预约演示
更新于:2025-09-09
Ferring Holding SA
更新于:2025-09-09
概览
标签
肿瘤
消化系统疾病
免疫系统疾病
三特异性NK细胞结合器
生物药
小分子化药
疾病领域得分
一眼洞穿机构专注的疾病领域
暂无数据
技术平台
公司药物应用最多的技术
暂无数据
靶点
公司最常开发的靶点
暂无数据
| 排名前五的药物类型 | 数量 |
|---|---|
| 小分子化药 | 9 |
| 合成多肽 | 9 |
| 激素 | 7 |
| 活菌制剂 | 3 |
| 单克隆抗体 | 2 |
关联
39
项与 Ferring Holding SA 相关的药物靶点 |
作用机制 IFNAR激动剂 [+1] |
在研适应症 |
非在研适应症 |
最高研发阶段批准上市 |
首次获批国家/地区 美国 |
首次获批日期2022-12-16 |
靶点- |
作用机制 细菌替代物 [+1] |
最高研发阶段批准上市 |
首次获批国家/地区 美国 |
首次获批日期2022-11-30 |
作用机制 硫酸软骨素蛋白聚糖调节剂 [+1] |
在研机构 |
原研机构 |
在研适应症 |
最高研发阶段批准上市 |
首次获批国家/地区 日本 |
首次获批日期2018-03-23 |
373
项与 Ferring Holding SA 相关的临床试验NCT07152730
An Open-label Trial Investigating the PK of Gonadotropin Releasing Hormone (GnRH) Administrated Subcutaneously Via the OmniPod Delivery System in Healthy Female Subjects
One of the Causes of Amenorrhea is hypothalamic amenorrhea, which is a deficiency in the amplitude and/or frequency of endogenous Gonadotropin-releasing hormone (GnRH) Pulses. The results of this leads to anovulation and cessation of the menstrual cycle. Pulsatile GnRH has been widely used in the United States (US), Canada, and Europe for almost 40 years in the treatment of primary hypothalamic amenorrhea. It has a favorable safety profile and a high degree of effectiveness in enabling ovulation and spontaneous pregnancy. At the moment there are no other GnRH products on the market, nor are there any other drugs marketed for induction of ovulation in women with primary hypothalamic amenorrhea in the US, creating a clear unmet medical need. The goal of this trial is to characterize the exposure variability of GnRH when administered via the OmniPod.
开始日期2025-10-07 |
申办/合作机构 |
NCT07153367
Using a Fixed Dosage of Follitropin Delta for Ovarian Stimulation for Intrauterine Insemination: Rekovelle for Intrauterine Successful Experience (RISE)
The aim of this study is to evaluate the use of a fixed dose of 3.66 mcg of follitropin delta (Rekovelle) on ovarian response during ovarian stimulation for intrauterine insemination (IUI). The assumption is that this fixed dose will be effective while minimizing the risk of multiple pregnancies. A recent study demonstrated the efficacy of follitropin delta for ovarian stimulation in IUI, and it has been approved in France for controlled ovarian stimulation in assisted reproductive techniques such as in vitro fertilization (IVF) and IVF with intracytoplasmic sperm injection (ICSI). With an adapted dose of Rekovelle for patients with regular menstrual cycles, ovarian stimulation is optimized from the first attempt, promoting the development of two follicles to improve pregnancy chances while reducing the risk of multiple pregnancies. This approach aims to achieve pregnancy faster and provide a more comfortable treatment experience.
开始日期2025-09-01 |
申办/合作机构 |
NCT07029451
Prospective, Post-authorisation, Multi-centre, Non-interventional Study to Investigate REKOVELLE in Chinese Women Undergoing Assisted Reproductive Technologies: Effectiveness, Safety, and Patterns of Use in Real-world Practice
This is a multi-centre prospective non-interventional trial to assess the pattern of use of REKOVELLE® in Women Undergoing In Vitro Fertilisation (IVF) or Intracytoplasmic Sperm Injection (ICSI) Procedures in Routine Clinical Practice.
There will be approximately 2500 patients initiated with REKOVELLE® treatment and enrolled in 30-50 sites in China. The total duration of the study will be approximately 35 months.
There will be approximately 2500 patients initiated with REKOVELLE® treatment and enrolled in 30-50 sites in China. The total duration of the study will be approximately 35 months.
开始日期2025-08-05 |
申办/合作机构 |
100 项与 Ferring Holding SA 相关的临床结果
登录后查看更多信息
0 项与 Ferring Holding SA 相关的专利(医药)
登录后查看更多信息
732
项与 Ferring Holding SA 相关的文献(医药)2025-10-01·JOURNAL OF CONTROLLED RELEASE
Impact of chemical structure, lipidation and formulation on luminal stability and intestinal absorption of GLP-1 analogues
Article
作者: Hugerth, Andreas ; Harun, Said ; Bergström, Christel A S ; Davies, Nigel ; Revell, Jefferson ; Englund, Maria ; Emeh, Prosper ; Santisteban Valencia, Zulma
Oral delivery of therapeutic peptides is limited by degradation by digestive proteases and poor gastrointestinal permeability. We have investigated how physicochemical properties, including degree of lipidation and degree of amino acid sequence modification, along with formulation with a permeation enhancer (PE), influence the enzymatic stability and intestinal absorption of glucagon-like peptide-1(GLP-1) receptor agonists. We compared four peptides: J211 (non-lipidated; modified), J229 (mono-lipidated; modified), MEDI7219 (bis-lipidated; modified), and semaglutide (mono-lipidated control; least modified). J211, J229 and MEDI7219 have similar amino acid modifications in the peptide sequence to reduce the number of labile proteolytic sites. An in vitro head-to-head comparison between MEDI7219 and semaglutide showed that MEDI7219 was more proteolytically stable (% remaining after 90 min) than semaglutide, which was degraded completely within 10 min. Notably, co-formulation with sodium caprate (C10) improved semaglutide stability, and at least doubled its half-life. Results from in vivo studies in rats following intraduodenal bolus administration, showed that in the absence of C10, the absorption of all the peptides was minimal, with cumulative fractions absorbed below 1 % for all four compounds. Co-formulation with C10 increased the bioavailability of the modified peptides by 35-40-fold, with J211, J229, and MEDI7219 reaching 7.5 %, 4 %, and 17.3 % respectively. Semaglutide's bioavailability improved by ∼200-fold, however bioavailability did not exceed 2 %. These results demonstrate that C10 enhances peptide absorption primarily by increasing intestinal permeability but also likely by improving enzymatic stability of a labile peptide like semaglutide. Furthermore, when comparing the three modified peptides, the degree of lipidation positively correlated with increased intestinal absorption in both the presence and absence of C10.
2025-09-04·EUROPEAN JOURNAL OF PHARMACEUTICAL SCIENCES
Diffusion of macromolecules in extracellular matrix mimetic hydrogels - effect of size and charge.
Article
作者: Pet, Eva ; Mojumdar, Enamul ; Hansson, Per ; Parlow, Julia ; Sjögren, Helen ; Smirnova, Anna
Subcutaneous (SC) injection is the primary alternative to oral administration for therapeutic proteins and peptides. However, bioavailability and absorption rate are often variable and difficult to predict. Therefore, there is a need for new biorelevant and predictive SC in vitro methods. In this study we systematically investigate the effect of size and charge of a macromolecule on its partitioning and diffusion within extracellular matrix (ECM) mimetic hydrogels in order to gain insight on interactions with the components of the ECM affecting the absorption of a drug after SC injection. Hydrogels consisting of either agarose, cross-linked collagen and hyaluronic acid (HA) or cross-linked HA, were made and equilibrated in solutions of FITC-dextrans of varying sizes (4 to 150 kDa) and model peptides of varying net charge (+2 to +9). Partitioning and diffusion coefficients within gel and solution were determined using confocal laser scanning microscopy and fluorescence recovery after photo bleaching (FRAP), and compared to theoretical models. Generally, the partitioning and diffusivities within the gels decreased with increasing molecular weight, which was in good agreement with models describing the effect of obstruction of the gel network corrected for heterogeneity in the gel structure. The cationic peptides were enriched in the oppositely charged gels and their diffusivities decreased with increasing peptide charge. The experimental results were in semi quantitative agreement with an electrostatic model presented in this work.
2025-09-01·North American Spine Society journal
Real-world treatment patterns and management gaps of lumbar disc herniation in the United States
Article
作者: Guo, Amy ; Bhandutia, Amit ; Yang, Min ; Gao, Yipeng ; Chauhan, Kinsuk ; Liu, Jiaxuan ; Liu, Qing ; Liu, Steven
Background:
Lumbar disc herniation (LDH) affects approximately 1% to 3% of the population annually and leads to substantial physical burden, quality-of-life burden, and productivity loss. Commonly used interventions, including pharmacological and epidural steroid injections (ESIs), have limited high-quality evidence to support their effectiveness in the long-term for treating LDH beyond symptom relief. In general, there is a lack of consensus for timing of treatment after LDH onset and limited data on real-world treatment of LDH. The objective of this study was to describe current real-world treatment patterns and inform gaps in clinical management of patients with LDH.
Methods:
A retrospective analysis was performed using data from January 01, 2018 through March 31, 2023 of a United States commercial health insurance claims database (IQVIA PharMetrics Plus). Patients aged 30-70 years with newly-diagnosed LDH and continuous insurance enrollment for ≥6 months before and ≥12 months after index (first) LDH diagnosis were included. Relevant billing codes were used to identify LDH, related treatments (nonpharmacologic, pharmacologic, invasive), and comorbidities. Demographic and clinical characteristics were summarized for the baseline (preindex) period. Treatment patterns were described over the follow-up period, up to 3 years after LDH diagnosis. Time from LDH diagnosis to ESI(s) and surgery(ies) were calculated.
Results:
A total of 1,086,552 patients with LDH were included, with a mean age of 50.8 years. Patients had a mean follow-up of 27 months after LDH diagnosis. Nearly 20% of patients with LDH underwent ESI, with half of this group undergoing multiple ESIs. Multiple ESIs were associated with a greater likelihood of surgical intervention and repeat surgical intervention compared to those who only underwent single ESI. LDH surgery was performed on 7.2% of patients, approximately 10% of whom had multiple surgeries during follow-up. A large subset (44.1%) of patients who underwent LDH surgery did not have any ESI prior to surgery. General limitations of claims data analyses can include data misclassification, missing claims for diagnoses and procedures that were conducted, missing clinical information (severity of condition, insights into clinical decision making), and some missing patient demographics and characteristics.
Conclusions:
In this study, approximately one quarter of patients with LDH underwent ESI and/or surgery after conservative treatment. Opportunities exist to provide more guideline-concordant care to patients with LDH. In addition, unmet needs exist in the current treatment options for patients with LDH, potentially including the need for other nonsurgical treatment options for patients who do not fully respond to conservative treatment.
384
项与 Ferring Holding SA 相关的新闻(医药)2025-09-05
Rising prevalence of genetic disorders, supportive regulatory frameworks, and advancements in gene and RNA-based therapies are fueling market expansion.
WILMINGTON, DE, UNITED STATES, September 5, 2025 /
EINPresswire.com
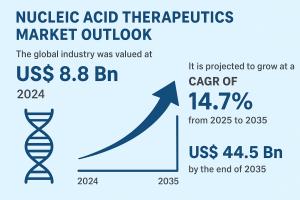
/ -- The global
nucleic acid therapeutics market
is entering a phase of accelerated growth, driven by strong innovation pipelines and favorable policy support. Valued at US$ 8.8 Bn in 2024, the market is projected to grow at a robust CAGR of 14.7% between 2025 and 2035, reaching US$ 44.5 Bn by 2035. Growing awareness of genetic diseases, widespread clinical trials, and rapid progress in RNA-based platforms are reshaping the future of precision medicine.
Market Introduction
Nucleic acid therapeutics represent one of the most transformative innovations in modern biotechnology. These therapies leverage engineered DNA and RNA molecules to correct or silence defective genes, introduce beneficial genetic material, or modulate protein expression. Core modalities include gene therapies, antisense oligonucleotides (ASOs), RNA interference (RNAi), aptamers, and messenger RNA (mRNA)-based therapeutics.
Their therapeutic applications extend across neuromuscular disorders, oncology, viral infections, ophthalmological diseases, metabolic syndromes, and autoimmune conditions, providing high specificity, targeted action, and the potential for one-time curative treatments. Unlike traditional pharmaceuticals, nucleic acid therapeutics address the root cause of diseases at the genetic level, minimizing systemic toxicity and enabling personalized medicine.
Dive Deeper into Data: Get Your In-Depth Sample Now -
https://www.transparencymarketresearch.com/sample/sample.php?flag=S&rep_id=86333
Analyst Viewpoint
Analysts at Transparency Market Research note that the nucleic acid therapeutics market is on the cusp of becoming a mainstream segment in the biopharmaceutical industry.
Key growth catalysts include:
Rising prevalence of rare and inherited disorders such as Duchenne muscular dystrophy, cystic fibrosis, and sickle cell anemia, which remain underserved by conventional treatments.
Regulatory acceleration through FDA Fast Track, Breakthrough Therapy, and EMA Priority Medicines (PRIME) designations, which shorten development timelines for critical therapies.
Challenges remain in terms of manufacturing scalability, delivery mechanisms, and cost-effectiveness, particularly for therapies requiring complex viral or lipid nanoparticle vectors. However, the success of mRNA vaccines during the COVID-19 pandemic has proven the scalability and adaptability of RNA-based platforms, catalyzing confidence among investors and policymakers. The increasing number of clinical trials, coupled with strategic collaborations, points to a market that is set to transform global healthcare by 2035.
Key Market Drivers
1. Rising Prevalence of Genetic Disorders
Genetic diseases affect millions worldwide, with a growing number being diagnosed through advancements in genomic sequencing and prenatal screening. This improved detection rate is expanding the eligible patient pool for nucleic acid therapeutics. As these therapies directly correct or silence defective genes, their adoption is expected to increase significantly in the coming years.
2. Regulatory Approvals and Expedited Pathways
Regulatory agencies such as the U.S. FDA and the European Medicines Agency (EMA) are creating streamlined approval frameworks for nucleic acid therapeutics, particularly in the area of rare and orphan diseases. Expedited review processes reduce time-to-market, encouraging pharmaceutical and biotech companies to increase investment in clinical development.
3. Advancements in RNA-based and Gene Therapies
Breakthroughs in antisense oligonucleotides, siRNA, and mRNA platforms are enabling therapies that target diseases previously considered untreatable. The integration of artificial intelligence, nanotechnology-driven delivery systems, and improved chemical modifications has enhanced drug stability, precision targeting, and reduced off-target effects, making RNA-based treatments commercially viable.
Segment Analysis
By Therapy Type
Antisense Oligonucleotides (ASOs): Largest segment due to their efficacy in modulating gene expression; widely used in neuromuscular and rare genetic disorders.
Small Interfering RNA (siRNA): Emerging as a strong segment with applications in oncology and metabolic diseases.
Gene Therapies: High growth potential owing to curative benefits in monogenic disorders.
Aptamers: Gaining momentum in targeted oncology treatments.
Others (including mRNA therapies): Strong future growth expected, particularly post-COVID-19 success.
By Delivery Method
Viral Vector-based Systems: Widely used but face challenges in immunogenicity and manufacturing scalability.
Non-viral Delivery Systems: Lipid nanoparticles and polymer-based carriers showing promising safety and efficiency.
By Route of Administration
Intravenous: Dominant due to systemic distribution.
Subcutaneous: Preferred for ease of administration and chronic therapies.
Others (Intrathecal, etc.): Specialized routes for targeted disease conditions.
By Therapeutic Area
Neuromuscular Disorders (DMD, SMA)
Metabolic Disorders
Cardiovascular Disorders
Ophthalmological Disorders
Oncological Disorders
Others (Infectious, Autoimmune, etc.)
By End-user
Hospitals (largest share due to advanced care infrastructure)
Academic and Research Institutes (driving clinical trials and innovation)
Specialty Centers (focusing on genetic and rare disorders)
Regional Insights
North America: Leads the global market due to strong biotech infrastructure, presence of leading companies, and early adoption of innovative therapies. Favorable reimbursement policies and active regulatory support enhance growth prospects.
Europe: Rapid adoption supported by EMA’s proactive stance on genetic therapies. Strong research ecosystems in Germany, the UK, and France are advancing innovation.
Asia Pacific: Poised for fastest growth with rising healthcare investments, supportive government initiatives, and a growing patient pool in China, India, and Japan.
Latin America & Middle East & Africa: Emerging regions where adoption is gradually increasing, though affordability and access remain challenges. Partnerships with global biotech firms are expected to enhance growth.
Key Players
Novartis AG
Pfizer, Inc.
Sanofi
Novo Nordisk A/S
AstraZeneca plc
Alnylam Pharmaceuticals, Inc.
Amgen Inc.
Sarepta Therapeutics, Inc.
Bluebird Bio, Inc.
CSL Behring LLC
Ferring Pharmaceuticals Inc.
Krystal Biotech, Inc.
PTC Therapeutics, Inc.
Jazz Pharmaceuticals plc
Astellas Pharma Inc.
Recent Developments
Novartis (Nov 2024): Acquired Kate Therapeutics, expanding its AAV-based gene therapy pipeline for neuromuscular diseases in a deal valued up to US$ 1.1 Bn.
Sarepta Therapeutics (Nov 2024): Entered into a licensing agreement with Arrowhead Pharmaceuticals for siRNA programs in muscle and rare pulmonary disorders, valued at US$ 825 Mn.
Market Trends
Accelerating investments in RNA-based and gene-editing platforms.
Rising demand for personalized and precision medicine approaches.
Continuous technological innovations in viral and non-viral delivery platforms.
Increasing collaborations between pharma companies, biotech firms, and academic institutions to fast-track clinical pipelines.
Future Outlook
The nucleic acid therapeutics market is expected to remain one of the fastest-growing sectors within biotechnology and pharmaceuticals through 2035.
Factors such as increasing prevalence of genetic disorders, regulatory acceleration, advancements in RNA and gene therapy platforms, and strong demand for personalized medicine will drive market expansion. With a projected CAGR of 14.7%, the sector is set to deliver groundbreaking treatments that could redefine modern healthcare.
Why Buy This Report?
Reliable market size forecasts and CAGR projections through 2035
In-depth assessment of market drivers, restraints, and opportunities
Comprehensive segmentation by therapy type, delivery method, therapeutic area, and region
Competitive landscape with profiles of leading companies and recent strategic developments
Analysis of emerging technologies and trends shaping future growth
Browse More Trending Research Reports:
Nucleic Acid Extraction Instruments & Reagents Market -
https://www.transparencymarketresearch.com/nucleic-acid-extraction-instruments-reagents-market.html
Oligonucleotide Synthesis Market -
https://www.transparencymarketresearch.com/oligonucleotide-synthesis-market.html
Aptamers Market -
https://www.transparencymarketresearch.com/aptamers-market.html
RNA Therapeutics Market -
https://www.transparencymarketresearch.com/rna-therapeutics-market.html
Small Interfering RNA (siRNA) Therapeutics Market -
https://www.transparencymarketresearch.com/small-interfering-rna-therapeutics-market-report.html
Dark Genome Therapeutics Market -
https://www.transparencymarketresearch.com/dark-genome-therapeutics-market.html
Nucleic Acid Aptamers Market -
https://www.transparencymarketresearch.com/nucleic-acid-aptamers.html
Ligase Enzymes Market -
https://www.transparencymarketresearch.com/ligase-enzymes-market.html
Thermal Cycler Market -
https://www.transparencymarketresearch.com/thermal-cycler-market.html
Electroporation Instruments Market -
https://www.transparencymarketresearch.com/electroporation-instruments-market.html
Legionella Testing Market -
https://www.transparencymarketresearch.com/legionella-testing-market.html
Cell Analysis Market -
https://www.transparencymarketresearch.com/cell-analysis-market.html
Molecular Diagnostics Market -
https://www.transparencymarketresearch.com/molecular-diagnostics-industry.html
Biobanking Market -
https://www.transparencymarketresearch.com/biobanking-market.html
Genetic Testing Services Market -
https://www.transparencymarketresearch.com/genetic-testing-services-market.html
Array Market -
https://www.transparencymarketresearch.com/global-array-market.html
Oligonucleotides Market -
https://www.transparencymarketresearch.com/oligonucleotides-market.html
Digital PCR & Quantitative PCR Market -
https://www.transparencymarketresearch.com/digital-pcr-quantitative-pcr-market.html
About Transparency Market Research
Transparency Market Research, a global market research company registered at Wilmington, Delaware, United States, provides custom research and consulting services. Our exclusive blend of quantitative forecasting and trends analysis provides forward-looking insights for thousands of decision makers. Our experienced team of Analysts, Researchers, and Consultants use proprietary data sources and various tools & techniques to gather and analyses information.
Our data repository is continuously updated and revised by a team of research experts, so that it always reflects the latest trends and information. With a broad research and analysis capability, Transparency Market Research employs rigorous primary and secondary research techniques in developing distinctive data sets and research material for business reports.
Contact:
Transparency Market Research Inc.
CORPORATE HEADQUARTER DOWNTOWN,
1000 N. West Street,
Suite 1200, Wilmington, Delaware 19801 USA
Tel: +1-518-618-1030
USA – Canada Toll Free: 866-552-3453
Website:
https://www.transparencymarketresearch.com
Email: sales@transparencymarketresearch.com
Follow Us: LinkedIn| Twitter| Blog | YouTube
Atil Chaudhari
Transparency Market Research Inc.
+1 518-618-1030
email us here
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability
for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this
article. If you have any complaints or copyright issues related to this article, kindly contact the author above.
并购引进/卖出寡核苷酸
2025-08-18
Male Hypogonadism revenue is expected to grow at a CAGR of 4.8% from 2025 to 2032, reaching nearly USD 5.32 Bn. by 2032.
Advancements in testosterone therapy and diagnostics are reshaping the male hypogonadism market, bringing health and vitality back to millions.”
— Dharati Raut
SAVANNAH, GA, UNITED STATES, August 18, 2025 /
EINPresswire.com
/ -- Stellar Market Research examines the growth rate of the
Male Hypogonadism Market
during the forecasted period 2025-2032
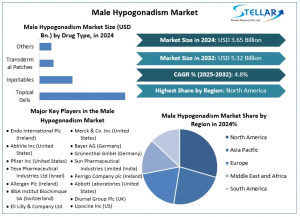
The Male Hypogonadism Market is projected to grow at a CAGR of approximately 4.8% over the forecast period. The Male Hypogonadism Market was valued at USD 3.65 billion in 2024 and is expected to reach USD 5.32 billion by 2032. More old people, higher rates of obesity, better tests, new testosterone treatments, more people knowing about it, growth in telehealth, and better access to health care are all major reasons driving the Male Hypogonadism market.
Male Hypogonadism Market Overview
The male hypogonadism market deals with finding and treating this issue. It grows as more people get old, more get long-term health problems, and more become aware of it. Better tools for finding the issue and new ways to treat it, like gels, shots, and under-skin devices, give more choices for care. More people can now get health care, especially in new markets, and things like health care from home help the market grow. The big companies in this area work on making treatments that are safe and fit the person. All in all, this market looks set to grow steadily, helped by new ideas and a stronger focus on the health and life quality of men.
To know the most attractive segments, click here for a free sample of the report:
https://www.stellarmr.com/report/req_sample/Male-Hypogonadism-Market/1780
Male Hypogonadism Market Dynamics
Drivers
Expansion of Healthcare Infrastructure
Growing health care setups in new markets, like Asia-Pacific, Middle East, and Latin America, are helping more men get checked and treated for low hormone issues. Money put into online health services, AI-based tests, better insurance, and new government rules are making care cheaper and reaching more people. These steps help the market grow by finding health problems sooner, making care fit each person, and spreading treatments wider in areas that didn't have much help before.
Innovation in Treatment Modalities
New forms of testosterone fix (TRT) are now out like FDA-ok'd pills (Jatenzo, TLANDO), nose gels (Natesto), and new skin gels and shots. These types give safer, easier, and one-of-a-kind care that make it better for men to keep up. New updates on FDA labels take away heart health risk alerts and this helps more men get TRT. This makes TRT more open and it works better for men with low hormone levels.
Advances in Diagnostic Technologies
Better ways to find out if a man has low testosterone now exist. Quick auto tests for free testosterone, more home blood and spit kits, and new point-of-care tools are here. Better scans like shear wave elastography and MRI make results more exact. These new things help find the issue sooner, with more right results, make it easy to get help, and lift the number of people getting treated. This helps the market grow by making it easier for people to take care of their health early on and reach more people in places with less help.
Restrain
Lack of Awareness and Social Stigma
Many men with low hormone levels are not diagnosed because their signs mix with old age and there is a bad view on hormone help and being 'manly.' Efforts to spread the word, online health help, and doctor education are making it easier to break these walls. Still, bad views keep many from getting a diagnosis and treatment, holding back market growth as lots of men don't seek the care they need soon enough.
Innovations and Developments
Technological innovation is a key factor propelling the Male Hypogonadism Market forward. Notable advancements include:
Ulo, a telehealth startup, gives custom at-home testosterone help. They offer lab checks, online talks, and send out monthly care from $159. This makes it easy, safe, and easy to reach with steady checks.
In February 2025, the FDA changed the tags on testosterone items. They took off heart risk notes due to new test data and put in blood pressure notes. This made things clearer about safety and lifted trust for users and providers.
Male Hypogonadism Market Segmentation
By Disease Type
By Disease Type, the Male Hypogonadism Market is further segmented into Klinefelter's Syndrome, Kallmann Syndrome, Pituitary Disorders, and Others. Klinefelter Syndrome dominates the male hypogonadism market since it shows up more (1 in 500 males), has better tests, and has new man hormone fixes. It made the most money in 2024, and the market will likely get much bigger by 2034. This growth comes from more people knowing about it and better ways to treat it.
Male Hypogonadism Market Regional Analysis
North America: North America dominates the Male Hypogonadism Market due its top-notch health care set-up, high knowing about the issue, strong insurance help, solid work in research and growth, good laws, and more use of telehealth. All these points make getting a check, getting to treatment, and new ways to treat better.
Europe: Europe is number two in the Male Hypogonadism Market. This is due to good health care, more people knowing about it, lower prices from biosimilar use, helpful rules, and big steps in main places. For example, Germany has special treatment spots and the UK quickly says yes to TRT, making it easy and cheap for more people.
Asia-Pacific: Asia-Pacific is third in the Male Hypogonadism Market due to fast health care upgrades, aid from the government in China, Japan, and India, more knowing about the issue, more ways to get care, and the rising need from a growing middle class.
Recent Developments:
Clarus Therapeutics got a lot of talk with its pill form of testosterone help, JATENZO
®
, and Marius Pharmaceuticals grew big by using special doctor plans. Both teams lead the way in easy, pill-based TRT help.
In September 2024, WellSync teamed up with Marius Pharmaceuticals to give out KYZATREX (a type of pill testosterone) on its web care site, making it easier for patients to get to testosterone change help.
To know the most attractive segments, click here for a free sample of the report:
https://www.stellarmr.com/report/req_sample/Male-Hypogonadism-Market/1780
Male Hypogonadism Market Competitive Landscape
The global and regional players in the Male Hypogonadism Market concentrate on developing and enhancing their capabilities, resulting in fierce competition. Notable players include:
Endo International Plc (Ireland)
AbbVie Inc (United States)
Pfizer Inc (United States)
Teva Pharmaceutical Industries Ltd (Israel)
Allergan Plc (Ireland)
IBSA Institut Biochimque SA (Switzerland)
Eli Lilly & Company Ltd (United States)
Ferring International Center SA (Switzerland)
Merck & Co. Inc (United States)
Bayer AG (Germany)
Related Reports:
PEEK Implants Market:
https://www.stellarmr.com/report/pEEK-implants-market/2742
Medical Device Design Development Services Market:
https://www.stellarmr.com/report/medical-device-design-development-services-market/2741
Iron Deficiency Anemia Therapy Market:
https://www.stellarmr.com/report/iron-deficiency-anemia-therapy-market/2740
North America Aesthetic Medical Devices Market:
https://www.stellarmr.com/report/north-america-aesthetic-medical-devices-market/2719
Veterinary Excipients Market:
https://www.stellarmr.com/report/veterinary-excipients-market/2716
About Stellar Market Research:
Stellar Market Research is a multifaceted market research and consulting company with professionals from several industries. Some of the industries we cover include medical devices, pharmaceutical manufacturers, science and engineering, electronic components, industrial equipment, technology and communication, cars and automobiles, chemical products and substances, general merchandise, beverages, personal care, and automated systems. To mention a few, we provide market-verified industry estimations, technical trend analysis, crucial market research, strategic advice, competition analysis, production and demand analysis, and client impact studies.
Contact Stellar Market Research:
S.no.8, h.no. 4-8 Pl.7/4, Kothrud,
Pinnac Memories Fl. No. 3, Kothrud, Pune,
Pune, Maharashtra, 411029
sales@stellarmr.com
Lumawant Godage
Stellar Market Research
+ +91 9607365656
email us here
Visit us on social media:
LinkedIn
Instagram
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability
for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this
article. If you have any complaints or copyright issues related to this article, kindly contact the author above.
2025-08-13
声明:因水平有限,错误不可避免,或有些信息非最及时,欢迎留言指出。本文仅作医疗健康相关药物介绍,非治疗方案推荐(若涉及);本文不构成任何投资建议。
8月12日,专注于生殖健康的临床阶段生物技术公司 Gameto宣布,该公司已筹集到4400万美元(约3.2亿人民币)的C轮融资。新一轮融资由Overwater Ventures领投,该风投公司由前Khosla Ventures投资人Kristina Simmons于2021年创立。
此前,公司已完成多轮融资,2024 年完成了 3300 万美元的 B 轮融资。2025 年又获得 4400 万美元的 C 轮融资,由 Overwater Ventures 领投,总融资额达到 1.27 亿美元。这些资金将用于支持其产品的研发和临床试验等。
新的资金将使Gameto能够完成对500名希望通过体外受精受孕的人进行的第三阶段研究。中期结果最快将于2026年底公布。
这家初创公司已经开发出卵巢支持细胞,可以筑巢和滋养人类卵子,为试管婴儿做好准备。在医生取卵之前,妇女通常要接受两周的艰苦激素治疗。Gameto公司希望通过提前收集卵子,并在其 “盘中卵巢 ”中使其成熟,从而将疗程缩短到几天。
首席执行官Radenkovic认为,更短的时间表将特别吸引那些希望捐赠卵子或冷冻卵子以备未来体外受精(IVF)使用的年轻女性。
“目前市面上还没有真正具有时代标志性的女性健康品牌。而Gameto将突破性科学与打造消费品牌的能力相结合,”Simmons表示,“我认为这是一个巨大的机遇。”
Gameto的程序是对一种名为体外成熟(IVM)的传统技术的新颖应用,该技术通过在营养培养液中培养卵子。尽管IVM可以缩短激素治疗方案,但由于妊娠成功率通常较低,因此不如传统体外受精(IVF)受欢迎。
该方法基于哈佛大学Church实验室的科学研究,该实验室成功将空白干细胞(具体为来自成年捐赠者的诱导多能干细胞,即iPSCs)转化为支持细胞。Gameto表示,这是首个在美国进行III期临床试验的iPSC衍生治疗方案。
这项双盲临床试验将比较使用Fertilo或传统体外成熟(IVM)处理的卵子,并评估12周时的怀孕率。拉Radenkovic表示,已有12个研究站点招募了约60名参与者,她预计首例胚胎将在8月底前完成移植。
Gameto及其投资者认为,Fertilo将对价值数十亿美元的激素注射市场造成重大冲击,该市场目前由Merck KGaA、Ferring Pharmaceuticals和Organon等少数公司主导,这些公司去年从体外受精激素中获得的收入超过20亿美元。
Gameto正在开发超越Fertilo的项目,包括缓解更年期症状的治疗方案,如脑雾、潮热、肌肉流失、骨质疏松、体重增加和阴道干涩。这项针对卵巢的抗衰老疗法获得了去年秋季政府提供的1000万美元拨款。
关于Gameto
Gameto 是一家专注于女性生殖健康领域的生物技术公司,总部位于美国纽约。成立于 2020 年,由 Dina Radenkovic、Pranam Chatterjee 和 Martin Varsavsky 共同创立,Dina Radenkovic 担任首席执行官。公司规模较小,员工数量在 11-50 人之间。
Gameto研发管线
参考资料:
endpoints
细胞疗法临床研究
100 项与 Ferring Holding SA 相关的药物交易
登录后查看更多信息
100 项与 Ferring Holding SA 相关的转化医学
登录后查看更多信息
组织架构
使用我们的机构树数据加速您的研究。
登录
或

管线布局
2026年05月22日管线快照
管线布局中药物为当前组织机构及其子机构作为药物机构进行统计,早期临床1期并入临床1期,临床1/2期并入临床2期,临床2/3期并入临床3期
药物发现
1
4
临床前
临床1期
3
4
临床2期
临床3期
4
1
申请上市
批准上市
20
45
其他
登录后查看更多信息
药物交易
使用我们的药物交易数据加速您的研究。
登录
或

转化医学
使用我们的转化医学数据加速您的研究。
登录
或

营收
使用 Synapse 探索超过 36 万个组织的财务状况。
登录
或

科研基金(NIH)
访问超过 200 万项资助和基金信息,以提升您的研究之旅。
登录
或

投资
深入了解从初创企业到成熟企业的最新公司投资动态。
登录
或

融资
发掘融资趋势以验证和推进您的投资机会。
登录
或

生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用





