预约演示
更新于:2025-05-07
Oligoarticular Arthritis
少关节关节炎
更新于:2025-05-07
基本信息
别名 Oligoarthritides、Oligoarthritis、Oligoarticular Arthritis + [4] |
简介 An arthritis affecting fewer than five separate joints. |
关联
11
项与 少关节关节炎 相关的药物靶点 |
作用机制 IL-6RA拮抗剂 |
原研机构 |
非在研适应症- |
最高研发阶段批准上市 |
首次获批国家/地区 韩国 |
首次获批日期2024-12-20 |
靶点 |
作用机制 IL-6RA拮抗剂 |
非在研适应症- |
最高研发阶段批准上市 |
首次获批国家/地区 欧盟 [+3] |
首次获批日期2023-09-15 |
靶点 |
作用机制 IL-6RA拮抗剂 |
原研机构 |
非在研适应症- |
最高研发阶段批准上市 |
首次获批国家/地区 中国 |
首次获批日期2023-01-16 |
4
项与 少关节关节炎 相关的临床试验CTR20244412
巴瑞替尼片在中国健康受试者中单中心、随机、开放、单剂量、两制剂、两周期、两序列、双交叉、空腹/餐后状态下生物等效性试验
主要研究目的
健康受试者空腹/餐后状态下,单次口服广州绿十字制药股份有限公司生产的受试制剂巴瑞替尼片(规格:4mg/片)或以Eli Lilly Nederland B.V.持有的参比制剂巴瑞替尼片(商品名:OLUMIANT®;规格:4mg/片),比较空腹/餐后状态下受试制剂与参比制剂在健康受试者体内的生物等效性。
次要研究目的
考察受试制剂和参比制剂在健康受试者中的安全性。
开始日期2024-12-11 |
申办/合作机构 |
NCT03797872
Clinical Effectiveness of Symptomatic Therapy Compared to Standard Step up Care for the Treatment of Low Impact Psoriatic Oligoarthritis: a 2 Arm Parallel Group Feasibility Study
POISE is a two arm interventional trial nested within a cohort (Trials Within Cohorts or TWiCs design). This tests less aggressive early therapy in patients newly diagnosed with low impact oligoarticular PsA. Arm 1 will receive standard step up therapy in the cohort and act as the control group. Arm 2 will receive local steroid injections to active joints and will be able to use non-steroidal anti-inflammatory drugs (NSAIDs) only
开始日期2019-04-17 |
申办/合作机构  University of Oxford University of Oxford [+1] |
NCT04154852
Prospective, Single-centre, Double-Blind, Randomised, Placebo-controlled Study Evaluating Efficacy of Adalimumab + Methotrextate Compared With Placebo + Methotrexate in Patients With Early Oligoarthritis (ADEOS)
The purpose of this study is to investigate adalimumab, a drug that is currently licensed for treatment of patients with rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis and inflammatory bowel disease (Crohn's disease). It is a protein (human monoclonal antibody) that is designed to block the effects of an inflammatory cytokine, tumour necrosis factor alpha (TNF alpha) which is a causative factor in joint inflammation. It is given as a subcutaneous injection.
Management of established rheumatoid arthritis has been transformed with the use of TNFantagonists, the first in the class of biological agents. The benefits in early RA are also continuing to emerge. Oligoarthritis represents a subgroup of early inflammatory arthritis that warrants more effective treatment strategy including the potential to modulate the disease course and halt further progression. The TNFantagonists offer the potential of achieving this, justifying this initial investigation.
Hence, this study is designed to establish the benefit of adalimumab in preventing progression of oligoarthritis. The primary aim of the study is to assess how many patients achieve remission i.e. no further evidence of joint inflammation or damage. The investigators will do this by including patients who present with inflammation of four or less joints who do not fulfil criteria for a definite arthritis condition such as rheumatoid arthritis. The investigators will perform clinical, laboratory (blood tests) and imaging assessments at regular intervals on these patients to check on safety of the study drug adalimumab and evaluate any changes in disease activity that may have resulted from use of adalimumab. All patients will receive treatment with a standard therapy (methotrexate) with randomisation of half of the patients to receive the active study drug, adalimumab as additional treatment for six months.
Management of established rheumatoid arthritis has been transformed with the use of TNFantagonists, the first in the class of biological agents. The benefits in early RA are also continuing to emerge. Oligoarthritis represents a subgroup of early inflammatory arthritis that warrants more effective treatment strategy including the potential to modulate the disease course and halt further progression. The TNFantagonists offer the potential of achieving this, justifying this initial investigation.
Hence, this study is designed to establish the benefit of adalimumab in preventing progression of oligoarthritis. The primary aim of the study is to assess how many patients achieve remission i.e. no further evidence of joint inflammation or damage. The investigators will do this by including patients who present with inflammation of four or less joints who do not fulfil criteria for a definite arthritis condition such as rheumatoid arthritis. The investigators will perform clinical, laboratory (blood tests) and imaging assessments at regular intervals on these patients to check on safety of the study drug adalimumab and evaluate any changes in disease activity that may have resulted from use of adalimumab. All patients will receive treatment with a standard therapy (methotrexate) with randomisation of half of the patients to receive the active study drug, adalimumab as additional treatment for six months.
开始日期2011-06-01 |
申办/合作机构  University of Leeds University of Leeds [+1] |
100 项与 少关节关节炎 相关的临床结果
登录后查看更多信息
100 项与 少关节关节炎 相关的转化医学
登录后查看更多信息
0 项与 少关节关节炎 相关的专利(医药)
登录后查看更多信息
1,264
项与 少关节关节炎 相关的文献(医药)2025-04-01·Annals of the Rheumatic Diseases
How to treat undifferentiated arthritis today or tomorrow? A consideration of treatment recommendations in light of current evidence
Review
作者: van der Helm-van Mil, Annette H M ; Aletaha, Daniel
2025-03-01·Best Practice & Research Clinical Rheumatology
Fungal arthritis
Review
作者: Kong, Kok Ooi ; Kong, Ying Xi
2025-02-21·Modern Rheumatology
Nationwide epidemiological survey of juvenile idiopathic arthritis during transition to young adulthood in Japan using the National Database of Designated Incurable Diseases of Japan
Article
作者: Matsushita, Masakazu ; Mori, Masaaki ; Inoue, Eisuke ; Miyamae, Takako ; Inoue, Yuzaburo ; Sakai, Ryoko ; Sugihara, Takahiko ; Yamaji, Ken ; Shimojo, Naoki ; Shimizu, Masaki ; Mitsunaga, Kanako
5
项与 少关节关节炎 相关的新闻(医药)2025-02-04
THOUSAND OAKS, Calif., Feb. 4, 2025 /PRNewswire/ -- Amgen (NASDAQ:AMGN) today announced financial results for the fourth quarter and full year of 2024 versus comparable periods in 2023.
"Robust growth in sales and earnings throughout 2024 reflects the momentum of our business. With strong performance globally, we are investing heavily in our rapidly advancing pipeline to deliver innovative therapies across our four therapeutic areas," said Robert A. Bradway, chairman and chief executive officer.
Key results include:
For the fourth quarter, total revenues increased 11% to $9.1 billion in comparison to the fourth quarter of 2023.
Product sales grew 11%, primarily driven by 14% volume growth. Excluding sales from our Horizon Therapeutics (Horizon) acquisition, product sales grew 10%, driven by volume growth of 15%.
Ten products delivered at least double-digit sales growth in the fourth quarter, including Repatha® (evolocumab), BLINCYTO® (blinatumomab), TEZSPIRE® (tezepelumab-ekko), EVENITY® (romosozumab-aqqg) and TAVNEOS® (avacopan).
Our performance included $1.2 billion of sales from our rare disease products, driven by several first-in-class, early-in-lifecycle medicines, including TEPEZZA® (teprotumumab-trbw), KRYSTEXXA® (pegloticase), UPLIZNA® (inebilizumab-cdon) and TAVNEOS®.
For the full year, total revenues increased 19% to $33.4 billion in comparison to the full year of 2023.
Product sales grew 19%, primarily driven by 23% volume growth, partially offset by 2% lower net selling price. Excluding sales from our Horizon acquisition, product sales grew 7%, driven by volume growth of 11%.
Ten products delivered at least double-digit sales growth for the full year, including Repatha®, TEZSPIRE®, EVENITY®, BLINCYTO® and TAVNEOS®.
21 products achieved record sales for the full year.
GAAP earnings per share (EPS) decreased 18% from $1.42 to $1.16 for the fourth quarter, primarily driven by mark-to-market losses on our equity investments, partially offset by higher revenues. For the full year, GAAP EPS decreased 39% from $12.49 to $7.56, primarily driven by higher operating expenses, including amortization expense from Horizon acquisition-related assets and incremental operating expenses from Horizon, and overall mark-to-market losses on our equity investments in 2024, partially offset by higher revenues.
For the fourth quarter, GAAP operating income increased from $1.3 billion to $2.3 billion, and GAAP operating margin increased 10.3 percentage points to 26.5%. For the full year, GAAP operating income decreased from $7.9 billion to $7.3 billion, and GAAP operating margin decreased 6.6 percentage points to 22.7%.
Non-GAAP EPS increased 13% from $4.71 to $5.31 for the fourth quarter, driven by higher revenues, partially offset by higher operating expenses. For the full year, non-GAAP EPS increased 6% from $18.65 to $19.84, driven by higher revenues, partially offset by higher operating expenses, including incremental operating expenses from Horizon, and higher interest expense.
For the fourth quarter, non-GAAP operating income increased from $3.7 billion to $4.0 billion, and non-GAAP operating margin decreased 0.4 percentage points to 46.3%. For the full year, non-GAAP operating income increased from $13.4 billion to $15.0 billion, and non-GAAP operating margin decreased 2.9 percentage points to 46.9%.
The Company generated $10.4 billion of free cash flow for the full year versus $7.4 billion in 2023, driven by business performance and timing of working capital items, primarily collections, partially offset by higher net interest expense.
References in this release to "non-GAAP" measures, measures presented "on a non-GAAP basis," "free cash flow" (computed by subtracting capital expenditures from operating cash flow), "EBITDA, or earnings before interest, taxes, depreciation and amortization" (computed by adding interest expense, provision for income taxes, and depreciation and amortization expense to GAAP net income) and "debt leverage ratio" (calculated as the ratio of GAAP total debt to EBITDA) refer to non-GAAP financial measures. Adjustments to the most directly comparable GAAP financial measures and other items are presented on the attached reconciliations. Refer to Non-GAAP Financial Measures below for further discussion.
Product Sales Performance
General Medicine
Repatha® (evolocumab) sales increased 45% year-over-year to $606 million in the fourth quarter, primarily driven by volume growth. Full year sales increased 36%, primarily driven by 43% volume growth, partially offset by 10% lower net selling price. For 2025, we expect lower declines in net selling price.
EVENITY® (romosozumab-aqqg) sales increased 36% year-over-year to $431 million in the fourth quarter and 35% for the full year, driven by volume growth.
Prolia® (denosumab) sales increased 5% year-over-year to $1.2 billion in the fourth quarter and 8% for the full year, driven by volume growth. For 2025, we expect sales erosion driven by biosimilar competition.
Rare Disease
Except for TAVNEOS®, the products listed below were added through the acquisition of Horizon on Oct. 6, 2023.
TEPEZZA® (teprotumumab-trbw) generated $460 million of sales in the fourth quarter and $1.9 billion for the full year. TEPEZZA is the first and only approved treatment for thyroid eye disease (TED) in the U.S. and Japan.
KRYSTEXXA® (pegloticase) generated $346 million of sales in the fourth quarter and $1.2 billion for the full year. KRYSTEXXA is the first and only FDA-approved treatment for chronic refractory gout.
UPLIZNA® (inebilizumab-cdon) generated $101 million of sales in the fourth quarter and $379 million for the full year. UPLIZNA is used to treat adults with neuromyelitis optica spectrum disorder (NMOSD).
TAVNEOS® (avacopan) generated $81 million of sales in the fourth quarter. Sales increased 84% year-over-year in the fourth quarter and 111% for the full year, primarily driven by volume growth. TAVNEOS is a first-in-class treatment for severe active anti-neutrophil cytoplasmic autoantibody-associated vasculitis (ANCA-associated vasculitis).
Ultra-Rare products, which consist of
RAVICTI® (glycerol phenylbutyrate),
PROCYSBI® (cysteamine bitartrate),
ACTIMMUNE®
(interferon gamma-1b),
QUINSAIR® (levofloxacin) and
BUPHENYL® (sodium phenylbutyrate), generated $214 million of sales in the fourth quarter and $758 million for the full year.
Inflammation
TEZSPIRE® (tezepelumab-ekko) sales increased 67% year-over-year to $296 million in the fourth quarter and 71% for the full year, primarily driven by volume growth.
Otezla® (apremilast) sales decreased 1% year-over-year to $624 million in the fourth quarter, driven by 7% lower net selling price, partially offset by 5% volume growth. Sales decreased 3% for the full year, primarily driven by 8% lower net selling price, partially offset by 3% volume growth.
Enbrel® (etanercept) sales were flat year-over-year at $1.0 billion in the fourth quarter as 7% favorable changes to estimated sales deductions were offset by lower net selling price. Full year sales decreased 10%, driven by lower net selling price. For 2025, we expect continued declining net selling price and relatively flat volumes.
We expect Otezla and Enbrel to follow the historical pattern of lower sales in the first quarter relative to subsequent quarters due to the impact of benefit plan changes, insurance reverification and increased co-pay expenses as U.S. patients work through deductibles.
AMJEVITA®/AMGEVITA™ (adalimumab) sales increased 84% year-over-year to $294 million in the fourth quarter and 22% for the full year, driven by volume growth, partially offset by lower net selling price.
Oncology
BLINCYTO® (blinatumomab) sales increased 58% year-over-year to $381 million in the fourth quarter and 41% for the full year, primarily driven by volume growth.
Vectibix® (panitumumab) sales decreased 2% year-over-year to $246 million in the fourth quarter, driven by 5% unfavorable foreign exchange impact and 4% lower volume, partially offset by higher net selling price. Sales increased 6% for the full year, driven by 8% higher net selling price and 4% volume growth, partially offset by unfavorable foreign exchange impact.
KYPROLIS® (carfilzomib) sales increased 6% year-over-year to $372 million in the fourth quarter and 7% for the full year, driven by volume growth outside the U.S.
LUMAKRAS®/LUMYKRAS™ (sotorasib) sales increased 10% year-over-year to $85 million in the fourth quarter, primarily driven by volume growth. Sales increased 25% for the full year, driven by volume growth and favorable changes to estimated sales deductions.
XGEVA® (denosumab) sales increased 6% year-over-year to $561 million in the fourth quarter, driven by volume growth. Sales increased 5% for the full year, driven by higher net selling price. For 2025, we expect sales erosion driven by biosimilar competition.
Nplate® (romiplostim) sales decreased 13% year-over-year to $337 million in the fourth quarter. Excluding a fourth quarter 2023 U.S. government order of $62 million, Nplate sales grew 4% year-over-year in the fourth quarter, driven by volume growth. Full year sales decreased 1%. U.S. government orders were $128 million in 2024 compared to $286 million in 2023. Excluding these U.S. government orders, Nplate sales grew 12% year-over-year for the full year, driven by 8% volume growth and 6% higher net selling price.
IMDELLTRA® (tarlatamab-dlle) generated $67 million of sales in the fourth quarter. Sales increased 86% quarter-over-quarter, driven by volume growth and inventory levels. IMDELLTRA is the first and only FDA-approved bispecific T-cell engager (BiTE
®) therapy for the treatment of extensive-stage small cell lung cancer (ES-SCLC).
MVASI® (bevacizumab-awwb) sales decreased 8% year-over-year to $173 million in the fourth quarter and 9% for the full year.
Established Products
Our established products, which consist of
EPOGEN® (epoetin alfa),
Aranesp® (darbepoetin alfa),
Parsabiv® (etelcalcetide) and
Neulasta® (pegfilgrastim), generated $500 million of sales in the fourth quarter. Sales decreased 29% year-over-year for the fourth quarter, driven by volume declines, unfavorable changes to estimated sales deductions and lower net selling price. Sales decreased 19% for the full year, driven by volume declines, lower net selling price and unfavorable changes to estimated sales deductions.
Product Sales Detail by Product and Geographic Region
Operating Expense, Operating Margin and Tax Rate Analysis
On a GAAP basis:
Total Operating Expenses decreased 2% year-over-year for the fourth quarter and increased 29% for the full year.
Cost of Sales as a percentage of product sales decreased 4.0 percentage points for the fourth quarter primarily driven by lower amortization expense from the fair value step-up of inventory acquired from Horizon, partially offset by changes in our sales mix, and higher profit share and royalty expense. For the full year, cost of sales as a percentage of product sales increased 8.7 percentage points driven by higher amortization expense from Horizon acquisition-related assets and, to a lesser extent, higher profit share and royalty expense, partially offset by the Puerto Rico excise tax.
Research & Development (R&D) expenses increased 12% for the fourth quarter driven by higher spend in later-stage clinical programs, partially offset by lower research and early pipeline spend. R&D expenses increased 25% for the full year driven by higher spend in later-stage clinical programs and marketed products support, including spend from Horizon-acquired programs.
Selling, General & Administrative (SG&A) expenses decreased 17% for the fourth quarter primarily driven by lower Horizon acquisition-related expenses. SG&A expenses increased 15% for the full year primarily driven by expenses from the acquired Horizon business and other commercial expenses, partially offset by lower Horizon acquisition-related expenses incurred in 2024.
Other operating expenses for the full year primarily consisted of impairment charges associated with in-process R&D (IPR&D) intangible assets related to our Teneobio, Inc. acquisition in 2021 and expenses related to cost-savings initiatives incurred in 2024.
Operating Margin as a percentage of product sales increased 10.3 percentage points to 26.5% for the fourth quarter and decreased 6.6 percentage points to 22.7% for the full year.
Tax Rate increased 9.8 percentage points in the fourth quarter and decreased 3.2 percentage points for the full year. The fourth quarter tax rate increase was related to deferred tax adjustments associated with the U.S. minimum tax on the earnings of our foreign subsidiaries and prior year favorable items, partially offset by the change in earnings mix as a result of the fourth quarter 2024 unrealized losses on our strategic equity investments (primarily BeiGene). The full year tax rate decrease was due to the change in earnings mix, including the net unrealized impacts of our strategic equity investments (primarily BeiGene), partially offset by the deferred tax adjustments associated with the U.S. minimum tax on the earnings of our foreign subsidiaries.
On a non-GAAP basis:
Total Operating Expenses increased 11% year-over-year for the fourth quarter and increased 24% for the full year.
Cost of Sales as a percentage of product sales increased 1.3 percentage points for the fourth quarter driven by changes in our sales mix, and higher profit share and royalty expense. Cost of sales as a percentage of product sales increased 0.9 percentage points for the full year driven by higher profit share and royalty expense, partially offset by the Puerto Rico excise tax.
R&D expenses increased 14% for the fourth quarter driven by higher spend in later-stage clinical programs, partially offset by lower spend in research and early pipeline. R&D expenses increased 25% for the full year driven by higher spend in later-stage clinical programs and marketed products support, including spend from Horizon-acquired programs.
SG&A expenses increased 3% for the fourth quarter driven by higher general and administrative expenses. SG&A expenses increased 23% for the full year primarily driven by expenses from the acquired Horizon business and other marketed product expenses.
Operating Margin as a percentage of product sales decreased 0.4 percentage points to 46.3% for the fourth quarter and decreased 2.9 percentage points to 46.9% for the full year.
Tax Rate decreased 1.1 percentage points for the fourth quarter and decreased 2.0 percentage points for the full year. The fourth quarter tax rate decrease was primarily due to the change in earnings mix and net favorable items as compared to the prior year. The full year tax rate decrease was primarily due to the change in earnings mix as a result of the inclusion of the Horizon business and net favorable items as compared to the prior year.
Cash Flow and Balance Sheet
The Company generated a record $4.4 billion of free cash flow in the fourth quarter of 2024 versus $0.3 billion in the fourth quarter of 2023, driven by timing of tax payments, timing of working capital, primarily collections, lower transaction expenses compared to the fourth quarter of 2023, which included significant costs tied to the closing of the Horizon acquisition, and business performance. The Company generated $10.4 billion of free cash flow for the full year 2024 versus $7.4 billion in 2023.
The Company's fourth quarter 2024 dividend of $2.25 per share was declared on October 25, 2024, and was paid on December 9, 2024, to all stockholders of record as of November 18, 2024, representing a 6% increase from the same period in 2023.
During the fourth quarter, there were no repayments or extinguishments of debt. For the full year 2024, the Company reduced principal debt outstanding by $4.5 billion.
For the fourth quarter and full year, the Company repurchased 0.7 million shares of common stock at a total cost of $200 million.
Cash and investments totaled $12.0 billion and debt outstanding totaled $60.1 billion as of December 31, 2024. Debt leverage was approximately 4.5 times EBITDA as of December 31, 2024.
2025 Guidance
For the full year 2025, the Company expects:
Total revenues in the range of $34.3 billion to $35.7 billion.
On a
GAAP basis, EPS in the range of $10.89 to $12.14, and a
tax rate in the range of 11.0% to 12.5%.
On a
non-GAAP basis, EPS in the range of $20.00 to $21.20, and a
tax rate in the range of 15.0% to 16.0%.
Capital expenditures to be approximately $2.3 billion.
Share repurchases not to exceed $500 million.
Fourth
Quarter Product and Pipeline Update
The Company provided the following updates on selected product and pipeline programs:
General Medicine
MariTide (maridebart cafraglutide, AMG 133)
MariTide is a differentiated peptide-antibody conjugate that activates the glucagon like peptide 1 (GLP-1) receptor and antagonizes gastric inhibitory polypeptide receptor (GIPR).
In November 2024, data were presented from Part 1 of a Phase 2 chronic weight management study in adults who are living with overweight or obesity, with or without Type 2 diabetes mellitus. MariTide demonstrated robust weight loss at 52 weeks without a weight loss plateau, significant improvements in cardiometabolic parameters, and is the first obesity treatment with monthly or less frequent dosing to demonstrate safe and effective weight loss in a Phase 2 study.
Part 2 of the Phase 2 chronic weight management study is ongoing in adults who are living with overweight or obesity, with or without Type 2 diabetes mellitus. Data readout is anticipated in H2 2025.
A Phase 2 study investigating MariTide for the treatment of Type 2 diabetes mellitus is enrolling adults living with and without obesity. Data readout is anticipated in H2 2025.
Planning for MARITIME, a broad Phase 3 program across multiple indications remains on track with the first studies expected to begin in H1 2025.
AMG 513
A Phase 1 study of AMG 513 in people living with obesity was placed on clinical hold by the U.S. Food and Drug Administration (FDA). Discussions are underway on a path forward to reopen the study.
Olpasiran (AMG 890)
Olpasiran is a potentially best-in-class small interfering ribonucleic acid (siRNA) molecule that reduces lipoprotein(a) (Lp(a)) synthesis in the liver.
The OCEAN(a)-Outcomes trial, a Phase 3 cardiovascular (CV) outcomes study, is ongoing in patients with atherosclerotic CV disease and elevated Lp(a).
A Phase 3 CV outcomes study in patients with elevated Lp(a) and at high risk for a CV event is expected to be initiated in H2 2025 / H1 2026.
Repatha
VESALIUS-CV, a Phase 3 CV outcomes study of Repatha, is ongoing in patients at high CV risk without prior myocardial infarction or stroke. Data readout is event driven and anticipated in H2 2025.
EVOLVE-MI, a Phase 4 study of Repatha administered within 10 days of an acute myocardial infarction to reduce the risk of CV events, is ongoing.
Rare Disease
TAVNEOS
A Phase 3, open-label study of TAVNEOS in combination with rituximab or a cyclophosphamide-containing regimen is enrolling patients from 6 years to < 18 years of age with active ANCA-associated vasculitis (Granulomatosis with Polyangiitis (GPA) / Microscopic Polyangiitis (MPA)).
TEPEZZA
Regulatory review is underway in multiple additional geographies including with the European Medicines Agency (EMA) where approval is anticipated in H2 2025.
A Phase 3 study of TEPEZZA in Japan is enrolling patients with chronic or low clinical activity score TED.
A Phase 3 study evaluating the subcutaneous route of administration of teprotumumab is enrolling patients with TED.
KRYSTEXXA
Data were presented in November 2024 from the AGILE study evaluating the safety, tolerability and efficacy of KRYSTEXXA administered with a shorter infusion duration in patients with uncontrolled gout receiving methotrexate as co-administration:
Safety and efficacy data from the 60-minute infusion duration cohort of the AGILE trial are similar to the MIRROR randomized clinical trial and current administration of KRYSTEXXA with methotrexate over at least 120 minutes.
U.S. Regulatory filing for AGILE is underway.
UPLIZNA
In January 2025, the FDA granted UPLIZNA Orphan Drug Designation for the treatment of generalized myasthenia gravis (gMG) based upon data from the Phase 3 MINT study. Regulatory filing activities are underway with submission anticipated to be complete in H1 2025.
In November 2024, data were presented and simultaneously published in the New England Journal of Medicine from the Phase 3 MITIGATE study evaluating UPLIZNA compared to placebo in patients with immunoglobin G4-related disease (IgG4-RD). In this study, UPLIZNA demonstrated a statistically significant 87% reduction in IgG4-RD flares, this primary endpoint and all key secondary endpoints were met. The safety results in the placebo-controlled period were consistent with the established safety profile of UPLIZNA.
The FDA accepted the regulatory submission for the Phase 3 MITIGATE study under priority review with a Prescription Drug User Fee Act (PDUFA) action date of April 3, 2025.
Dazodalibep
Dazodalibep is a fusion protein that inhibits CD40L.
Two Phase 3 studies of dazodalibep in Sjögren's disease are enrolling patients. The first study is in patients with moderate-to-severe systemic disease activity, and the second study is in patients with moderate-to-severe symptomatic burden and low systemic disease activity.
Daxdilimab
Daxdilimab is a fully human monoclonal antibody targeting immunoglobulin-like transcript 7 (ILT7).
A Phase 2 study of daxdilimab is ongoing in patients with moderate-to-severe active primary discoid lupus erythematosus refractory to standard of care.
A Phase 2 study of daxdilimab is ongoing in patients with dermatomyositis and antisynthetase inflammatory myositis.
Fipaxalparant
Fipaxalparant is a lysophosphatidic acid receptor 1 (LPAR1) antagonist.
A Phase 2 study of fipaxalparant in patients with diffuse cutaneous systemic sclerosis is complete. The study did not meet the primary or secondary endpoints. Further development of fipaxalparant in this indication will be discontinued.
Inflammation
TEZSPIRE
The Company is planning to initiate Phase 3 studies in patients with moderate-to-very severe chronic obstructive pulmonary disease (COPD) and a BEC ≥ 150 cells/µl. Study initiation is anticipated in H1 2025.
In December, the Company announced positive top-line results from the Phase 3 WAYPOINT trial in patients with chronic rhinosinusitis with nasal polyps:
Patients treated with TEZSPIRE had a statistically significant and clinically meaningful reduction in the size of nasal polyps and reduced nasal congestion compared to placebo.
The safety profile and tolerability of TEZSPIRE in the trial were consistent with the known profile of the medicine.
Regulatory submission is anticipated in H1 2025.
A Phase 3 study of TEZSPIRE is enrolling patients with eosinophilic esophagitis.
In severe asthma, the WAYFINDER Phase 3b study is complete and the PASSAGE Phase 4 real-world effectiveness study is ongoing. The SUNRISE Phase 3 study will be discontinued due to limited enrollment.
Rocatinlimab (AMG 451/KHK4083)
Rocatinlimab is a first-in-class T-cell rebalancing monoclonal antibody targeting the OX40 receptor.
The eight study ROCKET Phase 3 program evaluating rocatinlimab in patients with moderate-to-severe atopic dermatitis (AD) has enrolled over 3300 patients. Enrollment is now complete in seven studies.
Key milestones from the ROCKET Phase 3 program:
ROCKET SHUTTLE is a 24-week study evaluating rocatinlimab in combination with topical corticosteroids and/or topical calcineurin inhibitors in adult patients with moderate-to-severe AD. Data readout is anticipated in H1 2025.
ROCKET IGNITE is a 24-week study evaluating rocatinlimab monotherapy in adult patients with moderate-to-severe AD. Data readout is anticipated in H1 2025.
ROCKET ASCEND is a study evaluating rocatinlimab maintenance therapy in adult and adolescent patients with moderate-to-severe AD. Data readout is anticipated in H2 2025.
ROCKET ASTRO is a 52-week study evaluating rocatinlimab in adolescent patients with moderate-to-severe AD. Data readout is anticipated in H2 2025.
A Phase 2 study of rocatinlimab is enrolling patients with moderate-to-severe asthma.
A Phase 3 study of rocatinlimab is enrolling patients with prurigo nodularis.
Otezla
In November 2024, we made six data presentations at the American College of Rheumatology (ACR). Notable highlights include:
Otezla reduces axial inflammation in patients with psoriatic arthritis (PsA) as assessed by CANDEN Magnetic Resonance Imaging Scoring, results From the Phase 4 MOSAIC study.
FOREMOST oligoarticular PsA data presentations (4 posters), including 48-week data and data at 16 weeks showing that Otezla was associated with fewer patients progressing from < 4 to > 4 active joints when compared to placebo suggesting that Otezla reduced the progression from oligoarticular to polyarticular PsA.
Blinatumomab
Blinatumomab is a BiTE molecule targeting CD19.
A Phase 2 study of blinatumomab in autoimmune disease was initiated in patients with systemic lupus erythematosus (SLE).
Inebilizumab
Inebilizumab is a monoclonal antibody targeting CD19.
A Phase 2 study of inebilizumab in autoimmune disease was initiated in patients with SLE.
Ordesekimab (AMG 714/PRV-015)
Ordesekimab is a monoclonal antibody that binds interleukin-15.
A Phase 2b study of ordesekimab, conducted by Provention Bio, a Sanofi Company, in patients with nonresponsive celiac disease was completed as planned and did not meet primary or secondary endpoints. No safety concerns were noted.
AMG 104 (AZD8630)
AMG 104 is an inhaled anti-thymic stromal lymphopoietin (TSLP) fragment antigen-binding (Fab).
A Phase 2 study is enrolling patients with asthma.
Oncology
BLINCYTO
In December 2024, data from a Phase 3 study (AALL1731) conducted by the Children's Oncology Group, were presented and simultaneously published in the New England Journal of Medicine. These data demonstrated that BLINCYTO added to chemotherapy significantly improves disease-free survival in newly diagnosed pediatric patients with National Cancer Institute standard risk B-cell precursor acute lymphoblastic leukemia (B-ALL) of average or higher risk of relapse.
Golden Gate, a Phase 3 study of BLINCYTO alternating with low-intensity chemotherapy, is enrolling older adult patients with newly diagnosed Philadelphia chromosome (Ph)-negative B-ALL.
A Phase 1/2 study of subcutaneous blinatumomab is ongoing in the dose-expansion and optimization phase in adult patients with relapsed or refractory Ph-negative B-ALL. The Company is planning to advance blinatumomab subcutaneous administration to a potentially registration-enabling Phase 2 portion of this study with initiation in H2 2025.
IMDELLTRA
IMDELLTRA is a first-in-class delta-like ligand 3 (DLL3) targeting BiTE molecule.
In 2024, IMDELLTRA received accelerated approval in the U.S. for the treatment of adult patients with extensive-stage small cell lung cancer (ES-SCLC) with disease progression on or after platinum-based chemotherapy. Marketing authorizations have subsequently been granted in Japan and in additional countries, including Canada, Brazil, Israel and Great Britain.
The Company is advancing a comprehensive, global clinical development program across extensive-stage and limited-stage small cell lung cancer (SCLC):
DeLLphi-304, a Phase 3 study of IMDELLTRA in second-line ES-SCLC, is ongoing. Data readout is anticipated in H1 2025.
DeLLphi-305, a Phase 3 study of IMDELLTRA and durvalumab is enrolling patients with first-line ES-SCLC in the maintenance setting.
DeLLphi-306, a Phase 3 study of IMDELLTRA following concurrent chemoradiation therapy, is enrolling patients with limited-stage SCLC.
DeLLphi-308, a Phase 1b study evaluating subcutaneous tarlatamab, is enrolling patients with second line or later ES-SCLC.
DeLLphi-309, a Phase 2 study evaluating alternative intravenous dosing regimens in second-line ES-SCLC, was initiated.
DeLLphi-303, a Phase 1b study of IMDELLTRA in combination with a programmed cell death protein ligand-1 (PD-L1) inhibitor, carboplatin and etoposide or separately in combination with PD-L1 alone, is ongoing in patients with first-line ES-SCLC.
Xaluritamig (AMG 509)
Xaluritamig is a first-in-class bispecific T-cell engager targeting six-transmembrane epithelial antigen of prostate 1 (STEAP1).
A Phase 3 study of xaluritamig is enrolling patients with metastatic castrate resistant prostate cancer (mCRPC) who have previously been treated with taxane-based chemotherapy.
A Phase 1 monotherapy dose-expansion study of xaluritamig is enrolling mCRPC patients who have not yet received taxane-based chemotherapy and to enroll patients in a fully outpatient treatment setting to further improve administration convenience.
A Phase 1 combination of xaluritamig with enzalutamide or abiraterone is enrolling patients with mCRPC in dose-escalation and dose-expansion respectively.
A Phase 1b study evaluating neoadjuvant xaluritamig therapy prior to radical prostatectomy is enrolling patients with newly diagnosed localized intermediate or high–risk prostate cancer.
A Phase 1b study of xaluritamig is enrolling patients with high-risk biochemically recurrent prostate cancer after definitive therapy.
AMG 193
AMG 193 is a first-in-class small molecule methylthioadenosine (MTA)-cooperative protein arginine methyltransferase 5 (PRMT5) inhibitor.
A Phase 2 study evaluating the efficacy, safety, tolerability and pharmacokinetics of AMG 193 is enrolling patients with methylthioadenosine phosphorylase (MTAP)-null previously treated advanced non-small cell lung cancer (NSCLC).
A Phase 1/1b/2 study of AMG 193 is enrolling patients with advanced MTAP-null solid tumors in the dose-expansion portion of the study.
A Phase 1b study of AMG 193 alone or in combination with other therapies is enrolling patients with advanced MTAP-null thoracic malignancies.
A Phase 1b study of AMG 193 in combination with other therapies is enrolling patients with advanced MTAP-null gastrointestinal, biliary tract, and pancreatic cancers.
A Phase 1/2 study of AMG 193 in combination with IDE397, an investigational methionine adenosyltransferase 2A (MAT2A) inhibitor, is enrolling patients with advanced MTAP-null solid tumors.
Bemarituzumab
Bemarituzumab is a first-in-class fibroblast growth factor receptor 2b (FGFR2b) targeting monoclonal antibody.
FORTITUDE-101, a Phase 3 study of bemarituzumab plus chemotherapy, is ongoing in patients with first-line gastric cancer. Data readout is anticipated in H1 2025.
FORTITUDE-102, a Phase 1b/3 study of bemarituzumab plus chemotherapy and nivolumab is ongoing in patients with first-line gastric cancer. Phase 3 data readout is anticipated in H2 2025.
FORTITUDE-103, a Phase 1b/2 study of bemarituzumab plus oral chemotherapy regimens with or without nivolumab, is enrolling patients with first-line gastric cancer.
FORTITUDE-301, a Phase 1b/2 basket study of bemarituzumab monotherapy, is ongoing in patients with solid tumors with FGFR2b overexpression.
LUMAKRAS/LUMYKRAS
In January 2025, the FDA approved LUMAKRAS in combination with Vectibix as a targeted, biomarker-driven combination therapy for the treatment of adult patients with KRAS G12C-mutated metastatic colorectal cancer (mCRC), as determined by an FDA-approved test, who have received prior fluoropyrimidine-, oxaliplatin- and irinotecan-based chemotherapy.
CodeBreaK 301, a Phase 3 study of LUMAKRAS in combination with Vectibix and FOLFIRI, is enrolling patients with first-line KRAS G12C–mutated CRC.
Regulatory review by the EMA of the CodeBreaK 200 Phase 3 study of adults with previously treated locally advanced or metastatic KRAS G12C–mutated NSCLC concluded with the conditional status of marketing authorization maintained.
CodeBreaK 202 (CB202), a Phase 3 study of LUMAKRAS plus chemotherapy vs. pembrolizumab plus chemotherapy, is enrolling patients with first-line KRAS G12C–mutated and PD-L1 negative advanced NSCLC.
The ongoing CB202 study is being conducted to serve as confirmatory study and to support conversion of accelerated/conditional approval to full approval, in the US, EU and other regions where applicable.
Nplate
The primary analysis of a Phase 3 study of Nplate as supportive care in chemotherapy-induced thrombocytopenia in gastrointestinal malignancies is complete. The Company continues to follow patients through a planned final analysis in H1 2025. Data presentation at a medical congress is anticipated in mid-2025.
Biosimilars
A randomized, double-blind pharmacokinetic similarity study of ABP 206 compared with OPDIVO® (nivolumab) is enrolling patients with resected stage III or stage IV melanoma in the adjuvant setting. Data readout is anticipated in H2 2025.
A randomized, double-blind comparative clinical study of ABP 206 compared with OPDIVO is enrolling patients with treatment-naïve unresectable or metastatic melanoma.
A randomized, double-blind pharmacokinetic similarity study of ABP 234 compared with KEYTRUDA® (pembrolizumab) is enrolling patients with early-stage non-squamous non-small cell lung cancer as adjuvant treatment.
A randomized, double-blind combined pharmacokinetic/comparative clinical study of ABP 234 compared to KEYTRUDA is enrolling patients with advanced or metastatic non-squamous non-small cell lung cancer.
A randomized, double-blind, pharmacokinetic similarity/comparative clinical study of ABP 692 and OCREVUS® (ocrelizumab) was initiated and is currently enrolling patients with relapsing-remitting multiple sclerosis.
TEZSPIRE is being developed in collaboration with AstraZeneca.
AMG 104 is being developed in collaboration with AstraZeneca.
Rocatinlimab, formerly AMG 451/KHK4083, is being developed in collaboration with Kyowa Kirin.
Ordesekimab, formerly AMG 714 and also known as PRV-015, is being developed in collaboration with Provention Bio, a Sanofi Company. For the purposes of the collaboration, Provention Bio conducts a clinical trial and leads certain development and regulatory activities for the program.
Xaluritamig, formerly AMG 509, is being developed pursuant to a research collaboration with Xencor, Inc.
IDE397 is an investigational MAT2A inhibitor from IDEAYA Biosciences.
OPDIVO is a registered trademark of Bristol-Myers Squibb Company.
KEYTRUDA is a registered trademark of Merck & Co., Inc.
OCREVUS is a registered trademark of Genentech, Inc.
Non-GAAP Financial Measures
In this news release, management has presented its operating results for the fourth quarters and full years of 2024 and 2023, in accordance with U.S. Generally Accepted Accounting Principles (GAAP) and on a non-GAAP basis. In addition, management has presented its full year 2025 EPS and tax guidance in accordance with GAAP and on a non-GAAP basis. These non-GAAP financial measures are computed by excluding certain items related to acquisitions, divestitures, restructuring and certain other items from the related GAAP financial measures. Management has presented Free Cash Flow (FCF), which is a non-GAAP financial measure, for the fourth quarters and full years of 2024 and 2023. FCF is computed by subtracting capital expenditures from operating cash flow, each as determined in accordance with GAAP. Management has also presented Earnings Before Interest, Taxes, Depreciation and Amortization (EBITDA) and debt leverage ratio for 2024, both of which are non-GAAP financial measures. EBITDA is computed by adding interest expense, provision for income taxes, and depreciation and amortization expense to GAAP net income. Debt leverage ratio is calculated as the ratio of GAAP total debt to EBITDA.
The Company believes that its presentation of non-GAAP financial measures provides useful supplementary information to and facilitates additional analysis by investors. The Company uses certain non-GAAP financial measures to enhance an investor's overall understanding of the financial performance and prospects for the future of the Company's normal and recurring business activities by facilitating comparisons of results of normal and recurring business operations among current, past and future periods. The Company believes that FCF provides a further measure of the Company's liquidity. The Company believes its debt leverage ratio provides a supplemental operating metric for the full year period as it compares the amount of cash generated by our operations for the year.
The Company uses the non-GAAP financial measures set forth in the news release in connection with its own budgeting and financial planning internally to evaluate the performance of the business, including to allocate resources and to evaluate results relative to incentive compensation targets. The non-GAAP financial measures are in addition to, not a substitute for, or superior to, measures of financial performance prepared in accordance with GAAP.
About Amgen
Amgen discovers, develops, manufactures and delivers innovative medicines to help millions of patients in their fight against some of the world's toughest diseases. More than 40 years ago, Amgen helped to establish the biotechnology industry and remains on the cutting-edge of innovation, using technology and human genetic data to push beyond what's known today. Amgen is advancing a broad and deep pipeline that builds on its existing portfolio of medicines to treat cancer, heart disease, osteoporosis, inflammatory diseases and rare diseases.
In 2024, Amgen was named one of the "World's Most Innovative Companies" by Fast Company and one of "America's Best Large Employers" by Forbes, among other external recognitions. Amgen is one of the 30 companies that comprise the Dow Jones Industrial Average®, and it is also part of the Nasdaq-100 Index®, which includes the largest and most innovative non-financial companies listed on the Nasdaq Stock Market based on market capitalization.
For more information, visit Amgen.com and follow Amgen on X, LinkedIn, Instagram, YouTube and Threads.
Forward-Looking Statements
This news release contains forward-looking statements that are based on the current expectations and beliefs of Amgen. All statements, other than statements of historical fact, are statements that could be deemed forward-looking statements, including any statements on the outcome, benefits and synergies of collaborations, or potential collaborations, with any other company (including BeiGene, Ltd. or Kyowa Kirin Co., Ltd.), the performance of Otezla® (apremilast) (including anticipated Otezla sales growth and the timing of non-GAAP EPS accretion), our acquisitions of Teneobio, Inc., ChemoCentryx, Inc., or Horizon (including the prospective performance and outlook of Horizon's business, performance and opportunities and any potential strategic benefits, synergies or opportunities expected as a result of such acquisition, and any projected impacts from the Horizon acquisition on our acquisition-related expenses going forward), as well as estimates of revenues, operating margins, capital expenditures, cash, other financial metrics, expected legal, arbitration, political, regulatory or clinical results or practices, customer and prescriber patterns or practices, reimbursement activities and outcomes, effects of pandemics or other widespread health problems on our business, outcomes, progress, and other such estimates and results. Forward-looking statements involve significant risks and uncertainties, including those discussed below and more fully described in the Securities and Exchange Commission reports filed by Amgen, including our most recent annual report on Form 10-K and any subsequent periodic reports on Form 10-Q and current reports on Form 8-K. Unless otherwise noted, Amgen is providing this information as of the date of this news release and does not undertake any obligation to update any forward-looking statements contained in this document as a result of new information, future events or otherwise.
No forward-looking statement can be guaranteed and actual results may differ materially from those we project. Our results may be affected by our ability to successfully market both new and existing products domestically and internationally, clinical and regulatory developments involving current and future products, sales growth of recently launched products, competition from other products including biosimilars, difficulties or delays in manufacturing our products and global economic conditions. In addition, sales of our products are affected by pricing pressure, political and public scrutiny and reimbursement policies imposed by third-party payers, including governments, private insurance plans and managed care providers and may be affected by regulatory, clinical and guideline developments and domestic and international trends toward managed care and healthcare cost containment. Furthermore, our research, testing, pricing, marketing and other operations are subject to extensive regulation by domestic and foreign government regulatory authorities. We or others could identify safety, side effects or manufacturing problems with our products, including our devices, after they are on the market. Our business may be impacted by government investigations, litigation and product liability claims. In addition, our business may be impacted by the adoption of new tax legislation or exposure to additional tax liabilities. If we fail to meet the compliance obligations in the corporate integrity agreement between us and the U.S. government, we could become subject to significant sanctions. Further, while we routinely obtain patents for our products and technology, the protection offered by our patents and patent applications may be challenged, invalidated or circumvented by our competitors, or we may fail to prevail in present and future intellectual property litigation. We perform a substantial amount of our commercial manufacturing activities at a few key facilities, including in Puerto Rico, and also depend on third parties for a portion of our manufacturing activities, and limits on supply may constrain sales of certain of our current products and product candidate development. An outbreak of disease or similar public health threat, such as COVID-19, and the public and governmental effort to mitigate against the spread of such disease, could have a significant adverse effect on the supply of materials for our manufacturing activities, the distribution of our products, the commercialization of our product candidates, and our clinical trial operations, and any such events may have a material adverse effect on our product development, product sales, business and results of operations. We rely on collaborations with third parties for the development of some of our product candidates and for the commercialization and sales of some of our commercial products. In addition, we compete with other companies with respect to many of our marketed products as well as for the discovery and development of new products. Discovery or identification of new product candidates or development of new indications for existing products cannot be guaranteed and movement from concept to product is uncertain; consequently, there can be no guarantee that any particular product candidate or development of a new indication for an existing product will be successful and become a commercial product. Further, some raw materials, medical devices and component parts for our products are supplied by sole third-party suppliers. Certain of our distributors, customers and payers have substantial purchasing leverage in their dealings with us. The discovery of significant problems with a product similar to one of our products that implicate an entire class of products could have a material adverse effect on sales of the affected products and on our business and results of operations. Our efforts to collaborate with or acquire other companies, products or technology, and to integrate the operations of companies or to support the products or technology we have acquired, may not be successful. There can be no guarantee that we will be able to realize any of the strategic benefits, synergies or opportunities arising from the Horizon acquisition, and such benefits, synergies or opportunities may take longer to realize than expected. We may not be able to successfully integrate Horizon, and such integration may take longer, be more difficult or cost more than expected. A breakdown, cyberattack or information security breach of our information technology systems could compromise the confidentiality, integrity and availability of our systems and our data. Our stock price is volatile and may be affected by a number of events. Our business and operations may be negatively affected by the failure, or perceived failure, of achieving our environmental, social and governance objectives. The effects of global climate change and related natural disasters could negatively affect our business and operations. Global economic conditions may magnify certain risks that affect our business. Our business performance could affect or limit the ability of our Board of Directors to declare a dividend or our ability to pay a dividend or repurchase our common stock. We may not be able to access the capital and credit markets on terms that are favorable to us, or at all.
CONTACT: Amgen, Thousand Oaks
Elissa Snook, 609-251-1407 (media)
Justin Claeys, 805-313-9775 (investors)
Our GAAP diluted EPS guidance does not include the effect of GAAP adjustments triggered by events that may occur subsequent to this press release such as acquisitions, asset impairments, litigation, changes in fair value of our contingent consideration obligations and changes in fair value of our equity investments.
SOURCE Amgen
WANT YOUR COMPANY'S NEWS FEATURED ON PRNEWSWIRE.COM?
440k+
Newsrooms &
Influencers
9k+
Digital Media
Outlets
270k+
Journalists
Opted In
GET STARTED
上市批准并购财报临床2期临床结果
2025-02-04
THOUSAND OAKS, Calif.
,
Feb. 4, 2025
/PRNewswire/ --
Amgen
(NASDAQ:AMGN) today announced financial results for the fourth quarter and full year of 2024 versus comparable periods in 2023.
"Robust growth in sales and earnings throughout 2024 reflects the momentum of our business. With strong performance globally, we are investing heavily in our rapidly advancing pipeline to deliver innovative therapies across our four therapeutic areas," said Robert A. Bradway, chairman and chief executive officer.
Key results include:
References in this release to "non-GAAP" measures, measures presented "on a non-GAAP basis," "free cash flow" (computed by subtracting capital expenditures from operating cash flow), "EBITDA, or earnings before interest, taxes, depreciation and amortization" (computed by adding interest expense, provision for income taxes, and depreciation and amortization expense to GAAP net income) and "debt leverage ratio" (calculated as the ratio of GAAP total debt to EBITDA) refer to non-GAAP financial measures. Adjustments to the most directly comparable GAAP financial measures and other items are presented on the attached reconciliations. Refer to Non-GAAP Financial Measures below for further discussion.
Product Sales Performance
General Medicine
Rare Disease
Except for TAVNEOS
®
, the products listed below were added through the acquisition of Horizon on Oct. 6, 2023.
Inflammation
Oncology
Established Products
Product Sales Detail by Product and
$Millions, except percentages
Q4 '24
Q4 '23
YOY Δ
U.S
ROW
TOTAL
TOTAL
TOTAL
Repatha
®
$ 315
$ 291
$ 606
$ 417
45 %
EVENITY
®
325
106
431
318
36 %
Prolia
®
775
390
1,165
1,107
5 %
TEPEZZA
®(1)
456
4
460
448
3 %
KRYSTEXXA
®(1)
346
—
346
272
27 %
UPLIZNA
®(1)
93
8
101
65
55 %
TAVNEOS
®
76
5
81
44
84 %
Ultra-Rare products
(1)
205
9
214
164
30 %
TEZSPIRE
®
296
—
296
177
67 %
Otezla
®
514
110
624
629
(1 %)
Enbrel
®
1,008
7
1,015
1,015
— %
AMJEVITA
®
/AMGEVITA
™
153
141
294
160
84 %
BLINCYTO
®
245
136
381
241
58 %
Vectibix
®
134
112
246
251
(2 %)
KYPROLIS
®
236
136
372
350
6 %
LUMAKRAS
®
/LUMYKRAS
™
53
32
85
77
10 %
XGEVA
®
369
192
561
527
6 %
Nplate
®
221
116
337
386
(13 %)
IMDELLTRA
®
67
—
67
—
N/A
MVASI
®
108
65
173
188
(8 %)
EPOGEN
®
19
—
19
55
(65 %)
Aranesp
®
90
218
308
319
(3 %)
Parsabiv
®
39
36
75
89
(16 %)
Neulasta
®
72
26
98
239
(59 %)
Other products
(2)
294
67
361
295
22 %
Total product sales
$ 6,509
$ 2,207
$ 8,716
$ 7,833
11 %
N/A = not applicable
(1)
Horizon-acquired products, and the Ultra-Rare products consist of RAVICTI
®
, PROCYSBI
®
, ACTIMMUNE
®
,
QUINSAIR
®
and BUPHENYL
®
.
(2)
Consists of (i) Aimovig
®
, KANJINTI
®
, AVSOLA
®
, RIABNI
®
, PAVBLU
™
, NEUPOGEN
®
, WEZLANA
™
/WEZENLA
™
,
BEKEMV
™
, IMLYGIC
®
, Corlanor
®
and Sensipar
®
/Mimpara
™
, where Biosimilars total $218 million in Q4 '24
and $135 million in Q4 '23; and (ii) Horizon-acquired products including RAYOS
®
, PENNSAID
®
and DUEXIS
®
.
$Millions, except percentages
FY '24
FY '23
YOY Δ
US
ROW
TOTAL
TOTAL
TOTAL
Repatha
®
$ 1,139
$ 1,083
$ 2,222
$ 1,635
36 %
EVENITY
®
1,131
432
1,563
1,160
35 %
Prolia
®
2,885
1,489
4,374
4,048
8 %
TEPEZZA
®(1)
1,835
16
1,851
448
*
KRYSTEXXA
®(1)
1,185
—
1,185
272
*
UPLIZNA
®(1)
314
65
379
65
*
TAVNEOS
®
256
27
283
134
*
Ultra-Rare products
(1)
726
32
758
164
*
TEZSPIRE
®
972
—
972
567
71 %
Otezla
®
1,699
427
2,126
2,188
(3 %)
Enbrel
®
3,288
28
3,316
3,697
(10 %)
AMJEVITA
®
/AMGEVITA
™
202
559
761
626
22 %
BLINCYTO
®
800
416
1,216
861
41 %
Vectibix
®
519
526
1,045
984
6 %
KYPROLIS
®
948
555
1,503
1,403
7 %
LUMAKRAS
®
/LUMYKRAS
™
214
136
350
280
25 %
XGEVA
®
1,507
718
2,225
2,112
5 %
Nplate
®
970
486
1,456
1,477
(1 %)
IMDELLTRA
®
115
—
115
—
N/A
MVASI
®
449
278
727
800
(9 %)
EPOGEN
®
125
—
125
226
(45 %)
Aranesp
®
386
956
1,342
1,362
(1 %)
Parsabiv
®
203
153
356
362
(2 %)
Neulasta
®
318
113
431
848
(49 %)
Other products
(2)
1,115
230
1,345
1,191
13 %
Total product sales
$ 23,301
$ 8,725
$ 32,026
$ 26,910
19 %
* Change in excess of 100%
N/A = not applicable
(1)
Horizon-acquired products, and the Ultra-Rare products consist of RAVICTI
®
, PROCYSBI
®
, ACTIMMUNE
®
,
BUPHENYL
®
and QUINSAIR
®
.
(2)
Consists of (i)Aimovig
®
,KANJINTI
®
,RIABNI
®
,AVSOLA
®
,NEUPOGEN
®
,Corlanor
®
,
IMLYGIC
®
,BEKEMV
™
,
PAVBLU
™
, WEZLANA
™
/WEZENLA
™
and Sensipar
®
/Mimpara
™
, where Biosimilars total $725 million in FY '24
and $490 million in FY '23; and (ii) Horizon-acquired products including RAYOS
®
, PENNSAID
®
and DUEXIS
®
.
Operating Expense, Operating Margin and Tax Rate Analysis
On a GAAP basis:
On a non-GAAP basis:
$Millions, except percentages
GAAP
Non-GAAP
Q4 '24
Q4 '23
YOY Δ
Q4 '24
Q4 '23
YOY Δ
Cost of Sales
$ 3,112
$ 3,112
— %
$ 1,536
$ 1,278
20 %
% of product sales
35.7 %
39.7 %
(4.0) pts
17.6 %
16.3 %
1.3 pts
Research & Development
$ 1,724
$ 1,534
12 %
$ 1,698
$ 1,494
14 %
% of product sales
19.8 %
19.6 %
0.2 pts
19.5 %
19.1 %
0.4 pts
Selling, General & Administrative
$ 1,878
$ 2,274
(17 %)
$ 1,819
$ 1,764
3 %
% of product sales
21.5 %
29.0 %
(7.5) pts
20.9 %
22.5 %
(1.6) pts
Other
$ 61
$ 5
*
$ —
$ —
N/A
Total Operating Expenses
$ 6,775
$ 6,925
(2 %)
$ 5,053
$ 4,536
11 %
Operating Margin
operating income as % of product sales
26.5 %
16.2 %
10.3 pts
46.3 %
46.7 %
(0.4) pts
Tax Rate
19.8 %
10.0 %
9.8 pts
14.8 %
15.9 %
(1.1) pts
pts: percentage points
* change in excess of 100%
N/A = not applicable
$Millions, except percentages
GAAP
Non-GAAP
FY '24
FY '23
YOY Δ
FY '24
FY '23
YOY Δ
Cost of Sales
$ 12,858
$ 8,451
52 %
$ 5,736
$ 4,573
25 %
% of product sales
40.1 %
31.4 %
8.7 pts
17.9 %
17.0 %
0.9 pts
Research & Development
$ 5,964
$ 4,784
25 %
$ 5,878
$ 4,700
25 %
% of product sales
18.6 %
17.8 %
0.8 pts
18.4 %
17.5 %
0.9 pts
Selling, General & Administrative
$ 7,096
$ 6,179
15 %
$ 6,782
$ 5,518
23 %
% of product sales
22.2 %
23.0 %
(0.8) pts
21.2 %
20.5 %
0.7 pts
Other
$ 248
$ 879
(72 %)
$ —
$ —
N/A
Total Operating Expenses
$ 26,166
$ 20,293
29 %
$ 18,396
$ 14,791
24 %
Operating Margin
operating income as % of product sales
22.7 %
29.3 %
(6.6) pts
46.9 %
49.8 %
(2.9) pts
Tax Rate
11.3 %
14.5 %
(3.2) pts
14.5 %
16.5 %
(2.0) pts
pts: percentage points
N/A = not applicable
Cash Flow and Balance Sheet
$Billions, except shares
Q4 '24
Q4 '23
YOY Δ
FY '24
FY '23
YOY Δ
Operating Cash Flow
$ 4.8
$ 0.5
$ 4.2
$ 11.5
$ 8.5
$ 3.0
Capital Expenditures
$ 0.4
$ 0.2
$ 0.1
$ 1.1
$ 1.1
$ 0.0
Free Cash Flow
$ 4.4
$ 0.3
$ 4.1
$ 10.4
$ 7.4
$ 3.0
Dividends Paid
$ 1.2
$ 1.1
$ 0.1
$ 4.8
$ 4.6
$ 0.3
Share Repurchases
$ 0.2
$ —
$ 0.2
$ 0.2
$ —
$ 0.2
Average Diluted Shares (millions)
542
540
2
541
538
3
Note: Numbers may not add due to rounding
$Billions
12/31/24
12/31/23
YTD Δ
Cash and Investments
$ 12.0
$ 10.9
$ 1.0
Debt Outstanding
$ 60.1
$ 64.6
$ (4.5)
Note: Numbers may not add due to rounding
2025 Guidance
For the full year 2025, the Company expects:
Fourth Quarter Product and Pipeline Update
The Company provided the following updates on selected product and pipeline programs:
General Medicine
MariTide (maridebart cafraglutide, AMG 133)
AMG 513
Olpasiran (AMG 890)
Repatha
Rare Disease
TAVNEOS
TEPEZZA
KRYSTEXXA
UPLIZNA
Dazodalibep
Daxdilimab
Fipaxalparant
Inflammation
TEZSPIRE
Rocatinlimab (AMG 451/KHK4083)
Otezla
Blinatumomab
Inebilizumab
Ordesekimab (AMG 714/PRV-015)
AMG 104 (AZD8630)
Oncology
BLINCYTO
IMDELLTRA
Xaluritamig (AMG 509)
AMG 193
Bemarituzumab
LUMAKRAS/LUMYKRAS
Nplate
Biosimilars
TEZSPIRE is being developed in collaboration with AstraZeneca.
AMG 104 is being developed in collaboration with AstraZeneca.
Rocatinlimab, formerly AMG 451/KHK4083, is being developed in collaboration with Kyowa Kirin.
Ordesekimab, formerly AMG 714 and also known as PRV-015, is being developed in collaboration with
Provention Bio
, a
Sanofi Company
. For the purposes of the collaboration,
Provention Bio
conducts a clinical trial and leads certain development and regulatory activities for the program.
Xaluritamig, formerly AMG 509, is being developed pursuant to a research collaboration with Xencor, Inc.
IDE397 is an investigational MAT2A inhibitor from IDEAYA Biosciences.
OPDIVO is a registered trademark of Bristol-Myers Squibb Company.
KEYTRUDA is a registered trademark of Merck & Co., Inc.
OCREVUS is a registered trademark of
Genentech, Inc.
Non-GAAP Financial Measures
In this news release, management has presented its operating results for the fourth quarters and full years of 2024 and 2023, in accordance with U.S. Generally Accepted Accounting Principles (GAAP) and on a non-GAAP basis. In addition, management has presented its full year 2025 EPS and tax guidance in accordance with GAAP and on a non-GAAP basis. These non-GAAP financial measures are computed by excluding certain items related to acquisitions, divestitures, restructuring and certain other items from the related GAAP financial measures. Management has presented Free Cash Flow (FCF), which is a non-GAAP financial measure, for the fourth quarters and full years of 2024 and 2023. FCF is computed by subtracting capital expenditures from operating cash flow, each as determined in accordance with GAAP. Management has also presented Earnings Before Interest, Taxes, Depreciation and Amortization (EBITDA) and debt leverage ratio for 2024, both of which are non-GAAP financial measures. EBITDA is computed by adding interest expense, provision for income taxes, and depreciation and amortization expense to GAAP net income. Debt leverage ratio is calculated as the ratio of GAAP total debt to EBITDA.
The Company believes that its presentation of non-GAAP financial measures provides useful supplementary information to and facilitates additional analysis by investors. The Company uses certain non-GAAP financial measures to enhance an investor's overall understanding of the financial performance and prospects for the future of the Company's normal and recurring business activities by facilitating comparisons of results of normal and recurring business operations among current, past and future periods. The Company believes that FCF provides a further measure of the Company's liquidity. The Company believes its debt leverage ratio provides a supplemental operating metric for the full year period as it compares the amount of cash generated by our operations for the year.
The Company uses the non-GAAP financial measures set forth in the news release in connection with its own budgeting and financial planning internally to evaluate the performance of the business, including to allocate resources and to evaluate results relative to incentive compensation targets. The non-GAAP financial measures are in addition to, not a substitute for, or superior to, measures of financial performance prepared in accordance with GAAP.
About
Amgen
Amgen
discovers, develops, manufactures and delivers innovative medicines to help millions of patients in their fight against some of the world's toughest diseases. More than 40 years ago,
Amgen
helped to establish the biotechnology industry and remains on the cutting-edge of innovation, using technology and human genetic data to push beyond what's known today.
Amgen
is advancing a broad and deep pipeline that builds on its existing portfolio of medicines to treat cancer, heart disease, osteoporosis, inflammatory diseases and rare diseases.
In 2024,
Amgen
was named one of the "World's Most Innovative Companies" by
Fast Company
and one of "America's Best Large Employers" by Forbes, among other external recognitions.
Amgen
is one of the 30 companies that comprise the Dow Jones Industrial Average
®
, and it is also part of the Nasdaq-100 Index
®
, which includes the largest and most innovative non-financial companies listed on the
Nasdaq Stock Market
based on market capitalization.
For more information, visit
Amgen.com
and follow
Amgen
on
X
,
LinkedIn
,
Instagram
,
YouTube
and
Threads
.
Forward-Looking Statements
This news release contains forward-looking statements that are based on the current expectations and beliefs of
Amgen
. All statements, other than statements of historical fact, are statements that could be deemed forward-looking statements, including any statements on the outcome, benefits and synergies of collaborations, or potential collaborations, with any other company (including BeiGene, Ltd. or Kyowa Kirin Co., Ltd.), the performance of Otezla
®
(apremilast) (including anticipated Otezla sales growth and the timing of non-GAAP EPS accretion), our acquisitions of
Teneobio, Inc.
,
ChemoCentryx, Inc.
, or Horizon (including the prospective performance and outlook of Horizon's business, performance and opportunities and any potential strategic benefits, synergies or opportunities expected as a result of such acquisition, and any projected impacts from the Horizon acquisition on our acquisition-related expenses going forward), as well as estimates of revenues, operating margins, capital expenditures, cash, other financial metrics, expected legal, arbitration, political, regulatory or clinical results or practices, customer and prescriber patterns or practices, reimbursement activities and outcomes, effects of pandemics or other widespread health problems on our business, outcomes, progress, and other such estimates and results. Forward-looking statements involve significant risks and uncertainties, including those discussed below and more fully described in the
Securities and Exchange Commission
reports filed by
Amgen
, including our most recent annual report on Form 10-K and any subsequent periodic reports on Form 10-Q and current reports on Form 8-K. Unless otherwise noted,
Amgen
is providing this information as of the date of this news release and does not undertake any obligation to update any forward-looking statements contained in this document as a result of new information, future events or otherwise.
No forward-looking statement can be guaranteed and actual results may differ materially from those we project. Our results may be affected by our ability to successfully market both new and existing products domestically and internationally, clinical and regulatory developments involving current and future products, sales growth of recently launched products, competition from other products including biosimilars, difficulties or delays in manufacturing our products and global economic conditions. In addition, sales of our products are affected by pricing pressure, political and public scrutiny and reimbursement policies imposed by third-party payers, including governments, private insurance plans and managed care providers and may be affected by regulatory, clinical and guideline developments and domestic and international trends toward managed care and healthcare cost containment. Furthermore, our research, testing, pricing, marketing and other operations are subject to extensive regulation by domestic and foreign government regulatory authorities. We or others could identify safety, side effects or manufacturing problems with our products, including our devices, after they are on the market. Our business may be impacted by government investigations, litigation and product liability claims. In addition, our business may be impacted by the adoption of new tax legislation or exposure to additional tax liabilities. If we fail to meet the compliance obligations in the corporate integrity agreement between us and the U.S. government, we could become subject to significant sanctions. Further, while we routinely obtain patents for our products and technology, the protection offered by our patents and patent applications may be challenged, invalidated or circumvented by our competitors, or we may fail to prevail in present and future intellectual property litigation. We perform a substantial amount of our commercial manufacturing activities at a few key facilities, including in Puerto Rico, and also depend on third parties for a portion of our manufacturing activities, and limits on supply may constrain sales of certain of our current products and product candidate development. An outbreak of disease or similar public health threat, such as COVID-19, and the public and governmental effort to mitigate against the spread of such disease, could have a significant adverse effect on the supply of materials for our manufacturing activities, the distribution of our products, the commercialization of our product candidates, and our clinical trial operations, and any such events may have a material adverse effect on our product development, product sales, business and results of operations. We rely on collaborations with third parties for the development of some of our product candidates and for the commercialization and sales of some of our commercial products. In addition, we compete with other companies with respect to many of our marketed products as well as for the discovery and development of new products. Discovery or identification of new product candidates or development of new indications for existing products cannot be guaranteed and movement from concept to product is uncertain; consequently, there can be no guarantee that any particular product candidate or development of a new indication for an existing product will be successful and become a commercial product. Further, some raw materials, medical devices and component parts for our products are supplied by sole third-party suppliers. Certain of our distributors, customers and payers have substantial purchasing leverage in their dealings with us. The discovery of significant problems with a product similar to one of our products that implicate an entire class of products could have a material adverse effect on sales of the affected products and on our business and results of operations. Our efforts to collaborate with or acquire other companies, products or technology, and to integrate the operations of companies or to support the products or technology we have acquired, may not be successful. There can be no guarantee that we will be able to realize any of the strategic benefits, synergies or opportunities arising from the Horizon acquisition, and such benefits, synergies or opportunities may take longer to realize than expected. We may not be able to successfully integrate Horizon, and such integration may take longer, be more difficult or cost more than expected. A breakdown, cyberattack or information security breach of our information technology systems could compromise the confidentiality, integrity and availability of our systems and our data. Our stock price is volatile and may be affected by a number of events. Our business and operations may be negatively affected by the failure, or perceived failure, of achieving our environmental, social and governance objectives. The effects of global climate change and related natural disasters could negatively affect our business and operations. Global economic conditions may magnify certain risks that affect our business. Our business performance could affect or limit the ability of our Board of Directors to declare a dividend or our ability to pay a dividend or repurchase our common stock. We may not be able to access the capital and credit markets on terms that are favorable to us, or at all.
CONTACT:
Amgen
, Thousand Oaks
Elissa Snook, 609-251-1407 (media)
Justin Claeys, 805-313-9775 (investors)
Amgen Inc.
Consolidated Statements of Income - GAAP
(In millions, except per-share data)
(Unaudited)
Three months ended
December 31,
Twelve months ended
December 31,
2024
2023
2024
2023
Revenues:
Product sales
$ 8,716
$ 7,833
$ 32,026
$ 26,910
Other revenues
370
363
1,398
1,280
Total revenues
9,086
8,196
33,424
28,190
Operating expenses:
Cost of sales
3,112
3,112
12,858
8,451
Research and development
1,724
1,534
5,964
4,784
Selling, general and administrative
1,878
2,274
7,096
6,179
Other
61
5
248
879
Total operating expenses
6,775
6,925
26,166
20,293
Operating income
2,311
1,271
7,258
7,897
Other income (expense):
Interest expense, net
(747)
(821)
(3,155)
(2,875)
Other (expense) income, net
(782)
402
506
2,833
Income before income taxes
782
852
4,609
7,855
Provision for income taxes
155
85
519
1,138
Net income
$ 627
$ 767
$ 4,090
$ 6,717
Earnings per share:
Basic
$ 1.17
$ 1.43
$ 7.62
$ 12.56
Diluted
$ 1.16
$ 1.42
$ 7.56
$ 12.49
Weighted-average shares used in calculation of earnings per share:
Basic
537
535
537
535
Diluted
542
540
541
538
Amgen Inc.
Consolidated Balance Sheets - GAAP
(In millions)
December 31,
December 31,
2024
2023
(Unaudited)
Assets
Current assets:
Cash and cash equivalents
$ 11,973
$ 10,944
Trade receivables, net
6,782
7,268
Inventories
6,998
9,518
Other current assets
3,277
2,602
Total current assets
29,030
30,332
Property, plant and equipment, net
6,543
5,941
Intangible assets, net
27,699
32,641
Goodwill
18,637
18,629
Other noncurrent assets
9,930
9,611
Total assets
$ 91,839
$ 97,154
Liabilities and Stockholders' Equity
Current liabilities:
Accounts payable and accrued liabilities
$ 19,549
$ 16,949
Current portion of long-term debt
3,550
1,443
Total current liabilities
23,099
18,392
Long-term debt
56,549
63,170
Long-term deferred tax liabilities
1,616
2,354
Long-term tax liabilities
2,349
4,680
Other noncurrent liabilities
2,349
2,326
Total stockholders' equity
5,877
6,232
Total liabilities and stockholders' equity
$ 91,839
$ 97,154
Shares outstanding
537
535
Amgen Inc.
GAAP to Non-GAAP Reconciliations
(Dollars in millions)
(Unaudited)
Three months ended
December 31,
Twelve months ended
December 31,
2024
2023
2024
2023
GAAP cost of sales
$ 3,112
$ 3,112
$ 12,858
$ 8,451
Adjustments to cost of sales:
Acquisition-related expenses (a)
(1,576)
(1,834)
(7,122)
(3,842)
Certain net charges pursuant to our restructuring and cost-savings initiatives
—
—
—
(36)
Total adjustments to cost of sales
(1,576)
(1,834)
(7,122)
(3,878)
Non-GAAP cost of sales
$ 1,536
$ 1,278
$ 5,736
$ 4,573
GAAP cost of sales as a percentage of product sales
35.7 %
39.7 %
40.1 %
31.4 %
Acquisition-related expenses (a)
(18.1)
(23.4)
(22.2)
(14.3)
Certain net charges pursuant to our restructuring and cost-savings initiatives
0.0
0.0
0.0
(0.1)
Non-GAAP cost of sales as a percentage of product sales
17.6 %
16.3 %
17.9 %
17.0 %
GAAP research and development expenses
$ 1,724
$ 1,534
$ 5,964
$ 4,784
Adjustments to research and development expenses:
Acquisition-related expenses (b)
(26)
(28)
(86)
(55)
Certain net charges pursuant to our restructuring and cost-savings initiatives
—
(12)
—
(29)
Total adjustments to research and development expenses
(26)
(40)
(86)
(84)
Non-GAAP research and development expenses
$ 1,698
$ 1,494
$ 5,878
$ 4,700
GAAP research and development expenses as a percentage of product sales
19.8 %
19.6 %
18.6 %
17.8 %
Acquisition-related expenses (b)
(0.3)
(0.3)
(0.2)
(0.2)
Certain net charges pursuant to our restructuring and cost-savings initiatives
0.0
(0.2)
0.0
(0.1)
Non-GAAP research and development expenses as a percentage of product sales
19.5 %
19.1 %
18.4 %
17.5 %
GAAP selling, general and administrative expenses
$ 1,878
$ 2,274
$ 7,096
$ 6,179
Adjustments to selling, general and administrative expenses:
Acquisition-related expenses (c)
(59)
(510)
(314)
(648)
Certain net charges pursuant to our restructuring and cost-savings initiatives
—
—
—
(13)
Total adjustments to selling, general and administrative expenses
(59)
(510)
(314)
(661)
Non-GAAP selling, general and administrative expenses
$ 1,819
$ 1,764
$ 6,782
$ 5,518
GAAP selling, general and administrative expenses as a percentage of product sales
21.5 %
29.0 %
22.2 %
23.0 %
Acquisition-related expenses (c)
(0.6)
(6.5)
(1.0)
(2.4)
Certain net charges pursuant to our restructuring and cost-savings initiatives
0.0
0.0
0.0
(0.1)
Non-GAAP selling, general and administrative expenses as a percentage of product sales
20.9 %
22.5 %
21.2 %
20.5 %
GAAP operating expenses
$ 6,775
$ 6,925
$ 26,166
$ 20,293
Adjustments to operating expenses:
Adjustments to cost of sales
(1,576)
(1,834)
(7,122)
(3,878)
Adjustments to research and development expenses
(26)
(40)
(86)
(84)
Adjustments to selling, general and administrative expenses
(59)
(510)
(314)
(661)
Certain net charges pursuant to our restructuring and cost-savings initiatives (d)
(40)
(2)
(36)
(185)
Certain other expenses (e)
(21)
(3)
(212)
(694)
Total adjustments to operating expenses
(1,722)
(2,389)
(7,770)
(5,502)
Non-GAAP operating expenses
$ 5,053
$ 4,536
$ 18,396
$ 14,791
Three months ended
December 31,
Twelve months ended
December 31,
2024
2023
2024
2023
GAAP operating income
$ 2,311
$ 1,271
$ 7,258
$ 7,897
Adjustments to operating expenses
1,722
2,389
7,770
5,502
Non-GAAP operating income
$ 4,033
$ 3,660
$ 15,028
$ 13,399
GAAP operating income as a percentage of product sales
26.5 %
16.2 %
22.7 %
29.3 %
Adjustments to cost of sales
18.1
23.4
22.2
14.4
Adjustments to research and development expenses
0.3
0.4
0.2
0.3
Adjustments to selling, general and administrative expenses
0.6
6.5
1.0
2.6
Certain net charges pursuant to our restructuring and cost-savings initiatives (d)
0.5
0.1
0.1
0.7
Certain other expenses (e)
0.3
0.1
0.7
2.5
Non-GAAP operating income as a percentage of product sales
46.3 %
46.7 %
46.9 %
49.8 %
GAAP interest expense, net
$ (747)
$ (821)
$ (3,155)
$ (2,875)
Adjustments to interest expense, net:
Interest expense on acquisition-related debt (f)
—
19
—
807
Non-GAAP interest expense, net
$ (747)
$ (802)
$ (3,155)
$ (2,068)
GAAP other (expense) income, net
$ (782)
$ 402
$ 506
$ 2,833
Adjustments to other (expense) income, net
Interest income and other expenses on acquisition-related debt (f)
—
(18)
—
(625)
Net losses (gains) from equity investments (g)
875
(217)
182
(1,522)
Total adjustments to other (expense) income, net
875
(235)
182
(2,147)
Non-GAAP other income, net
$ 93
$ 167
$ 688
$ 686
GAAP income before income taxes
$ 782
$ 852
$ 4,609
$ 7,855
Adjustments to income before income taxes:
Adjustments to operating expenses
1,722
2,389
7,770
5,502
Adjustments to interest expense, net
—
19
—
807
Adjustments to other income, net
875
(235)
182
(2,147)
Total adjustments to income before income taxes
2,597
2,173
7,952
4,162
Non-GAAP income before income taxes
$ 3,379
$ 3,025
$ 12,561
$ 12,017
GAAP provision for income taxes
$ 155
$ 85
$ 519
$ 1,138
Adjustments to provision for income taxes:
Income tax effect of the above adjustments (h)
537
404
1,544
846
Other income tax adjustments (i)
(192)
(7)
(236)
(1)
Total adjustments to provision for income taxes
345
397
1,308
845
Non-GAAP provision for income taxes
$ 500
$ 482
$ 1,827
$ 1,983
GAAP tax as a percentage of income before taxes
19.8 %
10.0 %
11.3 %
14.5 %
Adjustments to provision for income taxes:
Income tax effect of the above adjustments (h)
0.7
6.1
5.1
2.0
Other income tax adjustments (i)
(5.7)
(0.2)
(1.9)
0.0
Total adjustments to provision for income taxes
(5.0)
5.9
3.2
2.0
Non-GAAP tax as a percentage of income before taxes
14.8 %
15.9 %
14.5 %
16.5 %
GAAP net income
$ 627
$ 767
$ 4,090
$ 6,717
Adjustments to net income:
Adjustments to income before income taxes, net of the income tax effect
2,060
1,769
6,408
3,316
Other income tax adjustments (i)
192
7
236
1
Total adjustments to net income
2,252
1,776
6,644
3,317
Non-GAAP net income
$ 2,879
$ 2,543
$ 10,734
$ 10,034
Note: Numbers may not add due to rounding
Amgen Inc.
GAAP to Non-GAAP Reconciliations
(In millions, except per-share data)
(Unaudited)
The following table presents the computations for GAAP and non-GAAP diluted earnings per share:
Three months ended
December 31, 2024
Three months ended
December 31, 2023
GAAP
Non-GAAP
GAAP
Non-GAAP
Net income
$ 627
$ 2,879
$ 767
$ 2,543
Weighted-average shares for diluted EPS
542
542
540
540
Diluted EPS
$ 1.16
$ 5.31
$ 1.42
$ 4.71
Twelve months ended
December 31, 2024
Twelve months ended
December 31, 2023
GAAP
Non-GAAP
GAAP
Non-GAAP
Net income
$ 4,090
$ 10,734
$ 6,717
$ 10,034
Weighted-average shares for diluted EPS
541
541
538
538
Diluted EPS
$ 7.56
$ 19.84
$ 12.49
$ 18.65
(a)
The adjustments related primarily to noncash amortization of intangible assets and fair value step-up of inventory acquired from business acquisitions.
(b)
For the three and twelve months ended December 31, 2024, the adjustments related primarily to acquisition-related costs related to our Horizon acquisition. For the three months ended December 31, 2023, the adjustments related primarily to acquisition-related costs related to our Horizon acquisition. For the twelve months ended December 31, 2023, the adjustments related primarily to noncash amortization of intangible assets acquired from business acquisitions.
(c)
For the three and twelve months ended December 31, 2024 and 2023, the adjustments related primarily to acquisition-related costs related to our Horizon acquisition.
(d)
For the three and twelve months ended December 31, 2024 and 2023, the adjustments related to separation costs associated with our restructuring plan and other cost-savings initiatives.
(e)
For the twelve months ended December 31, 2024, the adjustments related primarily to impairment charges for IPR&D intangible assets related to our
Teneobio, Inc.
acquisition from 2021. For the twelve months ended December 31, 2023, the adjustments related primarily to a net IPR&D intangible asset impairment charge for AMG 340.
(f)
For the three and twelve months ended December 31, 2023, the adjustments included (i) interest expense and income on senior notes issued in March 2023 and (ii) debt issuance costs and other fees related to our bridge credit and term loan credit agreements, incurred prior to the closing of our acquisition of Horizon.
(g)
For the three and twelve months ended December 31, 2024, the adjustments related primarily to our BeiGene equity fair value adjustment. For the twelve months ended December 31, 2023, the adjustments related primarily to our BeiGene equity fair value adjustment.
(h)
The tax effect of the adjustments between our GAAP and non-GAAP results takes into account the tax treatment and related tax rate(s) that apply to each adjustment in the applicable tax jurisdiction(s). Generally, the tax impact of adjustments, including the amortization of intangible assets and acquired inventory, gains and losses on our investments in equity securities and expenses related to restructuring and cost-savings initiatives, depends on whether the amounts are deductible in the respective tax jurisdictions and the applicable tax rate(s) in those jurisdictions. Due to these factors, the effective tax rate for the adjustments to our GAAP income before income taxes for the three and twelve months ended December 31, 2024, was 20.7% and 19.4%, respectively, compared to 18.6% and 20.3% for the corresponding periods of the prior year.
(i)
The adjustments related to certain acquisition items, prior period and other items excluded from GAAP earnings.
Amgen Inc.
Reconciliations of Cash Flows
(In millions)
(Unaudited)
Three months ended
December 31,
Twelve months ended
December 31,
2024
2023
2024
2023
Net cash provided by operating activities
$ 4,771
$ 538
$ 11,490
$ 8,471
Net cash used in investing activities
(402)
(27,089)
(1,046)
(26,204)
Net cash (used in) provided by financing activities
(1,407)
2,754
(9,415)
21,048
Increase (decrease) in cash and cash equivalents
2,962
(23,797)
1,029
3,315
Cash and cash equivalents at beginning of period
9,011
34,741
10,944
7,629
Cash and cash equivalents at end of period
$ 11,973
$ 10,944
$ 11,973
$ 10,944
Three months ended
December 31,
Twelve months ended
December 31,
2024
2023
2024
2023
Net cash provided by operating activities
$ 4,771
$ 538
$ 11,490
$ 8,471
Capital expenditures
(371)
(249)
(1,096)
(1,112)
Free cash flow
$ 4,400
$ 289
$ 10,394
$ 7,359
Amgen Inc.
Reconciliation of GAAP Net Income to EBITDA and Debt Leverage Ratio Calculation
(Dollars in millions)
(Unaudited)
Twelve months ended
December 31, 2024
GAAP Net Income
$ 4,090
Depreciation and amortization
5,592
Interest expense, net
3,155
Provision for income taxes
519
EBITDA
(a)
$ 13,356
As of December 31, 2024
Current portion of long-term debt
$ 3,550
Long-term debt
56,549
Total GAAP Debt
$ 60,099
As of December 31, 2024
Total GAAP Debt
$ 60,099
EBITDA
$ 13,356
Debt leverage ratio
4.5
(a)
2024 EBITDA includes amortization of inventory step-up of $2.4 billion and net losses from equity investments of $182 million.
Amgen Inc.
Reconciliation of GAAP EPS Guidance to Non-GAAP
EPS Guidance for the Year Ending December 31, 2025
(Unaudited)
GAAP diluted EPS guidance
$ 10.89
—
$ 12.14
Known adjustments to arrive at non-GAAP*:
Acquisition-related expenses (a)
9.06
—
9.11
Non-GAAP diluted EPS guidance
$ 20.00
—
$ 21.20
* The known adjustments are presented net of their related tax impact, which amount to approximately $1.54 per share.
(a) The adjustments include noncash amortization of intangible assets and fair value step-up of inventory acquired in business acquisitions.
Our GAAP diluted EPS guidance does not include the effect of GAAP adjustments triggered by events that may occur subsequent to this press release such as acquisitions, asset impairments, litigation, changes in fair value of our contingent consideration obligations and changes in fair value of our equity investments.
Reconciliation of GAAP Tax Rate Guidance to Non-GAAP
Tax Rate Guidance for the Year Ending December 31, 2025
(Unaudited)
GAAP tax rate guidance
11.0 %
—
12.5 %
Tax rate of known adjustments discussed above
3.5 %
—
4.0 %
Non-GAAP tax rate guidance
15.0 %
—
16.0 %
View original content to download multimedia:
https://www.prnewswire.com/news-releases/amgen-reports-fourth-quarter-and-full-year-2024-financial-results-302367906.html
SOURCE
Amgen
并购财报生物类似药上市批准引进/卖出
2024-06-28
FRIDAY, June 28, 2024 -- Rheumatoid arthritis (RA) patients with mono- or oligo-arthritis and high Patient Global Assessment (PGA) at diagnosis remain the most fatigued, according to a study published in the June issue of
Rheumatology
.
Anna M.P. Boeren, from the Leiden University Medical Center in the Netherlands, and colleagues studied Disease Activity Score (DAS) components at diagnosis in relation to the course of fatigue over a five-year period in two early RA cohorts. Data were included for 1,560 RA patients in the Leiden Early Arthritis Cohort and 415 in the tREACH cohort. Swollen joint count, tender joint count, erythrocyte sedimentation rate, and PGA (on a visual analogue scale) were assessed in relation to fatigue.
The researchers found that a more severe course of fatigue was seen in association with higher tender joint count and higher PGA at diagnosis. Mono- or oligo-arthritis at diagnosis was associated with patients remaining more fatigued. In contrast, there was an inverse association seen for the swollen joint count. Patients presenting with mono- or oligo-arthritis and PGA ≥50 mm remained the most fatigued over time (+20 mm versus polyarthritis with PGA <50 mm) on examination of combinations of characteristics, while no difference was seen for the DAS course over time. Fourteen percent of the early RA population were part of this subgroup. Similar findings were seen in the tREACH trial.
"Future studies on the efficacy of additional nonpharmacological treatments, such as cognitive behavioral therapy, preferentially within this subgroup of RA patients, are needed, because these patients have the highest unmet need," the authors write.
Abstract/Full Text
Whatever your topic of interest,
subscribe to our newsletters
to get the best of Drugs.com in your inbox.
临床结果
分析
对领域进行一次全面的分析。
登录
或
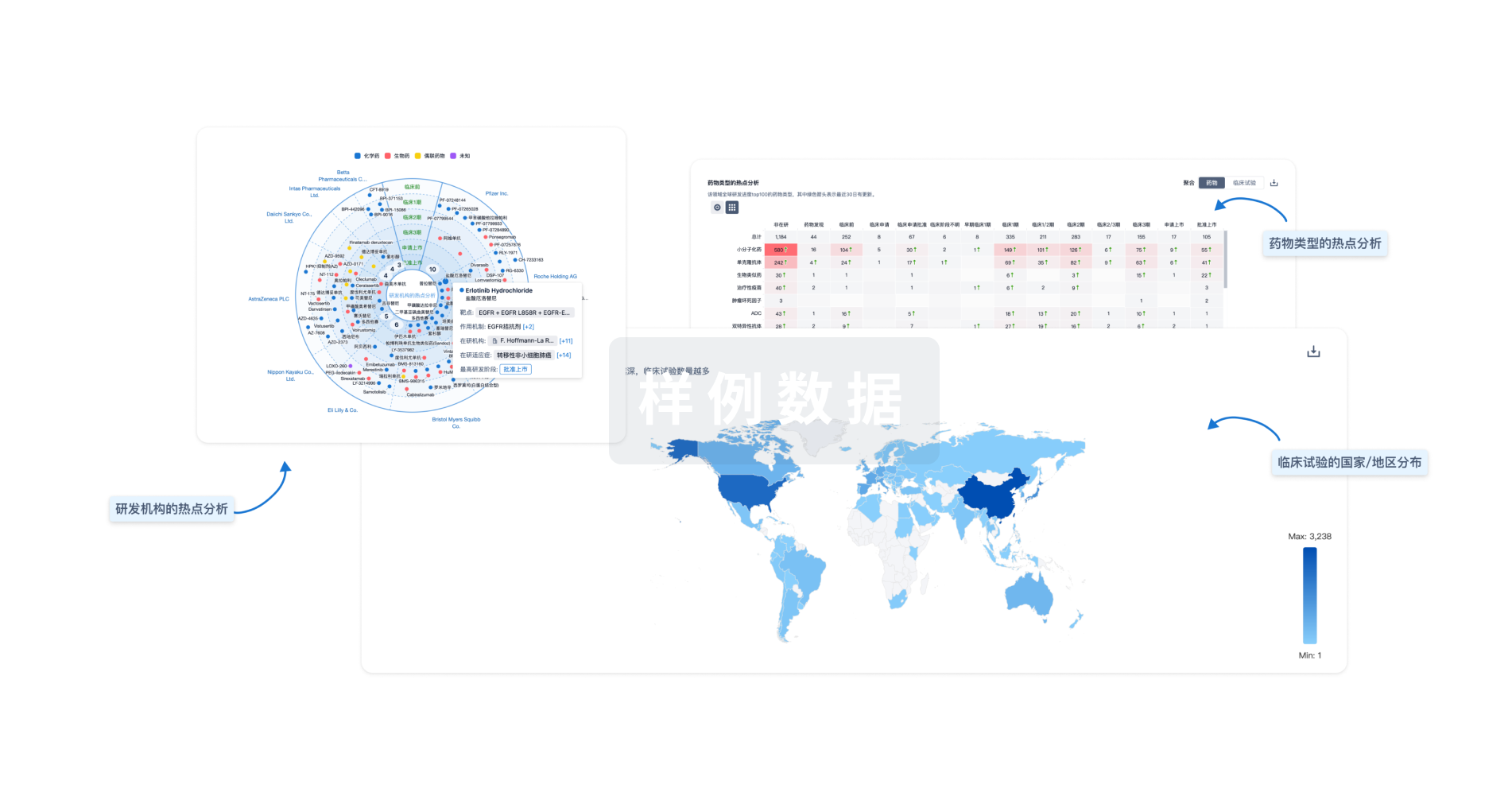
生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用


