预约演示
更新于:2026-03-08

AskBio Inc.
更新于:2026-03-08
概览
标签
心血管疾病
其他疾病
神经系统疾病
腺相关病毒基因治疗
疾病领域得分
一眼洞穿机构专注的疾病领域
暂无数据
技术平台
公司药物应用最多的技术
暂无数据
靶点
公司最常开发的靶点
暂无数据
| 排名前五的药物类型 | 数量 |
|---|---|
| 腺相关病毒基因治疗 | 8 |
关联
8
项与 AskBio Inc. 相关的药物作用机制 GDNF刺激剂 [+2] |
在研机构 |
在研适应症 |
非在研适应症 |
最高研发阶段临床2期 |
首次获批国家/地区- |
首次获批日期- |
靶点 |
作用机制 DUSP1 inhibitors [+1] |
在研机构 |
非在研适应症- |
最高研发阶段临床2期 |
首次获批国家/地区- |
首次获批日期- |
作用机制 GDNF modulators |
原研机构 |
在研适应症 |
非在研适应症- |
最高研发阶段临床2期 |
首次获批国家/地区- |
首次获批日期- |
13
项与 AskBio Inc. 相关的临床试验NCT07215403
Open-Label, Multi-Stage Study to Optimize the Intraputaminal Administration of AB-1005 Using a Prescriptive Infusion Algorithm (PIA)
This study is being done to test a new way of delivering AB-1005 into the brain. The goal is to make the procedure easier and quicker to perform, while providing similar amounts of drug to the part of the brain that needs treatment.
Technical (Stage 1) To test if the new delivery method (PIA-based infusion) can consistently deliver AB-1005 to the putamen using brain imaging by MRI.
Technical (Stage 2) To confirm the new delivery method works without brain imaging by MRI, using standard operating room tools including brain imaging by CT.
Safety (Stage 1 and 2) To assess the safety and tolerability of the new delivery method for AB-1005 up to 6 months after surgery
Technical (Stage 1) To test if the new delivery method (PIA-based infusion) can consistently deliver AB-1005 to the putamen using brain imaging by MRI.
Technical (Stage 2) To confirm the new delivery method works without brain imaging by MRI, using standard operating room tools including brain imaging by CT.
Safety (Stage 1 and 2) To assess the safety and tolerability of the new delivery method for AB-1005 up to 6 months after surgery
开始日期2026-05-01 |
申办/合作机构  AskBio Inc. AskBio Inc. [+1] |
NCT07282847
A Single-Arm, Open-Label, Dose-Escalation Study to Evaluate the Safety, Tolerability and Efficacy of a Single Intravenous Infusion of AB-1009 in Adult Participants With Late Onset Pompe Disease (LOPD)
This is a single-arm, open-label, dose-escalation study to evaluate the safety, tolerability and efficacy of a single intravenous infusion of AB-1009 in adult participants with late-onset Pompe disease (LOPD).
开始日期2026-02-10 |
申办/合作机构  AskBio Inc. AskBio Inc. [+1] |
NCT07081841
Long-Term Safety and Efficacy Follow-up of AB-1005 Gene Transfer Study Participants With Parkinson's Disease or Multiple System Atrophy
This is an observational, long term, follow-up study for participants with Parkinson's disease (PD) or Multiple System Atrophy-Parkinsonian subtype (MSA-P) who participated in prior interventional studies with AB-1005. It is intended to better understand the long-term safety of AB-1005, how well tolerated it is and how long lasting the effects are.
开始日期2025-09-04 |
申办/合作机构 |
100 项与 AskBio Inc. 相关的临床结果
登录后查看更多信息
0 项与 AskBio Inc. 相关的专利(医药)
登录后查看更多信息
20
项与 AskBio Inc. 相关的文献(医药)2023-10-01·Cellular oncology (Dordrecht)
Non-canonical role for the ataxia-telangiectasia-Rad3 pathway in STAT3 activation in human multiple myeloma cells
Article
作者: Nkwocha, Jewel ; Li, Lin ; Grant, Steven ; Hu, Xiaoyan ; Kmieciak, Maciej ; Sharma, Kanika ; Mann, Hashim ; Zhou, Liang
Abstract:
Purpose:
The goal of this study was to characterize the relationship between ATR and STAT3 interactions in human multiple myeloma (MM) cells.
Methods:
Various MM cell lines, including IL-6-dependent cells were exposed to ATR inhibitors and effects on STAT3 Tyr705 and Ser727 were monitored by WB analysis and ImageStream analysis. Parallel studies examined induction of cell death, STAT3 DNA binding activity, and expression of STAT3 downstream targets (BCL-XL, MCL-1, c-MYC). Validation was obtained in ATR shRNA knock-down cells, and in cells ectopically expressing BCL-XL, MCL-1, or c-MYC. Analogous studies were performed in primary MM cells and in a MM xenograft model.
Results:
Multiple pharmacologic ATR inhibitors inhibited STAT3 Tyr705 (but not Ser727) phosphorylation at low uM concentrations and down-regulated BCL-XL, MCL-1, c-MYC in association with cell death induction. Compatible results were observed in ATR shRNA knock-down cells. Cell death induced by ATR inhibitors was significantly attenuated in cells ectopically expressing constitutively active STAT3, BCL-XL, MCL-1, or c-MYC. Concordant results were observed in primary human MM cells and in an in vivo MM xenograft model.
Conclusions:
Collectively, these findings argue for a non-canonical role for the ATR kinase in STAT3 activation in MM cells, and suggest that STAT3 inactivation contributes to the lethal actions of ATR inhibitors in MM.
2023-05-09·Cancer research communications
Phase I Trial of Autologous RNA-electroporated cMET-directed CAR T Cells Administered Intravenously in Patients with Melanoma and Breast Carcinoma
Article
作者: Julia Tchou ; Payal D. Shah ; Amy Marshall ; Joanne Shea ; Andrea Brennan ; Lester Lledo ; E. John Wherry ; Simon F. Lacey ; Robert H. Vonderheide ; Xiaowei Xu ; Gabriela Plesa ; Tina Matlawski ; Ravi K. Amaravadi ; Joan Gilmore ; Robert Orlowski ; Lynn M. Schuchter ; Carl H. June ; Alexander C. Huang ; Irina Kulikovskaya ; Amanda Cervini ; Tara C. Mitchell ; Vanessa Gonzalez ; Gerald P. Linette ; Paul Zhang ; Karen Dengel
Purpose::
Treatments are limited for metastatic melanoma and metastatic triple-negative breast cancer (mTNBC). This pilot phase I trial (NCT03060356) examined the safety and feasibility of intravenous RNA-electroporated chimeric antigen receptor (CAR) T cells targeting the cell-surface antigen cMET.
Experimental Design::
Metastatic melanoma or mTNBC subjects had at least 30% tumor expression of cMET, measurable disease and progression on prior therapy. Patients received up to six infusions (1 × 10e8 T cells/dose) of CAR T cells without lymphodepleting chemotherapy. Forty-eight percent of prescreened subjects met the cMET expression threshold. Seven (3 metastatic melanoma, 4 mTNBC) were treated.
Results::
Mean age was 50 years (35–64); median Eastern Cooperative Oncology Group 0 (0–1); median prior lines of chemotherapy/immunotherapy were 4/0 for TNBC and 1/3 for melanoma subjects. Six patients experienced grade 1 or 2 toxicity. Toxicities in at least 1 patient included anemia, fatigue, and malaise. One subject had grade 1 cytokine release syndrome. No grade 3 or higher toxicity, neurotoxicity, or treatment discontinuation occurred. Best response was stable disease in 4 and disease progression in 3 subjects. mRNA signals corresponding to CAR T cells were detected by RT-PCR in all patients’ blood including in 3 subjects on day +1 (no infusion administered on this day). Five subjects underwent postinfusion biopsy with no CAR T-cell signals seen in tumor. Three subjects had paired tumor tissue; IHC showed increases in CD8 and CD3 and decreases in pS6 and Ki67.
Conclusions::
Intravenous administration of RNA-electroporated cMET-directed CAR T cells is safe and feasible.
Significance::
Data evaluating CAR T therapy in patients with solid tumors are limited. This pilot clinical trial demonstrates that intravenous cMET-directed CAR T-cell therapy is safe and feasible in patients with metastatic melanoma and metastatic breast cancer, supporting the continued evaluation of cellular therapy for patients with these malignancies.
2022-12-01·Molecular therapy. Nucleic acids
Non-uniform dystrophin re-expression after CRISPR-mediated exon excision in the dystrophin/utrophin double-knockout mouse model of DMD
Article
作者: Stenler, Sofia ; Hanson, Britt ; Ahlskog, Nina ; Svrzikapa, Nenad ; Weinberg, Marc S ; Coenen-Stass, Anna M L ; Roberts, Thomas C ; Wood, Matthew J A ; Chwalenia, Katarzyna
Duchenne muscular dystrophy (DMD) is the most prevalent inherited myopathy affecting children, caused by genetic loss of the gene encoding the dystrophin protein. Here we have investigated the use of the Staphylococcus aureus CRISPR-Cas9 system and a double-cut strategy, delivered using a pair of adeno-associated virus serotype 9 (AAV9) vectors, for dystrophin restoration in the severely affected dystrophin/utrophin double-knockout (dKO) mouse. Single guide RNAs were designed to excise Dmd exon 23, with flanking intronic regions repaired by non-homologous end joining. Exon 23 deletion was confirmed at the DNA level by PCR and Sanger sequencing, and at the RNA level by RT-qPCR. Restoration of dystrophin protein expression was demonstrated by western blot and immunofluorescence staining in mice treated via either intraperitoneal or intravenous routes of delivery. Dystrophin restoration was most effective in the diaphragm, where a maximum of 5.7% of wild-type dystrophin expression was observed. CRISPR treatment was insufficient to extend lifespan in the dKO mouse, and dystrophin was expressed in a within-fiber patchy manner in skeletal muscle tissues. Further analysis revealed a plethora of non-productive DNA repair events, including AAV genome integration at the CRISPR cut sites. This study highlights potential challenges for the successful development of CRISPR therapies in the context of DMD.
302
项与 AskBio Inc. 相关的新闻(医药)2026-03-06
2026年3月初,一则消息在罕见病群体中引发波澜:制药巨头拜耳在最新的财报中,正式为旗下一款名为ACTUS-101的庞贝病基因疗法画上了句号。这个决定并非一时兴起,而是企业在技术浪潮更迭中,一次冷静的“汰旧换新”。
一款已经进入临床阶段的在研药物,为何在投入巨资收购后仍被放弃?这背后,是拜耳对下一代疗法的全力押注,也折射出罕见病基因治疗领域研发策略的深刻变革。
01
从“并行”到“二选一”:一次基于技术的战略聚焦
故事要追溯到2020年,拜耳豪掷20亿美元收购了基因治疗公司AskBio,一并拿下了两款针对晚发型庞贝病的在研AAV基因疗法。其中,ACTUS-101作为“一代”产品,彼时已经率先进入临床。
然而,技术迭代的速度远超预期。到2026年1月,拜耳突然叫停了ACTUS-101的1期临床试验,不再招募新患者。此时,该试验仅完成了7名患者的入组。最终,在2025年第四季度的财报中,拜耳正式确认了ACTUS-101的“退役”。
摆在拜耳面前的本是两条腿走路的选项,为何最终选择壮士断腕?答案藏在另一款名为 AB-1009 的“二代”候选药身上。
02
直击病灶:二代疗法的“降维打击”
与被淘汰的ACTUS-101相比,AB-1009在设计理念和监管进展上都展现了明显的代际优势。这款新药已获得美国FDA授予的快速通道和孤儿药资格,并于2026年初在美国启动了1/2期临床试验。
那么,两代疗法之间最核心的差异究竟是什么?
我们可以用一个简单的比喻来理解:
一代疗法(ACTUS-101) :它像一个“外围补给站”,以肝脏为靶向器官。希望通过改造肝脏细胞,让它持续生产患者体内缺乏的GAA酶,再输送到全身。
二代疗法(AB-1009) :它更像一支“精准空降兵”,直接瞄准了问题的核心——肌肉组织。庞贝病正是由于GAA酶缺乏,导致糖原在肌肉细胞中大量堆积,造成进行性肌肉损伤。
AB-1009之所以更受青睐,在于其采用了 AAV8载体,能够更高效地将经过优化设计的GAA基因递送到肌肉。同时,它搭载了 组织特异性启动子 和经过 密码子优化的GAA基因,这就像给“空降兵”配备了精确的地图和更强的装备,旨在极大提升酶在肌肉组织中的表达效率,从根源上修复病灶。
正如该项目首席研究员、加州大学尔湾分校的Tahseen Mozaffar博士所言,AB-1009的目标是让患者自身成为“生产工厂”,减少对外源性、需定期输注的酶替代疗法的依赖。
值得一提的是,这款寄托厚望的新药背后,还有中国生物科技公司的身影。 信念医药 为其提供了专有的AAV衣壳技术及载体生产服务,成为其全球研发链条中的重要一环。
03
前路漫漫:庞贝病药物研发的“荆棘之路”
庞贝病,这个又称糖原贮积病Ⅱ型的罕见病,患病率约为4万至30万分之一。患者因GAA基因突变,面临进行性肌肉损伤,严重者可累及呼吸和心脏,危及生命。目前,全球范围内尚无能够根治此病的疗法,标准治疗方案酶替代疗法也仅能缓解病情。
正因如此,庞贝病成为众多药企渴望攻克的堡垒,但这条路上布满荆棘。拜耳的调整并非个例:
Denali Therapeutics 的一款酶替代疗法,在2025年底因临床前研究中的免疫反应问题被FDA叫停。
罗氏 在2024年综合考虑市场竞争和现有成熟疗法后,放弃了其庞贝病基因疗法的开发。
安斯泰来 的基因疗法也曾因受试者出现神经损伤,在2022年被FDA紧急叫停,数月后才得以恢复。
这些挫折共同描绘出罕见病新药研发的残酷现实:技术迭代快,安全性格栅高,容错率极低。
04
结语:聚焦AB-1009,希望与挑战并存
拜耳果断放弃一代疗法,将所有资源集中于AB-1009,是一场基于技术判断的豪赌。对于患者而言,这无疑是一个积极的信号——更精准、更高效的疗法正在加速走向临床。
当然,我们必须保持理性的期待。AB-1009的1/2期临床试验刚刚启动,其安全性和有效性数据仍是未知数。它能否真正成为突破性的疗法,为绝望中的庞贝病患者带去转机,还需等待后续临床试验结果的揭晓。
对于我们每一个人,这个故事也提供了一个观察窗口:在充满不确定性的创新药研发领域,懂得“舍”,有时比盲目“取”更需要智慧,也更能代表未来的方向。
2026-03-05
·药时代
出海机会来了!
欧美药企、VC公司委托药时代BD 团队帮助寻找:靶向TL1A之口服小分子抑制剂、口服多肽抑制剂、口服抗体等在研口服创新药产品,全球权益或大中华区之外权益,License 或 NewCo,目前所处阶段不限。
请感兴趣的公司立即联系:药时代BD团队
BD@drugtimes.cn
正文共:1500字
预计阅读时间:5分钟
2026年3月4日,拜耳发布2025年财报,全年销售额达456亿欧元,同比下降2.2%;不计特殊项目的息税折旧摊销前利润97亿欧元,同比下降4.5%;核心每股收益4.91欧元,同比下降2.8%。制药业务收入178.29亿欧元,同比下降1.7%。
与此同时,拜耳对其罕见病基因治疗管线做出重要调整:终止一项名为ACTUS-101的早期临床阶段在研药物,转而集中资源推进另一款同类候选药物AB-1009。
这一决策源于,拜耳对2020年以20亿美元收购AskBio公司所得资产的战略优化。通过该交易,拜耳获得两款基于腺相关病毒(AAV)载体的基因疗法,均针对晚发型庞贝病(Pompe disease)。
其中代号为ACTUS-101的疗法在被收购前已进入临床试验阶段。然而今年1月,拜耳释放出调整信号,宣布停止该疗法Ⅰ期试验的新患者入组。根据美国国立卫生研究院的临床试验数据库,该研究此前共招募了7名受试者。
从研发逻辑上看,这种调整并不罕见。在罕见病基因疗法领域,企业往往会同时推进多个候选项目,但当其中一项显现出更大潜力时,资源便会逐渐向更具前景的方案倾斜。
拜耳选择集中资源,加快推动另一款庞贝病基因疗法AB-1009的研发进程,也在意料之中。与靶向肝脏的ACTUS-101不同,AB-1009采用AAV8载体直接靶向肌肉组织,这正是庞贝病患者发生进行性肌肉变性的核心病灶。同时,其搭载的GAA基因经过密码子优化,并搭配组织特异性启动子,可显著提升酶在肌肉组织中的表达效率。
值得注意的是,中国企业信念医药为其提供了专有的AAV衣壳技术及载体生产服务,成为该疗法研发的重要技术支撑。
这款候选药物已获得美国FDA授予的快速通道资格和孤儿药认定,并于今年初在美国启动Ⅰ/Ⅱ期临床试验。
加州大学尔湾分校医疗中心ALS与神经肌肉中心主任、该项目主要研究者Tahseen Mozaffar医学博士在今年1月的公告中表示:这款在研基因疗法旨在修复庞贝病患者的基因突变,探索其能否提升患者体内缺乏的酸性-α-葡糖苷酶生成量,进而减少患者对外源性酶替代疗法的依赖。
庞贝病又称糖原贮积病Ⅱ型,是由GAA基因突变引发的罕见常染色体隐性代谢病,人群患病率为1/4万~1/30万,核心特征为进行性肌肉变性,还可累及心脏、呼吸等多个系统,严重时会导致患者呼吸衰竭、心力衰竭。根据发病年龄可分为婴儿型和晚发型,其中婴儿型患者病情进展迅速,若无有效治疗常于1岁左右死亡。
目前全球范围内尚无根治庞贝病的方法,临床标准治疗为酶替代疗法(ERT),也是唯一获批的特异性治疗手段。该疗法需要通过定期输注外源性酸性α-葡萄糖苷酶(GAA)来缓解症状,但患者通常需终身用药,且对部分病例效果有限。正因如此,基因疗法被认为是一种具备“一次性治愈”潜力的新方向。如果能够通过一次治疗,使患者体内稳定产生所需的GAA酶,理论上可以从根本上改变疾病进展。不过,庞贝病的基因疗法仍处于临床研究阶段,目前全球尚无产品获批上市。
这足以见得,庞贝病基因疗法的临床转化之路并不平坦。过去几年间,多家生物制药公司在这一领域遭遇波折。2024年12月,Denali Therapeutics的酶替代疗法因临床前小鼠研究显示不良免疫反应风险,被FDA叫停临床试验计划;罗氏在评估市场竞争格局后,也于2024年选择放弃其庞贝病基因疗法项目。罗氏彼时坦言:“尽管目前尚无获批的庞贝病基因疗法,但多个机构正在评估不同的研究性治疗方案。”
同样,安斯泰来的同类药物亦遇挑战,2022年下半年,因一名受试者出现神经损伤,FDA紧急叫停其临床试验,直至2023年初才解除临床限制。
值得关注的是,FDA近期发布了一项关于个性化基因治疗的监管路径草案,提出以“小样本数据”作为支持产品获批的“合理机制框架”,为基因治疗领域的开发注入了新的政策动力。
参考资料:
1.https://www.fiercebiotech.com/biotech/bayer-ditches-one-aav-gene-therapy-pompe-disease-focus-rival-option
2.其他公开资料
封面图来源:即梦AI
恒瑞开年王炸,生物制药创新大会刷屏!
2026-03-02
赋能本土创新,一对一深度链接 |“2026 H1艾伯维中国合作日”Final Call
2026-03-02
刚刚!阿斯利康中国肿瘤架构大调整
2026-03-03
强生猛将潘昉玥空降默沙东中国,执掌肿瘤事业部!
2026-03-03
前默克中国张巍加入费森尤斯卡比,任总裁兼董事长
2026-03-03
版权声明/免责声明
本文为原创文章。
本文仅作信息交流之目的,不提供任何商用、医用、投资用建议。
文中图片、视频、字体、音乐等素材或为药时代购买的授权正版作品,或来自微信公共图片库,或取自公司官网/网络,部分素材根据CC0协议使用,版权归拥有者,药时代尽力注明来源。
如有任何问题,请与我们联系。
衷心感谢!
药时代官方网站:www.drugtimes.cn
联系方式:
电话:13651980212
微信:27674131
邮箱:contact@drugtimes.cn
点击查看更多精彩内容!
基因疗法孤儿药并购财报引进/卖出
2026-03-05
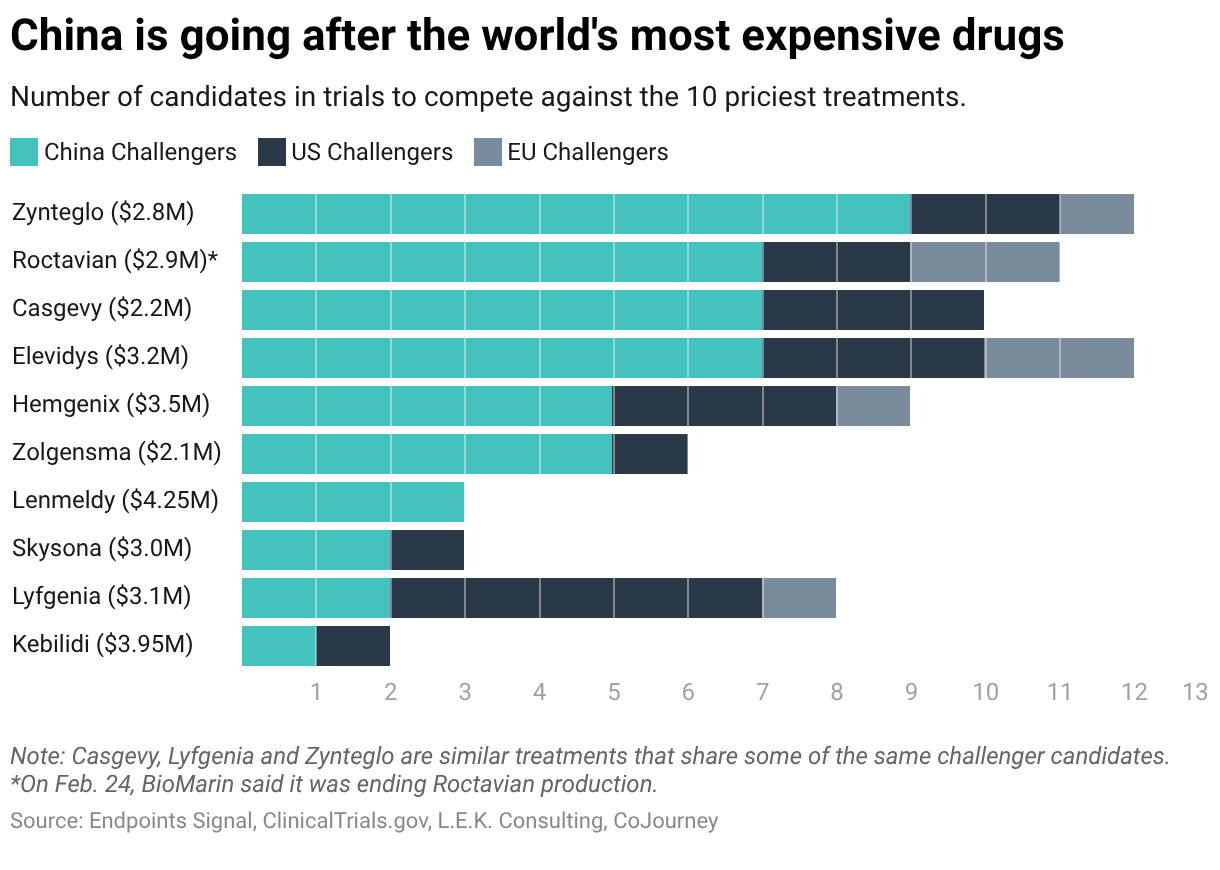
Last April, Chinese regulators approved Belief BioMed’s gene therapy for the bleeding disorder hemophilia B, setting up a challenge to one of the priciest medicines.
The treatment marked the first hemophilia gene therapy in China to be developed and manufactured entirely by a domestic company. Belief BioMed priced it at $350,000, one-tenth the cost of a rival therapy that sells for $3.5 million in the US.
In the year since, it’s become clear that Belief BioMed’s approval is a bellwether: In a global race to develop cheaper and potentially better alternatives to the world’s 10 most expensive medicines, China has pulled ahead.
An
Endpoints News
analysis of clinical trial databases shows that China accounts for 48 of the 77 programs targeting this group of ultra-expensive gene therapies. China’s total is more than double the number in the US and seven times as many as Europe.
Even if only a fraction of these lower-cost alternatives reach the market, it could upend the economics of a gene therapy system built on multimillion-dollar prices.
Shanghai-based Belief BioMed’s 161,000-square-foot facility — the equivalent of three football fields — stands as tangible proof that China’s gene therapy ambitions have gone beyond lab science and into industrial execution.
The company plans to dose its first commercial hemophilia B patient this month, and it’s working to secure approval in the US and additional countries.
“We want to be a global gene therapy company,” said Xiao Xiao, the co-founder and chief science officer of Belief BioMed. “No drug is only for one nation.”
In its expansive pipeline, the company is also conducting clinical trials of two therapies that could compete with top-priced therapies for hemophilia A and the rare disorder Duchenne muscular dystrophy. Belief BioMed is joined by Chinese companies like Shanghai Vitalgen BioPharma and CorrectSequence Therapeutics in chasing many of the most expensive therapies on the market.
For most of the pharmaceutical industry, direct competition is an expected part of the business. That wasn’t necessarily the case with gene therapies.
These medicines, which replace defective genes, require intricate scientific design and manufacturing. A 2024
analysis in the
Journal of Law and the Biosciences
argued that know-how is much tougher to reproduce compared to other parts of drug development.
And it was assumed that gene therapies would cure certain rare conditions with a limited market, discouraging competition. But some first-mover therapies had slow uptake, were
far from cures
or
mainly reached wealthy countries
.
Belief BioMed illustrates how China industrialized gene therapy development, as part of the
country’s wider rise in biotech
.
Trained in China, Xiao’s 35-year career took him from the University of Pittsburgh to the North Carolina company AskBio. He contributed fundamental science behind using AAV vectors to deliver gene therapies. He also navigated scientific challenges and high costs that caused many early US efforts to struggle.
Xiao went back to China more than a decade ago upon seeing Chinese patients unable to afford hemophilia B treatments. While patients in wealthy countries get preventative annual therapy covered by insurance, many in
China only receive treatment
after dangerous bleeding events.
Xiao and his colleagues founded Belief BioMed in 2018 to develop a gene therapy, in the hope that a one-time treatment could transform a chronic condition into a curable disease.
At the time, the Dutch company uniQure’s hemophilia B therapy was in clinical trials, and would be
approved in the US
as Hemgenix in 2022 with a $3.5 million list price, part of a wave of multimillion-dollar gene therapies. (The medicine has since been approved in Europe, Canada, Saudi Arabia, and in some Asia-Pacific countries, though not China.) Xiao sought to develop a less costly — and maybe even better — therapy for China and the larger world.
Belief’s therapy, like Hemgenix, uses an AAV vector to deliver a working copy of the factor IX gene to cells to produce long-term clotting. Xiao’s team engineered a next-generation capsid that’s designed to improve delivery and reduce immune reactions for greater safety and efficacy.
In both early and late-stage studies, a single infusion of Belief’s medicine greatly reduced or eliminated bleeding episodes in the year following treatment, according to findings published
last November in
Nature Medicine
. The clinical results so far appear comparable to Hemgenix, though Belief must dose more commercial patients before making a final comparison.
CSL Behring, which commercializes uniQure’s hemophilia B therapy, said in a statement that it’s confident in the value of Hemgenix given its long-term data. It’s working to expand the therapy to countries where it’s not available.
Belief’s bigger breakthrough may lie less in science and more in its decision to build its own facility, showing how China can independently develop and commercialize gene therapies.
Gene therapy production is notoriously fragile. Scientists grow large batches of specially engineered cells in bioreactors and insert DNA instructions into them. The cells’ own machinery then produces and assembles the viral particles that carry the therapeutic gene.
Belief does everything in-house, including producing the genetic material and vectors, to reduce errors that commonly plague the field. In 2022, for instance, US-based Regenxbio publicly said that
a third-party manufacturing delay
pushed back the start of a study for its Duchenne muscular dystrophy gene therapy.
In addition, Belief sought to rethink gene therapy manufacturing processes that had slowly evolved in the US and Europe. The company designed high-yield cells that churn out larger quantities of therapeutic virus, reworked gene insertion steps to reduce waste, and stripped out costly ingredients embedded in earlier-generation processes.
The streamlined manufacturing process drove down costs, Xiao said. So did China’s system for
quickly testing medicines
in humans. The result, he said, is an economic model that makes it possible to pursue gene therapies for conditions that others have deemed too risky or given up on, such as hemophilia A.
In February, US-based BioMarin said it would withdraw its hemophilia A therapy from the market after
failing to find a buyer
. That’s but one example of Western drugmakers pulling back from the field amid challenging economics.
Other Chinese companies echo Belief’s philosophy to be better and less expensive. YolTech Therapeutics is in clinical trials with a program that could compete with Casgevy, a $2.2 million gene therapy for sickle cell disease.
Rather than removing a patient’s cells and editing them in the lab, as companies in the US have already done, the Shanghai-based biotech
edits cells directly
in the bone marrow with a simple transfusion. Zi Jun Emma Wang, the chief technology officer and co-founder of YolTech, said it’s too early to talk about pricing but there’s potential to be much lower in cost than Casgevy.
“With our cost of goods being an order of magnitude lower than what’s typically the cost for cell therapy, we believe there’s a huge potential,” Wang said.
Helen Chen, head of LEK Consulting’s healthcare practice in Shanghai, recalled visiting Chinese companies 15 years ago that had lists of the world’s top-selling drugs in a bid to potentially compete with them.
“The difference now is that the competition from China is coming out much faster,” Chen said. “Before it was kind of an amusing lab exercise.”
China has the right conditions for gene therapy development, but not necessarily for commercialization, posing business challenges.
In China, it can still be difficult to get payer coverage for these treatments
.
The Chinese government recently expanded insurance
coverage for innovative medicines
. But gene therapies, even when relatively discounted, remain a tough sell. There’s no alternative but to design a lower-cost gene therapy from the beginning.
“We know that in China if you have a $3 million drug, nobody is able to afford it,” said Lijun Wang, the CEO of CoJourney, a company that manufactures gene therapies.
US and European challengers, by contrast, aren’t as bound by cost during development. Their payers balk at high gene therapy costs, but more often relent because the medicines replace a lifetime of chronic care.
After months of reimbursement struggles that delayed patient dosing, Belief recently secured a breakthrough when a city-level program known as Huiminbao agreed to partly cover the costs for its $350,000 therapy. The company partnered with Takeda China on domestic commercialization.
The next test lies abroad. The
FDA is skeptical
of studies from patients in China. But the company has engaged the FDA with its Chinese clinical and manufacturing data, hoping to avoid or minimize expensive US trials. US approval would grant access to the world’s largest pharmaceutical market, and speed approval elsewhere.
Xiao said the therapy’s cost in other markets would be lower than Hemgenix, but added it’s premature to give a concrete price. A discounted gene therapy could be welcome amid
slow uptake for hemophilia B gene therapies
, particularly in low-income countries that struggle to afford them.
“It could have a huge impact,” said hemophilia researcher Glenn Pierce, who has worked with companies like Biogen and Bayer. But, he added, even if it was one-tenth the price, Belief’s therapy could still be too expensive in poorer countries.
Already, China’s growing ability to make low-cost gene therapies is driving worldwide patients to Asia,
as Endpoints previously reported
.
Xiao said that compared to when the company was founded, China is bursting with gene therapy manufacturers. Investors are warming up to the space, aiming to be picks and shovels in a potential gene therapy gold rush.
“We had no choice but to bite the bullet and build out manufacturing. Now it’s everywhere,” Xiao said.
基因疗法上市批准
100 项与 AskBio Inc. 相关的药物交易
登录后查看更多信息
100 项与 AskBio Inc. 相关的转化医学
登录后查看更多信息
组织架构
使用我们的机构树数据加速您的研究。
登录
或

管线布局
2026年06月10日管线快照
管线布局中药物为当前组织机构及其子机构作为药物机构进行统计,早期临床1期并入临床1期,临床1/2期并入临床2期,临床2/3期并入临床3期
药物发现
2
1
临床前
临床1期
1
4
临床2期
其他
7
登录后查看更多信息
当前项目
| 药物(靶点) | 适应症 | 全球最高研发状态 |
|---|---|---|
NAN-101 ( DUSP1 ) | 非缺血性心肌病 更多 | 临床2期 |
AAV2-GDNF(University of Lund) ( GDNF family ) | 帕金森病 更多 | 临床2期 |
Ametefgene parvec ( GDNF family ) | 帕金森病(青年型、早发型) 更多 | 临床2期 |
LION-101 ( FKRP ) | 肢带型肌营养不良2I型 更多 | 临床1/2期 |
ACTUS-101 ( α-glucosidase ) | II型糖原贮积病 更多 | 临床1期 |
登录后查看更多信息
药物交易
使用我们的药物交易数据加速您的研究。
登录
或

转化医学
使用我们的转化医学数据加速您的研究。
登录
或

营收
使用 Synapse 探索超过 36 万个组织的财务状况。
登录
或

科研基金(NIH)
访问超过 200 万项资助和基金信息,以提升您的研究之旅。
登录
或

投资
深入了解从初创企业到成熟企业的最新公司投资动态。
登录
或

融资
发掘融资趋势以验证和推进您的投资机会。
登录
或

生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用


