预约演示
更新于:2025-05-07
Bacterial respiratory infection
细菌性呼吸道感染
更新于:2025-05-07
基本信息
别名 Bacterial respiratory infection、Bacterial respiratory infection (disorder)、bacterial infections respiratory + [3] |
简介- |
关联
3
项与 细菌性呼吸道感染 相关的药物靶点 |
作用机制 DprE1抑制剂 [+1] |
非在研适应症 |
最高研发阶段临床2期 |
首次获批国家/地区- |
首次获批日期1800-01-20 |
靶点- |
作用机制- |
在研机构- |
在研适应症- |
最高研发阶段无进展 |
首次获批国家/地区- |
首次获批日期1800-01-20 |
16
项与 细菌性呼吸道感染 相关的临床试验NCT06188988
The Interaction Between Viral Infections and the Development of the Airway Microbiome in Young Children With Cystic Fibrosis
Cystic fibrosis (CF) is the most common hereditary life-threatening condition in Belgium. Because of a dysfunctional cystic fibrosis transmembrane conductance regulator (CFTR) channel, chloride is unable to move to the cell surface and mucus becomes more viscous. Consequently, CF patients are not able to clear their lungs efficiently, and trapped bacteria can lead to chronic infection and inflammation of the lungs, and ultimately respiratory failure.
CF lung disease starts at birth due to muco-inflammatory processes and is associated with a significantly altered microbial colonization of the infant airways compared to infants without CF. Additionally, young children with CF suffer from viral infections as often as their healthy peers, but the episodes are more severe and often prolonged. Moreover, frequent viral infections in children with CF contribute towards a more pathogenic airway microbiome at a young age. Although this link has been previously reported, the exact mechanisms by which this occurs need to be elucidated.
A pulmonary exacerbation in CF is characterized by an increase in respiratory symptoms, general symptoms and a decline in lung function. Most young children with CF suffer from a mean of 4 exacerbations per year for which antibiotics are prescribed. Despite the current novel therapies in CF, treatment of respiratory infections stay relevant and is a greater challenge with increasing survival.
The key objective of this study is to gain insights into the mechanisms by which viral infections leading to pulmonary exacerbations induce a more pathogenic microbiome in young children with CF.
About forty participants will be recruited at the paediatric CF clinic of the Antwerp University Hospital. Inclusion criteria are an age of less than 5 years and a diagnosis of CF. There are no exclusion criteria. Duration of the study is 1 year to cover for seasonality of clinical symptoms. Study visits are scheduled at 3-month intervals corresponding with the regular follow up, or unscheduled during an acute pulmonary exacerbation. From all participants, two oropharyngeal swabs (for microbiome analysis and for immunological/mucin analysis) will be collected at set time points. For the linking of the laboratory data to the clinical characteristics, we will examine demographics, environmental exposures, and disease markers of CF. Next to the collection of the oropharyngeal swabs, a history, physical examination, and technical investigations will be performed at the study visits.
CF lung disease starts at birth due to muco-inflammatory processes and is associated with a significantly altered microbial colonization of the infant airways compared to infants without CF. Additionally, young children with CF suffer from viral infections as often as their healthy peers, but the episodes are more severe and often prolonged. Moreover, frequent viral infections in children with CF contribute towards a more pathogenic airway microbiome at a young age. Although this link has been previously reported, the exact mechanisms by which this occurs need to be elucidated.
A pulmonary exacerbation in CF is characterized by an increase in respiratory symptoms, general symptoms and a decline in lung function. Most young children with CF suffer from a mean of 4 exacerbations per year for which antibiotics are prescribed. Despite the current novel therapies in CF, treatment of respiratory infections stay relevant and is a greater challenge with increasing survival.
The key objective of this study is to gain insights into the mechanisms by which viral infections leading to pulmonary exacerbations induce a more pathogenic microbiome in young children with CF.
About forty participants will be recruited at the paediatric CF clinic of the Antwerp University Hospital. Inclusion criteria are an age of less than 5 years and a diagnosis of CF. There are no exclusion criteria. Duration of the study is 1 year to cover for seasonality of clinical symptoms. Study visits are scheduled at 3-month intervals corresponding with the regular follow up, or unscheduled during an acute pulmonary exacerbation. From all participants, two oropharyngeal swabs (for microbiome analysis and for immunological/mucin analysis) will be collected at set time points. For the linking of the laboratory data to the clinical characteristics, we will examine demographics, environmental exposures, and disease markers of CF. Next to the collection of the oropharyngeal swabs, a history, physical examination, and technical investigations will be performed at the study visits.
开始日期2023-11-01 |
申办/合作机构- |
NCT05706935
Evaluation of the MeMed BV® Test in Adult Emergency Department Patients With Fever and Acute Respiratory Symptoms
The goal of this observational study is to compare the results of a serum biomarker called the MeMed BV®, which is approved to help differentiate between bacterial and viral respiratory infections, to clinical diagnoses of adult emergency department patients presenting with recent fever and signs or symptoms of a respiratory infection. Active participation is completed during the emergency department visit and includes drawing blood, obtaining a sputum sample, and answering survey questions.
开始日期2023-02-06 |
NCT05712330
Exploratory Study of Respiratory Bacterial Infections or Superinfections and Colonizations in Patients With Spinal Muscular Atrophy Under NIV
SMA (Spinal Muscular Atrophy) is a rare neuromuscular disease characterized by motoneuron damage. Symptoms consist of respiratory involvement with numerous respiratory infections and eventually respiratory failure, for which NIV (Non Invasive Ventilation) is often used. Ventilation machines are in close contact with the respiratory tract of patients. They contain heated water to humidify the circuit. These humid and warm environments are conducive to the development of bacteria such as Pseudomonas aeruginosa. In this context, it is interesting to look for the presence or absence of bacteria, in comparison with the respiratory ecology of the patients. The aim is to highlight the microbiological role of NIV on the occurrence of respiratory bacterial infections or secondary infections in patients with SMA. To do this, samples are taken from the machines, and ECBCs are performed on patients during respiratory physiotherapy sessions.
开始日期2023-01-24 |
100 项与 细菌性呼吸道感染 相关的临床结果
登录后查看更多信息
100 项与 细菌性呼吸道感染 相关的转化医学
登录后查看更多信息
0 项与 细菌性呼吸道感染 相关的专利(医药)
登录后查看更多信息
667
项与 细菌性呼吸道感染 相关的文献(医药)2025-05-01·Microbial Pathogenesis
Scutellaria baicalensis stem and leaf combat chicken-derived respiratory bacterial infection
Article
作者: Wang, Xinghai ; Wang, Xilin ; Dong, Hong ; Yu, Feike ; Yu, Xiaohan ; Guo, Ning ; Liu, Xiaoye ; Wu, Shihai ; Xu, Xiaofeng
2025-04-01·Infection
Can nCD64 and mCD169 biomarkers improve the diagnosis of viral and bacterial respiratory syndromes in the emergency department? A prospective cohort pilot study
Article
作者: Pivetta, Eliana ; Pontoni, Elisa ; Zanus-Fortes, Agnese ; Arcidiacono, Domenico ; De Rosa, Rita ; Bove, Tiziana ; Pellis, Tommaso ; Tascini, Carlo ; Avolio, Manuela ; Cugini, Francesco ; Valentini, Giada ; Sagnelli, Vincenzo ; Venturini, Sergio ; Fabro, Giovanni Del ; Santin, Veronica ; Reffo, Ingrid ; De Santi, Laura ; Basaglia, Giancarlo ; Tedeschi, Rosamaria ; Marangone, Anna ; Pratesi, Chiara ; Bramuzzo, Igor ; Vattamattahil, Kathreena ; Crapis, Massimo ; Callegari, Astrid ; Ziraldo, Barbra ; Doretto, Paolo ; Orso, Daniele
2025-03-04·Microbiology Spectrum
Comparing sputum, nasopharyngeal swabs, and combined samples for respiratory bacterial detection using multiplex PCR
Article
作者: Cho, Yunjung ; Nam, Myung-Hyun ; Yun, Seung Gyu ; Kim, Keun Ju ; Lee, Chang Kyu
12
项与 细菌性呼吸道感染 相关的新闻(医药)2024-10-29
IND2025主会场授课已被预定,大会第一天下午茶歇前授课时间段预定开始,咨询热线:177 0186 0390。点击图片,查看IND2024会后报告
文章来源:分子诊断实验室
儿童呼吸道感染病原包括病毒、细菌、真菌等。《儿童呼吸道感染病原体核酸检测专家共识》作为全球首个以儿童为主、涵盖全面的呼吸道感染病原学诊断应用标准,对这些病原体作了详细全面的介绍。
常见呼吸道病毒
病毒是儿童呼吸道感染的主要病原体,儿童呼吸道感染病毒种类至少涉及7个病毒科共200多个型别的病毒。
1.1 人呼吸道合胞病毒(human respiratory syncytial virus,HRSV)
HRSV是全球引起5岁以下儿童急性下呼吸道感染(acute lower respiratory tract infection,ALRTI)最重要的病毒病原体,占所有儿童ALRTI的22%。据估计,全球每年大约有270万~380万5岁以下儿童因HRSV感染住院,引起约4.8万~7.5万人死亡。
HRSV属于肺炎病毒科,正肺病毒属,为非节段性单股负链RNA病毒。目前,ON1和BA9基因型为我国及全球很多国家的优势流行基因型。
HRSV感染呈全球广泛流行,其流行受地理位置、温度和湿度等因素影响。在北半球国家和地区,HRSV的流行存在明显的流行季,主要集中于11月至次年2月的冬季和早春季节;在热带和亚热带,HRSV在潮湿的雨季感染率出现明显增高。我国北方地区HRSV流行季开始于第41周(10月中旬),结束于次年第20周(5月中旬),持续33周。
HRSV感染在不同年龄人群临床表现差异很大,毛细支气管炎和肺炎是婴幼儿HRSV感染的重要疾病,严重者可发生呼吸衰竭,甚至死亡,年长儿HRSV感染可表现为症状轻微的上呼吸道感染或中耳炎。
1.2 人流感病毒(human influenza virus,HIFV)
HIFV是急性呼吸道感染的重要病原,全人群人普遍易感,儿童和老年人感染后危害最为严重,每年可引起季节性流行,亦可引起暴发疫情。
HIFV可分为甲、乙、丙,共3型。甲型流感病毒依据其血凝素(hemagglutinin,HN)和神经氨酸酶(neuraminidase,NA)的亚型组合可进一步分为多种亚型,目前HN有18个亚型(H1~H18),NA有11个亚型(N1~N11)。乙型流感病毒根据HA的抗原性可分为Victoria和Yamagata两个种系。引起季节性流行的HIFV主要有甲型H1N1、H3N2亚型和乙型的Victoria和Yamagata系4个亚型HIFV。
HIFV在全球呈广泛流行,但不同地区的HIFV型别和流行规模有所不同。我国甲型HIFV在北方省份以冬季为流行高峰,南方省份每年4-6月份出现流行高峰,而中部省份则呈现每年1-2月和6-8月双流行峰。
HIFV感染后引起的临床表现轻重不一,包括无症状感染、上下呼吸道感染,重症病例可以迅速进展为肺炎,亦可并发心肌炎、脑膜炎或脑炎,甚至危及生命。
1.3 人副流感病毒(human parainfluenza virus,HPIVs)
HPIVs主要导致儿童和青少年呼吸道感染性疾病,是仅次于HRSV的第二大引起儿童呼吸道感染住院的病原体。
HPIVs属于副黏病毒科(Paramyxoviridae),有4个血清型(HPIV1、2、3和4),HPIV1和HPIV3属于呼吸道病毒属(Respirovirus genus),HPIV2和HPIV4属于腮腺炎病毒属(Rubulavirus genus)。通常HPIVs的流行是散发的,无明显的季节性。
HPIVs感染可引起鼻炎、咽炎、喉炎、气管支气管炎、细支气管炎和肺炎等,也可引发鼻窦炎和中耳炎。
1.4 人鼻病毒(human rhinovirus,HRV)
HRV长期以来被认为是"普通感冒"的主要病原体,主要引起上呼吸道感染。据统计,每年有30%~50%的呼吸道感染疾病与HRV感染有关。儿童是HRV的易感人群,据报道,有60%~80%在急诊科就诊的哮喘发作患儿伴有HRV感染,在3岁以下被诊断为毛细支气管炎的住院儿童病例中,有大约50%的婴儿感染或合并感染HRV,而几乎所有婴儿在出生1年后均会经历至少1次的HRV感染。
HRV属小核糖核酸RNA病毒科,肠道病毒属。截至目前已经发现了A、B、C 3个种共169个型别。HRV在全球范围内流行,一年四季均可发病。
HRV感染后主要表现为普通感冒症状,可引起喘息和哮喘急性加重。引起婴幼儿的支气管炎和毛细支气管炎。
1.5 人腺病毒(human adenovirus,HAdV)
HAdV可分为7个种(A~G)和至少104个基因型,其中与呼吸道感染相关的HAdV主要型别为B种(HAdV-3,7,11,14,16,21,50,55),C种(HAdV-1,2,5,6)和E种(HAdV-4)。HAdV-3和HAdV-7型是引起我国儿童呼吸道HAdV感染最常见的血清型,HAdV-55是引起成人HAdV社区型肺炎最常见的型别。
HAdV感染一年四季均可发生,在我国北方以冬春季节多见,南方以春夏季节常见。HAdV传染性较强,常可引起暴发流行。
HAdV感染可表现为上下呼吸道感染。HAdV是儿童重症肺炎的重要病原体,导致危重患者出现休克、呼吸衰竭、弥散性血管内凝血等,甚至导致死亡。
1.6 人偏肺病毒(human metapneumovirus,HMPV)
HMPV是导致儿童呼吸道感染的重要病毒病原之一。在急性呼吸道感染住院患者中,HMPV感染约为6.24%。
2017年国际病毒分类委员会将HMPV种属划分为肺病毒科(Pneumoviridae),偏肺病毒属。根据HMPV F、G蛋白基因序列的差异,将HMPV分为A1、A2a、A2b、A2c、B1和B2共6个基因型。
HMPV在热带地区全年活跃,在温带气候地区,HMPV季节性流行发生在冬季和春季,在3月和4月达到顶峰,其流行高峰与HRSV重叠或稍晚于后者。
HMPV感染患者临床症状与HRSV感染相似,严重者可发展为细支气管炎和肺炎。有研究表明儿童HMPV感染可能与中枢神经系统疾病相关,如癫痫、脑炎等。
1.7人冠状病毒(human coronavirus,HCoV)
HCoV是人类上呼吸道感染常见病原体。普通感冒中有15%~30%是由HCoV引起的。HCoV属于套式病毒目(Nidovirales),冠状病毒科(Coronaviridae),冠状病毒属(Coronavirus),分为4个属:α-HCoV、β-HCoV、γ-HCoV和δ-HCoV。HCoV可分为普通人冠状病毒(HCoV-NL63、HCoV-HUK1、HCoV-OC43、HCoV-N229E)及高致病性冠状病毒[严重急性呼吸综合征(severe acute respiratory syndrome,SARS)-CoV、中东呼吸综合征(Middle East respiratory syndrome,MERS)-CoV和严重急性呼吸综合征冠状病毒2 (severe acute respiratory syndrome coronavirus 2,SARS-CoV-2)等]。
1.7.1普通HCoV
引起儿童呼吸道感染的常见普通HCoV有4种,分别为α属的HCoV-229E、HCoV-NL63,β属的HCoV-OC43、HCoV-HKU1。
HCoV感染呈全年流行。不同型别的HCoV在不同地区流行特点具有差异性,流行高峰随地理区域有所不同,有一定的季节差异。北京地区HCoV-OC43感染多发于春季(4月和5月),HCoV-229E的流行高峰是在2月。在上海地区HCoV-OC43感染多发于夏秋季,HCoV-NL63感染多发生于秋冬季节。在中国香港地区HCoV-OC43感染主要发生在秋冬季,HCoV-HKU1在冬季检出更多,HCoV-229E多发生于冬春季节,HCoV-NL63感染多发生于夏秋季节。
这4种HCoV通常引起上呼吸道感染,多为自限性,但在婴幼儿、老年人或免疫缺陷患者也可引起严重的下呼吸道感染,表现为喘息、气促等。
1.7.2高致病性冠状病毒
SARS是由SARS-CoV感染引起的一种严重的致死性传染病,于2002年11月,在我国广东省首次报道。随后,SARS病例数在我国大幅增加,并在全球蔓延,超过30个国家和地区相继出现病例。2003年7月,世界卫生组织(World Health Organization,WHO)宣布其流行结束。全球SARS病例数达到8 437例,死亡813例,病死率约为10%。SARS-CoV感染起病迅速,常见的早期症状包括发热、畏寒、咳嗽、身体不适、肌肉疼痛和头痛等,可发展为进行性呼吸困难和低氧血症,严重者可造成急性呼吸窘迫综合征(ARDS),患者常因肺部损伤导致呼吸衰竭而死亡。与成人相比,儿童发病率低,且多为散发,患儿一般与SARS成人有密切接触史,未见在学校、托幼机构内流行;临床表现较轻,预后较好,一般没有远期严重并发症,未见死亡病例报道。
MERS是由MERS-CoV引起的一种严重的致死性传染病,于2012年9月在沙特阿拉伯王国首次报道。截至2020年,全球约27个国家报道过该疾病,其中沙特阿拉伯、阿拉伯联合酋长国和韩国疫情最为严重。截至2019年11月30日,全球总计2 494例实验室确诊病例和858例死亡病例。我国于2015年5月29日确诊了首例由韩国输入的病例。MERS-CoV的潜伏期为2~14 d,平均5~7 d,起病急,病情进展迅速;早期主要表现为发热、畏寒、乏力、头痛、肌痛等,随后出现咳嗽、胸痛、呼吸困难,部分病例还可出现呕吐、腹痛、腹泻等症状;重症病例多在7 d内进展为重症肺炎,可发生ARDS、急性肾衰竭,甚至多脏器功能衰竭等危及生命。MERS致死率较高,死亡率约为37%。老年人、免疫力较差或有基础疾病的患者病情较重,病程进展较快,预后较差。儿童发病率低,且大多数为无症状感染,死亡率较低,预后较好。
SARS-CoV-2是引起新型冠状病毒肺炎(coronavirus disease 2019,COVID-19)的病毒病原体。自2020年3月11日,WHO宣布COVID-19大流行,至今已1.5年的时间。目前,全球正处于第3次高峰的高位平台期。截至2021年11月29日,全球累计报告确诊病例超过2.5亿,死亡病例515万多例,病死率约为2.1%。
SARS-CoV-2属于冠状病毒科,β冠状病毒属。依据SARS-CoV-2的流行程度,WHO于2021年2月25日提出了关切变异株(variant of concern,VOC)和关注变异株(variant of interest,VOI)的定义。目前,WHO界定的VOC为4种,分别为Alpha(B.1.1.7及其亚分支)、Beta(B.1.351及其亚分支)、Gamma(P.1及其亚分支)和Delta(B.1.617.2及其亚分支)变异株;VOI为5种,分别为Eta(B.1.525)、Iota(B.1.526)、Kappa(B.1.617.1)、Lambda(C.37和C.37.1)和Mu(B.1.621和B.1.621.1)变异株。目前,Delta变异株为全球优势流行毒株。
SARS-CoV-2主要传播途径为呼吸道飞沫和密切接触传播,接触病毒污染的物品也可造成感染,在相对封闭的环境中暴露于高浓度气溶胶情况下存在经气溶胶传播可能。病毒在宿主外不能繁殖,但可在短期内存活,存活时间主要与湿度、温度和材质有关。在相同的材质下,低温干燥的环境有利于病毒的存活。因此,污染了SARS-CoV-2的冷链产品可通过国际贸易远距离跨国、跨地区和跨大洋传播。
SARS-CoV-2感染的临床表现差异很大,从无症状到死亡。确诊病例临床可分为轻型、普通型、重型、危重型。轻型患者症状很轻微,无肺炎表现;普通型有发热和呼吸道症状,影像有肺炎证据;重症患者气促、缺氧、肺部影像学进展迅速;危重型会出现呼吸衰竭、休克或合并其他脏器衰竭。
1.8人博卡病毒(human bocavirus,HBoV)
HBoV属于细小病毒科,细小病毒亚科,博卡病毒属。目前为止发现4个基因型HBoV(HBoV1~4)。
HBoV所引起的呼吸道感染疾病四季均可发生,季节性不明显。
HBoV呼吸道感染所致临床表现与呼吸道其他病毒相比无特异性,轻者可无症状,或发热、流涕、咳嗽等,重者可致喘息、呼吸困难等,甚至死亡。
1.9人肠道病毒(human enterovirus,HEV)
HEV在急性呼吸道感染中阳性检出率为4.7%~16.2%,EV-D68是公共卫生关注的一种肠道病毒病原体。
HEV属小核糖核酸RNA病毒科,肠道病毒属。基因组结构为单股正链RNA,可分为HEV-A(n=25),HEV-B(n=63)、HEV-C(n=23)和HEV-D(n=5)4组。
HEV全年散发,各月份均有检出。但在急性呼吸道感染中,HEV在我国流行具有明显的季节性,高峰期主要集中在4-7月,年龄分布主要在5岁以下。
HEV大多数血清型对人类可致病,既可表现为无症状或上下呼吸道感染,也可引起包括急性出血性结膜炎、病毒性脑炎、急性迟缓性麻痹、病毒性心肌炎等严重疾病。
常见细菌
引起儿童呼吸道感染的常见细菌有A族链球菌、肺炎链球菌、金黄色葡萄球菌、流感嗜血杆菌、百日咳鲍特菌、大肠埃希菌、肺炎克雷伯菌、结核分枝杆菌、铜绿假单胞菌等。这些常见细菌性呼吸道感染季节性并不明显,可能多见于季节变换的时期。
2.1 A族链球菌
具有β溶血性特点,也称为A组β溶血性链球菌,是致病力最强的链球菌。其主要致病因子包括链球菌M蛋白、溶血素和胞外酶等。在呼吸系统主要引起上呼吸道感染,包括咽扁桃体炎、化脓性中耳炎、鼻窦炎等,较少引起肺炎和胸膜炎。该菌可致链球菌感染后肾小球肾炎和急性风湿热。
2.2 肺炎链球菌
定植在人上呼吸道,可通过飞沫传播,其侵袭力主要为荚膜,溶血毒素(haemolysin,HL)和NA也是其主要致病因子,多引起呼吸道疾病;根据肺炎链球菌表面荚膜多糖抗原的差异,可分为不同血清型,目前已知有100种血清型,目前引起我国儿童感染的主要有19F、19A、23F、14、6A和6B型等;可以引起上下呼吸道感染,是儿童社区获得性肺炎最常见的细菌性病原。WHO建议将肺炎链球菌疫苗纳入婴儿常规免疫计划,对所有<2岁的儿童进行肺炎链球菌疫苗接种,以预防由该菌引起的多种感染。
2.3 流感嗜血杆菌
是一种多形性革兰阴性杆菌,常定植于人上呼吸道,有3种主要抗原成分,型特异性荚膜多糖抗原(M抗原)、型特异性菌体抗原(S抗原)和种特异性菌体抗原(R抗原),分为可分型(有荚膜)和不可分型(无荚膜)两类菌株,有荚膜流感嗜血杆菌分为a、b、c、d、e、f 6个血清型,其中b血清型致病力最强,f型次之。目前全球范围内致病型别以无荚膜型最为常见,是急性中耳炎、急性上颌窦炎、迁延性细菌性支气管炎、肺炎的重要病因。疫苗可以预防b型流感嗜血杆菌疾病。
2.4 金黄色葡萄球菌
金黄色葡萄球菌在显微镜下排列成葡萄串状,无芽孢、鞭毛,大多数无荚膜,常见于皮肤表面和上呼吸道黏膜,约30%的人可在鼻腔,特别是鼻前庭后部携带该细菌;产生并分泌外毒素休克综合征毒素1(toxic shock syndrome toxin-1,TSST-1)和肠毒素,特别是杀白细胞素(panton-valentine leukocidin,PVL),是一种具有强效细胞溶解及炎症活性的打孔毒素,另外HL、表皮剥脱素和耐热核酸酶也是金黄色葡萄球菌的毒力因子。金黄色葡萄球菌致病特点之一是引起化脓,造成组织坏死和脓肿。除了可以引起上呼吸道感染外,还可以导致支气管炎、肺炎、肺脓肿、肺大疱、胸膜炎等。由于广泛使用抗生素,耐药性(mecA)的传播越来越受到医疗机构的重视。
2.5 百日咳鲍特菌
是百日咳的病原体,人类是其唯一宿主,通过呼吸道飞沫传播,传染性强,易感人群主要集中在1岁以下婴儿,其致病物质包括荚膜、菌毛、内毒素和多种生物活性物质。百日咳毒素是主要的致病毒素。细菌附着在上呼吸道的纤毛上,释放毒素,从而损害纤毛并导致气道肿胀。尽管全球范围常年维持较高的疫苗接种率,近一二十年来多个国家地区出现了明显的百日咳再现。百日咳鲍特菌导致肺炎的比例为25%~88%,部分合并肺不张,极少见侵袭致胸膜炎。
2.6 大肠埃希菌
为革兰阴性杆菌,是一大群多样的细菌,大多数位于肠道内的大肠埃希菌为非致病性菌株,部分大肠埃希菌群含有毒力因子,可发生胃肠道外传播及致病;目前基于主要表面抗原可分为约190个血清群,某些种类的大肠埃希菌会导致腹泻,部分种类可致尿路感染,在呼吸系统常致新生儿和小婴儿肺炎,可伴有脓胸,较少致肺脓肿,多在医院环境下发生。
2.7 肺炎克雷伯菌
是肠杆菌科克雷伯杆菌属的一种细菌,属于人类口腔和肠道正常菌群,在致病性克雷伯杆菌中,肺炎克雷伯杆菌感染最为多见,是社区和医院获得性肺炎的重要细菌性病原,尤其是碳青霉烯耐药和高毒力肺炎克雷伯菌,肺部可形成空洞、单个或多个脓腔,与其他细菌比较,并发菌血症机会多。临床实验室分离的肺炎克雷伯菌主要分为3个亚种,分别为肺炎克雷伯菌肺炎亚种、肺炎克雷伯菌硬鼻亚种和肺炎克雷伯菌臭鼻亚种。
2.8 结核分枝杆菌
结核病是由一群关系密切的分枝杆菌引起的,统称为结核分枝杆菌复合群,包含至少13个成员,最常见的为人型、牛型、非洲型及鼠型,对人致病主要为人型,其次为牛型,在呼吸道主要导致原发性肺结核、血型播散性肺结核、支气管内膜结核及胸膜炎等。耐多药结核分枝杆菌是由至少对异烟肼和利福平这两种最有效的结核病药物有抗药性的病菌引起,成为危害人类健康的重要病原。
2.9 铜绿假单胞菌
是一种非发酵革兰阴性杆菌,在自然界分布广泛,各种水、空气、正常人的皮肤、呼吸道和肠道等均有本菌存在。为条件致病菌,是医院内感染的主要病原菌之一。主要的致病因子包括绿脓菌素、脂多糖、弹性蛋白酶和外毒素A,外毒素A是最主要的致病因子,经常引起术后伤口感染,也可引起褥疮、脓肿、化脓性中耳炎等;亦是呼吸机相关性肺炎的常见原因。
非典型病原
儿童呼吸道感染的非典型病原体主要包括肺炎支原体(Mycoplasma pneumoniae,MP)、肺炎衣原体(Chlamydia pneumonia,CP)、沙眼衣原体(Chlamydia trachomatis)及嗜肺军团菌(Legionella pneumophila,LP)。
3.1 支原体
是一类无细胞壁、多形性、可通过滤菌器的原核细胞型微生物。是目前所知的能在无生命培养基中繁殖的最小的微生物。其属于柔膜体纲支原体目下的支原体属。其中MP是呼吸道感染的常见病原体之一。全球感染率达9.6%~66.7%,且有逐年增高的趋势。全年发病,冬季更多见,每3~7年发生一次大流行。MP感染以学龄儿童多见,潜伏期为2~3周,突出表现为阵发性刺激性咳嗽,以夜间为重,咳少量黏痰或黏液脓性痰,有时痰中带血,也可有呼吸困难、胸痛。发热可持续2~3周,体温正常后仍可遗有咳嗽。严重MP感染可出现肺内肺外并发症的重症肺炎。
3.2 衣原体
是一群体积较小,能通过细菌滤器,细胞内专性寄生,并有独特发育周期的原核细胞型微生物,属于衣原体目衣原体科衣原体属。根据衣原体的抗原构造和DNA同源性的特点,将衣原体属分为4个种,包括沙眼衣原体、CP、鹦鹉热衣原体和家畜衣原体。能引起人类致病的主要是前3种。其中沙眼衣原体可分3个生物亚种,可引起结膜炎和婴幼儿肺炎。CP只有1个血清型,即TWAR组,CP传播方式主要是人与人之间经飞沫或呼吸道分泌物传播,常可呈地方性、散发性流行。CP引起的呼吸道感染约70%是无症状的或仅具有轻度症状,约30%为社区获得性肺炎。
3.3 军团菌
系需氧革兰阴性杆菌,广泛存在于湖泊和河流等天然水源及土壤中,也可在冷热水管道等人造水系统中繁殖。目前已知军团菌属有58个种和3个亚种,70个血清型,能引起人类疾病的约有30种,其中与人类疾病关系最密切的是LP,已确认有15个血清型,其中l型和6型最为常见,在欧美国家感染病例中,1型占80%。LP在自然界存活时间长,常在宿主免疫能力下降时依靠胞内寄生功能产生致病性,全年均可发病,夏秋季多,各年龄段均可以发病,成人发病高于儿童,男多于女。LP能引起社区获得性肺炎,特别是重症肺炎,还可引起医院内感染和旅游性肺炎,其潜伏期大约为2~14 d。可能出现前驱疾病,症状包括头痛、肌痛、虚弱和厌食。除了一些免疫功能低下的患者外,通常会出现发热,并且通常伴有相对的心动过缓。肺炎患者出现胃肠道和神经系统表现应提示军团病。胃肠道症状可能很突出,包括腹泻、恶心、呕吐和腹痛。约50%的患者咳嗽时会产生脓痰,并可能出现胸膜炎性胸痛。头痛可能很突出,并伴有意识模糊、癫痫发作和局灶性神经系统表现。
非典型病原
真菌是一种具有真核、能产孢、无叶绿体的真核生物。目前儿童呼吸道真菌感染的常见种类为假丝酵母菌属(Candida)、荚膜组织胞浆菌(Histoplasma capsulatum)、皮炎芽生菌(Blastomyces dermatitidis)、球孢子菌属(Coccidioides)、曲霉菌(Aspergillus)和新型隐球菌(Cryptococcus neoformans)等。真菌侵袭性感染常见于免疫功能受损时,尤其是各种免疫功能缺陷病、中性粒细胞减少、接受长期免疫抑制治疗及人类免疫缺陷病毒(HIV)感染的儿童;某些特殊情况下,如糖尿病酮症酸中毒患者的血清可刺激毛霉菌的生长。呼吸道真菌感染无明显季节特点。
由于全球气候变化和人类活动范围的扩大,真菌感染的流行病学也在发生变化。组织胞浆菌病和球孢子菌病在北美比较多见,芽生菌病和孢子丝菌病在北美和世界其他地区引起散发性病例,东南亚地区马尔尼菲青霉菌(Penicillium marneffei)病在免疫缺陷(如获得性免疫缺陷综合征)患儿中比较常见,而在南美洲,副球孢子菌经常引起皮肤和骨关节的感染。
4.1 假丝酵母菌感染
假丝酵母菌属菌体呈圆形或卵圆形,直径3~6 μm,革兰阳性,着色不均匀。可形成芽生孢子和假菌丝,经培养假菌丝中间或顶端可形成厚膜孢子。具有侵袭性的菌株在体内易形成假菌丝。
多种假丝酵母菌都能引起临床症状,但儿童以白色假丝酵母菌(Candida albicans)感染最常见。临床表现不一,可表现为局部黏膜感染,也可表现为广泛性播散,累及多系统器官。口咽假丝酵母菌病(鹅口疮)常见于小婴儿,也可见于使用抗菌药物、吸入性糖皮质激素(治疗哮喘或鼻炎)、化疗或放疗的年龄较大儿童以及有细胞免疫缺陷的儿童。侵袭性假丝酵母菌病尚与气管插管、中心静脉置管等有关。
4.2 荚膜组织胞浆菌感染
荚膜组织胞浆菌在自然界广泛分布,在被鸟类或蝙蝠粪便污染的土壤中最容易增殖。35 ℃培养时,可见直径3~5 μm,大小均匀的梨形孢子,人体感染荚膜组织胞浆菌后,一般可在巨噬细胞内或其周围发现其孢子,直径约2~4 μm,卵圆形,大小均一。
大多数病例是在较大量的暴露后数周发病,临床表现较轻,除发热外,体格检查通常无明显异常,但也可能发现啰音或实变证据。某些急性组织胞浆菌病患者可表现出流感样症状,伴呼吸道症状和影像学异常。症状通常在数周内缓解,但疲劳和乏力可能持续数月,尤其是病情更严重者。X线影像学通常显示局灶性浸润及纵隔或肺门淋巴结大。组织胞浆菌病经常被误诊为社区获得性肺炎,只有在经验性抗菌药物治疗无效后才会考虑该诊断。
4.3 隐球菌属感染
隐球菌在世界范围内广泛分布,其中新型隐球菌感染最为多见,其为圆形或卵圆形,单芽,厚壁,有宽阔、折光性的胶质样荚膜。隐球菌大小相差很大,一般直径多为4~7 μm,有些直径可达20 μm。周围荚膜由黏多糖组成,厚约3~5 μm。
隐球菌已成为一个重要的真菌感染病原体,可见于免疫功能正常的儿童。中枢神经系统和肺部是两个最常受累的器官,可表现为致命性脑膜脑炎和/或肺炎。咳嗽、呼吸困难和胸痛是最常见的肺部感染症状,但胸部影像学显示肺部结节或隐球菌性肉芽肿的儿童也可能无呼吸道症状。除了累及中枢神经系统和肺部,隐球菌还可累及皮肤、软组织、骨、关节、骨髓和淋巴结等,腹腔内隐球菌病少见。
4.4 曲霉菌感染
曲霉菌是一种典型的丝状菌,主要以枯死的植物、动物的排泄物及动物尸体为营养源。曲霉菌丝有隔膜,直径3~6 μm,分支呈锐角45°,菌丝两侧平行。
曲霉菌感染多见于宿主免疫抑制情况下。侵袭性曲霉菌病最常累及肺部。患儿表现出多种症状和体征,包括发热、胸痛、呼吸急促、咳嗽和/或咯血。中性粒细胞减少的肺曲霉菌病患儿存在典型三联征:发热、胸痛和咯血。胸部影像学检查常见肺结节和/或浸润影。烟曲霉亦可引起变应性支气管肺曲霉病(allergic bronchopulmonary aspergillosis,ABPA),急性期主要为咳嗽、喘息、咯痰、咯血及发热。可咯出棕色痰栓。曲霉尚可以引起中枢神经系统感染。
4.5 毛霉菌(Mucor)感染
毛霉菌在土壤、粪便、禾草及空气等环境中存在。菌丝粗大不规则,无隔、多核、分枝状,直径约5~60 μm,不产生定形菌落。
毛霉菌感染在儿童中发病率逐渐升高,与免疫受损或其他免疫抑制患儿生存率的提高有关。毛霉菌病的特征为菌丝侵犯血管系统导致宿主组织梗死和坏死。糖尿病酮症患儿容易发生鼻脑型毛霉菌病或肺毛霉菌病,这是一种罕见但通常致死的真菌感染。鼻脑型毛霉菌病表现为急性鼻窦炎,伴发热、鼻充血、脓性鼻分泌物、头痛和鼻窦区疼痛。所有鼻窦均受累,并蔓延至邻近结构。肺毛霉菌病主要表现为发热伴咯血,亦有肺梗死及血栓形成。
4.6 马尔尼菲蓝状菌感染
马尔尼菲蓝状菌,原名马尔尼菲青霉菌,是双相型真菌,在30 ℃时呈现为菌丝形态,而在37 ℃时则为酵母形态。在细胞内菌体呈卵圆形、椭圆形,其大小形状一致,直径约2 μm。而游离于组织细胞外的菌则具有多形性,大小形差异大,直径为1~8 μm。
马尔尼菲蓝状菌可引起全身性真菌感染,主要来自东南亚和中国南方,在免疫功能低下者中会发生蓝状菌病。马尔尼菲蓝状菌的传播方式仍不明确。症状和体征不一,轻则为单纯性皮肤病变,重则发生呼吸衰竭和循环衰竭,儿童和成人的表现相似。呼吸系统受累患儿可出现咳嗽、发热、呼吸困难和胸痛,胸片可显示弥漫性网状结节浸润、局限性或弥漫性肺泡浸润。
END
免责声明:本文仅作知识交流与分享及科普目的,不涉及商业宣传,不作为相关医疗指导或用药建议。文章如有侵权请联系删除。
TDS2025检测自动化建设与应用论坛主会场,大会第一天下午茶歇前授课时间段预定开始,咨询热线:177 0186 0390。点击图片,查看TDS2024会后报告
临床申请临床结果
2024-10-20
·动脉网
近来方兴未艾的鼻喷防护领域又出了一个大新闻!
近日,一项发表在Advanced Materials上的研究结果表明,一种全新的鼻喷剂有望阻断包括新冠病毒、甲流病毒在内的多种通过空气传播或飞沫传播的传染病病原体。对于近年来饱受新冠肺炎、流感等呼吸系统传染病困扰的大众来说,这无疑是一个好消息。
这只是鼻喷防护产品近来快速进展的冰山一角,在人类抗击呼吸系统传染病的道路上,新近崛起的鼻喷防护产品可能将在未来发挥越来越重要的作用。
非活性成分配方打造,
可防新冠甲流
这项引发高度关注的研究是由美国哈佛大学布莱根妇女医院的研究团队完成。在这项研究中,团队采用的名为Profi的病原体捕获及中和鼻喷剂(PCANS)在临床前研究中取得了极为突出的防护效果。
根据研究显示,在重症甲流小鼠模型中,给与单次暴露前剂量的Profi可使暴露在25倍致死剂量甲流病毒中的小鼠肺部病毒滴度降低大于99.99%,且肺部炎症细胞和细胞因子正常,最终确保小鼠100%存活率。相比之下,对照组的存活率为0,小鼠无一存活。
在基于3D打印的人鼻模型的实验室化验和测试中,Prioi成功阻断及中和了新冠病毒、甲流病毒、腺病毒、呼吸道合胞病毒、大肠杆菌和肺炎克雷伯菌等主要的呼吸系统传染病病原体。根据研究,其捕获飞沫的能力是单独粘液的两倍之高。
同时,Profi也具有较长的效力,研究显示单次剂量可在小鼠鼻腔存留长达8小时,并至少保证4小时的防护效果。
值得一提的是,Profi完全由非活性成分配方组成。所谓药物非活性成分即药用辅料,是指生产药品和调配处方时使用的赋形剂和附加剂;是除活性成分或前体以外,在安全性方面已进行合理评估,一般包含在药物制剂中的物质。
根据研究团队透露,Profi的配方使用了FDA非活性成分数据库中的成分开发而成,这些成分以前曾用于批准的鼻腔喷雾剂,或来自FDA的公认安全清单。Profi阻断病原体的方式很好理解,通过鼻喷雾剂在鼻腔内形成凝胶状基质捕获呼吸道飞沫,从而固定病原体并将其有效中和。
通俗来说,Profi相当于为鼻腔提供了一层“隐形口罩”。
值得一提的是,由于Profi采用非活性成分配方,不需要通过FDA药物上市前的复杂审批,可以实现快速上市。这也是研究团队最初选择非活性成分进行研发的主要目的。研究团队的几位核心成员共同创立了秋田生物科学(Akita Biosciences),并在2023年10月推出了Profi,将其作为鼻腔卫生化妆品进行销售。
此次的研究结果也为后续的进一步临床试验奠定了基础,证明鼻腔防护类产品具有巨大的潜力。
蓝帆医疗蓝狙士负责人赵鑫向动脉网表示,研究展示了跨学科合作的力量能够将材料科学与医学相结合,开发出可能对抗病毒和细菌性呼吸道感染的创新解决方案:“这代表了一种趋势,即在应对全球健康威胁时,采用多学科、多角度的方法来寻找解决方案,从而提升公共卫生安全,保护人类免受呼吸道病原体的侵害。”
疫情崛起的鼻喷防护,
多种路径殊途同归
事实上,关于此类鼻腔防护类产品的基础研究已有十多年历史。BHM(英国伯明翰生物科技公司)旗下产品采用了类似的技术路线,其CEO徐士哲向动脉网介绍道,以卡拉胶为例,这种提取自红海藻的天然物质对某些病毒蛋白具有特异性,可显著抑制免疫缺陷病毒和逆转录病毒,使病毒载量显著降低,其抑制作用也经过全球十余年的临床验证有效。基于这些研究,一直有将其作用于鼻腔起到防护的设想。
新冠肺炎疫情极大推动了鼻喷防护类产品的研发进度。随着后疫情时代的来临,多种呼吸系统传染病轮流登场,使人防不胜防,大众需要更多的常态化防护手段。佩戴口罩当然是最佳选择,但佩戴口罩在舒适性上也确有不足,导致佩戴意愿降低。
在这种背景下,顺应了大众对于兼顾舒适和防护需求的鼻喷防护类产品很快发展起来,在很短的时间内,市面上出现了多款鼻喷防护产品。由于其所起到的作用与口罩类似,也被形象地称为“液体口罩”。不过,鼻喷防护并不能保证百分百防护,因此,它并不能完全取代口罩,而是可以作为口罩的一种补充。
需要指出的是,鼻喷防护类产品也有不同的种类,虽然结局都是殊途同归,但其原理可以说是大相径庭。总体而言,按照作用机制,鼻喷防护类产品可以主要分为物理类和中和类。Profi就属于物理类防护,主要原理是以物理成膜,主要通过吸附的方式阻隔病原体侵入呼吸系统。
除了吸附,物理隔绝类产品中也有另外一种作用机制,即通过某种物理成分杀灭抑制病原体。这其中,NO(一氧化氮)已有几十年药理学、毒性和安全性研究,证明其具有的抗微生物特性可以直接杀死上呼吸道病毒,防止病毒增殖并扩散到肺部。
2021年,加拿大一家名为SaNOtize的生物企业就推出了以一氧化氮为主要成分的NONS(一氧化氮鼻喷剂),用于新冠肺炎治疗。临床研究证明,接受NONS治疗的新冠肺炎患者可在24小时内将新冠病毒RNA载量降低95%以上,并在72小时内将新冠病毒RNA载量降低99%以上。
中和类则是另外一种路径的鼻喷防护类产品。其核心逻辑是作用于病原体本身,通过抢先结合病原体(如新冠病毒)的方式,使其失去入侵人体的能力。中和类也有几种技术路线,主要包括小分子多肽和抗体大分子(如纳米抗体及Igm抗体)等。目前,几家国内药企推出的产品均采用中和路线。
几种市售鼻喷防护产品指标
两类产品各有千秋,都具有一定的防护作用。相对而言,中和类的鼻喷防护产品主要针对特定病原体开发,对于目标病原体有极强的中和效果,但对于非目标病原体则不具有中和作用。此外,一旦病毒发生变异,也存在失效的可能。
这其中,采用多肽路线的产品由于作用于病毒的区域不一样,理论上可能具有一定的广谱性。以针对新冠病毒的多肽类产品为例,主要作用于新冠病毒刺突蛋白的HR1区域,这一区域受病毒变异相对较小,理论上对于病毒变异有更好的效果。
基于其原理,物理类产品在广谱性上明显占优,一般都可以针对多种病原体进行防护,尤其对于一些尚未被发现的病原体理论上同样有效。另一方面,由于对鼻腔环境的改变相对较小,物理类产品在鼻腔中的持久性上也要更好一些。
徐士哲表示:“排除异物是鼻腔天生的作用。鼻喷阻断剂要想达到最好的效果,最大的技术挑战主要是能在鼻腔黏膜上停留足够久的时间,且不会改变鼻腔环境。这恰恰是物理类产品研发的重点方向。目前,BHM在真实环境中可以存留六小时,这是实际临床在志愿者鼻腔测试得到的数据。”
“事实上,卡拉胶在全球已有十余年应用,但是BHM的专利配方加入结冷胶可以延长卡拉胶在鼻腔停留的时间,并且增强物理阻断的作用。我们刚刚做完关于结冷胶在鼻喷阻断剂中的重要性。在研究中,我们发现结冷胶可以和鼻腔黏膜产生更好的结合作用,可以在鼻腔停留更久的时间,这些结果很快我们就会公开发表。”他补充道。
此外,这种技术作为一种安全、非药性并且非特异性的技术平台,也可作为一种药物载体,通过与药企合作研发,将药物有效成分与载体集成,使药物能持久、稳定地覆盖在用药处,保证药效的长期、精准的释放。
当然,无论何种路线,对于当下流行病原体的及时测试都必不可少。赵鑫就表示,以新冠病毒为例,每当出现新的变异株时,各品牌最好都能及时进行相关检测,以保证对新病毒中和的有效性。据介绍,蓝帆医疗对于国内多波次主要流行的新冠病毒变异株及其亚分支均进行了及时检测试验,证明其产品对以上毒株均具有显著抑制效果。”
鼻腔防护刚刚起步,
仍待规范但需求巨大
作为一种近年快速崛起的产品,鼻喷防护产品也面临创新产品所面临的不少问题。行业人士在与动脉网交流时均表示,由于历史较短,此类产品目前暂时没有明确的监管指南。这为产品的研发和推广带来了一些困难。
徐士哲表示,虽然在外在表现形式上都属于鼻喷剂,但物理类和中和类内在机制的不同决定了两者其实属于不同的分支。理论上而言,一旦产品的功效宣称达到需要监管的程度,采用单纯物理阻断形式的物理类产品在全球范围都应属于医疗器械。中和类产品则由于具有药理作用,应属于药品或药械合一。
“我们的产品在国内属于新产品,因此在上市的时候需要分类界定和临床试验,需要比较长的时间。但根据我们做的分类界定,鼻喷阻断剂作用原理是物理阻隔,不会产生药理作用,属于医疗器械,这点跟国外是一样的。我们正准备在国内进行临床注册为二类医疗器械,从而可以实现国内生产以降低成本。”他向动脉网介绍。
赵鑫也表示,鼻喷阻断类产品属于一个全新行业,国内尚无具体执行标准及监管指南。目前,整个行业正在摸索中前进,未来会逐渐规范,蓝帆医疗也在努力推动相关标准的制定。
另一个难题则来自临床试验,鼻喷类产品的临床试验难度较大。无论是临床试验的设计还是目标的达成,都比常规的临床试验更困难。根据一项研究统计,自国家药品监督管理局药物临床试验登记与信息公示平台开放注册日开始至2023年3月近十年时间,国内一共也只登记了80项鼻用喷雾剂注册临床试验,其中,超过七成都是在2019年之后注册。
徐士哲表示,从动物实验开始,鼻喷剂就面临不小的难度:“首先,动物的鼻腔结构就和人体有巨大的差异,以Profi的研究为例,无法采用将病原体通过空气传播进入小鼠鼻腔的形式进行试验,而是采取了滴入小鼠鼻腔的形式。同样,注入Profi也是采取滴入的形式而非鼻喷入小鼠鼻腔的形式。”
“即使进入到人体试验,试验也无法控制各种变量,‘制造’出试验需要的环境。受试者仍然需要继续生活,面临各种不同的环境和生活习惯,这些都会对鼻喷剂的临床试验造成巨大困扰,想要拿到预期的结果很有挑战性。”他补充说道。
然而,市场并不等人——耗费数年时间完成临床试验后再上市很有可能会错过最佳时期。新冠肺炎疫情期间,国内曾有多家药企开发新冠病毒抗体类鼻喷防护产品,并先后进入临床试验阶段。但动脉网查询公开信息发现,相关产品的后续信息并不多。
其中,翰宇药业在2024年初明确宣布国家药品审评中心批准其1.1类创新药广谱预防用HY3000鼻喷雾剂进行临床三期实验,是为数不多在今年仍有官方消息确认仍在继续进行的项目。
可想而知,此类产品的临床试验难度颇大。
因此,目前多数已上市的鼻喷防护类产品往往以消费类产品的形式上市,避免提及可能招致监管的字眼。另一方面,则同步进行临床试验。以翰宇药业为例,其采用相关专利的大健康产品——翰护融合抑制肽防护喷雾剂已经上市。
不过,这并未影响大众对于鼻喷防护类产品的巨大需求。在电商上搜索相关产品,单价介乎100-300元的多款鼻喷防护产品皆有不错的销量。蓝帆医疗的蓝狙士在京东上有超过5万条评价,据介绍,复产品复率也高达60%以上。
这也进一步推动了相关企业的研发热情。BHM目前就正开发新配方,以更有效地阻隔空气中的过敏源,预防过敏性鼻炎,或将在明年年底推出。蓝帆医疗则将整体围绕上呼吸道系统进行产品布局和规划,已推出针对鼻炎的两款产品,并正在开发防流感的新品,预计年底前推出。
写在最后
在后疫情时代,大众正面临一个更加复杂的状况,呼吸系统传染病的威胁并未随着疫情的结束而减少。相反,它们正以“温水煮青蛙”的方式更频繁地威胁公共卫生。
人类又该如何适应共存呢?也许这个答案就是转变墨守的防疫观念,以更安全、更有效的方式,帮助全年龄阶段的每个人阻隔传染源,防止感染。方兴未艾的鼻喷防护类产品,假以时日也将有望成为终结呼吸道传染病的一种有力武器。
* 参考资料:
Joseph J, Baby HM, Quintero JR, Kenney D, Mebratu YA, Bhatia E, Shah P, Swain K, Lee D, Kaur S, Li XL, Mwangi J, Snapper O, Nair R, Agus E, Ranganathan S, Kage J, Gao J, Luo JN, Yu A, Park D, Douam F, Tesfaigzi Y, Karp JM, Joshi N. Toward a Radically Simple Multi-Modal Nasal Spray for Preventing Respiratory Infections. Adv Mater. 2024 Sep 24:e2406348. doi: 10.1002/adma.202406348. Epub ahead of print. PMID: 39318086.
Darren Incorvaia, Fiercebiotech.com, Nasal spray neutralizes army of respiratory diseases and protects mice from deadly influenza
《21世纪经济报道》:《6款在研国产新冠预防鼻喷药物,谁能率先“撞线”?》
*封面图片来源:123rf
如果您认同文章中的观点、信息,或想进一步讨论,请与我们联系;也可加入动脉网行业社群,结交更多志同道合的好友。
近
期
推
荐
声明:动脉网所刊载内容之知识产权为动脉网及相关权利人专属所有或持有。未经许可,禁止进行转载、摘编、复制及建立镜像等任何使用。
动脉网,未来医疗服务平台
2024-07-11
Tailwind is evaluating inhaled AP-PA02 in non-cystic fibrosis bronchiectasis (NCFB), its second patient population, following successful evaluation in patients with cystic fibrosis (SWARM-P.a.) in 2023
Phase 2 Topline data anticipated in 2H 2024 followed by potential initiation of a pivotal bronchiectasis trial in 2025
LOS ANGELES, July 11, 2024 /PRNewswire/ -- Armata Pharmaceuticals, Inc. (NYSE American: ARMP) ("Armata" or the "Company"), a clinical-stage biotechnology company focused on pathogen-specific bacteriophage therapeutics for antibiotic-resistant and difficult-to-treat bacterial infections, today announced that it has achieved full enrollment in its Tailwind Phase 2 clinical study of inhaled AP-PA02 in patients with NCFB and chronic pulmonary Pseudomonas aeruginosa (P. aeruginosa) infection. The last patient final follow-up visit is scheduled for August 7, 2024. Armata anticipates topline data from the Tailwind study in the second half of 2024.
"We are pleased to have efficiently enrolled this important study according to our internal timelines as we continue to add to the growing body of evidence demonstrating the value of phage therapy in treating chronic bacterial respiratory infections," stated Dr. Deborah Birx, Chief Executive Officer of Armata. "In this study, we enrolled a cohort of patients without prior or current inhaled antibiotics, allowing for the evaluation of phage-only therapy compared to placebo, in addition to a cohort of patients receiving inhaled phage plus antibiotics. We look forward to sharing topline data as soon as it becomes available and plan to meet with the FDA shortly thereafter on the design of a pivotal Phase 3 study, which we are planning to initiate in 2025. We remain committed to the execution of rigorously designed randomized trials that, if successful, will support registration of our phage candidates."
"The success of our completed SWARM-P.a. study of inhaled AP-PA02 in cystic fibrosis patients with chronic pulmonary P. aeruginosa infection provided important learnings that we incorporated into the design of Tailwind," stated Mina Pastagia, MD, MS, Chief Medical Officer of Armata. "Notably, the Tailwind study includes an optimized dosing regimen that gives us confidence in our ability to demonstrate clinical safety, while evaluating the durability of P. aeruginosa reduction in the lungs. We look forward to evaluating the clinical data from Tailwind as we work diligently to introduce a sorely needed pathogen-specific class of anti-infectives to patients with bronchiectasis and chronic P. aeruginosa infections."
The Tailwind study is a Phase 2, multi-center, double blind, randomized, placebo-controlled trial evaluating the safety, tolerability, and efficacy of inhaled AP-PA02 as monotherapy, as well as in combination with inhaled antibiotics in subjects with NCFB and chronic pulmonary P. aeruginosa infection. The primary endpoint is P. aeruginosa recovery in sputum following multiple doses of AP-PA02 administered by inhalation.
For more information:
About Armata Pharmaceuticals, Inc.
Armata is a clinical-stage biotechnology company focused on the development of pathogen-specific bacteriophage therapeutics for the treatment of antibiotic-resistant and difficult-to-treat bacterial infections using its proprietary bacteriophage-based technology. Armata is developing and advancing a broad pipeline of natural and synthetic phage candidates, including clinical candidates for Pseudomonas aeruginosa, Staphylococcus aureus, and other pathogens. Armata is committed to advancing phage therapy with drug development expertise that spans bench to clinic including in-house phage specific cGMP manufacturing.
Forward Looking Statements
This communication contains "forward-looking" statements as defined by the Private Securities Litigation Reform Act of 1995. These statements relate to future events, results or to Armata's future financial performance and involve known and unknown risks, uncertainties and other factors which may cause Armata's actual results, performance or events to be materially different from any future results, performance or events expressed or implied by the forward-looking statements. In some cases, you can identify these statements by terms such as "anticipate," "believe," "could," "estimate," "expect," "intend," "may," "plan," "potential," "predict," "project," "should," "will," "would" or the negative of those terms, and similar expressions. These forward-looking statements reflect management's beliefs and views with respect to future events and are based on estimates and assumptions as of the date of this communication and are subject to risks and uncertainties including risks related to Armata's development of bacteriophage-based therapies; ability to staff and maintain its production facilities under fully compliant current Good Manufacturing Practices; ability to meet anticipated milestones in the development and testing of the relevant product; ability to be a leader in the development of phage-based therapeutics; ability to achieve its vision, including improvements through engineering and success of clinical trials; ability to successfully complete preclinical and clinical development of, and obtain regulatory approval of its product candidates and commercialize any approved products on its expected timeframes or at all; and Armata's estimates regarding anticipated operating losses, capital requirements and needs for additional funds. Additional risks and uncertainties relating to Armata and its business can be found under the caption "Risk Factors" and elsewhere in Armata's filings and reports with the SEC, including in Armata's Annual Report on Form 10-K, filed with the SEC on March 21, 2024, and in its subsequent filings with the SEC.
Armata expressly disclaims any obligation or undertaking to release publicly any updates or revisions to any forward-looking statements contained herein to reflect any change in Armata's expectations with regard thereto or any change in events, conditions or circumstances on which any such statements are based.
Media Contacts:
At Armata:
Pierre Kyme
Armata Pharmaceuticals, Inc.
[email protected]
310-665-2928 x234
Investor Relations:
Joyce Allaire
LifeSci Advisors, LLC
[email protected]
212-915-2569
SOURCE Armata Pharmaceuticals, Inc.
临床2期临床3期
分析
对领域进行一次全面的分析。
登录
或
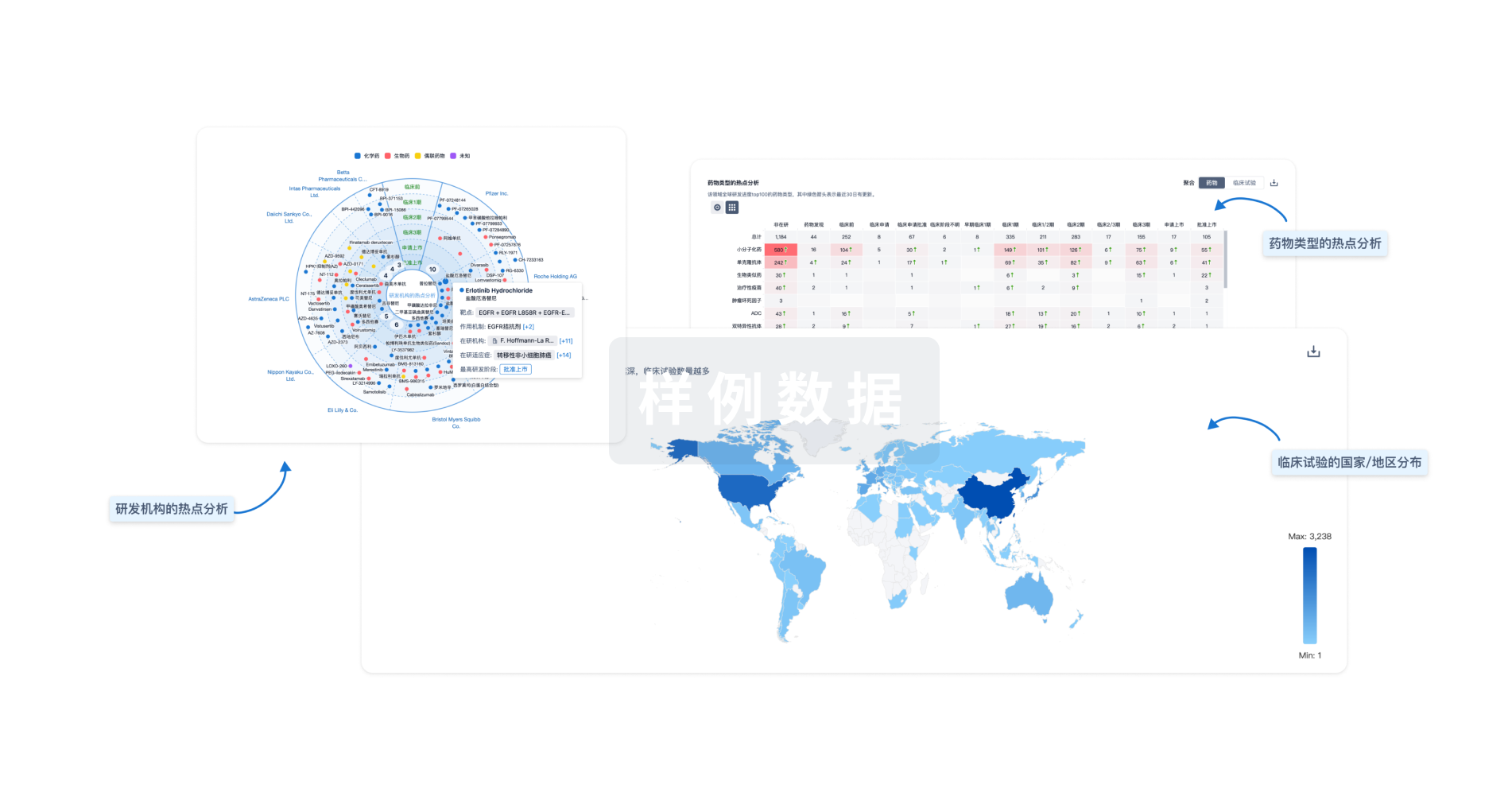
生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用



