:
Collaborative agreement revenue
46
49
WAINUA joint development revenue
10
11
Total research and development revenue
56
60
Total revenue
$
132
$
119
Commercial revenue for the first quarter of 2025 increased 28% compared to the same period in 2024, driven in part by revenue from U.S. product sales from the launch of TRYNGOLZA. Higher royalty revenues from SPINRAZA, WAINUA and QALSODY also contributed to the year over year increase.
The remainder of the Company’s revenue came from programs under its R&D collaborations, reflecting the value that Ionis’ pipeline and technology continues to generate.
Operating Expenses
SG&A expenses increased in the first quarter of 2025 compared to the same period in 2024 primarily due to the launches of WAINUA and TRYNGOLZA, and advancing launch preparation activities for donidalorsen. This increase was partially offset by a decrease in R&D expenses as several late-stage studies ended. Overall, this led to a slight increase in total operating expenses.
Balance Sheet
As of March 31, 2025 , Ionis’ cash, cash equivalents and short-term investments were $2.1 billion , compared to $2.3 billion at December 31, 2024 . Ionis’ working capital decreased over the same period primarily due to the Company’s lower cash and short-term investments balance. Ionis generated $280 million from the global license of sapablursen in the second quarter.
Webcast
Management will host a conference call and webcast to discuss Ionis’ first quarter 2025 results at 11:30 a.m. Eastern time on Wednesday, April 30, 2025 . Interested parties may access the webcast here. A webcast replay will be available for a limited time at the same address. To access the Company’s first quarter 2025 earnings slides click here.
Ionis’ Marketed Medicines
INDICATION for TRYNGOLZA™ (olezarsen)
TRYNGOLZA™ (olezarsen) was approved by the U.S. Food and Drug Administration as an adjunct to diet to reduce triglycerides in adults with familial chylomicronemia syndrome (FCS).
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
TRYNGOLZA is contraindicated in patients with a history of serious hypersensitivity to TRYNGOLZA or any of the excipients in TRYNGOLZA. Hypersensitivity reactions requiring medical treatment have occurred.
WARNINGS AND PRECAUTIONS
Hypersensitivity Reactions
Hypersensitivity reactions (including symptoms of bronchospasm, diffuse erythema, facial swelling, urticaria, chills and myalgias) have been reported in patients treated with TRYNGOLZA. Advise patients on the signs and symptoms of hypersensitivity reactions and instruct patients to promptly seek medical attention and discontinue use of TRYNGOLZA if hypersensitivity reactions occur.
ADVERSE REACTIONS
The most common adverse reactions (incidence >5% of TRYNGOLZA-treated patients and >3% higher frequency than placebo) were injection site reactions, decreased platelet count and arthralgia.
Please see full Prescribing Information for TRYNGOLZA.
INDICATION for WAINUA™ (eplontersen)
WAINUA injection, for subcutaneous use, 45 mg is indicated for the treatment of the polyneuropathy of hereditary transthyretin-mediated amyloidosis in adults.
IMPORTANT SAFETY INFORMATION for WAINUA™ (eplontersen)
WARNINGS AND PRECAUTIONS
Reduced Serum Vitamin A Levels and Recommended Supplementation WAINUA leads to a decrease in serum vitamin A levels. Supplement with recommended daily allowance of vitamin A. Refer patient to an ophthalmologist if ocular symptoms suggestive of vitamin A deficiency occur.
ADVERSE REACTIONS
Most common adverse reactions (≥9% in WAINUA-treated patients) were vitamin A decreased (15%) and vomiting (9%).
Please see link to U.S. Full Prescribing Information for WAINUA.
For more information about SPINRAZA and QALSODY, visit https://www.spinraza.com/ and https://www.qalsody.com/, respectively. QALSODY is approved under accelerated approval based on reduction in plasma neurofilament light chain (NfL) observed in patients treated with QALSODY. Continued approval may be contingent upon verification of clinical benefit in confirmatory trial(s).
About Ionis Pharmaceuticals, Inc.
For three decades, Ionis has invented medicines that bring better futures to people with serious diseases. Ionis currently has six marketed medicines and a leading pipeline in neurology, cardiology and select areas of high patient need. As the pioneer in RNA-targeted medicines, Ionis continues to drive innovation in RNA therapies in addition to advancing new approaches in gene editing. A deep understanding of disease biology and industry-leading technology propels our work, coupled with a passion and urgency to deliver life-changing advances for patients. To learn more about Ionis, visit Ionis.com and follow us on X (Twitter), LinkedIn and Instagram.
Ionis’ Forward-looking Statement
This press release includes forward-looking statements regarding Ionis’ business, financial guidance and the therapeutic and commercial potential of our commercial medicines, additional medicines in development and technologies. Any statement describing Ionis’ goals, expectations, financial or other projections, intentions or beliefs is a forward-looking statement and should be considered an at-risk statement. Such statements are subject to certain risks and uncertainties including those inherent in the process of discovering, developing and commercializing medicines that are safe and effective for use as human therapeutics, and in the endeavor of building a business around such medicines. Ionis’ forward-looking statements also involve assumptions that, if they never materialize or prove correct, could cause its results to differ materially from those expressed or implied by such forward-looking statements. Although Ionis’ forward-looking statements reflect the good faith judgment of its management, these statements are based only on facts and factors currently known by Ionis. Except as required by law, we undertake no obligation to update any forward-looking statements for any reason. As a result, you are cautioned not to rely on these forward-looking statements. These and other risks concerning Ionis' programs are described in additional detail in Ionis' annual report on Form 10-K for the year ended December 31, 2024 , and most recent Form 10-Q, which are on file with the Securities and Exchange Commission . Copies of these and other documents are available from the Company.
In this press release, unless the context requires otherwise, “Ionis,” “Company,” “we,” “our” and “us” all refer to Ionis Pharmaceuticals and its subsidiaries.
Ionis Pharmaceuticals ® is a registered trademark of Ionis Pharmaceuticals, Inc. TRYNGOLZATM is a trademark of Ionis Pharmaceuticals, Inc. Akcea Therapeutics ® is a registered trademark of Akcea Therapeutics, Inc. TEGSEDI® is a registered trademark of Akcea Therapeutics, Inc. WAYLIVRA® is a registered trademark of Akcea Therapeutics, Inc. SPINRAZA® and QALSODY® are registered trademarks of Biogen. WAINUATM is a registered trademark of the AstraZeneca group of companies.
IONIS PHARMACEUTICALS, INC.
SELECTED FINANCIAL INFORMATION
Condensed Consolidated Statements of Operations
(In Millions, Except Per Share Data)
Three months ended March 31 ,
2025
2024
(unaudited)
Revenue:
Commercial revenue:
Product sales, net
$
6
$
-
Royalty revenue
64
49
Other commercial revenue
6
10
Total commercial revenue
76
59
Research and development revenue:
Collaborative agreement revenue
46
49
WAINUA joint development revenue
10
11
Total research and development revenue
56
60
Total revenue
132
119
Expenses:
Cost of sales
1
2
Research, development and patent
201
214
Selling, general and administrative
76
53
Total operating expenses
278
269
Loss from operations
(146
)
(150
)
Other income (expense):
Interest expense related to the sale of future royalties
(19
)
(18
)
Other income, net
18
25
Loss before income tax expense
(147
)
(143
)
Income tax expense
-
-
Net loss
$
(147
)
$
(143
)
Basic and diluted net loss per share
$
(0.93
)
$
(0.98
)
Shares used in computing basic and diluted net loss per share
159
146
IONIS PHARMACEUTICALS, INC.
Reconciliation of GAAP to Non-GAAP Basis:
Condensed Consolidated Operating Expenses, Loss From Operations, and Net Loss
(In Millions)
Three months ended March 31 ,
2025
2024
(unaudited)
As reported research, development and patent expenses according to GAAP
$
201
$
214
Excluding compensation expense related to equity awards
(20
)
(22
)
Non-GAAP research, development and patent expenses
$
181
$
192
As reported selling, general and administrative expenses according to GAAP
$
76
$
53
Excluding compensation expense related to equity awards
(9
)
(9
)
Non-GAAP selling, general and administrative expenses
$
67
$
44
As reported operating expenses according to GAAP
$
278
$
269
Excluding compensation expense related to equity awards
(29
)
(31
)
Non-GAAP operating expenses
$
249
$
238
As reported loss from operations according to GAAP
$
(146
)
$
(150
)
Excluding compensation expense related to equity awards
(29
)
(31
)
Non-GAAP loss from operations
$
(117
)
$
(119
)
As reported net loss according to GAAP
$
(147
)
$
(143
)
Excluding compensation expense related to equity awards and related tax effects
(29
)
(31
)
Non-GAAP net loss
$
(118
)
$
(112
)
Reconciliation of GAAP to Non-GAAP Basis
As illustrated in the Selected Financial Information in this press release, non-GAAP operating expenses, non-GAAP loss from operations, and non-GAAP net loss were adjusted from GAAP to exclude compensation expense related to equity awards and the related tax effects. Compensation expense related to equity awards are non-cash. These measures are provided as supplementary information and are not a substitute for financial measures calculated in accordance with GAAP. Ionis reports these non-GAAP results to better enable financial statement users to assess and compare its historical performance and project its future operating results and cash flows. Further, the presentation of Ionis’ non-GAAP results is consistent with how Ionis’ management internally evaluates the performance of its operations.
IONIS PHARMACEUTICALS, INC.
Condensed Consolidated Balance Sheets
(In Millions)
March 31 , 2025
December 31 , 2024
(unaudited)
Assets:
Cash, cash equivalents and short-term investments
$
2,145
$
2,298
Contracts receivable
40
92
Other current assets
234
230
Property, plant and equipment, net
103
94
Right-of-use assets
159
162
Other assets
132
127
Total assets
$
2,813
$
3,003
Liabilities and stockholders’ equity:
Current portion of deferred contract revenue
$
81
$
79
Other current liabilities
169
229
1.75% convertible senior notes, net
566
565
0% convertible senior notes, net
629
629
Liability related to sale of future royalties, net
536
542
Long-term lease liabilities
159
162
Long-term obligations, less current portion
56
52
Long-term deferred contract revenue
141
157
Total stockholders’ equity
476
588
Total liabilities and stockholders’ equity
$
2,813
$
3,003
Key 2025 and 2026 Value Driving Events(1)
New Product Launches
Program
Indication
2025
2026
Donidalorsen ( U.S. )
HAE
•
TRYNGOLZA ( U.S. )
FCS
Achieved
WAINZUA (EU)
ATTRv-PN
Achieved
Olezarsen ( U.S. )
sHTG
•
Zilganersen ( U.S. )
Alexander disease
•
Regulatory Actions
Program
Indication
Regulatory Action
2025
2026
Donidalorsen
HAE
U.S. approval decision
•
EU approval decision
•
TRYNGOLZA
FCS
EU approval decision
•
Olezarsen
sHTG
U.S. submission
•
U.S. approval decision
•
Zilganersen
Alexander disease
U.S. submission
•
U.S. approval decision
•
Nusinersen
(higher dose)
SMA
U.S. and EU submissions
Achieved
U.S. approval decision
•
WAINZUA
ATTRv-PN
EU approval decision
Achieved
Pelacarsen
Lp(a)- CVD
U.S. submission
•
Bepirovirsen
HBV
Regulatory submission(s)
•
Regulatory decision(s)
•
Key Phase 3 Clinical Events
Program
Indication
Event
2025
2026
Olezarsen
sHTG
CORE, CORE2 and Essence data
•
Zilganersen
Alexander disease
Phase 3 data
•
ION582
Angelman syndrome
Phase 3 study start
•
Phase 3 enrollment completion
•
Pelacarsen
Lp(a)-CVD
Lp(a) HORIZON data
•
Bepirovirsen
HBV
B-Well data
•
Eplontersen
ATTR-CM
CARDIO-TTRansform data
•
Sefaxersen
IgAN
IMAGINATION data
•
Ulefnersen
FUS-ALS
FUSION data
•
Timing expectations based on current assumptions and subject to change.
•
Indicates that the milestone is anticipated in the respective year.
View source version on businesswire.com: https://www.businesswire.com/news/home/20250430859857/en/
Ionis Investor Contact: D. Wade Walke , Ph.D. IR@ionis.com 760-603-2331
Ionis Media Contact: Hayley Soffer media@ionis.com 760-603-4679
Source: Ionis Pharmaceuticals, Inc.
临床结果上市批准临床3期财报
2025-04-11
IDF 2025微专辑扫描二维码可查看更多内容编者按:4月7~10日,IDF 2025世界糖尿病大会在泰国曼谷盛大召开。在本次极具影响力的国际学术盛会上,东南大学糖尿病研究所孙子林教授研究团队带来6项前沿研究成果。系列研究围绕糖尿病及相关并发症展开多维度探索:新兴指标空腹血糖差值、体重调整腰围指数在糖尿病人群中的预测价值、血小板衍生生长因子(PDGF)在胰岛纤维化进程中的作用机制、血清视黄醇水平与糖尿病及合并症之间的关联,以及裸眼视力与糖尿病发病风险的关联、老年人握力与DR的性别特异性关联分析。本刊特邀孙子林教授及5位团队核心成员做客“遇见IDF·每日精粹”直播间,系统介绍研究过程、成果及临床意义,并从临床应用和未来研究方向等角度对研究进行深度点评。(一)使用1,5-AG和HbA1c计算的FPG差值作为糖尿病及糖尿病前期患者糖尿病视网膜病变发生的预测指标BA2025-1474作者:侯乐乐,周潇滢,邱山虎,孙子林研究介绍者:侯乐乐01背景糖尿病视网膜病变(DR)是糖尿病常见且严重的微血管并发症,与血糖波动密切相关。近年来,“血糖差”(glucose gap)的概念逐渐受到关注,其定义为空腹血浆葡萄糖(FPG)与由糖化血红蛋白(HbA1c)或1,5-脱水葡萄糖醇(1,5-AG)推算的平均血糖(eAG)之间的差值,被认为是血糖波动的潜在标志物。02目的本研究旨在探讨使用HbA1c和1,5-AG计算的空腹血糖差(FPG-GAP)与新发糖尿病视网膜病变(NDDR)风险之间的关联,并分析其在糖尿病及糖尿病前期人群中的预测价值。03方法对糖尿病及糖尿病前期患者进行随访,评估NDDR发生情况。FPG-GAP定义为FPG与HbA1c计算的eAG或1,5-AG之间的差值。采用限制性立方样条(RCS)分析FPG-GAP与NDDR风险之间的非线性关系。研究根据FPG-GAP=0作为拐点进行数据分层,并分别对FPG-GAP<0和FPG-GAP≥0组进行分段回归分析。04结果研究发现,无论FPG-GAP为正或负偏离0,均与NDDR风险增加相关(见图1)。具体而言,在1,5-AG计算的FPG-GAP≥0组中,FPG-GAP每增加1 mmol/L,NDDR风险增加约32%。HbA1c计算的FPG-GAP呈现U型关系,提示低血糖和高血糖均可能增加NDDR风险。调整年龄、性别、收缩压、体重指数(BMI)和甘油三酯等混杂因素后,这些相关性仍然显著。图1. (A)和(B)分别展示了基于1,5-AG和HbA1c计算的FPG-GAP与NDDR风险的RCS函数分析结果05结论FPG-GAP偏离0显著增加NDDR发生风险,提示其可能是血糖波动的标志物,并可作为预测NDDR风险的潜在指标。专家点评孙子林 教授东南大学糖尿病研究所近年来,基于传统糖代谢指标派生的新型评估指标为糖尿病研究注入创新活力,但其实用性仍有待验证。HbA1c和1.5AG是反映阶段血糖的重要指标,前者反映2~3个月平均血糖,后者反映过去一周平均血糖及波动,二者各有优劣,例如HbA1c无法捕捉血糖波动情况。连续血糖监测(CGM)作为新兴的血糖监测手段,虽能实时追踪血糖变化,但因佩戴不便、成本高,难以完全取代传统指标。在这项研究中,1,5-AG和HbA1c计算的FPG差值可有效反映血糖波动,或能成为预测DR等微血管病变发生发展的潜在指标。不过在临床应用中,1.5AG检测易受药物等因素干扰,而HbA1c控制目标因个体差异尚无统一标准。未来仍需更多循证研究,以推动这些派生指标在临床中的科学应用与精准转化。 (二)利用单细胞测序技术探究胰岛巨噬细胞与星状细胞的相互作用BA2025-1158作者:过妍,王焕,王倩倩,蔡珍生,刘钰,邱山虎,孙子林研究介绍者:过妍01背景胰岛纤维化是1型和2型糖尿病的终末期改变。糖尿病患者胰岛纤维化可影响胰岛素分泌,加速疾病进展。血小板衍生生长因子(platelet derived growth factor,PDGF)是巨噬细胞分泌的一种重要的有丝分裂因子,在机体损伤时促进细胞增殖和迁移。PDGF/PDGFR通路调节多器官纤维化,是纤维化疾病的重要介质之一。然而,PDGF/PDGFR在促进胰岛纤维化中的作用和机制尚不清楚。02目的探讨高脂肪饮食影响下小鼠胰岛巨噬细胞和星状细胞之间的相互作用。03方法正常饮食和高脂饮食小鼠中的单细胞数据经过质量控制和标准化处理后导入Seurat R 工具包。分别对T细胞、B细胞、巨噬细胞和单核细胞进行免疫细胞注释聚类,并对胰岛星状细胞进行聚类和降维处理。使用CellChatDB.mouse分析免疫细胞与胰岛星状细胞之间的相互作用。04结果将免疫细胞分为巨噬细胞、单核细胞、B细胞和T细胞。将星状细胞分为成纤维细胞样星状细胞和其他类型星状细胞。小鼠巨噬细胞和星状细胞之间显示出显著的相互作用,巨噬细胞可以影响星状细胞PDGFR,促进细胞增殖。05结论高脂小鼠胰岛巨噬细胞释放PDGF,促进胰岛星状细胞增殖、迁移和胰岛纤维化。专家点评孙子林 教授东南大学糖尿病研究所我们团队一直关注胰岛纤维化的病理生理过程,以及其在糖尿病发生发展中的作用。1型和2型糖尿病晚期均存在胰岛纤维化特征。既往研究证实,胰岛星状细胞活化可促进胰岛纤维化,而解决该细胞活化问题或可为糖尿病治疗提供潜在靶点。该项研究进一步深化了这一认知:其一,首次应用单细胞测序技术,证实胰岛星状细胞存在亚群,发现其中具有促纤维化的细胞亚群,解释了该细胞从生理功能向促纤维化病理状态的转变;其二,将胰岛星状细胞活化的细胞学与分子机制相结合,从巨噬细胞与星状细胞相互作用角度展开研究,发现巨噬细胞既能促进星状细胞活化引发纤维化,又能加剧胰岛炎症,双重作用破坏胰岛β细胞功能,这有助于我们更全面理解胰岛功能受损及衰竭过程。(三)中国糖尿病人群中体重调整腰围指数的价值:一项横断面和前瞻性队列研究BA2025-0457作者:王焕,过妍,蔡珍生,王倩倩,邱山虎,孙子林研究介绍者:王焕01背景与目的肥胖是糖尿病的主要风险因素。传统肥胖指标如BMI应用广泛,但无法区分脂肪分布。而腰围(WC)能更准确地反映腹部脂肪堆积情况。近期,体重调整腰围指数(WWI)作为一种通过腰围除以体重平方根计算得出的指标,被引入以更精确地衡量肥胖对代谢健康的影响。本研究旨在探讨WWI与中国人群糖尿病风险的关系,并评估其在糖尿病诊断中的潜力。02方法本研究采用横断面和前瞻性队列设计,数据来源于中国多个省份的常住居民。横断面分析研究了WWI与糖尿病患病率之间的关联,而队列研究则评估了WWI预测糖尿病发展的能力。研究人群为年龄20至70岁的居民,排除既往诊断为糖尿病或其他严重疾病的患者。测量身高、体重、腰围。通过空腹血糖(FPG)、2小时餐后血糖(2h-PG)、HbA1c诊断糖尿病,并计算WWI[WWI=腰围(cm)/√体重(kg)]。采用逻辑回归分析相关关系,通过ROC曲线评估筛查和预测效果。03结果WWI越高,糖尿病风险越大,且WWI与FPG、2h-PG和HbA1c呈强相关性。在调整年龄、性别、吸烟状况、饮酒状况和基线BMI后,结果显示WWI与糖尿病之间存在显著的非线性关系(总体P值<0.001,非线性P值=0.055)。此外,WWI在预测糖尿病方面的AUC(曲线下面积)显著优于BMI,且与FPG结合使用时,AUC达到0.80。04结论WWI能有效反映中心型肥胖,与糖尿病患病率和发病率密切相关。WWI越高,糖尿病风险显著增加。其预测能力在不同人群中均表现一致,使其成为糖尿病筛查和预测的有前景的工具。专家点评孙子林 教授东南大学糖尿病研究所我国推出为期3年的“体重管理年”,但体重管理并非单纯减重,需规避单纯减重误区。传统体重及BMI并不能揭示肥胖的本质,而更科学的体重调整腰围指数等简易指标,能精准反映中心性肥胖与疾病关联,值得在全球尤其是不发达地区推广。此外,中西方糖尿病特征存在差异,中国人群胰岛β细胞功能缺陷出现早、更易受代谢损伤,加之饮食偏好碳水且胃排空更快,导致餐后高血糖比例高,因此糖尿病筛查与治疗,包括药物选择及生活方式管理等,需结合这些特点制定个性化方案。 (四)血清维生素A(视黄醇)水平与糖尿病风险及管理之间的相关性研究BA2025-1322作者:王焕,过妍,蔡珍生,王倩倩,邱山虎,孙子林研究介绍者:王焕01背景与目的尽管众多研究已探讨了影响糖尿病风险的各种因素,但维生素A在糖尿病发病和进展中的具体作用仍不明确。当前研究主要关注视黄醇结合蛋白4(RBP4)与糖脂代谢的关系,往往忽视了视黄醇本身的直接作用。此外,RBP4水平可能受炎症等因素影响,导致其变化不一致。本研究旨在专门探讨血清视黄醇与糖尿病及其合并症之间的关联。02方法本研究为回顾性分析,以确诊的2型糖尿病(T2DM)患者作为病例组,并匹配健康个体作为对照组。收集参与者的基本信息,并使用高效液相色谱-质谱法(HPLC-MS)测量血清视黄醇及其相关代谢产物。同时记录糖尿病患者的血糖指标,以探索血清视黄醇水平、糖尿病风险及潜在合并症之间的关系。03结果结果显示,T2DM患者的血清视黄醇水平显著高于对照组。进一步分析发现,视黄醇水平升高是糖尿病的独立风险因素,视黄醇水平越高,糖尿病风险越大。此外,研究还发现血清视黄醇水平升高与高尿酸水平之间存在显著正相关,且在调整年龄、体重和HbA1c后,这种相关性仍然高度显著。最高视黄醇四分位数的高尿酸血症风险最高(β=89.63,P<0.001,95%CI:62.54~116.71,与最低四分位数相比)。尽管直接因果关系需要进一步研究,但本研究发现血清视黄醇水平较高的患者更容易发生并发症。此外,视黄醇在识别糖尿病患者高尿酸血症方面表现出显著较高的曲线下面积(AUC=0.79)。04结论本研究揭示了血清视黄醇水平与糖尿病风险及管理之间的密切关系,强调了视黄醇本身在糖尿病发病机理中的重要作用。它填补了维生素A及其代谢产物与糖尿病关系理解上的空白,为T2DM的治疗提供了新的见解。专家点评孙子林 教授东南大学糖尿病研究所我们团队在研究胰岛星状细胞活化与糖尿病关系时,受限于人体实验,只能依赖动物实验,研究比较受限。随着HPLC质谱技术突破检测瓶颈,我们尝试以循环维生素A为替代指标,探索其与糖尿病的关联,并与胰岛星状细胞活化研究相结合,试图为研究开辟新路径。这一研究亦契合“病从口入,亦可病从口出”理念,测量维生素A指标或能为精准饮食干预提供指引。以维生素A缺乏引发夜盲症为例,佐证了营养素补充对健康的重要性。未来可通过营养补充剂干预研究,探索其在糖尿病一级、二级预防及代谢指标控制中的价值。尽管背后机制比较复杂,但通过这些临床现象观察,有望为疾病防控提供新方向。(五)裸眼视力与糖尿病发病风险的关联性分析:一项基于中介效应分析的前瞻性队列研究BA2025-0229作者:王文娟,朱一鸣,周潇莹,胡适,董力榕,丁瑜芝,王多劳,邱山虎,孙子林研究介绍者:王文娟01背景糖尿病与视力下降存在显著相关性,其病理进程在合并DR的患者中尤为突出。然而,目前尚未有前瞻性队列研究阐明裸眼视力下降是否可以作为糖尿病发生的危险因素。02方法本研究基于前瞻性队列设计,纳入5223例基线无糖尿病的中国成年人,进行为期3年的随访。采用国际标准视力表检测双眼裸眼视力,并进行最小分辨角对数转换进行量化分析。糖尿病诊断根据美国糖尿病协会(ADA)标准。以糖尿病确诊时间或随访时间作为生存时间(月),进行Cox比例风险回归分析和中介效应分析。03结果随访过程中,共观察到536例糖尿病新发病例,糖尿病组参与者视力显著低于非糖尿病组(0.10±0.20 vs. 0.08±0.31 logMAR;P=0.017)。Cox回归分析显示:相较于最佳视力组,最低视力组的糖尿病风险比为1.64(95%CI:1.22~2.21)。亚组分析显示视力下降与糖尿病发生风险不受性别、体重状态、生活方式因素等影响(所有Pinteraction≤0.14)。中介效应分析提示收缩压、甘油三酯-葡萄糖指数可能介导了视力下降与糖尿病发生风险的关联,其介导效应分别为31.9%、9.2%。04结论中国成人视力下降与糖尿病发生风险增加有关,而收缩压升高及胰岛素抵抗可能是视力下降引起糖尿病发生风险增加的重要原因。专家点评孙子林 教授东南大学糖尿病研究所通过该项研究,我们发现不同于传统认知,视力不好或许是糖尿病发生的诱因,而非糖尿病导致视力下降。中介效应分析显示,血压、血脂、胰岛素抵抗指数等因素在其中发挥作用。这可能与维生素A缺乏等致视力下降的病因,和糖尿病存在共同病理生理机制有关。此外,视力不佳者因活动少(如儿童因看书多近视、体力活动少)或运动困难易导致肥胖,进而引发血压异常、胰岛抵抗与血糖升高。该研究提醒,加强儿童近视防控,或能同步改善糖尿病发病风险,为糖尿病预防提供新思路,但还需更多数据验证其实际效果。 (六)老年人群握力与糖尿病性视网膜病的性别特异性关联:一项横断面研究BA2025-1184作者:顾金金,邱山虎,徐瑛,丁瑜芝,周潇滢,刘琰,袁扬,孙子林研究介绍者:顾金金01目的本研究旨在探讨握力(HGS)与DR之间的关联,重点关注性别特异性差异。02方法本次横断面研究于2020年4月至2021年1月开展,纳入来自中国八个省份392例老年糖尿病患者(≥60岁)。通过数字液压测力计测量HGS,并根据早期DR研究(ETDRS)标准对DR进行分类。采用逻辑回归分析低HGS(男性<28 kg,女性<18 kg)与DR之间的关系,并控制多个协变量。03结果男性DR的患病率显著高于女性(男性18.99%,女性13.25%)。在男性中,低HGS与DR的发生率显著相关(比值比:6.03,95%CI:1.74~20.89,P=0.0045),该结果在调整年龄、民族、社会经济因素、心血管健康、血糖控制、肾功能和脂质谱后仍然显著。在女性中未发现显著关联,敏感性分析结果一致。04结论低HGS与中国老年男性DR风险的增加独立相关,但在女性中未见相关性。提示在评估老年糖尿病群体中HGS对DR风险影响时需要考虑性别差异。专家点评孙子林 教授东南大学糖尿病研究所肌肉力量与糖尿病视网膜病变的关联看似“不搭”,但其背后的潜在联系值得深入探讨。从代谢层面看,肌肉力量差往往意味着运动和体力活动少,易引发肥胖、高血糖等代谢性疾病,而这些疾病会增加视网膜病变风险;从肌肉内分泌学角度,肌肉分泌的多种因子(如肌肉因子、运动因子)中,可能存在影响视网膜健康的“好因子”与“坏因子”,部分因子或许会间接影响血管内皮生长因子(VEGF)等视网膜病变相关指标,比如血管内皮生长因子(VEGF)水平,这些都需要进一步深入探究。该研究为横断面研究,只能表明二者存在相关性,无法确定因果关系。此外,视网膜病变或提示全身微血管病变,肌肉是否存在微血管问题也值得进一步研究。后续可通过文献研究挖掘潜在联系,助力糖网筛查新方向。 专家简介孙子林 教授东南大学二级教授、特聘教授、主任医师、博士生导师、国家重点研发计划首席科学家东南大学糖尿病研究所所长、江苏省医学领军人才、全国优秀科技工作者中国微循环学会副理事长兼秘书长中国微循环学会糖尿病与微循环专业委员会主任委员中国卫生信息和健康医疗大数据学会糖尿病专业委员会主任委员中国健康促进与教育学会糖尿病教育与管理分会副主任委员中华医学会内分泌分会委员江苏省医师协会内分泌与代谢科分会会长担任BMC medicine、Diabetes and Metabolism Journal、《中华医学杂志》、《中华糖尿病杂志》、《中国糖尿病杂志》、《中华健康管理杂志》、《中国全科医学》等杂志编委主要研究方向:糖尿病微血管并发症发病机制和防治策略,糖尿病胰岛纤维化的细胞和分子机制,糖尿病筛查、教育和管理模式研究2025 IDF现场声明:本文仅供医疗卫生专业人士了解最新医药资讯参考使用,不代表本平台观点。该等信息不能以任何方式取代专业的医疗指导,也不应被视为诊疗建议,如果该信息被用于资讯以外的目的,本站及作者不承担相关责任。最新《国际糖尿病》读者专属微信交流群建好了,快快加入吧!扫描左边《国际糖尿病》小助手二维码(微信号:guojitnb),回复“国际糖尿病读者”,ta会尽快拉您入群滴!(来源:《国际糖尿病》编辑部)版权声明版权属《国际糖尿病》所有。欢迎个人转发分享。其他任何媒体、网站未经授权,禁止转载。
临床结果临床研究申请上市
2025-03-28
CAMBRIDGE, Mass. and MUMBAI, India, March 28, 2025 /PRNewswire/ -- Particles for Humanity and Hexagon Nutrition Limited are excited to announce their new partnership to help address vitamin A deficiency (VAD) in Africa. Hexagon Nutrition has licensed the rights to manufacture and distribute Particles for Humanity's lead product, PFH-VAP, for fortifying bouillon cubes across Africa. PFH-VAP, a more stable formulation of dry vitamin A palmitate, is designed for use in large scale food fortification in geographies with hot and humid climates. Particles for Humanity partnered with Hexagon because of their shared commitment to ensuring all people have access to adequate nutrition to grow and thrive.
"By joining forces with Hexagon Nutrition, we can accelerate the production and distribution of this critical product, ensuring it reaches those who need vitamin A the most. Combining Particles for Humanity's research and development expertise with Hexagon's established global distribution network advances a shared goal: improving lives with stable vitamin A," said Sherri Oberg, CEO at Particles for Humanity.
In West Africa, bouillon is consumed by 80% to 99% of all households. In many of these countries, bouillon consumption remains high in the lowest income communities, where VAD is most prevalent. Children without enough vitamin A have decreased rates of growth and development and an increased risk of infection and death from serious illness. Vitamin A is also a leading cause of night blindness in women and can become permanent in severe cases. Though widespread consumption makes bouillon an ideal vehicle for combatting VAD, commercial forms of vitamin A often degrade in hot and humid conditions like those found in parts of West Africa. PFH-VAP was designed to address this issue. As a result, fortifying bouillon with PFH-VAP could save thousands of child-lives annually and improve the lives of millions of children and women in Nigeria alone.
Under this new partnership, Hexagon Nutrition will have 3-year exclusive rights to manufacture and distribute PFH-VAP for bouillon fortification. Together Hexagon Nutrition and Particles for Humanity are taking an important step to expand access to lifesaving micronutrients in Africa.
"Improving nutrition globally is at the core of our business. PFH-VAP will be another strong asset in our product portfolio. We are excited to leverage Hexagon Nutrition's ingredient manufacturing and premixing expertise to advance innovation and reduce the burden of malnutrition," said Vikram Kelkar, Managing Director of Hexagon Nutrition Limited.
Particles for Humanity transforms early-stage medical technology into products for people living in low and lower-middle income countries. Its rigorous product development process is based on end-user input. Particles for Humanity is funded solely with philanthropy and is focused on financially sustainable product opportunities. Visit or contact [email protected]
Hexagon Nutrition is a differentiated and research-oriented pure-play nutrition company offering clinical nutrition solutions products, micronutrient premixes, and therapeutic products. Hexagon Nutrition has presence across retail pharmacies, hospitals, and prominent e-commerce players. Hexagon markets products across India and exports to more than 70 countries.
SOURCE Particles for Humanity
WANT YOUR COMPANY'S NEWS FEATURED ON PRNEWSWIRE.COM?
440k+
Newsrooms &
Influencers
9k+
Digital Media
Outlets
270k+
Journalists
Opted In
GET STARTED
引进/卖出
分析
对领域进行一次全面的分析。
登录
或
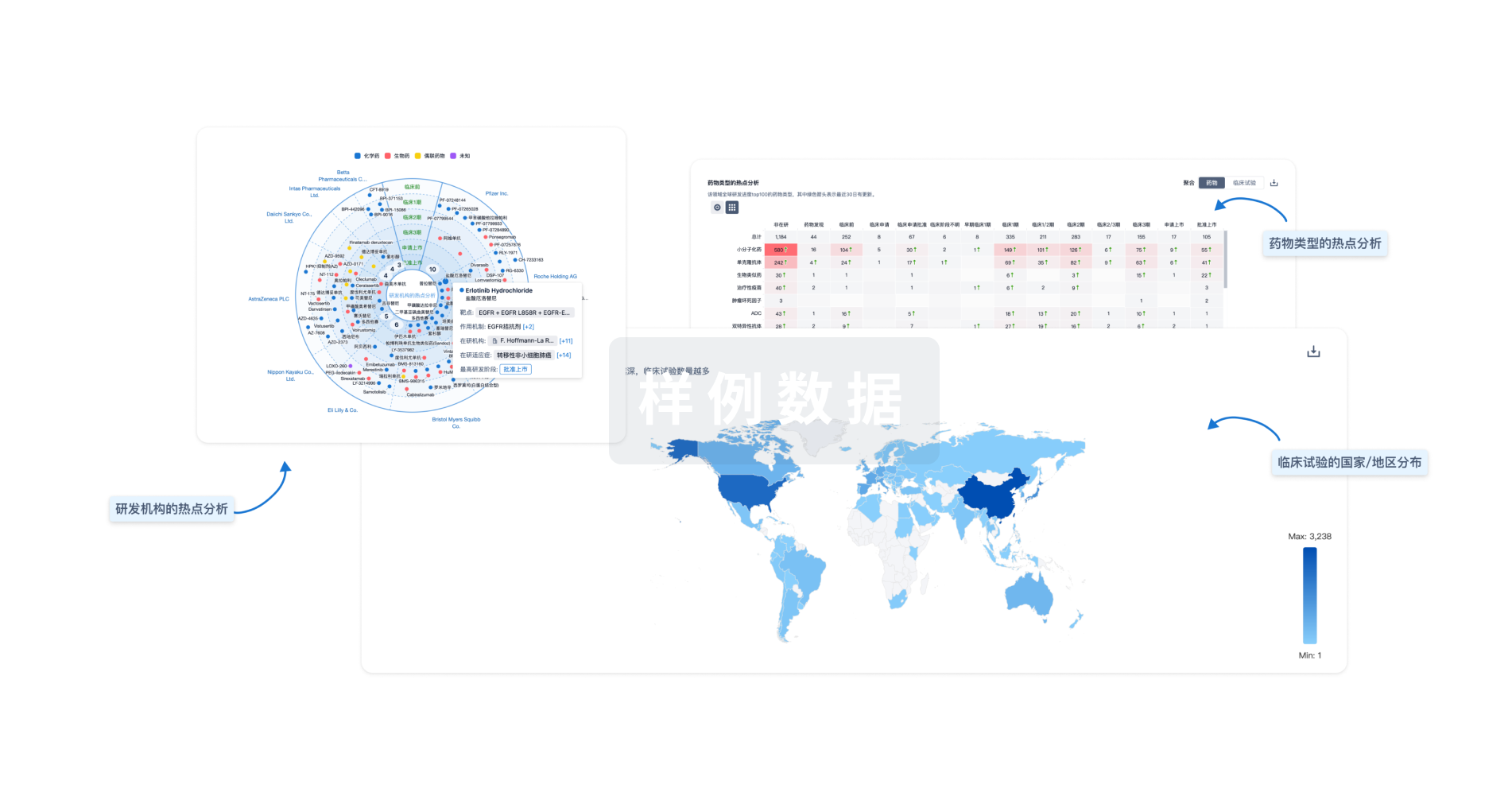
生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用





