预约演示
更新于:2025-05-07
Refractory Cytopenia of Childhood
儿童顽固性细胞减少症
更新于:2025-05-07
基本信息
别名 Refractory Cytopenia of Childhood、Refractory cytopenia of childhood |
简介 A myeloid disorder that occurs in childhood and is characterized by persistent cytopenia, dysplastic changes in at least two hematopoietic lineages or in 10% or more of cells in one hematopoietic lineage, less than 5% blasts in bone marrow, and less than 2% blasts in peripheral blood, absence of bone marrow fibrosis, and absence of history of prior cytotoxic chemotherapy or radiation therapy. In a subset of cases, somatic mutations or cytogenetic abnormalities can be identified. Other cases are the result of pre-existing disorders with germline mutations, including Fanconi anemia, Shwachman-Diamond syndrome, and dyskeratosis congenita. |
关联
3
项与 儿童顽固性细胞减少症 相关的药物作用机制 TPO receptor激动剂 |
在研机构 |
原研机构 |
最高研发阶段批准上市 |
首次获批国家/地区 澳大利亚 |
首次获批日期2008-08-08 |
靶点 |
作用机制 PD-1抑制剂 |
在研机构 |
原研机构 |
非在研适应症- |
最高研发阶段临床3期 |
首次获批国家/地区- |
首次获批日期1800-01-20 |
靶点- |
作用机制 免疫刺激剂 |
在研机构- |
在研适应症- |
最高研发阶段终止 |
首次获批国家/地区- |
首次获批日期1800-01-20 |
6
项与 儿童顽固性细胞减少症 相关的临床试验NCT04420494
Umbilical Cord Blood Treatment for Refractory Immune Cytopenia: a Single-arm Prospective Study
Immune-related hematocytopenia is a type of immunity Inflammatory cytopenia-mediated diseases, hormones and immunosuppressants are its first-line treatment. However, conventional immunosuppressants are ineffective or have a high recurrence rate. And some patients are not effective for these treatments, due to infection of blood cells, bleeding, decreased quality of life, and even severe death. There is currently no effective method for such patients. This study intends to recruit IRIC patients, give cord blood infusion, observe its efficacy and safety, and detect changes in inflammation-related indicators before and after treatment. There are no relevant reports at China and abroad. This study can provide new treatment options for patients with IRIC.
开始日期2022-04-20 |
申办/合作机构 |
NCT04478227
Single Arm Prospective Open Label Pilot Study Evaluating Short-Term Safety and Efficacy of Romiplostim in Children With Inherited and Acquired Disorders of Hematopoietic Failure
This is an open label, prospective Pilot interventional study will investigate the safety and efficacy of Romiplostim, thrombopoietin (TPO) mimetic, in children (ages: 0 to 21 years) with broad scope of bone marrow failure disorders including acquired and inherited conditions as a first line of therapy along with standard of care.
开始日期2020-08-18 |
申办/合作机构  University of Iowa University of Iowa [+1] |
ChiCTR1900023509
Allogeneic Hematopoietic Stem Cell Transplantation with Risk-adapted Conditioning Regimens for Children with Aplastic Anemia or Refractory Cytopenia of Childhood: a Single-Center Prospective Trial
开始日期2017-01-01 |
申办/合作机构- |
100 项与 儿童顽固性细胞减少症 相关的临床结果
登录后查看更多信息
100 项与 儿童顽固性细胞减少症 相关的转化医学
登录后查看更多信息
0 项与 儿童顽固性细胞减少症 相关的专利(医药)
登录后查看更多信息
74
项与 儿童顽固性细胞减少症 相关的文献(医药)2024-12-01·Bone Marrow Transplantation
Accurate donor and recipient selection and a short time to transplant offer excellent outcomes in upfront hematopoietic stem cell transplantation from matched unrelated donors for pediatric severe aplastic anemia and refractory cytopenia of childhood. A study of the Spanish Pediatric Group for Hematopoietic Cell Transplantation and Cell Therapy (GETH-TC)
Letter
作者: Bueno, David ; Quintero, Victor ; Gonzalez-Vicent, Marta ; Diaz-de-Heredia, Cristina ; Verdu, Jaime ; Lopez-Duarte, Monica ; Fuentes, Carolina ; Alonso, Laura ; Uria-Oficialdegui, M Luz ; Panesso, Melissa ; Murillo-Sanjuan, Laura ; Gomez, Graciela ; Trabazo, Maria ; Benitez-Carabante, M Isabel
2024-06-01·Experimental Hematology
Of gains and losses: SAMD9/SAMD9L and monosomy 7 in myelodysplastic syndrome
Review
作者: Cammenga, Jörg
2024-03-01·International Journal of Hematology
Recent advances in the diagnosis and treatment of pediatric acquired aplastic anemia
Review
作者: Yoshida, Nao
14
项与 儿童顽固性细胞减少症 相关的新闻(医药)2023-04-10
Merck (NYSE: MRK), known as MSD outside of the United States and Canada, and Eisai today provided updates on two Phase 3 trials, LEAP-003 and LEAP-017 investigating KEYTRUDA, Merck’s anti-PD-1 therapy, plus LENVIMA, the orally available multiple receptor tyrosine kinase inhibitor discovered by Eisai.
LEAP-003: Merck and Eisai are discontinuing the Phase 3 LEAP-003 trial evaluating KEYTRUDA plus LENVIMA for the first-line treatment of adults with unresectable or metastatic melanoma. This decision is based on the recommendation of an independent Data Monitoring Committee (DMC), which reviewed data from a planned interim analysis and determined KEYTRUDA plus LENVIMA did not demonstrate an improvement in overall survival (OS), one of the study’s dual primary endpoints, versus KEYTRUDA alone. Merck and Eisai are informing study investigators of the decision and advising them to reach out to patients in the study regarding treatment. At an earlier interim analysis, the trial’s other dual primary endpoint, progression-free survival (PFS), showed a statistically significant improvement in the KEYTRUDA plus LENVIMA arm versus the KEYTRUDA plus placebo arm.
LEAP-017: The Phase 3 LEAP-017 trial evaluating KEYTRUDA plus LENVIMA did not meet its primary endpoint of OS for the treatment of patients with unresectable and metastatic colorectal cancer that is mismatch repair proficient (pMMR) or not microsatellite instability-high (MSI-H) who experienced disease progression on, or became intolerant to, prior therapy. In the final pre-specified analysis of OS, a trend toward improvement was observed with KEYTRUDA plus LENVIMA versus regorafenib or TAS-102 (trifluridine and tipiracil hydrochloride); however, these results did not meet statistical significance per the pre-specified statistical analysis plan. A trend toward improvement was also observed in key secondary endpoints of PFS, objective response rate (ORR) and duration of response (DOR) with KEYTRUDA plus LENVIMA versus regorafenib or TAS-102; however, per the pre-specified statistical analysis plan these results were not tested for statistical significance.
In both the LEAP-003 and LEAP-017 trials, the safety profile of KEYTRUDA plus LENVIMA was consistent with previously reported data on the combination. A full evaluation of the data from these studies, including pre-planned key subgroup analyses, is ongoing. The companies will work with investigators to share the results with the scientific community.
“We are grateful to all the investigators, patients and their families for their participation in these studies, and we will continue to evaluate KEYTRUDA plus LENVIMA across different types of cancer where additional treatment options are needed. We remain fully committed to building on existing treatments as part of our efforts to help as many appropriate patients with cancer as we can,” said Dr. Gregory Lubiniecki, Vice President, Global Clinical Development, Merck Research Laboratories.
“With the LEAP-003 and LEAP-017 trials, we set out to help improve outcomes for patients with two difficult-to-treat advanced cancers, melanoma and colorectal cancer,” said Corina Dutcus, M.D., Senior Vice President, Clinical Development, Oncology at Eisai Inc. “While these results are different from our initial expectation, insights from both studies will help contribute to our understanding of KEYTRUDA plus LENVIMA. We remain confident in LENVIMA as a pillar of Eisai’s oncology portfolio and will continue to evaluate its potential in ongoing trials within the LEAP program.”
KEYTRUDA plus LENVIMA is approved in the U.S., the EU, Japan and other countries for the treatment of advanced renal cell carcinoma (RCC) and certain types of advanced endometrial carcinoma. Lenvatinib is marketed as KISPLYX® for advanced RCC in the EU. Results from the LEAP-003 and LEAP-017 trials do not affect the current approved indications for the KEYTRUDA plus LENVIMA combination. Merck and Eisai are studying the KEYTRUDA plus LENVIMA combination through the LEAP (LEnvatinib And Pembrolizumab) clinical program in multiple tumor types, including but not limited to endometrial carcinoma, hepatocellular carcinoma, melanoma, non-small cell lung cancer, RCC, head and neck cancer, colorectal cancer, gastric cancer and esophageal cancer, across more than 10 clinical trials.
About LEAP-003
LEAP-003 is a randomized, placebo-controlled Phase 3 trial (ClinicalTrials.gov, NCT03820986) evaluating KEYTRUDA plus LENVIMA versus KEYTRUDA alone for the first-line treatment of adults with unresectable or metastatic melanoma. The dual primary endpoints are OS and PFS, as assessed by Response Evaluation Criteria in Solid Tumors version 1.1 (RECIST v1.1). Key secondary endpoints include ORR and DOR, both as assessed by RECIST v1.1, and safety. The study enrolled 674 patients who were randomized 1:1 to receive:
KEYTRUDA was administered for up to 35 cycles (approximately two years) or until protocol-specified discontinuation criteria were met. After completing two years of combination therapy, LENVIMA may have been administered as a single agent until protocol-specified discontinuation criteria were met.
About melanoma
Melanoma, the most serious form of skin cancer, is characterized by the uncontrolled growth of melanocytes, pigment producing cells. The rates of melanoma have been rising over the past few decades, with nearly 325,000 new cases diagnosed worldwide in 2020. In the U.S., skin cancer is one of the most common types of cancer diagnosed. Although melanoma accounts for only 1% of skin cancers, it accounts for a large majority of skin cancer deaths. It is estimated there will be nearly 100,000 new cases of melanoma diagnosed and approximately 8,000 deaths resulting from the disease in the U.S. in 2023. The five-year survival rates from 2012-2018 are estimated to be 71% for regional disease and 32% for distant disease.
About LEAP-017
LEAP-017 is a randomized, open-label, Phase 3 trial (ClinicalTrials.gov, NCT04776148) evaluating KEYTRUDA plus LENVIMA versus regorafenib or TAS-102 for patients with unresectable and metastatic colorectal cancer that is pMMR or not MSI-H who have received and progressed on or after, or became intolerant to, prior treatment. Patients must have been previously treated for colorectal cancer and have shown disease progression as defined by RECIST v1.1 on or after, or could not tolerate, standard treatment. The standard treatment must include all of the following agents, if approved and locally available in the country where the participant is randomized:
The primary endpoint is OS and key secondary endpoints include PFS, ORR and DOR, according to RECIST v1.1 per blinded independent central review (BICR). The study enrolled 480 patients randomized 1:1 to receive:
KEYTRUDA was administered for up to 18 cycles (approximately two years), or until protocol-specified discontinuation criteria were met. After completing two years of combination therapy, LENVIMA may have been administered as a single agent until protocol-specified discontinuation criteria were met.
About colorectal cancer
Colorectal cancer can be referred to as colon cancer or rectal cancer, depending on where the cancer starts. Colorectal cancer often begins with growths on the inner lining of the colon or rectum called polyps, which can change into cancer over time. Colorectal cancer is the third most commonly diagnosed cancer and the second most common cause of cancer-related death worldwide. It is estimated there were more than 1,880,000 new cases of colorectal cancer globally in 2020. In the United States, it is estimated there will be approximately 107,000 new cases of colon cancer and approximately 46,000 new cases of rectal cancer, resulting in more than 52,000 deaths from colorectal cancer in 2023. The five-year relative survival rates in the U.S. for metastatic colon cancer and rectal cancer (stage IV) are estimated to be 13% and 17%, respectively.
About KEYTRUDA® (pembrolizumab) injection, 100 mg
KEYTRUDA is an anti-programmed death receptor-1 (PD-1) therapy that works by increasing the ability of the body’s immune system to help detect and fight tumor cells. KEYTRUDA is a humanized monoclonal antibody that blocks the interaction between PD-1 and its ligands, PD-L1 and PD-L2, thereby activating T lymphocytes which may affect both tumor cells and healthy cells.
Merck has the industry’s largest immuno-oncology clinical research program. There are currently more than 1,600 trials studying KEYTRUDA across a wide variety of cancers and treatment settings. The KEYTRUDA clinical program seeks to understand the role of KEYTRUDA across cancers and the factors that may predict a patient's likelihood of benefitting from treatment with KEYTRUDA, including exploring several different biomarkers.
About the Merck and Eisai strategic collaboration
In March 2018, Eisai and Merck, known as MSD outside the United States and Canada, through an affiliate, entered into a strategic collaboration for the worldwide co-development and co-commercialization of LENVIMA. Under the agreement, the companies will jointly develop, manufacture and commercialize LENVIMA, both as monotherapy and in combination with Merck’s anti-PD-1 therapy KEYTRUDA.
In addition to ongoing clinical studies evaluating the KEYTRUDA plus LENVIMA combination across several different tumor types, the companies have jointly initiated new clinical studies through the LEAP (LEnvatinib And Pembrolizumab) clinical program and are evaluating the combination in multiple tumor types across more than 10 clinical trials.
About Merck
At Merck, known as MSD outside of the United States and Canada, we are unified around our purpose: We use the power of leading-edge science to save and improve lives around the world. For more than 130 years, we have brought hope to humanity through the development of important medicines and vaccines. We aspire to be the premier research-intensive biopharmaceutical company in the world – and today, we are at the forefront of research to deliver innovative health solutions that advance the prevention and treatment of diseases in people and animals. We foster a diverse and inclusive global workforce and operate responsibly every day to enable a safe, sustainable and healthy future for all people and communities. For more information, visit www.merck.com and connect with us on Twitter, Facebook, Instagram, YouTube and LinkedIn.
About Eisai
Eisai’s Corporate Concept is “to give first thought to patients and people in the daily living domain, and to increase the benefits that health care provides.” Under this Concept [also known as our human health care (hhc) Concept], we aim to effectively achieve social good in the form of relieving anxiety over health and reducing health disparities. With a global network of R&D facilities, manufacturing sites and marketing subsidiaries, we strive to create and deliver innovative products to target diseases with high unmet medical needs, with a particular focus in our strategic areas of Neurology and Oncology.
In addition, our continued commitment to the elimination of neglected tropical diseases (NTDs), which is a target (3.3) of the United Nations Sustainable Development Goals (SDGs), is demonstrated by our work on various activities together with global partners.
SOURCE: Merck

临床结果加速审批上市批准
2023-02-17
In KEYNOTE-859, KEYTRUDA combined with chemotherapy also showed statistically significant improvements in progression-free survival and objective response rate, the trial’s key secondary endpoints, compared with chemotherapy alone
Merck (NYSE: MRK), known as MSD outside of the United States and Canada, today announced results from the pivotal Phase 3 KEYNOTE-859 trial investigating KEYTRUDA, Merck’s anti-PD-1 therapy, in combination with fluoropyrimidine- and platinum-containing chemotherapy for the first-line treatment of patients with human epidermal growth factor receptor 2 (HER2)-negative locally advanced unresectable or metastatic gastric or gastroesophageal junction (GEJ) adenocarcinoma. After a median follow-up of 31.0 months (range, 15.3-46.3 months), KEYTRUDA in combination with chemotherapy significantly improved overall survival (OS), reducing the risk of death by 22% (HR=0.78 [95% CI, 0.70-0.87]; p<0.0001) compared to chemotherapy alone for these patients, regardless of PD-L1 expression. Median OS was 12.9 months (95% CI, 11.9-14.0) for KEYTRUDA plus chemotherapy versus 11.5 months (95% CI, 10.6-12.1) for chemotherapy alone. These data are being presented today during a European Society for Medical Oncology (ESMO) Virtual Plenary and are being submitted to regulatory authorities worldwide.
“Most gastric cancers go undetected until an advanced stage, at which point the five-year survival rate is just 6%, one of the lowest across cancer types,” said Dr. Sun Young Rha, director and professor of medical oncology in the Department of Internal Medicine, Songdang Institute for Cancer Research, Yonsei University College of Medicine, Yonsei University Health System, Seoul, Korea. “The positive overall survival results observed in this study are very encouraging and demonstrate the potential of KEYTRUDA in combination with chemotherapy to extend the lives of patients with HER2-negative locally advanced unresectable or metastatic gastric or gastroesophageal junction adenocarcinoma.”
Results from the KEYNOTE-859 trial were generally consistent across pre-specified subgroups. In addition to an improvement in OS, results showed KEYTRUDA plus chemotherapy significantly improved progression-free survival (PFS) and objective response rate (ORR), which were both secondary endpoints of the trial, versus chemotherapy alone. Specifically, KEYTRUDA plus chemotherapy reduced the risk of disease progression or death by 24% (HR=0.76 [95% CI, 0.67-0.85]; p<0.0001) with a median PFS of 6.9 months (95% CI, 6.3-7.2) for KEYTRUDA plus chemotherapy compared to 5.6 months (95% CI, 5.5-5.7) for chemotherapy alone. The ORR was 51.3% (95% CI, 47.7-54.8), with a complete response (CR) rate of 9.5% and a partial response (PR) rate of 41.8%, for patients who received KEYTRUDA plus chemotherapy and 42.0% (95% CI, 38.5-45.5), with a CR rate of 6.2% and a PR rate of 35.7%, for those who received chemotherapy alone (p=0.00009). Median duration of response (DOR), another secondary endpoint of the trial, was 8.0 months (range, 1.2+-41.5+) for KEYTRUDA plus chemotherapy versus 5.7 months (range, 1.3+-34.7+) for chemotherapy alone.
“Eighty percent of patients with locally advanced unresectable or metastatic gastric or gastroesophageal junction adenocarcinoma have HER2-negative disease. Based on these new data, we hope to expand the use of KEYTRUDA and provide an option that, in combination with chemotherapy, may help more of these patients live longer,” said Dr. Scot Ebbinghaus, vice president, clinical research, Merck Research Laboratories. “These results also reinforce our commitment to research in gastrointestinal cancers, where we continue to evaluate a number of KEYTRUDA-based combinations, including in earlier stages of disease.”
Grade 3-5 treatment-related adverse events (TRAEs) occurred in 59.4% of patients receiving KEYTRUDA plus chemotherapy and 51.1% of patients receiving chemotherapy alone; TRAEs led to death in eight (1.0%) versus 16 (2.0%) patients, respectively. No new safety signals were identified. Grade 3-5 immune-mediated adverse events (AEs) occurred in 7.9% of patients receiving KEYTRUDA plus chemotherapy and 1.7% of patients receiving chemotherapy alone; immune-mediated AEs led to death in one patient (0.1%) in each of the treatment arms.
KEYTRUDA is approved in the U.S. in combination with trastuzumab, fluoropyrimidine- and platinum-containing chemotherapy, for the first-line treatment of patients with locally advanced unresectable or metastatic HER2-positive gastric or GEJ adenocarcinoma. This indication is approved under accelerated approval based on tumor response rate and durability of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in the confirmatory trials.
Merck has an extensive clinical development program evaluating KEYTRUDA in gastrointestinal cancers, which includes KEYNOTE-811 in first-line advanced HER2-positive gastric cancer, KEYNOTE-585 in early-stage gastric cancer and further exploration in advanced/metastatic gastric cancer in combination with a tyrosine kinase inhibitor in LEAP-015. Merck is continuing to study KEYTRUDA for multiple uses in hepatobiliary, esophageal, pancreatic, colorectal and biliary tract cancers.
About KEYNOTE-859
KEYNOTE-859 is a randomized, double-blind Phase 3 trial (ClinicalTrials.gov, NCT03675737) evaluating KEYTRUDA in combination with chemotherapy compared to placebo in combination with chemotherapy for the first-line treatment of patients with HER2-negative locally advanced unresectable or metastatic gastric or GEJ adenocarcinoma. The primary endpoint is OS, and secondary endpoints include PFS, ORR, DOR and safety. The trial enrolled 1,579 patients who were randomized to receive KEYTRUDA (200 mg every three weeks for up to approximately two years) in combination with fluoropyrimidine- and platinum-containing chemotherapy (n=785), or placebo in combination with chemotherapy (n=787). All patients received investigator’s choice of chemotherapy (5-FU plus cisplatin [FP] or capecitabine plus oxaliplatin [CAPOX]).
About gastric cancer
Gastric (stomach) cancer tends to develop slowly over many years and rarely causes early symptoms, resulting in most cases going undetected until an advanced stage. More than 70% of patients with gastric cancer develop advanced-stage disease. Most gastric cancers are adenocarcinomas (about 90-95%), which develop from cells in the innermost lining of the stomach (known as the mucosa). The majority of gastric cancers are HER2-negative, affecting approximately four out of every five patients. Gastric cancer is the fifth most diagnosed cancer and the fourth leading cause of cancer death worldwide, with approximately 1.1 million patients diagnosed and 768,000 deaths from the disease globally in 2020. In the U.S., it is estimated there will be approximately 26,500 patients diagnosed with gastric cancer and 11,000 deaths from the disease in 2023. The five-year survival rate for patients diagnosed with gastric cancer at an advanced stage is only 6%.
About KEYTRUDA® (pembrolizumab) injection, 100 mg
KEYTRUDA is an anti-programmed death receptor-1 (PD-1) therapy that works by increasing the ability of the body’s immune system to help detect and fight tumor cells. KEYTRUDA is a humanized monoclonal antibody that blocks the interaction between PD-1 and its ligands, PD-L1 and PD-L2, thereby activating T lymphocytes which may affect both tumor cells and healthy cells.
Merck has the industry’s largest immuno-oncology clinical research program. There are currently more than 1,600 trials studying KEYTRUDA across a wide variety of cancers and treatment settings. The KEYTRUDA clinical program seeks to understand the role of KEYTRUDA across cancers and the factors that may predict a patient's likelihood of benefitting from treatment with KEYTRUDA, including exploring several different biomarkers.
Selected KEYTRUDA® (pembrolizumab) Indications in the U.S.
Melanoma
KEYTRUDA is indicated for the treatment of patients with unresectable or metastatic melanoma.
KEYTRUDA is indicated for the adjuvant treatment of adult and pediatric (12 years and older) patients with stage IIB, IIC, or III melanoma following complete resection.
Non-Small Cell Lung Cancer
KEYTRUDA, in combination with pemetrexed and platinum chemotherapy, is indicated for the first-line treatment of patients with metastatic nonsquamous non-small cell lung cancer (NSCLC), with no EGFR or ALK genomic tumor aberrations.
KEYTRUDA, in combination with carboplatin and either paclitaxel or paclitaxel protein-bound, is indicated for the first-line treatment of patients with metastatic squamous NSCLC.
KEYTRUDA, as a single agent, is indicated for the first-line treatment of patients with NSCLC expressing PD-L1 [tumor proportion score (TPS) ≥1%] as determined by an FDA-approved test, with no EGFR or ALK genomic tumor aberrations, and is:
KEYTRUDA, as a single agent, is indicated for the first-line treatment of patients with NSCLC expressing PD-L1 [tumor proportion score (TPS) ≥1%] as determined by an FDA-approved test, with no EGFR or ALK genomic tumor aberrations, and is:
stage III where patients are not candidates for surgical resection or definitive chemoradiation, or metastatic.
KEYTRUDA, as a single agent, is indicated for the treatment of patients with metastatic NSCLC whose tumors express PD-L1 (TPS ≥1%) as determined by an FDA-approved test, with disease progression on or after platinum-containing chemotherapy. Patients with EGFR or ALK genomic tumor aberrations should have disease progression on FDA-approved therapy for these aberrations prior to receiving KEYTRUDA.
KEYTRUDA, as a single agent, is indicated as adjuvant treatment following resection and platinum-based chemotherapy for adult patients with Stage IB (T2a ≥4 cm), II, or IIIA NSCLC.
Head and Neck Squamous Cell Cancer
KEYTRUDA, in combination with platinum and fluorouracil (FU), is indicated for the first-line treatment of patients with metastatic or with unresectable, recurrent head and neck squamous cell carcinoma (HNSCC).
KEYTRUDA, as a single agent, is indicated for the first-line treatment of patients with metastatic or with unresectable, recurrent HNSCC whose tumors express PD-L1 [Combined Positive Score (CPS) ≥1] as determined by an FDA-approved test.
KEYTRUDA, as a single agent, is indicated for the treatment of patients with recurrent or metastatic HNSCC with disease progression on or after platinum-containing chemotherapy.
Classical Hodgkin Lymphoma
KEYTRUDA is indicated for the treatment of adult patients with relapsed or refractory classical Hodgkin lymphoma (cHL).
KEYTRUDA is indicated for the treatment of pediatric patients with refractory cHL, or cHL that has relapsed after 2 or more lines of therapy.
Primary Mediastinal Large B-Cell Lymphoma
KEYTRUDA is indicated for the treatment of adult and pediatric patients with refractory primary mediastinal large B-cell lymphoma (PMBCL), or who have relapsed after 2 or more prior lines of therapy.
KEYTRUDA is not recommended for treatment of patients with PMBCL who require urgent cytoreductive therapy.
Urothelial Carcinoma
KEYTRUDA is indicated for the treatment of patients with locally advanced or metastatic urothelial carcinoma (mUC):
Non-muscle Invasive Bladder Cancer
KEYTRUDA is indicated for the treatment of patients with Bacillus Calmette-Guerin-unresponsive, high-risk, non-muscle invasive bladder cancer (NMIBC) with carcinoma in situ with or without papillary tumors who are ineligible for or have elected not to undergo cystectomy.
Microsatellite Instability-High or Mismatch Repair Deficient Cancer
KEYTRUDA is indicated for the treatment of adult and pediatric patients with unresectable or metastatic microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR) solid tumors, as determined by an FDA-approved test, that have progressed following prior treatment and who have no satisfactory alternative treatment options.
This indication is approved under accelerated approval based on tumor response rate and durability of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in the confirmatory trials. The safety and effectiveness of KEYTRUDA in pediatric patients with MSI-H central nervous system cancers have not been established.
Microsatellite Instability-High or Mismatch Repair Deficient Colorectal Cancer
KEYTRUDA is indicated for the treatment of patients with unresectable or metastatic MSI-H or dMMR colorectal cancer (CRC) as determined by an FDA-approved test.
Gastric Cancer
KEYTRUDA, in combination with trastuzumab, fluoropyrimidine- and platinum-containing chemotherapy, is indicated for the first-line treatment of patients with locally advanced unresectable or metastatic HER2-positive gastric or gastroesophageal junction (GEJ) adenocarcinoma.
This indication is approved under accelerated approval based on tumor response rate and durability of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in the confirmatory trials.
Esophageal Cancer
KEYTRUDA is indicated for the treatment of patients with locally advanced or metastatic esophageal or gastroesophageal junction (GEJ) (tumors with epicenter 1 to 5 centimeters above the GEJ) carcinoma that is not amenable to surgical resection or definitive chemoradiation either:
Cervical Cancer
KEYTRUDA, in combination with chemotherapy, with or without bevacizumab, is indicated for the treatment of patients with persistent, recurrent, or metastatic cervical cancer whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-approved test.
KEYTRUDA, as a single agent, is indicated for the treatment of patients with recurrent or metastatic cervical cancer with disease progression on or after chemotherapy whose tumors express PD-L1 (CPS ≥1) as determined by an FDA-approved test.
Hepatocellular Carcinoma
KEYTRUDA is indicated for the treatment of patients with hepatocellular carcinoma (HCC) who have been previously treated with sorafenib. This indication is approved under accelerated approval based on tumor response rate and durability of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in the confirmatory trials.
Merkel Cell Carcinoma
KEYTRUDA is indicated for the treatment of adult and pediatric patients with recurrent locally advanced or metastatic Merkel cell carcinoma (MCC). This indication is approved under accelerated approval based on tumor response rate and durability of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in the confirmatory trials.
Renal Cell Carcinoma
KEYTRUDA, in combination with axitinib, is indicated for the first-line treatment of adult patients with advanced renal cell carcinoma (RCC).
KEYTRUDA is indicated for the adjuvant treatment of patients with RCC at intermediate-high or high risk of recurrence following nephrectomy, or following nephrectomy and resection of metastatic lesions.
Endometrial Carcinoma
KEYTRUDA, as a single agent, is indicated for the treatment of patients with advanced endometrial carcinoma that is MSI-H or dMMR, as determined by an FDA-approved test, who have disease progression following prior systemic therapy in any setting and are not candidates for curative surgery or radiation.
Tumor Mutational Burden-High Cancer
KEYTRUDA is indicated for the treatment of adult and pediatric patients with unresectable or metastatic tumor mutational burden-high (TMB-H) [≥10 mutations/megabase] solid tumors, as determined by an FDA-approved test, that have progressed following prior treatment and who have no satisfactory alternative treatment options. This indication is approved under accelerated approval based on tumor response rate and durability of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in the confirmatory trials. The safety and effectiveness of KEYTRUDA in pediatric patients with TMB-H central nervous system cancers have not been established.
Cutaneous Squamous Cell Carcinoma
KEYTRUDA is indicated for the treatment of patients with recurrent or metastatic cutaneous squamous cell carcinoma (cSCC) or locally advanced cSCC that is not curable by surgery or radiation.
Triple-Negative Breast Cancer
KEYTRUDA is indicated for the treatment of patients with high-risk early-stage triple-negative breast cancer (TNBC) in combination with chemotherapy as neoadjuvant treatment, and then continued as a single agent as adjuvant treatment after surgery.
KEYTRUDA, in combination with chemotherapy, is indicated for the treatment of patients with locally recurrent unresectable or metastatic TNBC whose tumors express PD-L1 (CPS ≥10) as determined by an FDA-approved test.
Merck’s focus on cancer
Our goal is to translate breakthrough science into innovative oncology medicines to help people with cancer worldwide. At Merck, the potential to bring new hope to people with cancer drives our purpose and supporting accessibility to our cancer medicines is our commitment. As part of our focus on cancer, Merck is committed to exploring the potential of immuno-oncology with one of the largest development programs in the industry across more than 30 tumor types. We also continue to strengthen our portfolio through strategic acquisitions and are prioritizing the development of several promising oncology candidates with the potential to improve the treatment of advanced cancers. For more information about our oncology clinical trials, visit www.merck.com/clinicaltrials.
About Merck
At Merck, known as MSD outside of the United States and Canada, we are unified around our purpose: We use the power of leading-edge science to save and improve lives around the world. For more than 130 years, we have brought hope to humanity through the development of important medicines and vaccines. We aspire to be the premier research-intensive biopharmaceutical company in the world – and today, we are at the forefront of research to deliver innovative health solutions that advance the prevention and treatment of diseases in people and animals. We foster a diverse and inclusive global workforce and operate responsibly every day to enable a safe, sustainable and healthy future for all people and communities. For more information, visit www.merck.com and connect with us on Twitter, Facebook, Instagram, YouTube and LinkedIn.
Merck’s focus on cancer
Our goal is to translate breakthrough science into innovative oncology medicines to help people with cancer worldwide. At Merck, the potential to bring new hope to people with cancer drives our purpose and supporting accessibility to our cancer medicines is our commitment. As part of our focus on cancer, Merck is committed to exploring the potential of immuno-oncology with one of the largest development programs in the industry across more than 30 tumor types. We also continue to strengthen our portfolio through strategic acquisitions and are prioritizing the development of several promising oncology candidates with the potential to improve the treatment of advanced cancers. For more information about our oncology clinical trials, visit www.merck.com/clinicaltrials.
About Merck
At Merck, known as MSD outside of the United States and Canada, we are unified around our purpose: We use the power of leading-edge science to save and improve lives around the world. For more than 130 years, we have brought hope to humanity through the development of important medicines and vaccines. We aspire to be the premier research-intensive biopharmaceutical company in the world – and today, we are at the forefront of research to deliver innovative health solutions that advance the prevention and treatment of diseases in people and animals. We foster a diverse and inclusive global workforce and operate responsibly every day to enable a safe, sustainable and healthy future for all people and communities. For more information, visit www.merck.com and connect with us on Twitter, Facebook, Instagram, YouTube and LinkedIn.
SOURCE: Merck
内容来源于网络,如有侵权,请联系删除。

临床结果加速审批上市批准免疫疗法
2022-12-13
·生物谷
使用无创或微创的方式实现对各种癌症精准快速的诊断是无数科研工作者、医生和病人的期望。但之前,我们并没有很好的技术来实现这一点。由于癌症起病隐匿,发展速度快,所以许多病人往往在癌症的中晚期才被确诊,从而错过了最佳治疗期,这往往给患者的家庭带来了沉重的经济负担和巨大的心理压力。
同时,每年又有大量新的癌症患者被确诊,这也给社会带来不小的压力,而与此相对的是:大多数的癌症在发展的早期被发现后都能被很好的治疗,倘若患者能够在癌症的早期就被筛查到身体异常,这将极大地提高患者的生存率,降低家庭和社会的负担。但遗憾的是,虽然对一些常见的癌症,如乳腺癌、肺癌、肝癌等有对应的检测方式和金标准,但目前主流的检测方法很难做到对各种类型的早期癌症做到精确快速的筛查。
近日,PNAS的一篇文章:Noninvasive detection of any-stage cancer using free glycosaminoglycans 报告了一种新的,以前未经测试的方法,它可以很容易地同时发现多种类型的早期癌症,包括之前很难检测到的癌症类型。其实早在2021年,Science和Nature的子刊就报道过一种基于基因组学驱动的液体活检技术(MCED),用于检测早期癌症的发生,简单来讲就是使用患者的血液样本分析其体液中的肿瘤成分,包括循环的肿瘤细胞和无细胞结构的肿瘤DNA,再通过检测基因表达的甲基化DNA基因组测序手段,将血液检测结合机器学习等技术,来寻找体内是否存在癌症的异常甲基化变化以及预测癌症在体内所处的位置。检测结果会在样本到达实验室后的10个工作日内出来。然而,上述的这种方法并不是完美无缺,这些先进的液体活检所需的复杂检测增加了测量的成本,对于普通人来说,筛查的价格可能令人望而却步。更重要的是,只有少数液体活检验证了其在MCED的外部代表性人群中的I期敏感性。为了优化体液检测早期癌症的方法,填补当前液体活检平台的信息空白,这项新的方法研究的不再是基因组学和蛋白质组学,而是将癌症代谢作为一种可识别的癌症标志。在作者之前的研究中通过肿瘤代谢的系统生物学泛癌性分析发现糖胺聚糖(GAG)的生物合成具有癌症特异性重编程的特点,作者在肾细胞癌(RCC)中发现血浆和尿液游离的GAG组(GAGomes)显著改变,这一发现促使作者研究不同癌症类型的游离GAGomes,并开发一种标准化的高通量超高效液相色谱结合三重四极杆质谱(UHPLC-MS/MS)方法来测量游离的GAGomes。
图一:GAGome的开发研究概述和总结分析3
首先,研究人员进行了一项病例对照研究,如图一所示,包括979名受试者,这其中有代表了14种癌症类型的553名癌症患者,以及426名健康受试者,他们具有相似的人口统计学特征,作者在实验室使用标准化的UHPLLC-MS / MS试剂盒测量了96%受试者血浆中的游离GAGome,其包括硫酸软骨素(CS)、硫酸肝素(HS)和透明质酸(HA)的17个二糖亚单位的浓度和GAGome的特征,如CS和HS电荷,这导致每种液体共有39个特征。在这39个特征中,作者发现6个血浆和17个尿液的GAGome特征是可被检测到的。
图二:血浆、尿液和不同阶段/分级组的综合评分的特异性为 95% 的敏感性3
接下来,作者将每种癌症类型中可检测到的GAGome特征与健康受试者的基线生理水平进行比较。敏感性分析表明,这个比较模型的建立是恰当可行的。作者认为对于一个给定的GAGome特征,其与正常生理水平的差异具有生理学意义,在确认所有癌症类型都具有与基线生理水平不同的通用GAGome特征后,他们又探索了使用GAGomes来区分任意的癌症病人和健康受试者,检测到最多的癌症类型是血浆评分最高的NHL,CRC和慢性淋巴细胞白血病(CLL),尿液联合评分最高的为RCC和非小细胞肺癌(NSCLC),如图二所示。总之,这些发现表明,游离GAGomes与早期和晚期癌症的生理水平显着不同,可用于MCED。鉴于不同癌症类型中存在独特的游离GAGomes,作者还探讨了这些模式是否可用于识别癌症类型,结果发现使用游离GAGomes预测推定的癌症位置(PCL)的预测准确率为89.2%,同时,生存分析表明,未检测到游离GAGome MCED评分的患者具有更好的预后和更少的侵袭性癌症表型,并且其与肿瘤分期和分级没有明显的关系。 进一步的,作者研究了任意癌症类型的游离GAGomes的改变是否与体内癌症的发作和进展在机械上相关。
图三:小鼠癌症进展期间血浆和游离GAGomes的变化3
对BALB / c小鼠进行肾腺癌肿瘤细胞原位诱导,第7天切除含有肿瘤的肾脏,第20天处死小鼠,在这个过程中,对小鼠的尿液和血浆游离的GAGome进行了纵向测量,主成分分析显示,血浆游离GAGomes和尿液中游离GAGomes的较小程度的改变与从基线(第0天)到局部生长(第6天)到术后切除(第8天)到转移(第20天)的进展一致,这与人类癌症样本中观察到的模式也是相符的。综上所述,此项研究证明了游离GAGomes的代谢性质及其检测不良cfDNA脱落物癌症类型的能力,能够极大地补充基于基因组生物标志物的液体活检,为多模态MCED方法铺平了道路。这种方法可能会进一步增加对I期癌症的敏感性,甚至达到仅通过早期发现就可以大幅抑制癌症死亡率的水平。事实上,通过血液检测实现癌症早筛的概念并不是近年才出现的,就在不久前,美国公司Theranos创始人伊丽莎白·霍姆斯因“滴血验癌”的骗局被法院以欺诈罪判处135个月监禁,曾经估值高达90亿美元的血液检测公司轰然倒塌,这场惊天骗局也让我们从一个侧面了解到市场对于实现癌症快速准确的早筛有多大的渴望。虽然在癌症早筛这条路上,血液检测这个方法“栽了个大跟头”,但我们仍有理由相信,快速便捷准确的早筛手段一定会在不远的以后来到我们的身边,为人民的身体安全保驾护航。
参考文献:1. Heitzer, E., Haque, I.S., Roberts, C.E.S. et al. Current and future perspectives of liquid biopsies in genomics-driven oncology. Nat Rev Genet 20, 71–88 (2019).2. D. Crosby et al., Early detection of cancer. Science 375, eaay9040 (2022)3. Bratulic S, Limeta A, Dabestani S, et al. Noninvasive detection of any-stage cancer using free glycosaminoglycans. Proc Natl Acad Sci U S A. 2022;119(50):e2115328119.撰文 | 之予编辑 | cici精彩推荐:1、为什么冬季新冠猖獗?科学家揭露首个生物学机制!2、抗老只能靠基因?Nature Aging新研究:年轻做的事都“刻”在基因里,影响晚年基因活动,甚至缩短寿命!3、不粘锅有划痕致癌不能用?最新研究揭示:微小划痕即可释放百万级有毒微塑料,或可造成永久伤害!4、喝茶养生是真的!加奶也行!最新研究:每天1-6杯红茶,心血管病风险降低42%、抗癌长寿不痴呆!5、闻闻肉味就能瘦?Nature子刊:「气味疗法」可促进脂质代谢轻松减重,或是减肥困难户福音!
内容来源于网络,如有侵权,请联系删除。
临床研究
分析
对领域进行一次全面的分析。
登录
或
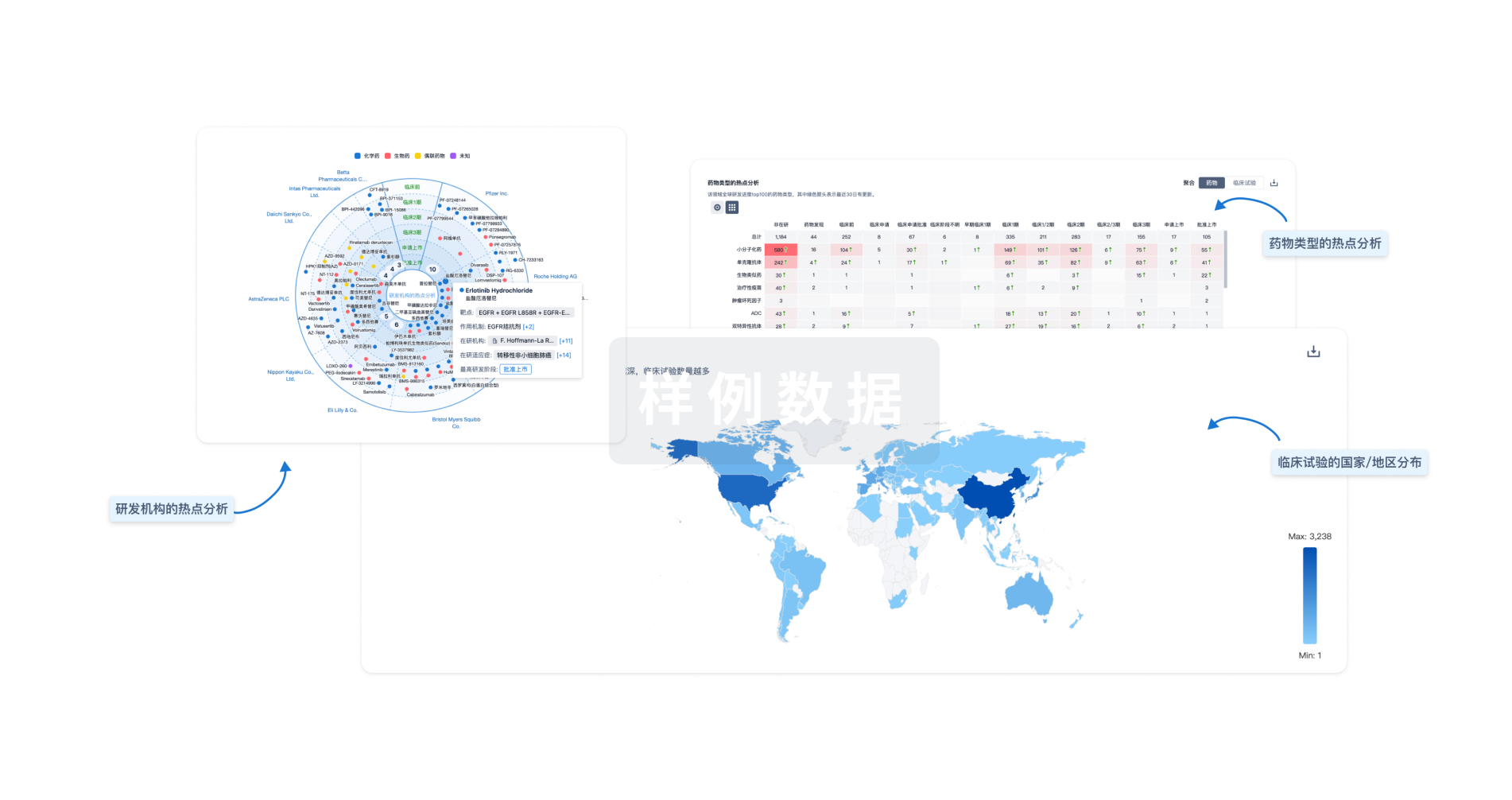
生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用



