预约演示
更新于:2025-05-07
Niemann-Pick Diseases
尼曼匹克症
更新于:2025-05-07
基本信息
别名 HISTIOCYTOSIS, LIPID、LIPIDOSIS, SPHINGOMYELIN、NIEMANN-PICK DISEASE + [52] |
简介 A group of autosomal recessive disorders in which harmful quantities of lipids accumulate in the viscera and the central nervous system. They can be caused by deficiencies of enzyme activities (SPHINGOMYELIN PHOSPHODIESTERASE) or defects in intracellular transport, resulting in the accumulation of SPHINGOMYELINS and CHOLESTEROL. There are various subtypes based on their clinical and genetic differences. |
关联
48
项与 尼曼匹克症 相关的药物作用机制 calcium channel调节剂 |
在研机构 |
原研机构 |
最高研发阶段批准上市 |
首次获批国家/地区 美国 |
首次获批日期2024-09-24 |
作用机制 TFE3 agonists [+3] |
最高研发阶段批准上市 |
首次获批国家/地区 美国 |
首次获批日期2024-09-20 |
88
项与 尼曼匹克症 相关的临床试验NCT06869499
Niemann-PID: Study of the Prevalence of Acid Sphingomyelinase Deficiency/Niemann Pick AB and B Disease in Patients with Diffuse Interstitial Lung Disease
The goal of this clinical trial is to optimise and facilitate screening for Acid SphingoMyelinase Deficiency (ASMD) disease, by evaluating acid sphingomyelinase activity and, where appropriate, LysoSM levels in a cohort of 200 participants with diffuse interstitial lund disease (ILD) at risk of developing ASMD disease.
ILD is common in the general population, so in order to limit the number of differential diagnoses, the population to be studied will be restricted to participants aged between 15 years and 3 months and 60 years, with ILD plus ground-glass opacities on chest CT scan certified by a pulmonologist/radiologist or internist, AND splenomegaly or splenectomy, and/or thrombocytopenia, and/or low HDL cholesterol, and/or parental consanguinity which increase the sensitivity of ASMD screening.
In this clinical trail, two procedures are added, participants will be asked for :
* a blood sample to measure the acid sphingomyelinase enzyme activity and LysoSM, if required.
* a follow-up visit at 6 months
ILD is common in the general population, so in order to limit the number of differential diagnoses, the population to be studied will be restricted to participants aged between 15 years and 3 months and 60 years, with ILD plus ground-glass opacities on chest CT scan certified by a pulmonologist/radiologist or internist, AND splenomegaly or splenectomy, and/or thrombocytopenia, and/or low HDL cholesterol, and/or parental consanguinity which increase the sensitivity of ASMD screening.
In this clinical trail, two procedures are added, participants will be asked for :
* a blood sample to measure the acid sphingomyelinase enzyme activity and LysoSM, if required.
* a follow-up visit at 6 months
开始日期2025-04-01 |
申办/合作机构 |
CTIS2023-505832-36-01
OPEN-LABEL EXTENSION STUDY TO ASSESS THE SAFETY AND LONG-TERM EFFECTIVENESS OF ORALLY ADMINISTERED EFAVIRENZ IN PATIENTS DIAGNOSED WITH ADULT OR LATE-ONSET JUVENILE TYPE C NIEMANN-PICK DISEASE WITH COGNITIVE IMPAIRMENT - HUB-NEU-2023-01
开始日期2024-05-16 |
NCT06258577
Screening for Gaucher Disease and Acid Sphingomyelinase Deficiency From Taiwanese Candidates With Splenomegaly and/or Thrombocytopenia
High-risk screening for Gaucher disease and Acid Sphingomyelinase Deficiency in patients with splenomegaly and/or thrombocytopenia in Taiwan
开始日期2024-05-01 |
申办/合作机构 中国医药大学附设医院 [+1] |
100 项与 尼曼匹克症 相关的临床结果
登录后查看更多信息
100 项与 尼曼匹克症 相关的转化医学
登录后查看更多信息
0 项与 尼曼匹克症 相关的专利(医药)
登录后查看更多信息
3,796
项与 尼曼匹克症 相关的文献(医药)2025-07-01·Physiological Reviews
TPCs: FROM PLANT TO HUMAN
Review
作者: Grimm, Christian ; Klingl, Yvonne Eileen ; Jaślan, Dawid ; Petrauskas, Arnas
2025-06-01·Molecular Genetics and Metabolism Reports
Benefits of early intervention with olipudase alfa in symptomatic children with acid sphingomyelinase deficiency: A sibling case-comparison study
Article
作者: Ganesh, Jaya ; Simpson, William L ; Mason, Lauren Mackenzie ; Ting, Andrew ; Bier, Louise ; Diaz, George A ; Freeman, Mary ; Sinha, Drew B
2025-06-01·Molecular Genetics and Metabolism
The value of MR spectroscopy and MR elastography in assessing hepatic involvement of chronic visceral acid sphingomyelinase deficiency in adults
Article
作者: Nederveen, Aart J ; Eskes, Eline C B ; Wassenaar, Nienke P M ; Hollak, Carla E M ; van den Berg-Faaij, Sandra A M ; Sjouke, Barbara
372
项与 尼曼匹克症 相关的新闻(医药)2025-04-21
Highlights 54.8% Total Stockholder Return Under Refreshed Board and Management Team
Zevra Continues Growth Trajectory into a Global Commercial Rare Disease Company with Consistent Execution, New Product Launches, and Significant Financial Flexibility to Execute Strategic Plan
Urges Stockholders to Defend Board Independence from Majority Influence by Individual 2.8% Stockholder, Mangless, Who Had Three Nominees Elected to the Board in 2023
Mangless Has Not Detailed any Strategies, Plans, or New Ideas to Improve Zevra’s Business and Appears Solely Focused on Replacing Two Highly Qualified Directors
Board and Management Urge Stockholders to Vote "FOR" Zevra’s Highly-Qualified and Experienced Director Nominees on the WHITE Proxy Card
CELEBRATION, Fla., April 21, 2025 (GLOBE NEWSWIRE) -- Zevra Therapeutics, Inc. (NasdaqGS: ZVRA) (“Zevra”, or “the Company”), a commercial-stage company focused on providing therapies for people living with rare disease, today announced it has filed its definitive proxy statement with the U.S. Securities and Exchange Commission (the “SEC”) in connection with the Company’s 2025 Annual Meeting of Stockholders (“Annual Meeting”), which is scheduled to take place on May 29, 2025. Stockholders of record as of April 4, 2025 will be entitled to vote at the Annual Meeting.
Zevra’s Board of Directors strongly recommends that stockholders vote "FOR" the Company's two highly-qualified directors up for re-election on the WHITE proxy card – Wendy L. Dixon, Ph.D and Tamara A. Favorito, both of whom bring extensive public company board experience and life sciences management expertise to the Zevra Board. Both of these independent directors have contributed valuable perspectives that have helped accelerate the Company’s progress and execution of its strategic goals.
In conjunction with the definitive proxy filing, Zevra is mailing a letter to stockholders detailing the decisive action the Company has taken over the past two years to transform Zevra into a stronger, growth-oriented commercial organization, which is poised to continue to drive significant stockholder value. The letter also addresses the attempt by an individual stockholder, Daniel J. Mangless, to replace two highly qualified directors with his nominees who could disrupt the clear progress underway and jeopardize Zevra’s ability to deliver long-term value for stockholders.
As stockholders consider this important election, it is critical to remember that:
The proactive and refreshed Zevra Board and management team have a proven track record of success, as demonstrated by the continued progress and significant stockholder value creation during the last two years. Seven of eight directors have joined the Board since 2023 and Zevra’s refreshed management team has launched two rare disease therapies, rapidly expanded the Company’s commercial capabilities, and positioned its pipeline for continued success. The ongoing execution of Zevra’s strategy is designed to continue driving value over the coming months and years. Zevra’s Board nominees are proven, experienced public company leaders and directors who have the requisite company and industry expertise and fulfill the essential requirements for effective governance of a rapidly growing commercial-stage rare disease company. During their tenure, the Board’s nominees have initiated and overseen substantial improvements to the Company. Despite owning just 2.8% of the Company and having already had three nominees elected to the Board in 2023, Mr. Mangless is now actively seeking two additional Board seats without providing any clear strategy or explanation of what he intends to accomplish or how he proposes to increase stockholder value. If Mr. Mangless’ two nominees are elected this year, his nominees would have five Board seats in total, representing a majority of the Board. This latest attempt raises questions about the amount of undue influence one stockholder should have on the Board. In his own proxy filing, Mr. Mangless acknowledges that if his two nominees are elected, the five nominees that he will have added to the Board “will potentially be able to implement any actions that they may believe are necessary...” Mr. Mangless has not disclosed any specific strategies, plans, or new ideas to improve Zevra’s business – in fact, he has expressed agreement with the recent actions that Zevra has taken, including the onboarding of Zevra’s current leadership, yet he still appears focused on replacing two highly-qualified directors with his nominees who lack the necessary expertise, independence, and proper business judgment and acumen to serve Zevra's stockholders effectively. Mr. Mangless’ nominees are a proxy solicitor with no relevant Board or industry experience, and a former CEO, both of whom have previously destroyed significant stockholder value. The Zevra Board believes that adding his nominees will put stockholders‘ investments in Zevra at risk. Mr. Mangless’ nominees, Arthur Regan and Dr. Travis Mickle, have track records of destroying stockholder value in public company leadership roles. During Regan’s tenure as a director at US Wats, US Wats’ stock price fell 63.9%. While Dr. Mickle was CEO of Zevra, its stock price plummeted 97.4%.1
Zevra's letter to stockholders and other materials regarding the Board's recommendation for the 2025 Annual Meeting can be found on the Company’s investor relations website at https://investors.zevra.com/.
The full text of the letter follows:
Dear Fellow Stockholders,
The Zevra Board of Directors and management team have taken decisive action over the past two years, transforming the Company and driving significant stockholder value creation.
This transformation began in October 2023, when your Board appointed Neil McFarlane as President and CEO of Zevra, following a thorough process to find the right leader to accelerate Zevra’s progress and harness our pipeline of innovative rare disease therapies. Since then, we have launched two rare disease therapies and have been rapidly expanding Zevra’s commercial capabilities. We completed a comprehensive review of our development pipeline to facilitate investment in the most promising, highest return opportunities. At the same time, we have added significant expertise to support the Company’s growth and help us continue to drive operational excellence across our commercial and pre-commercial programs. Further, we have strengthened Zevra’s balance sheet, providing the Company with ample financial flexibility to continue investing in growth, without needing incremental funding from the capital markets.
In late 2024, your Board and management team detailed a clear and comprehensive five-year strategic plan to drive long-term growth and patient impact. We are making significant progress as we successfully drive towards our clear priorities and achieve our vision of positioning Zevra as a leading global rare disease company.
The results of our execution speak for themselves: Our strategy has enabled Zevra to deliver superior returns for our stockholders. During the last 1-year and 2-year periods, your Board and management team have delivered total stockholder returns of 54.1% and 14.8%, respectively, all in excess of the comparable biotech sector and the Russell 2000. As seen below, since Mr. McFarlane was appointed CEO in October 2023, Zevra has achieved impressive total stockholder returns of 54.8%.2
Against this backdrop of significant commercial progress and superior returns, you have an important decision to make at our upcoming Annual Meeting on May 29, 2025. Your Board has two independent directors up for re-election, Wendy L. Dixon, Ph.D., who has served as a director since April 2023, and Tamara A. Favorito, who has served as a director and Chair of the Audit Committee since August 2021 and as our Board Chair since May 2023. Dr. Dixon and Ms. Favorito bring extensive public company board experience, deep knowledge of Zevra, and life sciences management expertise to the Zevra Board. Both directors have contributed valuable perspectives that have helped accelerate the Company’s progress and execution of its strategic goals.
Meanwhile, Daniel J. Mangless, an individual stockholder, has nominated Travis C. Mickle, Ph.D., Zevra’s former President and CEO, and Arthur C. Regan, a proxy solicitor, for election to your Board. We believe that adding a former CEO, who presided over a period of significant stockholder value destruction during his tenure at Zevra and cannot be considered independent under SEC and stock exchange rules, as well as a proxy solicitor with no industry experience risks significant disruption to Zevra’s ongoing progress and ability to continue delivering sector-leading returns to stockholders. Further, their appointments would distract from the Company’s continued disciplined execution of its five-year strategic plan and value-creation initiatives.
Zevra’s Board recommends that stockholders reject these efforts by Mr. Mangless. Please discard any blue proxy card sent to you by Mr. Mangless.
To elect the Zevra Board's nominees, we encourage you to vote today – no matter how many or how few shares you own –online or by signing and dating the enclosed WHITE proxy card and returning it in the postage-paid envelope provided.
As you consider this important election, it is critical to remember that:
Your proactive and refreshed Zevra Board and management team have a proven track record of success, as demonstrated by the continued progress and significant stockholder value creation during the last two years. Seven of eight directors have joined the Board since 2023, and Zevra’s refreshed management team executed the launch of two rare disease therapies, rapidly expanded the Company’s commercial capabilities and positioned its pipeline for continued success. The ongoing execution of Zevra’s strategy is designed to continue driving value over the coming months and years. Zevra’s Board nominees are proven, experienced public company leaders and directors who have the requisite company and industry expertise and fulfill the essential requirements for effective governance of a rapidly growing commercial-stage rare disease company. During their tenure, the Board’s nominees have initiated and overseen substantial improvements to the Company. Despite owning just 2.8% of the Company and having already had three nominees elected to the Board in 2023, Mr. Mangless is now actively seeking two additional Board seats without providing any clear strategy or explanation of what he intends to accomplish or how he proposes to increase stockholder value. If Mr. Mangless’ two nominees are elected this year, his nominees would have five Board seats in total, representing a majority of the Board. This latest attempt raises questions about the amount of undue influence one stockholder should have on the Board. In his own proxy filing, Mr. Mangless acknowledges that if his two nominees are elected, the five nominees that he will have added to the Board “will potentially be able to implement any actions that they may believe are necessary...” Mr. Mangless has not disclosed any strategies, plans, or new ideas to improve Zevra’s business – in fact, he has expressed agreement with the recent actions that Zevra has taken, including the onboarding of Zevra’s current leadership, yet he still appears focused on replacing two highly-qualified directors with his nominees who lack the necessary expertise, independence, and proper business judgment and acumen to serve Zevra's stockholders effectively. Mr. Mangless’ nominees are a proxy solicitor with no relevant Board or industry experience, and a former CEO, both of whom have previously destroyed significant stockholder value. The Zevra Board believes that adding his nominees will put stockholders‘ investments in Zevra at risk. Mr. Mangless’ nominees, Arthur Regan and Dr. Travis Mickle, have track records of destroying stockholder value in public company leadership roles. During Regan’s tenure as a director at US Wats, US Wats’ stock price fell 63.9%. While Dr. Mickle was CEO of Zevra, its stock price plummeted 97.4%.3
We ask you a simple question: Would you rather have a proven team with a clear strategy, strong momentum, relevant expertise, and a track record of increasing stockholder value, or risk distraction, disruption, and potential destruction of the Company's value by a single stockholder with unqualified director nominees and no stated business strategy? We urge you to stand with management and reject the blue proxy card with Mr. Mangless’ nominees.
Your Board and Management Team are Actively Delivering Significant Progress as we Continue Zevra’s Transformation into a Leading Rare Disease Company
Over the past two years, your Board and management team have been actively transforming Zevra into a more efficient, more focused organization that is better positioned to create value for our patients and stockholders. In the third quarter of 2024, we introduced our five-year strategic plan, built on four key pillars that have informed our priorities and investments as we continue to accelerate Zevra’s commercial momentum: (1) Commercial Excellence, (2) Pipeline and Innovation, (3) Talent and Culture, and (4) Corporate Foundation.
1. Delivering Commercial Excellence – In 2024, we successfully commercialized and launched Zevra’s first two products. Since then, we have maintained our active focus on driving best-in-class commercial excellence.
In September 2024, we received approval from the U.S. Food and Drug Administration for MIPLYFFA®, the first approved product in the U.S. for the treatment of Niemann-Pick disease type C (NPC). Alongside this approval, we received a Rare Pediatric Disease Priority Review Voucher (PRV), which Zevra recently sold for $150 million to further enhance our financial flexibility. MIPLYFFA became available to patients within eight weeks following approval, and with the support of the NPC community and strong execution by our team, we were able to quickly ensure a smooth transition for all U.S. Expanded Access Program (EAP) participants to MIPLYFFA as a commercial product. Eight months earlier, in January 2024, we launched OLPRUVA®, Zevra’s first-ever commercial product, for people suffering from certain urea cycle disorders (UCDs), and we have continued its growth while accelerating the establishment of our commercial footprint.
2. Enhancing the Value of Zevra’s Pipeline and Innovation – Throughout 2024, your Board and management team conducted a comprehensive, data-driven review to optimize and curate Zevra’s portfolio. This review allowed us to focus our resources on development projects where Zevra is uniquely positioned to create the most value for patients and our stockholders – ensuring that we are investing in Zevra’s most promising, highest return opportunities.
Launched Phase 3 DiSCOVER trial: Based on the results of our review, in mid-2024, we reinitiated recruitment for the Phase 3 DiSCOVER trial of celiprolol for the treatment of Vascular Ehlers-Danlos Syndrome (VEDS) as a potential treatment for a patient population with high unmet need. Maximizing the Value of KP1077: In June, we announced positive topline results from our KP1077 Phase 2 trial in idiopathic hypersomnia at the 2024 Annual SLEEP Meeting. At the end of the third quarter, we completed our end-of-Phase 2 meeting. The FDA and Zevra reached alignment on a Phase 3 trial design and indicated that a single pivotal study with appropriate confirmatory evidence would be sufficient to submit a New Drug Application (NDA). As part of our pipeline review, we have determined that the best path forward to maximize the value of KP1077 is to explore strategic alternatives for advancing its clinical development and future commercialization.
3. Optimizing Leadership Team with the Right Talent: As we have continued our momentum, we have also evolved our leadership team to ensure we have the right talent to support our continued growth into a leading rare disease commercial company.
In June 2024, we enhanced our executive leadership team with the appointments of Rahsaan Thompson as Chief Legal Officer, Secretary and Compliance Officer, and Alison Peters as Chief People Officer; we also consolidated our development and scientific functions under Adrian Quartel, our Chief Medical Officer.
4. Strengthening Zevra’s Corporate Foundation: We have continued our approach of disciplined capital allocation, focusing our investments on the highest return activities and actively enhancing our financial flexibility.
In April 2024, we bolstered Zevra’s balance sheet by restructuring the Company’s debt, using a new credit facility provided by premier biotech investors. In September 2024, we secured a Rare Pediatric Disease Priority Review Voucher (PRV) with the approval of MIPLYFFA, which we sold this April for approximately $150 million, providing additional non-dilutive capital to fuel our commercial launches of MIPLYFFA and OLPRUVA, and further the development of celiprolol. We believe this transaction provides us with important financial flexibility to deliver on our strategic plan without undue reliance on incremental funding from the capital markets.
In the coming quarters, your Board and management team intend to maintain focus on accelerating Zevra’s momentum – continuing to drive value for both rare disease patients and our stockholders.
Your Board and Management Team Have a Clear Path Forward to Continue Zevra’s Momentum
Looking ahead, we have a clear strategic plan and path forward to continue Zevra’s growth trajectory and realize our bright future. Our priorities in the coming year include:
Expanding adoption of MIPLYFFA. We are actively focused on increasing awareness and adoption of MIPLYFFA. Most recently, in February 2025, we launched efforts to provide integral educational and testing resources to NPC treatment teams and physicians in the U.S. Our market access team is also working tirelessly to continuously engage with payors to formalize coverage so that all patients may have access to this life-changing treatment. Securing regulatory approval of MIPLYFFA in the EU. We remain on track to file a marketing authorization application (MAA) in the second half of 2025. This approval would further expand our addressable market to the estimated 1,100 people living with NPC in the EU. Expanding adoption of OLPRUVA for the adult-onset population for whom OLPRUVA's portability and ease of administration may be most beneficial. Our efforts are also focused on supporting patients facing insurance coverage challenges. Accelerating enrollment in the Phase 3 DiSCOVER trial for celiprolol, by expanding our outreach and education efforts within the patient community and among treating physicians. Accelerating enrollment and completion of the Phase 3 trial will advance efforts towards making this potential treatment available. There are currently no treatments approved in the U.S. for VEDS and the approximately 7,500 people living with this disease.
With a strong pipeline of innovative products, multiple upcoming milestones in the coming quarters, and your management team’s continued disciplined execution, we are confident that Zevra is positioned for long-term success.
Your Board and Management Team Already Have Fresh Perspectives to Drive Stockholder Value
The Zevra Board and management team have been dramatically refreshed since 2023. Seven out of the eight current directors joined the Board in the past two years. Since his appointment as President and CEO in October 2023, Mr. McFarlane’s leadership has delivered significant stockholder value creation. Since then, we have added more talent and expertise to support Zevra’s continued growth, as evidenced by our June 2024 executive appointments.
Your Board is composed of directors who are exceptionally qualified, independent, and committed to acting in the best interests of all Zevra stockholders. They provide rigorous oversight of the Company's progress, and we are confident they are the right directors to continue driving superior stockholder value.
With the re-election of the two directors your Board has nominated, Dr. Dixon and Ms. Favorito, your Board would continue to be comprised of eight highly-qualified directors, seven of whom are independent and all of whom bring important perspectives and expertise to oversee the continued execution of Zevra’s strategy.
We Actively Engaged with Mangless to Try to Avoid a Costly, Potentially Distracting Proxy Contest – Yet He is Intent on Replacing Two Highly-Qualified Directors
Over the past several months, we have actively engaged with Mr. Mangless in an attempt to avoid a costly proxy contest that could distract from our continued strong execution. Despite his 2.8% ownership of the Company and having already had three of his nominees elected to the Board in 2023, Mr. Mangless appears intent on trying to secure two additional seats on the Board without providing any specific strategy or plan to meaningfully improve Zevra’s business. Mr. Mangless only expresses dissatisfaction with the Company’s governance structure, without disclosing any expertise or experience regarding the corporate governance practices that are typical for companies of similar size, stage, and/or therapeutic focus to Zevra.
Mr. Mangless’ proxy states that he previously shared suggestions about “increasing the diversity of the Board,” but has not described how either of the candidates he has nominated would promote diversity or otherwise align with his own suggestion.
As a company dedicated to developing breakthrough therapies for rare diseases, we operate in a highly specialized and complex environment. Success for stockholders, employees, and the patients we help requires an experienced Board with an appropriate mix of perspectives, including deep scientific and commercial expertise, regulatory acumen, financial experience, and the ability to oversee a long-term vision.
Despite the significant and recent Board refreshment at Zevra, Mr. Mangless is actively trying to replace two highly-qualified directors who bring valuable industry, operational, and financial expertise to the Board, both of whom have substantial experience in leadership roles and as public company directors:
Wendy L. Dixon, Ph.D., has served as a director since April 2023 and is a member of the Company’s Audit Committee. Dr. Dixon has more than 40 years of biopharmaceutical industry experience in drug development, with leadership roles in regulatory affairs and commercial capabilities. Dr. Dixon has held a number of leadership roles at leading publicly-traded companies, including serving as Chief Marketing Officer and President of Global Marketing at Bristol-Myers Squibb, senior Vice President of Marketing at Merck, and executive management positions at West Pharmaceuticals, Osteotech, and Centocor, in addition to roles at SmithKline and French (now GlaxoSmithKline) in marketing, regulatory affairs, project management, and as a biochemist.
Tamara A. Favorito has served as a director and Chair of the Audit Committee since August 2021, and as our Board Chair since May 2023. Ms. Favorito has more than 30 years of industry experience, including 20 years as a Chief Financial Officer for multiple publicly-traded life science companies, including Immunic, Inc., Signal Genetics, Inc. (now Viridian Therapeutics, Inc.), and Favrille, Inc. (now MMR Global, Inc.). Ms. Favorito is the only director who was on the Board prior to 2023, giving her a deeper understanding of the Company’s history and an important perspective as the Company continues its path forward.
Mangless’ Proposal to Add a Proxy Solicitor and a Former CEO with Value-Destructive Track Records Meaningfully Risks Disruption to Zevra’s Continued Execution of its Strategic Plan
Mr. Mangless is proposing to replace two of your highly-qualified directors with his two nominees. Neither of Mr. Mangless’ nominees would bring any additive skills or experience to the Board.
We have significant concerns about the potential disruption and distraction Mr. Mangless’ nominees could bring to Zevra’s operations. More specifically:
Mr. Mangless’ nominee, Mr. Regan, runs a five-person proxy solicitation firm, has no industry experience or expertise in life sciences, and is distinctly ill-suited to serve as a director of a leading commercial rare disease company like Zevra. Mr. Regan's only experience as a director of a public operating company was over 25 years ago at US Wats, Inc. (“US Wats”), a telecommunications company where he served as corporate secretary and director. As of April 2000, subsequent to a merger, US Wats appears to have abruptly ceased complying with its reporting requirements, a responsibility that seems directly tied to Mr. Regan as the company’s corporate secretary and director at that time, and the company’s SEC registration was subsequently revoked. Furthermore, during Mr. Regan’s tenure as a director, US Wats stockholder value was destroyed, with the stock price falling 63.9%.
Mr. Regan’s only other public company director-level experience appears to have taken place for a little over a year starting in 2017, when he was nominated by an activist investor as a trustee of a closed-end mutual fund, Madison Strategic Sector Premium Fund. During that contested election, Mr. Regan was also performing proxy solicitation services for the activist, leading independent proxy advisor Glass Lewis to determine during the proxy contest that “we see very little value in the prospective election of...Mr. Regan.” On August 22, 2017, Madison Strategic Sector Premium Fund held an Annual Meeting of stockholders of the fund, at which Mr. Regan was elected as a trustee. By October 2018, Madison Strategic Sector Premium Fund merged with another fund, deregistered from the SEC and delisted from the stock exchange on which it had been listed. Mr. Regan is inexperienced and wholly unqualified to serve as a director of a highly specialized rare disease company like Zevra. Specifically, we believe that Mr. Regan’s erratic and arguably unprofessional nature, evident in his communications on his personal blog and online posts, would create serious risk to Zevra’s reputation, performance, and momentum if he were elected to the Board. Mr. Mangless has also nominated Dr. Mickle, who cannot be considered independent under SEC and stock exchange rules due to his former roles as CEO and President of the Company until his resignation in 2023 and his consulting arrangement with the Company until 2024. While our current management team has delivered a strong 54.8% total stockholder return, Dr. Mickle's leadership led to stockholder value destruction at Zevra – resulting in the share price plummeting 97.4% during his tenure, substantially underperforming both industry benchmarks and the broader market.
Further, it would be highly unusual for a former CEO to serve as a director of that same company, so soon after departing as CEO. This could create a scenario where Dr. Mickle seeks to micromanage the executive team instead of focusing on oversight, or even to pursue a return to his previous role as CEO. We view this potential outcome as negative to Zevra and its stockholders, especially given the substantial increase in stockholder value delivered by the current management team, as compared to the destruction of stockholder value under Dr. Mickle’s leadership.
Your Board and management team believe the addition of one or both of Mr. Mangless’ nominees would distract from management's continued successful execution of our strategic plan. In the absence of any stated strategy or ideas for increasing stockholder value, we believe Mr. Mangless and his nominees will only disrupt the clear progress underway and risk our ability to deliver short- and long-term value for you.
PROTECT THE VALUE OF YOUR INVESTMENT IN ZEVRA:VOTE THE WHITE PROXY CARD TODAY
Your Board and management team have a clear path forward to continue delivering value for all Zevra stockholders as we advance the Company’s transformation into a leading rare disease company. Your refreshed Zevra Board is active, engaged, and delivering results. Our delivery of significant stock price appreciation, strong execution, and operational successes demonstrate that we have the right leadership team and strategic vision in place. Maintaining this momentum is critical to Zevra's continued transformation into a leading rare disease company and to our continued efforts in delivering sustained value for you, our valued stockholders.
Whether or not you plan to attend the Annual Meeting, you have an opportunity to protect your investment in Zevra by voting the WHITE proxy card. Your vote is extremely important, no matter how many or how few shares you own. We urge you to vote today, either online or by signing and dating the enclosed WHITE proxy card and returning it in the postage-paid envelope provided. The Board strongly urges you to discard and not to sign or return any blue proxy card sent to you by Mr. Mangless.
This is an important moment for Zevra’s future, and we hope we can count on your support. For all of the reasons we have outlined, we encourage you to vote on the WHITE proxy card "FOR" the re-election of Wendy Dixon, Ph.D. and Tamara A. Favorito to the Company’s Board as Class I directors to hold office until the 2028 Annual Meeting of Stockholders.
We are excited about the opportunities ahead for Zevra. Thank you for your continued support.
Sincerely,
The Zevra Board of Directors
About Zevra Therapeutics, Inc.
Zevra Therapeutics, Inc. is a commercial-stage company combining science, data, and patient need to create transformational therapies for rare diseases with limited or no treatment options. Our mission is to bring life-changing therapeutics to people living with rare diseases. With unique, data-driven development and commercialization strategies, the Company is overcoming complex drug development challenges to make new therapies available to the rare disease community.
For more information, please visit www.zevra.com or follow us on X and LinkedIn.
Cautionary Note Concerning Forward-Looking Statements
This press release may contain forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. Forward-looking statements include all statements that do not relate solely to historical or current facts, including without limitation statements regarding the Company’s actions to enhance stockholder value, the Company’s plans with respect to director candidates nominated by stockholders; the Company’s strategic, financial, operational, and product development objectives; and the timing of any of the foregoing. Forward-looking statements are based on information currently available to Zevra and its current plans or expectations. They are subject to several known and unknown uncertainties, risks, and other important factors that may cause our actual results, performance, or achievements to be materially different from any future results, performance, or achievements expressed or implied by the forward-looking statements, as described in detail in the "Risk Factors" section of Zevra’s Annual Report on Form 10-K for the year ended December 31, 2024, and Zevra’s other filings with the SEC. While we may elect to update such forward-looking statements at some point in the future, except as required by law, we disclaim any obligation to do so, even if subsequent events cause our views to change. Although we believe the expectations reflected in such forward-looking statements are reasonable, we cannot assure that such expectations will prove correct. These forward-looking statements should not be relied upon as representing our views as of any date after the date of this press release.
Additional Information and Where to Find It
Zevra has filed with the SEC a definitive proxy statement on Schedule 14A, containing a form of WHITE proxy card, with respect to its solicitation of proxies for the 2025 Annual Meeting of Stockholders.
INVESTORS AND SECURITY HOLDERS ARE URGED TO READ CAREFULLY AND IN THEIR ENTIRETY THE PROXY STATEMENT (INCLUDING ANY AMENDMENTS OR SUPPLEMENTS THERETO) FILED BY ZEVRA, AND ANY OTHER RELEVANT DOCUMENTS TO BE FILED BY ZEVRA WITH THE SEC WHEN THEY BECOME AVAILABLE, BECAUSE THEY WILL CONTAIN IMPORTANT INFORMATION ABOUT ANY SOLICITATION.
Investors and security holders may obtain copies of these documents and other documents filed with the SEC by Zevra free of charge through the website maintained by the SEC at www.sec.gov. Copies of the documents filed by Zevra are also available free of charge by accessing Zevra’s investor relations website at investors.zevra.com.
Participants in the Solicitation
Zevra, its directors, executive officers, and employees may be deemed to be participants in the solicitation of proxies with respect to a solicitation by Zevra. Information about Zevra’s executive officers and directors is available under the heading “Information about our Executive Officers and Directors” in Part I of Zevra’s Annual Report on Form 10-K for the year ended December 31, 2024, which was filed with the SEC on March 12, 2025 and under the headings “Proposal 1: Election of Directors,” “Executive Officers,” “Security Ownership of Certain Beneficial Owners and Management” and “Executive Compensation,” and “Director Compensation” in Zevra’s definitive proxy statement on Schedule 14A for its annual meeting of stockholders held in 2025, which was filed with the SEC on April 21, 2025. To the extent holdings of our directors and executive officers of Zevra securities reported in such definitive proxy statement change, such changes will be reflected on Statements of Change in Ownership on Forms 3, 4 or 5 filed with the SEC. These documents are available free of charge at the SEC’s website at www.sec.gov. Copies of the documents are also available free of charge by accessing Zevra’s investor relations website at investors.zevra.com.
Contacts
For investor inquiries
Nichol Ochsner+1 (732) 754-2545nochsner@zevra.com
For media inquiries
FTI ConsultingTanner Kaufman / Kyla MacLennan / Mike Gaudreauzevratherapeutics@fticonsulting.com
1 Source: FactSet Research Systems Inc.2 Source: FactSet Research Systems Inc. as of 4/17/25 3 Source: FactSet Research Systems Inc.
Photos accompanying this announcement are available at https://www.globenewswire.com/NewsRoom/AttachmentNg/6d5679f5-0a5d-493b-b14b-3c2f48df6987
https://www.globenewswire.com/NewsRoom/AttachmentNg/ce75ec49-2edd-4b5e-b286-a2dc3de3a0d9
https://www.globenewswire.com/NewsRoom/AttachmentNg/e861f5be-f8ec-4a72-a9cf-f736c0847ee9
https://www.globenewswire.com/NewsRoom/AttachmentNg/edf0ce9f-f81b-4444-9d54-36c2be2ea69d
https://www.globenewswire.com/NewsRoom/AttachmentNg/011661ce-8fcf-4fc4-937a-a2bc1f608cc7
临床3期临床结果高管变更临床2期上市批准
2025-04-20
·精准药物
1引言创新与患者需求驱动药物审批2025年1月美国食品药品监督管理局(FDA)药物评价与研究中心(CDER)的2024年度报告《Advancing Health Through Innovation: New Drug Therapy Approvals 2024》展示了其在推动安全有效药物上市方面的核心作用。报告不仅列举了50种新药的批准情况,还深入分析了加速审批机制、罕见病治疗药物的进展以及生物类似药的里程碑式成就。这份报告不仅是对2024年药物审批工作的总结,更是对全球医药创新生态的深刻洞察。主要章节包括:• 执行摘要:概述年度审批亮点与整体趋势• 2024年CDER创新药物批准:按类别(首创新药、罕见病药物等)详细列出• 加速开发与审评路径的应用:分析快速通道、突破性疗法等工具的使用• PDUFA目标达成情况:评估审评时效性• 新适应症与儿科扩展:已批准药物的新用途及儿童患者覆盖• 生物类似药批准:推动市场竞争与患者可及性• 其他重要审批:新剂型、非处方药转换等创新22024年新药审批亮点创新与效率的平衡01执行摘要2024年,CDER共批准50种创新药物(含新分子实体与生物制剂),同比2022年增加了13例获批新药,而比2023年少5例。下图显示,从2015年到2024年,CDER平均每年有47种新药获批。(图片源于FDA官网)其中:• 52%(26种)针对罕见病,覆盖尼曼-匹克病C型、杜氏肌营养不良等无药可治的疾病。• 48%(24种)为“首创新药”(First-in-Class),具有全新药理机制,如抗精神分裂药物Cobenfy(首个毒蕈碱受体激动剂)。• 66%(33种)通过加速审评路径(快速通道、突破性疗法等)获批,缩短上市时间。• 94%的申请在PDUFA目标日期前完成审评,体现高效流程管理。重点领域:感染性疾病、癌症、神经系统疾病(如阿尔茨海默病新药Kisunla)、心血管疾病及孤儿药。022024年CDER创新药物批准1.首创新药(First-in-Class)2024年,CDER批准了50种新药(novel drugs),其中包括24种首创新药(first-in-class drugs)。这些药物覆盖了从阿尔茨海默病到罕见遗传病的广泛疾病领域。(图片源于FDA官网)例如:• Anktiva:首个膀胱内灌注疗法,用于卡介苗(BCG)无应答的非肌层浸润性膀胱癌。• Cobenfy:首个毒蕈碱受体激动剂,通过调节乙酰胆碱信号改善精神分裂症状。• Lumisight:术中荧光成像剂,辅助乳腺癌手术精准切除。• Rezdiffra:加速批准的首个MASH(代谢功能障碍相关脂肪性肝炎)治疗药物。衍生思考:首创新药占比高达48%,表明基础研究向临床转化的效率正在提升。然而,首创新药的高昂研发成本是否会导致其可及性受限?如何在鼓励创新与保障患者可负担性之间找到平衡,是未来政策制定的关键。2.罕见病药物2024年批准的26种罕见病药物中,包括针对尼曼-皮克病C型(Niemann-Pick disease type C)和WHIM综合征的疗法。这些疾病此前几乎无药可用,新药的批准为患者带来了希望。• Aqneursa:治疗C型尼曼匹克病患者的神经症状,填补空白。• Crenessity:经典先天性肾上腺皮质增生症的首个靶向疗法。• Ojemda:儿童低级别胶质瘤的RAF突变靶向药。• Xolremdi:WHIM综合征(免疫缺陷罕见病)的首个疗法。衍生思考:罕见病药物的高额研发成本和小市场规模导致其价格居高不下。以Zolbetuximab(Vyloy)为例,其治疗胃癌的年费用可能超过30万美元。这种“孤儿药悖论”是否会加剧医疗资源分配的不平等?如何缓解这一矛盾也需要思考。3.其他重要创新药物• Kisunla:阿尔茨海默病新药,靶向淀粉样蛋白斑块。• Zevtera:广谱抗生素,覆盖金黄色葡萄球菌血流感染等重症。• Exblifep:复合抗生素,治疗复杂尿路感染。03加速开发与审评路径的应用66%的新药通过了加速审批通道(如快速通道、突破性疗法等),CDER通过四大加速工具提升效率:• 快速通道(Fast Track):44%新药获此资格,允许滚动提交与优先沟通。快速通道通过提高FDA和药物开发人员之间的沟通水平以及使CDER能够滚动审查药物申请的部分内容来加快新药和生物制剂的开发和审查。• 突破性疗法(Breakthrough Therapy):36%新药入选,提供高强度指导。突破性疗法的指定包括所有快速通道项目的特点,并在药物开发过程中提供强化的FDA指导,包括高级管理人员的参与。• 优先审评(Priority Review):56%申请缩短至6个月内完成。如果CDER确定药物治疗严重疾病,并且如果批准,将在治疗、诊断或预防严重疾病的安全性或有效性方面提供显着改进,则药物将获得优先审查。• 加速批准(Accelerated Approval):14%新药基于替代终点提前上市,需后续验证(如Rezdiffra)。与传统的审批途径相比,该计划能以更快的速度将某些满足不了医疗需求(unmet medical needs)的药物推向市场。对于治疗严重疾病的新药来说,加速审批可能是一种选择,这种新药与现有疗法相比具有显著优势。对于符合加速审批条件的药物,在确定其安全性和有效性时,将不以直接的临床疗效为依据,而是以两个替代终点之一为依据:(1) 有可能预测临床疗效的替代终点( a surrogate endpoint that is reasonably likely to predict clinical benefit);或 (2) 有可能预测临床疗效的中间临床终点(an intermediate clinical endpoint that is reasonably likely to predict clinical benefit)。使用这些终点可以使药物的研究时间比传统审批的典型时间更短,并根据这些研究结果获得加速审批。但重要的是,对于通过加速审批途径批准的产品,FDA要求进行批准后研究,以确认临床获益,而且,除其他外,FDA还可能通过加速程序从市场上撤回未能确认临床获益的加速审批产品。典型案例:• Imdelltra(小细胞肺癌):通过加速批准上市,基于肿瘤缓解率数据。• Ziihera(胆道癌):基于客观缓解率加速批准,后续需验证生存获益。衍生思考:加速审批机制虽然缩短了药物上市时间,但也可能降低对长期安全性和有效性的要求。以加速审批的药物为例,后续确认性试验失败的概率是否高于传统审批路径?如何在加速审批与科学严谨性之间建立动态平衡需要申请人和监管长远考量。04审评效率与PDUFA目标达成• 94%的新药在PDUFA目标日期前获批,反映CDER审评流程的高度可预测性。• 74%的申请在“首轮审评”中通过,归因于早期与申办方的清晰沟通。• 68%新药在美国率先获批,彰显其全球药物创新领导地位。05新适应症与儿科扩展1.新适应症批准• Enhertu:从HER2阳性乳腺癌扩展至所有HER2高表达实体瘤。• Wegovy:减肥药新增心血管事件风险降低适应症。• Xolair:哮喘药扩展用于IgE介导的食物过敏。2.儿科人群扩展• Dupixent(达必妥):嗜酸性食管炎治疗年龄下限降至1岁。• Farxiga(达格列净):2型糖尿病适应症扩展至10岁以上儿童。• Veklury(瑞德西韦):COVID-19治疗覆盖新生儿(出生时体重至少1.5公斤的患者)。3生物类似药降低成本与扩大可及性的双刃剑2024年,CDER批准了18种生物类似药,创下历史新高。其中包括9种可互换生物类似药(interchangeable biosimilars),如Bkemv(eculizumab-aeeb)和Jubbonti(denosumab-bbdz)。这些药物可在无需医生干预的情况下替代原研药,类似于仿制药的模式。2024年CDER批准18种生物类似药,创年度纪录,涉及8个参照产品:• Bkemv(首款可互换的依库珠单抗类似药):用于PNH与aHUS。• Jubbonti/Wyost(地诺单抗类似药):治疗骨质疏松与癌症骨事件。• Opuviz/Yesafili(阿柏西普类似药):眼底疾病治疗。衍生思考:生物类似药的推广虽然降低了治疗成本,但也引发了对原研药企业研发动力的担忧。以Humira(修美乐)为例,其好几个种生物类似药的获批是否会导致原研药企业减少对下一代药物的研发投入?这种“创新-竞争”悖论是否需要通过专利补偿或市场独占期延长来缓解?其他重要审批• Neffy:首个鼻用肾上腺素,急救过敏性反应。• Aurlumyn:冻伤治疗新药,减少截肢风险。• Tecentriq Hybreza:首个皮下注射PD-L1抗体,提升患者便利性。4新药审批趋势与未来展望01罕见病治疗的突破2024年罕见病药物占比过半,反映监管机构对未满足医疗需求的重视。基因疗法与精准靶向药(如Xolremdi针对WHIM综合征)成为趋势,但定价与可及性仍是挑战。未来需探索医保覆盖与全球准入策略。02加速审评的平衡加速路径虽加快新药上市,但依赖替代终点可能增加疗效不确定性(如Rezdiffra需后续验证)。需强化上市后研究监管,确保风险收益比透明化。03生物类似药的竞争格局生物类似药数量激增,但市场渗透率受专利诉讼与医生偏好影响。但需加强公众教育与政策支持。04儿科用药的扩展儿科适应症扩展体现“儿童不是小大人”的研发理念,但儿童临床试验仍面临招募难与伦理争议。自适应试验设计与真实世界证据或成突破口。05阿尔茨海默病的希望与争议Kisunla等淀粉样蛋白靶向药获批,延缓认知衰退,但疗效幅度有限且副作用显著。未来需探索多靶点联合疗法与早期干预策略。06监管建立/优化动态审批框架例如推动全球监管协调,通过跨国审批机制降低重复性试验成本。对罕见病药物实施分级定价策略,根据患者收入水平调整费用。— 结语 —2024年新药审批展现了FDA在创新与效率间的平衡,既推动突破性疗法上市,又坚守安全性与科学性。未来,随着AI辅助审评、真实世界数据应用深化,监管科学有望进一步提速,但核心仍在于以患者为中心的价值医疗导向。参考来源:https://www.fda.gov/drugs/novel-drug-approvals-fda/novel-drug-approvals-2024声明:发表/转载本文仅仅是出于传播信息的需要,并不意味着代表本公众号观点或证实其内容的真实性。据此内容作出的任何判断,后果自负。若有侵权,告知必删!长按关注本公众号 粉丝群/投稿/授权/广告等请联系公众号助手 觉得本文好看,请点这里↓
突破性疗法加速审批孤儿药生物类似药
2025-04-18
·同写意
以“医药健康产业的变革与新生”为主题,汇聚行业领袖,聚焦创新药械和消费医疗,探讨产业变革,交流商业保险与AI技术带来的机遇,助力行业新生。火热报名中1引言创新与患者需求驱动药物审批2025年1月美国食品药品监督管理局(FDA)药物评价与研究中心(CDER)的2024年度报告《Advancing Health Through Innovation: New Drug Therapy Approvals 2024》展示了其在推动安全有效药物上市方面的核心作用。报告不仅列举了50种新药的批准情况,还深入分析了加速审批机制、罕见病治疗药物的进展以及生物类似药的里程碑式成就。这份报告不仅是对2024年药物审批工作的总结,更是对全球医药创新生态的深刻洞察。主要章节包括:• 执行摘要:概述年度审批亮点与整体趋势• 2024年CDER创新药物批准:按类别(首创新药、罕见病药物等)详细列出• 加速开发与审评路径的应用:分析快速通道、突破性疗法等工具的使用• PDUFA目标达成情况:评估审评时效性• 新适应症与儿科扩展:已批准药物的新用途及儿童患者覆盖• 生物类似药批准:推动市场竞争与患者可及性• 其他重要审批:新剂型、非处方药转换等创新22024年新药审批亮点创新与效率的平衡01执行摘要2024年,CDER共批准50种创新药物(含新分子实体与生物制剂),同比2022年增加了13例获批新药,而比2023年少5例。下图显示,从2015年到2024年,CDER平均每年有47种新药获批。(图片源于FDA官网)其中:• 52%(26种)针对罕见病,覆盖尼曼-匹克病C型、杜氏肌营养不良等无药可治的疾病。• 48%(24种)为“首创新药”(First-in-Class),具有全新药理机制,如抗精神分裂药物Cobenfy(首个毒蕈碱受体激动剂)。• 66%(33种)通过加速审评路径(快速通道、突破性疗法等)获批,缩短上市时间。• 94%的申请在PDUFA目标日期前完成审评,体现高效流程管理。重点领域:感染性疾病、癌症、神经系统疾病(如阿尔茨海默病新药Kisunla)、心血管疾病及孤儿药。022024年CDER创新药物批准1.首创新药(First-in-Class)2024年,CDER批准了50种新药(novel drugs),其中包括24种首创新药(first-in-class drugs)。这些药物覆盖了从阿尔茨海默病到罕见遗传病的广泛疾病领域。(图片源于FDA官网)例如:• Anktiva:首个膀胱内灌注疗法,用于卡介苗(BCG)无应答的非肌层浸润性膀胱癌。• Cobenfy:首个毒蕈碱受体激动剂,通过调节乙酰胆碱信号改善精神分裂症状。• Lumisight:术中荧光成像剂,辅助乳腺癌手术精准切除。• Rezdiffra:加速批准的首个MASH(代谢功能障碍相关脂肪性肝炎)治疗药物。衍生思考:首创新药占比高达48%,表明基础研究向临床转化的效率正在提升。然而,首创新药的高昂研发成本是否会导致其可及性受限?如何在鼓励创新与保障患者可负担性之间找到平衡,是未来政策制定的关键。2.罕见病药物2024年批准的26种罕见病药物中,包括针对尼曼-皮克病C型(Niemann-Pick disease type C)和WHIM综合征的疗法。这些疾病此前几乎无药可用,新药的批准为患者带来了希望。• Aqneursa:治疗C型尼曼匹克病患者的神经症状,填补空白。• Crenessity:经典先天性肾上腺皮质增生症的首个靶向疗法。• Ojemda:儿童低级别胶质瘤的RAF突变靶向药。• Xolremdi:WHIM综合征(免疫缺陷罕见病)的首个疗法。衍生思考:罕见病药物的高额研发成本和小市场规模导致其价格居高不下。以Zolbetuximab(Vyloy)为例,其治疗胃癌的年费用可能超过30万美元。这种“孤儿药悖论”是否会加剧医疗资源分配的不平等?如何缓解这一矛盾也需要思考。3.其他重要创新药物• Kisunla:阿尔茨海默病新药,靶向淀粉样蛋白斑块。• Zevtera:广谱抗生素,覆盖金黄色葡萄球菌血流感染等重症。• Exblifep:复合抗生素,治疗复杂尿路感染。03加速开发与审评路径的应用66%的新药通过了加速审批通道(如快速通道、突破性疗法等),CDER通过四大加速工具提升效率:• 快速通道(Fast Track):44%新药获此资格,允许滚动提交与优先沟通。快速通道通过提高FDA和药物开发人员之间的沟通水平以及使CDER能够滚动审查药物申请的部分内容来加快新药和生物制剂的开发和审查。• 突破性疗法(Breakthrough Therapy):36%新药入选,提供高强度指导。突破性疗法的指定包括所有快速通道项目的特点,并在药物开发过程中提供强化的FDA指导,包括高级管理人员的参与。• 优先审评(Priority Review):56%申请缩短至6个月内完成。如果CDER确定药物治疗严重疾病,并且如果批准,将在治疗、诊断或预防严重疾病的安全性或有效性方面提供显着改进,则药物将获得优先审查。• 加速批准(Accelerated Approval):14%新药基于替代终点提前上市,需后续验证(如Rezdiffra)。与传统的审批途径相比,该计划能以更快的速度将某些满足不了医疗需求(unmet medical needs)的药物推向市场。对于治疗严重疾病的新药来说,加速审批可能是一种选择,这种新药与现有疗法相比具有显著优势。对于符合加速审批条件的药物,在确定其安全性和有效性时,将不以直接的临床疗效为依据,而是以两个替代终点之一为依据:(1) 有可能预测临床疗效的替代终点( a surrogate endpoint that is reasonably likely to predict clinical benefit);或 (2) 有可能预测临床疗效的中间临床终点(an intermediate clinical endpoint that is reasonably likely to predict clinical benefit)。使用这些终点可以使药物的研究时间比传统审批的典型时间更短,并根据这些研究结果获得加速审批。但重要的是,对于通过加速审批途径批准的产品,FDA要求进行批准后研究,以确认临床获益,而且,除其他外,FDA还可能通过加速程序从市场上撤回未能确认临床获益的加速审批产品。典型案例:• Imdelltra(小细胞肺癌):通过加速批准上市,基于肿瘤缓解率数据。• Ziihera(胆道癌):基于客观缓解率加速批准,后续需验证生存获益。衍生思考:加速审批机制虽然缩短了药物上市时间,但也可能降低对长期安全性和有效性的要求。以加速审批的药物为例,后续确认性试验失败的概率是否高于传统审批路径?如何在加速审批与科学严谨性之间建立动态平衡需要申请人和监管长远考量。04审评效率与PDUFA目标达成• 94%的新药在PDUFA目标日期前获批,反映CDER审评流程的高度可预测性。• 74%的申请在“首轮审评”中通过,归因于早期与申办方的清晰沟通。• 68%新药在美国率先获批,彰显其全球药物创新领导地位。05新适应症与儿科扩展1.新适应症批准• Enhertu:从HER2阳性乳腺癌扩展至所有HER2高表达实体瘤。• Wegovy:减肥药新增心血管事件风险降低适应症。• Xolair:哮喘药扩展用于IgE介导的食物过敏。2.儿科人群扩展• Dupixent(达必妥):嗜酸性食管炎治疗年龄下限降至1岁。• Farxiga(达格列净):2型糖尿病适应症扩展至10岁以上儿童。• Veklury(瑞德西韦):COVID-19治疗覆盖新生儿(出生时体重至少1.5公斤的患者)。3生物类似药降低成本与扩大可及性的双刃剑2024年,CDER批准了18种生物类似药,创下历史新高。其中包括9种可互换生物类似药(interchangeable biosimilars),如Bkemv(eculizumab-aeeb)和Jubbonti(denosumab-bbdz)。这些药物可在无需医生干预的情况下替代原研药,类似于仿制药的模式。2024年CDER批准18种生物类似药,创年度纪录,涉及8个参照产品:• Bkemv(首款可互换的依库珠单抗类似药):用于PNH与aHUS。• Jubbonti/Wyost(地诺单抗类似药):治疗骨质疏松与癌症骨事件。• Opuviz/Yesafili(阿柏西普类似药):眼底疾病治疗。衍生思考:生物类似药的推广虽然降低了治疗成本,但也引发了对原研药企业研发动力的担忧。以Humira(修美乐)为例,其好几个种生物类似药的获批是否会导致原研药企业减少对下一代药物的研发投入?这种“创新-竞争”悖论是否需要通过专利补偿或市场独占期延长来缓解?其他重要审批• Neffy:首个鼻用肾上腺素,急救过敏性反应。• Aurlumyn:冻伤治疗新药,减少截肢风险。• Tecentriq Hybreza:首个皮下注射PD-L1抗体,提升患者便利性。4新药审批趋势与未来展望01罕见病治疗的突破2024年罕见病药物占比过半,反映监管机构对未满足医疗需求的重视。基因疗法与精准靶向药(如Xolremdi针对WHIM综合征)成为趋势,但定价与可及性仍是挑战。未来需探索医保覆盖与全球准入策略。02加速审评的平衡加速路径虽加快新药上市,但依赖替代终点可能增加疗效不确定性(如Rezdiffra需后续验证)。需强化上市后研究监管,确保风险收益比透明化。03生物类似药的竞争格局生物类似药数量激增,但市场渗透率受专利诉讼与医生偏好影响。但需加强公众教育与政策支持。04儿科用药的扩展儿科适应症扩展体现“儿童不是小大人”的研发理念,但儿童临床试验仍面临招募难与伦理争议。自适应试验设计与真实世界证据或成突破口。05阿尔茨海默病的希望与争议Kisunla等淀粉样蛋白靶向药获批,延缓认知衰退,但疗效幅度有限且副作用显著。未来需探索多靶点联合疗法与早期干预策略。06监管建立/优化动态审批框架例如推动全球监管协调,通过跨国审批机制降低重复性试验成本。对罕见病药物实施分级定价策略,根据患者收入水平调整费用。— 结语 —2024年新药审批展现了FDA在创新与效率间的平衡,既推动突破性疗法上市,又坚守安全性与科学性。未来,随着AI辅助审评、真实世界数据应用深化,监管科学有望进一步提速,但核心仍在于以患者为中心的价值医疗导向。参考来源:https://www.fda.gov/drugs/novel-drug-approvals-fda/novel-drug-approvals-2024
突破性疗法加速审批孤儿药生物类似药上市批准
分析
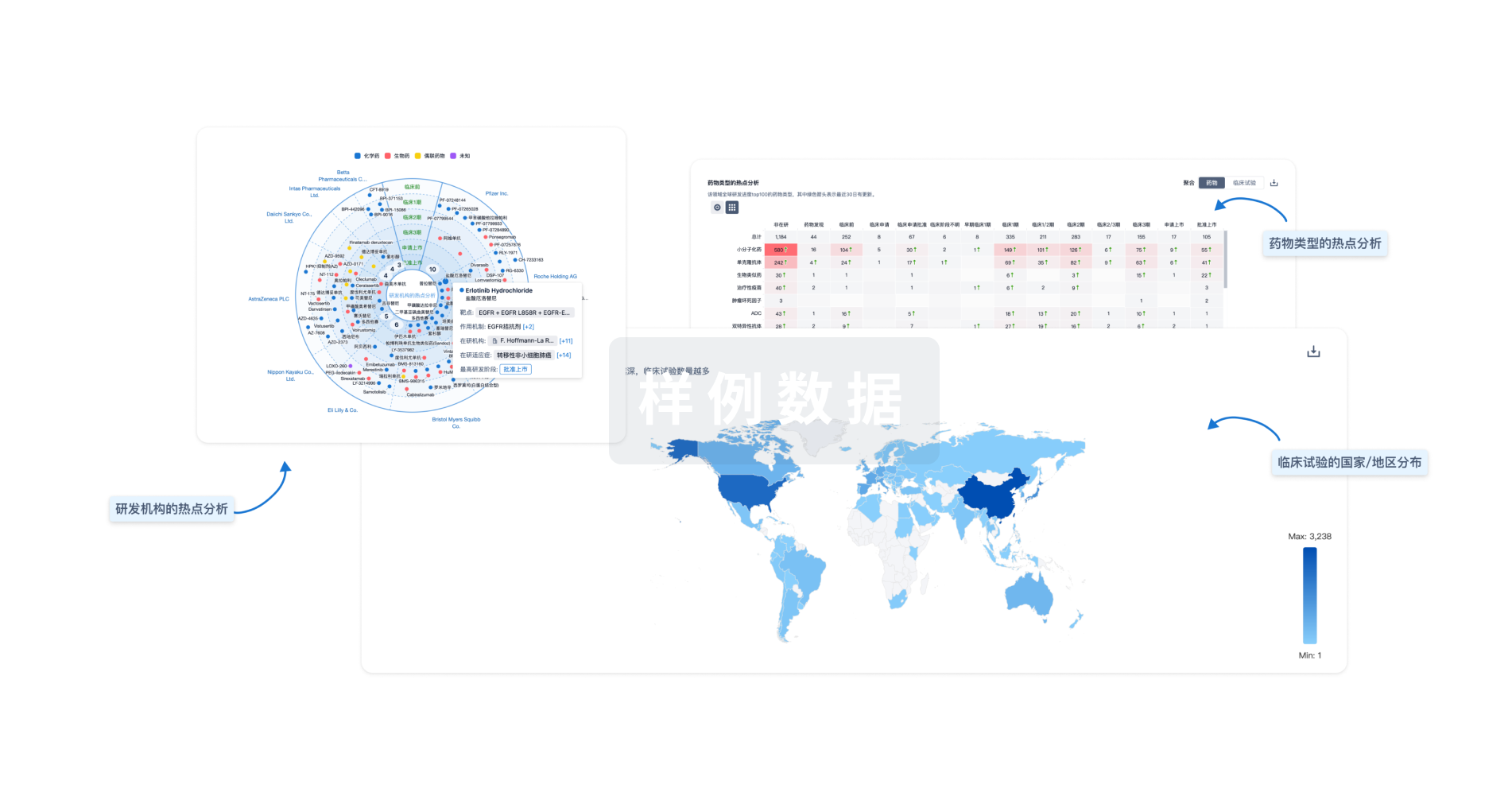
对领域进行一次全面的分析。
登录
或
生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用






