预约演示
更新于:2025-05-07
Growth Disorders
生长障碍
更新于:2025-05-07
基本信息
别名 Disorder, Growth、Growth Disorder、Growth Disorders + [18] |
简介 Deviations from the average values for a specific age and sex in any or all of the following: height, weight, skeletal proportions, osseous development, or maturation of features. Included here are both acceleration and retardation of growth. |
关联
61
项与 生长障碍 相关的药物靶点 |
作用机制 GHR激动剂 |
非在研适应症- |
最高研发阶段批准上市 |
首次获批国家/地区 加拿大 |
首次获批日期2021-10-26 |
靶点 |
作用机制 NPRB激动剂 |
非在研适应症- |
最高研发阶段批准上市 |
首次获批国家/地区 欧盟 [+3] |
首次获批日期2021-08-26 |
作用机制 PARP1抑制剂 [+1] |
在研机构 |
原研机构 |
在研适应症 |
最高研发阶段批准上市 |
首次获批国家/地区 中国 |
首次获批日期2021-04-30 |
429
项与 生长障碍 相关的临床试验NCT06927310
A Multicenter, Randomized, Open-Label, Active-Controlled Phase III Clinical Study to Evaluate the Efficacy and Safety of Inpegsomatropin Injection, s.c Once a Week, Compared With Recombinant Human Growth Hormone (rhGH) Injection in Children With Idiopathic Short Stature (ISS).
This is a multicenter, randomized, open-label, active-controlled Phase III clinical trial to evaluate the efficacy and safety of Inpegsomatropin injection,once a week,compared with recombinant human growth hormone (rhGH) in children with idiopathic short stature (ISS). It plans to enroll 300 children with ISS, who will be randomized , stratified by gender and age, and assigned to either the experimental group or the positive controlled group. Each participant will undergo a screening period (up to 12 weeks), a treatment period (52 weeks), and a post-treatment follow-up period (5 weeks). And the safety and efficacy will be evaluated.
开始日期2025-04-07 |
申办/合作机构 |
NCT06676215
Efficacy of Amino Acid Supplementation on Gut Function and Nutritional Status in FIlipino Children Below Three Years Old
The goal of this intervention study is to determine the effects of indispensable amino acid (IAA) supplementation on children of either sex, aged 18 to 36 months. The main questions it aims to answer are:
* Does IAA supplementation affect biomarkers of Environmental Enteric Dysfunction (EED), through the Dual Sugar Lactulose/Rhamnose (L/R) test, plasma intestinal fatty acid binding protein, plasma lipopolysaccharide binding protein, fecal myeloperoxidase, fecal neopterin, and gut microbiota?
* Does IAA supplementation impact amino acid digestion, as assessed by a dual-stable isotope tracer?
* What is the weight gain and the changes in length-for-Age z-scores (LAZ) and body composition?
The researchers will compare the effects of consumption of standard complementary food with added IAA against a control group that has no added IAA through the assessment of the various biomarkers.
Participants will:
* answer questions regarding personal, demographic, anthropometric, and food intake information.
* provide biological samples, including breath, urine, stool, blood, and saliva.
* consume solutions or meals that contain sugars and/or stable isotope tracers.
* visit the FNRI and receive house visits by study staff.
* consume complementary food with or without supplement daily for 28 days.
* Does IAA supplementation affect biomarkers of Environmental Enteric Dysfunction (EED), through the Dual Sugar Lactulose/Rhamnose (L/R) test, plasma intestinal fatty acid binding protein, plasma lipopolysaccharide binding protein, fecal myeloperoxidase, fecal neopterin, and gut microbiota?
* Does IAA supplementation impact amino acid digestion, as assessed by a dual-stable isotope tracer?
* What is the weight gain and the changes in length-for-Age z-scores (LAZ) and body composition?
The researchers will compare the effects of consumption of standard complementary food with added IAA against a control group that has no added IAA through the assessment of the various biomarkers.
Participants will:
* answer questions regarding personal, demographic, anthropometric, and food intake information.
* provide biological samples, including breath, urine, stool, blood, and saliva.
* consume solutions or meals that contain sugars and/or stable isotope tracers.
* visit the FNRI and receive house visits by study staff.
* consume complementary food with or without supplement daily for 28 days.
开始日期2025-02-01 |
申办/合作机构 |
NCT06666777
Speech Therapy and Parenting for Early Socio-communicative Skills in Children with Neurodevelopmental Disabilities
Every year, millions of children are diagnosed with neurodevelopmental disabilities. This term covers a wide range of conditions, from genetic syndromes to brain injuries such as cerebral palsy. Children with neurodevelopmental disabilities often struggle in multiple areas, including language development. While standard speech therapy mainly focuses on understanding and producing words, these children may also have difficulties with the social and communicative skills needed for language. The parent-child relationship is especially important for helping kids develop in their early years.
This clinical trial aims to find out if an intervention focused on early social and communication skills, and involving parents, can help children with neurodevelopmental disabilities. The study includes children aged 6 months to 5 years. It seeks to answer two key questions:
* Does this intervention improve social and communication skills better than standard speech therapy?
* Does this intervention affect how parents interact with their child?
To find the answers, the study will compare two groups: one group will get the parent-involved intervention that focuses on early communication skills, while the other group will get standard speech therapy.
In the first intervention, therapists will guide parents in observing and supporting their child's social and communication behaviors during various activities like playtime and snack time. In contrast, the standard speech therapy will focus on traditional goals, such as improving the child's ability to vocalize, understand, and use words, without involving parents.
Both interventions will follow the same schedule-eight weekly sessions, each lasting 45 minutes, over two months.
Before and after the interventions, the children and parents will:
* Have an assessment of the child's language, social, and communication development.
* Participate in a 10-minute video recording of parent-child playtime, which will be used to study parenting behavior.
This clinical trial aims to find out if an intervention focused on early social and communication skills, and involving parents, can help children with neurodevelopmental disabilities. The study includes children aged 6 months to 5 years. It seeks to answer two key questions:
* Does this intervention improve social and communication skills better than standard speech therapy?
* Does this intervention affect how parents interact with their child?
To find the answers, the study will compare two groups: one group will get the parent-involved intervention that focuses on early communication skills, while the other group will get standard speech therapy.
In the first intervention, therapists will guide parents in observing and supporting their child's social and communication behaviors during various activities like playtime and snack time. In contrast, the standard speech therapy will focus on traditional goals, such as improving the child's ability to vocalize, understand, and use words, without involving parents.
Both interventions will follow the same schedule-eight weekly sessions, each lasting 45 minutes, over two months.
Before and after the interventions, the children and parents will:
* Have an assessment of the child's language, social, and communication development.
* Participate in a 10-minute video recording of parent-child playtime, which will be used to study parenting behavior.
开始日期2025-01-15 |
申办/合作机构- |
100 项与 生长障碍 相关的临床结果
登录后查看更多信息
100 项与 生长障碍 相关的转化医学
登录后查看更多信息
0 项与 生长障碍 相关的专利(医药)
登录后查看更多信息
20,021
项与 生长障碍 相关的文献(医药)2025-12-31·Journal of Dermatological Treatment
Effect of amniotic fluid on hair follicle growth
Article
作者: Yurtsever, Bulut ; Aygun, Elif Ganime ; Cakirsoy, Didem ; Ovali, Ercument ; Tumentemur, Gamze
2025-12-31·Veterinary Quarterly
Developing a novel TaqMan qPCR assay for optimizing
Salmonella
Pullorum detection in chickens
Article
作者: Li, Yan ; Wang, Haijian ; Kehrenberg, Corinna ; Yu, Longhai ; Wang, Hao ; Müller, Anja ; Kang, Xiamei ; Yue, Min
2025-12-31·Global Health Action
Trends in the prevalence of concurrent anaemia and stunting among infants and young children in Rwanda: a cross-sectional study from 2010 to 2020
Article
作者: Gbadamosi, Mojeed Akorede ; Nemerimana, Mathieu
448
项与 生长障碍 相关的新闻(医药)2025-05-05
COPENHAGEN, Denmark, May 5, 2025 /PRNewswire/ -- Dawn Health, a global leader in digital health, has secured a funding round of €11.5 million from its existing investors: Chr. Augustinus Fabrikker, the Export and Investment Fund of Denmark (EIFO), and Trifork. The investment is aimed at supporting the company's strategy to deliver its platform and product suite to global pharma companies through a SaaS model, while continuing to invest in further offerings within the Dawn Product Suite.
Since 2021, Dawn Health has been dedicated to developing a best-in-class platform designed specifically to accommodate the needs and use cases of the pharmaceutical industry. The Dawn Platform and Product Suite have already been widely adopted by five global industry leaders, including Merck and Novartis. The Dawn Platform is currently used in areas such as oncology, multiple sclerosis, and rare pediatric conditions like growth disorders. It helps patients manage their treatment, report symptoms, and stay in close contact with their healthcare team.
The Dawn Platform and Product Suite empower pharma companies, patients, and healthcare professionals to improve outcomes and patient care by leveraging advanced capabilities in AI, data, evidence generation, clinical integrations, personalization, and connected health. By improving both data collection and analytics, these capabilities ultimately benefit patients and pharma companies alike, positioning the Dawn Platform as the foundation for therapy companions, disease management programs, and real-world evidence (RWE) solutions that enable the next generation of digital health.
"Our ambition is to be the global leader in digital health, powering pharma's next-generation products – and ultimately improving the lives of patients worldwide," said Alexander Mandix Hansen, CEO of Dawn Health. "This funding allows us to bring our proven platform to more markets and deepen our impact."
This next phase reinforces Dawn Health's position as a trusted partner to pharma companies, delivering valuable, scalable, regulatory-grade digital health products that evolve with the needs of modern medicine.
"Since the major investment in December 2021, Dawn Health has grown its revenue significantly and expanded its footprint in global pharma. With more than 100 employees, unique solutions, and a strong regulatory infrastructure, we are prepared to further accelerate our growth," says Chairman Lars Marcher.
About Dawn Health
Dawn Health is a global leader in digital health, specializing in the development of Software as a Medical Device (SaMD), Digital Therapeutics (DTx), and connected health solutions. Accelerating the launch of digital solutions to market, the Dawn Health product suite drives innovation to change the lives of people with chronic conditions. Through close partnerships with the life sciences industry, Dawn Health creates digital health products that transform patient care through an empathetic and human-centric approach.
Logo -
WANT YOUR COMPANY'S NEWS FEATURED ON PRNEWSWIRE.COM?
440k+
Newsrooms &
Influencers
9k+
Digital Media
Outlets
270k+
Journalists
Opted In
GET STARTED
2025-05-01
EPIDIOLEX has significant market potential due to the growing demand for alternative therapies and its effectiveness in treating rare seizure disorders like Lennox-Gastaut syndrome and Dravet syndrome. As awareness around the benefits of CBD in treating neurological conditions increases, EPIDIOLEX stands to benefit from expanding indications and market reach.
LAS VEGAS, May 1, 2025 /PRNewswire/ -- DelveInsight's '
EPIDIOLEX Market Size, Forecast, and Market Insight Report' highlights the details around EPIDIOLEX, the first FDA-approved prescription cannabidiol (CBD) to treat seizures associated with Lennox-Gastaut syndrome (LGS), Dravet syndrome, or tuberous sclerosis complex (TSC) in patients 1 year of age or older. The report provides product descriptions, patent details, and competitor products (marketed and emerging therapies) of EPIDIOLEX. The report also highlights the historical and forecasted sales from 2020 to 2034 segmented into 7MM [the United States, the EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan].
Jazz Pharmaceuticals' EPIDIOLEX (cannabidiol) Overview
EPIDIOLEX (formerly known as GWP42003-P and marketed as EPIDYOLEX in Europe) is the first plant-based, prescription cannabis-derived oral medication developed by GW Pharmaceuticals. It represents a new category of antiepileptic drugs with a unique mechanism of action. The active compound, cannabidiol (CBD), is a naturally occurring cannabinoid found in the Cannabis sativa L. plant. While the exact way EPIDIOLEX works to reduce seizures in humans remains unclear, it is distinct from traditional anticonvulsants. CBD does not act through CB1 receptors or by blocking voltage-gated sodium channels, which are typical pathways for other antiepileptic drugs.
Instead, CBD is thought to work by influencing multiple internal pathways, potentially producing a combined anticonvulsant effect. These include enhancing neuronal inhibition (through synaptic and extrasynaptic GABA channels), affecting intracellular calcium levels (via TRPV channels, VDAC, and GPR55), and reducing inflammation, possibly via adenosine modulation. Importantly, CBD does not directly bind to or activate the CB1 or CB2 receptors at levels relevant to its seizure-reducing effects. Ongoing research is examining other possible mechanisms such as interactions with adenosine, glycine, GABAergic systems, and serotonin receptors.
EPIDIOLEX has received regulatory approval in numerous countries, including the U.S. (FDA), Europe (European Commission), Great Britain (MHRA), Australia (Therapeutic Goods Administration), Switzerland (Swissmedic), Israel (Ministry of Health), and New Zealand (Medicines and Medical Devices Safety Authority). It is formulated as an oral solution containing highly purified CBD. In the U.S., it is approved to treat seizures linked to Lennox-Gastaut syndrome (LGS), Dravet syndrome (DS), and tuberous sclerosis complex (TSC) in patients aged one year and older. In the European Union, under the name Epidyolex, it is approved as an add-on treatment with clobazam for LGS and DS in patients two years and older, and for TSC-associated seizures as well. Both the FDA and EMA have granted Orphan Drug Designation (ODD) for EPIDIOLEX/EPIDYOLEX for these conditions.
EPIDIOLEX/EPIDYOLEX net product sales rose by
15% to
USD 972.4 million in 2024 and by
14% to
USD 275.0 million in the fourth quarter of 2024, compared to the corresponding periods in 2023.
Learn more about EPIDIOLEX/EPIDYOLEX projected market size for DEE @
EPIDIOLEX Market Potential
Developmental and epileptic encephalopathies (DEE) encompass a group of severe neurological conditions marked by developmental delays and persistent epilepsy. These disorders typically present early in life and are linked to significant cognitive deficits. According to DelveInsight's analysis, there were approximately
287K diagnosed prevalent cases of DEE across the 7MM in 2024. This number is projected to increase due to the rise in neurological disorders, including epilepsy, as well as environmental and maternal health factors contributing to a higher DEE prevalence throughout the forecast period (2025−2034).
The US FDA has approved a range of medications for conditions like LGS, Dravet Syndrome, and CDKL5 Deficiency Disorder, including various drug classes such as sodium channel modulators, GABA receptor modulators, calcium channel blockers, receptor blockers, and others. Some of the available anti-epileptic drugs (AEDs) include
FINTEPLA, EPIDIOLEX, TOPAMAX, BANZEL, LAMICTAL, FELBATOL, ONFI, KLONOPIN, DIACOMIT, SABRIL, and more.
In 2024, the total market size for DEE in the 7MM was around
USD 2.3 billion, and this figure is expected to grow by 2034, driven by wider adoption of approved therapies due to label expansions and the introduction of emerging treatments.
Discover more about the DEE market in detail @
Developmental and Epileptic Encephalopathy Market Report
Emerging Competitors of EPIDIOLEX
Some of the drugs in the pipeline include
EPX-100 (Harmony Biosciences),
Bexicaserin (Lundbeck),
Zorevunersen (Stoke Therapeutics/Biogen),
Relutrigine (Praxis Precision Medicines), and others. In August 2024, the FDA removed a partial clinical hold as the company advances toward a Phase III registrational study of zorevunersen (STK-001) in children and adolescents with Dravet syndrome.
In
July 2024, Longboard Pharmaceuticals announced that the FDA had granted Breakthrough Therapy designation for its investigational drug bexicaserin for the treatment of seizures associated with DEEs for patients 2 years of age or older.
To know more about the number of competing drugs in development, visit @
EPIDIOLEX Alternatives
Key Milestones of EPIDIOLEX
In
August 2024, Jazz Pharmaceuticals reported top-line results from a Phase 3 open-label, single-arm study conducted in Japan, assessing the safety and effectiveness of its cannabidiol oral solution (marketed globally as EPIDIOLEX/EPIDYOLEX) as an add-on therapy for seizures linked to Lennox-Gastaut syndrome (LGS), Dravet syndrome (DS), or tuberous sclerosis complex (TSC). While the trial did not achieve its primary efficacy goal—defined as a pre-specified percentage reduction in seizure frequency during the up-to-16-week treatment period compared to baseline in Japanese pediatric patients—numerical improvements were noted in both the primary and several secondary measures. The study did not identify any new safety concerns.
In
May 2021, Jazz Pharmaceuticals acquired EPIDIOLEX as part of the acquisition of GW Pharmaceuticals plc, or GW, which expanded its growing neuroscience business with a global, high-growth childhood-onset epilepsy franchise.
In
April 2021, EPIDYOLEX was approved for adjunctive therapy of seizures associated with TSC for patients 2 years of age and older in the EU.
In
July 2020, EPIDYOLEX was approved for the treatment of seizures associated with TSC in patients one year of age and older. FDA also approved the expansion of all existing indications, LGS and DS, to patients one year of age and older.
In
September 2019, the European Commission approved EPIDYOLEX for use as adjunctive therapy of seizures associated with LGS or DS, in conjunction with clobazam, for patients two years of age and older.
In
June 2018, EPIDIOLEX was approved in the US for the treatment of seizures associated with two rare and severe forms of epilepsy, LGS and DS, in patients two years of age and older.
EPIDIOLEX Launch Status
EPIDIOLEX was launched on November 1, 2018, in the US market after FDA approval for the treatment of seizures associated with LGS or Dravet syndrome in patients two years of age and older.
EPIDYOLEX was launched in 2019 in Germany and the UK market for use as adjunctive therapy for seizures associated with LGS or Dravet syndrome, in conjunction with clobazam, for patients two years of age and older (GW Pharmaceuticals, 2019b).
In 2021, EPIDYOLEX was launched in Spain.
In the first quarter of 2022, the company launched EPIDYOLEX for LGS, Dravet syndrome, and TSC in Ireland, for TSC in Scotland and Wales.
In the third quarter of 2022, the company launched EPIDYOLEX for TSC in Italy and Switzerland.
In the fourth quarter of 2022, the company successfully completed the pricing and reimbursement process and commercial launch of EPIDYOLEX in France.
Discover how EPIDIOLEX/EPIDYOLEX is shaping the DEE treatment landscape @
EPIDIOLEX CBD
EPIDIOLEX Market Dynamics
EPIDIOLEX, the first
FDA-approved cannabidiol (CBD)-based medication for the treatment of rare forms of epilepsy, has had a significant impact on the pharmaceutical market since its approval in 2018. The drug is primarily used to treat seizures associated with Dravet syndrome and Lennox-Gastaut syndrome, two rare and severe forms of epilepsy. The market dynamic for EPIDIOLEX is shaped by several factors, including
increasing awareness of its therapeutic potential, the
expanding medical cannabis industry, and the
growing demand for alternative treatments to traditional anti-epileptic drugs.
One of the main drivers of the EPIDIOLEX market is the
rising acceptance of CBD-based therapies. As medical cannabis becomes more mainstream and research into its medicinal properties continues to grow, patients and healthcare providers are increasingly turning to EPIDIOLEX as a treatment option. The
growing body of clinical evidence supporting its effectiveness and safety has reinforced this trend. Additionally, as the stigma around cannabis-derived medications continues to fade, it opens up more
opportunities for EPIDIOLEX in both pediatric and adult patient populations.
However, the market for EPIDIOLEX is not without its challenges.
High treatment costs and insurance coverage issues can limit accessibility for some patients, especially in markets outside the United States. Despite its clinical efficacy, the
price point for EPIDIOLEX remains a point of contention, as it may not be affordable for all families, especially when compared to other conventional anti-epileptic drugs. Furthermore,
competition from other cannabinoid-based therapies, as well as
new developments in the broader epilepsy drug market, could impact the drug's market share over time.
Looking ahead, the market dynamics for EPIDIOLEX will likely continue to evolve as the
global regulatory landscape changes and
new clinical trials further establish its benefits in other neurological conditions. With increasing research into CBD's potential applications, there may be opportunities for EPIDIOLEX to expand its indications, providing a solid foundation for its continued growth. However, market growth will also depend on the
ability to address pricing concerns, improve insurance coverage, and navigate potential competitive pressures from both traditional and emerging therapies.
Dive deeper to get more insight into EPIDIOLEX/EPIDYOLEX's strengths & weaknesses relative to competitors @
EPIDIOLEX Market Drug Report
Table of Contents
Related Reports
Developmental and Epileptic Encephalopathies Market
Developmental and Epileptic Encephalopathies Market Insights, Epidemiology, and Market Forecast – 2034 report deliver an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key DEE companies, including
vid Therapeutics, Stoke Therapeutics, Biogen, Harmony Biosciences, Lundbeck (Longboard Pharmaceuticals), Praxis Precision Medicines, SK Life Science, Cerecin Neurosciences, Encoded Therapeutics, Praxis Precision Medicines, among others.
Developmental and Epileptic Encephalopathies Pipeline
Developmental and Epileptic Encephalopathies Pipeline Insight
– 2025 report provides comprehensive insights about the pipeline landscape, pipeline drug profiles, including clinical and non-clinical stage products, and the key DEE companies, including
Denovo Biopharma, Cantex Pharmaceuticals, CNS Pharmaceuticals, CANbridge Pharmaceuticals, Vaximm, Inovio Pharmaceuticals, Mustang Bio, Bullfrog AI Holdings, Cantex, Chimeric Therapeutics, Philogen, Boehringer Ingelheim, Photonamic GmbH, Berg Pharma, Beyond Bio, Genenta Science, Polaris Pharmaceuticals, Telix Pharmaceuticals, Shanghai Simnova Biotechnology, NEONC Technologies, among others.
Dravet Syndrome Market
Dravet Syndrome Market Insights, Epidemiology, and Market Forecast – 2034 report deliver an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key Dravet syndrome companies including
Biocodex, GW Pharmaceuticals, Zogenix, Ovid Therapeutics, PTC Therapeutics, among others.
Lennox-Gastaut Syndrome Market
Lennox-Gastaut Syndrome Market Insights, Epidemiology, and Market Forecast – 2034 report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key Lennox-Gastaut syndrome companies, including
GlaxoSmithKline, Meda Pharmaceuticals, Roche, Lundbeck, Greenwich Biosciences, Janssen Pharmaceuticals, Eisai, Zogenix, Takeda, Ovid Therapeutics, among others.
About DelveInsight
DelveInsight is a leading Business Consultant and Market Research firm focused exclusively on life sciences. It supports pharma companies by providing comprehensive end-to-end solutions to improve their performance. Get hassle-free access to all the healthcare and pharma market research reports through our subscription-based platform PharmDelve
.
Contact Us
Shruti Thakur
[email protected]
+14699457679
Logo:
SOURCE DelveInsight Business Research, LLP
WANT YOUR COMPANY'S NEWS FEATURED ON PRNEWSWIRE.COM?
440k+
Newsrooms &
Influencers
9k+
Digital Media
Outlets
270k+
Journalists
Opted In
GET STARTED
上市批准临床3期并购突破性疗法孤儿药
2025-05-01
First Quarter 2025 Total Revenues of $745 million (+15% Y/Y and +17% at Constant Currency Y/Y)
First Quarter 2025 GAAP Diluted Earnings Per Share (EPS) of $0.95 (+107% Y/Y)
First Quarter 2025 Non-GAAP Diluted EPS of $1.13 (+59% Y/Y)
Conference Call and Webcast Scheduled Today at 4:30 p.m. ET
SAN RAFAEL, Calif., May 1, 2025 /PRNewswire/ -- BioMarin Pharmaceutical Inc. (NASDAQ: BMRN) today announced financial results for the first quarter ended March 31, 2025.
"During the first quarter, we saw continued high demand for our innovative medicines resulting in strong revenue growth and profitability," said Alexander Hardy, President and Chief Executive Officer of BioMarin. "Products in our pipeline also advanced according to plan. In April, we shared positive top-line results from the Phase 3 PALYNZIQ® study for the treatment of adolescents with phenylketonuria between the ages of 12 and 17. Also in April, we were pleased to conclude enrollment in the pivotal study in hypochondroplasia with VOXZOGO, keeping us on track to launch in 2027, should data be supportive."
Mr. Hardy continued, "For the remainder of 2025, we look forward to continued momentum in our global expansion of VOXZOGO® for achondroplasia. Across our Enzyme Therapies, we plan to build upon strong PALYNZIQ performance in the quarter, as well as initiatives to drive uptake of our other therapies to reach an even greater number of patients around the world. In addition to our strong financial outlook, we expect to advance multiple new indications with VOXZOGO in our CANOPY clinical program, share early clinical results from both BMN 351 for Duchenne Muscular dystrophy and BMN 333, our long-acting C-type natriuretic peptide, as well as execute on our business development strategy. We are delivering strong growth and profitability while we continue to implement BioMarin's new strategy and operating model. We look forward to seeing the benefits of this transformation flow through our results in the coming quarters and beyond."
First Quarter 2025 Financial Highlights
Total Revenues for the first quarter of 2025 were $745 million, an increase of 15% compared to the same period in 2024, driven by strong 40% year-over-year VOXZOGO revenue growth from new patients initiating therapy across all regions. In the quarter, revenues from BioMarin's Enzyme Therapies (ALDURAZYME®, BRINEURA®, NAGLAZYME®, PALYNZIQ and VIMIZIM®) increased 8% compared to the first quarter of 2024, driven by a combination of increased patient demand and the timing of large government orders in all regions. The increase was partially offset by lower KUVAN® product revenues attributed to continued generic competition as a result of the loss of market exclusivity.
GAAP Net Incom
e increased by $97 million to $186 million in the first quarter of 2025 compared to the same period in 2024, an increase of 109%, primarily attributed to higher gross profit driven by the factors noted above. The increase was also attributed to lower operating expenses following the termination of certain early stage development programs following the company's 2024 strategic portfolio review and focused ROCTAVIAN strategy announced in the second half of 2024. These increases were partially offset by higher tax provision primarily due to increase in taxable income.
Non-GAAP Income increased by $81 million to $221 million in the first quarter of 2025 compared to the same period in 2024, representing 58% growth. The increase in Non-GAAP Income was primarily due to the factors noted above.
First Quarter 2025 Business Highlights
Innovation
Skeletal Conditions: In March 2025, BioMarin presented new data demonstrating favorable safety and strong adherence in real-world clinical practice with VOXZOGO in children with achondroplasia under the age of 3 years old at the 2025 American College of Medical Genetics and Genomics (ACMG) Annual Meeting. No treatment-related adverse events nor any dose interruptions were reported among 63 children followed for up to 23.7 months. These real-world findings further validate VOXZOGO's established safety profile and reinforce the therapeutic benefit seen in clinical studies. The study's safety results, including in infants as young as 1 month old, add to the growing body of evidence supporting early treatment initiation with VOXZOGO, consistent with new international treatment guidelines published in the journal Nature Reviews Endocrinology earlier this year.
In April 2025, BioMarin completed enrollment in its pivotal Phase 3 study with VOXZOGO in hypochondroplasia and the company is on track to share topline data in 2026, with a potential launch in 2027. BioMarin plans to leverage its multiyear track record treating children with achondroplasia, a related condition, to raise awareness and treat children with hypochondroplasia across the globe. The CANOPY clinical program is continuing to advance VOXZOGO in additional new indications, including idiopathic short stature, Noonan syndrome, Turner syndrome, and SHOX deficiency.
With BMN 333, BioMarin's long-acting C-type natriuretic peptide (CNP), the company enrolled multiple cohorts of healthy volunteers in its first-in-human study, with initial pharmacokinetic (PK) data expected by year-end. Detailed data from this study is expected to be presented at a scientific forum in the first half of 2026. Pre-clinical data with BMN 333 demonstrated sustained 100 pM concentrations for free CNP, representing an approximate 2-3 fold increase versus published data in an analogous pre-clinical model for other long-acting CNP analogs.
Additionally, BioMarin recently met with FDA and reached agreement on an overall clinical development plan for BMN 333 in achondroplasia. Assuming the Phase 1 data are supportive, the company plans to initiate a registration-enabling study in 2026, supporting a previously disclosed target for 2030 approval. BioMarin plans to seek similar agreements with additional global regulators in the coming months.
The company announced in April that its pivotal study with PALYNZIQ for the treatment of adolescents between the ages of 12 and 17 met its primary efficacy endpoint, demonstrating a statistically significant lowering in blood Phe levels. These data will support the planned submission of applications in the second half of 2025 to expand PALYNZIQ age eligibility in the United States and Europe.
Other Clinical Pipeline Programs: BMN 351, BioMarin's next generation oligonucleotide for Duchenne Muscular Dystrophy, and BMN 349, an oral therapeutic for Alpha-1 antitrypsin deficiency (AATD)-associated liver disease, continue to advance. Initial data for BMN 351 is anticipated to be presented at a scientific congress in the second half of 2025 (including muscle dystrophin levels from the 6 mg/kg cohort after 25 weeks of dosing).
During a recent strategic portfolio assessment of R&D programs, BioMarin determined that the evolving profile for BMN 370, a pre-clinical candidate for the treatment of von Willebrand disease, did not meet its threshold for further development and commercialization. The program has been discontinued and impacted employees have been redeployed within BioMarin.
Growth
Total VOXZOGO revenue in the first quarter increased 40% compared to the same period in 2024, representing continued strong global demand since its commercial launch in 2021. As of the end of the quarter, children with achondroplasia in 49 countries around the world were being treated with VOXZOGO.
In the U.S., BioMarin is investing in focused initiatives to drive continued expansion. These efforts include increasing field personnel to broaden the prescriber base and adding awareness platforms to drive adoption of VOXZOGO treatment. This is expected to begin increasing the rate of U.S. expansion in the second half of the year. Outside of the U.S. (OUS), from where the majority of VOXZOGO revenue is generated, uneven ordering patterns, consistent with BioMarin's other brands, were observed. This OUS dynamic is expected to result in VOXZOGO full-year revenues being more weighted towards the second half of 2025.
Enzyme Therapies revenues grew 8% in the first quarter Y/Y, driven by strong continued demand for PALYNZIQ. Strong PALYNZIQ performance as well as solid growth from BioMarin's other enzyme treatments are expected to continue throughout 2025.
Value Commitment
In the first quarter of 2025, BioMarin delivered strong results across the business. Total revenues for the first quarter grew 15% Y/Y. First quarter GAAP Operating Margin of 30.0% expanded 16.4 percentage points Y/Y while GAAP Diluted EPS of $0.95 increased 107% Y/Y. First quarter Non-GAAP Operating Margin of 35.7% expanded 11.9 percentage points Y/Y while Non-GAAP Diluted EPS of $1.13 increased 59% Y/Y. These measures of profitability increased at rates faster than revenue growth, representing the company's focus on operational efficiency.
During the quarter, BioMarin continued to realize the benefits of cost transformation initiatives implemented in 2024, resulting in a decrease in GAAP and Non-GAAP R&D and SG&A expenses Y/Y. Throughout the remainder of 2025, BioMarin expects to increase investments in VOXZOGO indication expansion, clinical pipeline development, and commercialization initiatives supporting the company's Skeletal Conditions and Enzyme Therapies business units.
The company generated operating cash flows totaling $174 million in first quarter 2025, an increase of 271% compared to first quarter 2024. Total cash and investments at the end of the first quarter were approximately $1.8 billion, and with anticipated increasing profitability, BioMarin is positioned to generate increasing operating cash flow into the future.
Today, the company reaffirmed its previously communicated 2025 full-year financial guidance, which reflects the impact of tariffs that have already been enacted but does not reflect the impact of potential future pharmaceutical tariffs. BioMarin has immaterial exposure to U.S. tariffs for China, Mexico and Canada across its global supply chain operations and product sales.
Forward-Looking Non-GAAP Financial Information
BioMarin does not provide guidance for GAAP reported financial measures (other than revenue) or a reconciliation of forward-looking Non-GAAP financial measures to the most directly comparable GAAP reported financial measures because the company is unable to predict with reasonable certainty the financial impact of changes resulting from its strategic portfolio and business operating model reviews; potential future asset impairments; gains and losses on investments; and other unusual gains and losses without unreasonable effort. These items are uncertain, depend on various factors, and could have a material impact on GAAP reported results for the guidance period. As such, any reconciliations provided would imply a degree of precision that could be confusing or misleading to investors.
2025 Full-Year Financial Guidance (in millions, except % and EPS amounts)
BioMarin will host a conference call and webcast to discuss first quarter 2025 financial results today, Thursday, May 1, 2025, at 4:30 p.m. ET. This event can be accessed through this link or on the investor section of the BioMarin website at .
About BioMarin
BioMarin is a global biotechnology company dedicated to translating the promise of genetic discovery into medicines that make a profound impact on the life of each patient. The San Rafael, California-based company, founded in 1997, has a proven track record of innovation with eight commercial therapies and a strong clinical and preclinical pipeline. Using a distinctive approach to drug discovery and development, BioMarin pursues treatments that offer new possibilities for patients and families around the world navigating rare or difficult to treat genetic conditions. To learn more, please visit .
Forward-Looking Statements
This press release and the associated conference call and webcast contain forward-looking statements about the business prospects of BioMarin Pharmaceutical Inc. (BioMarin), including, without limitation, statements about: future financial performance, including the expectations of Total Revenues, Non-GAAP Operating Margin percentage, Non-GAAP Diluted EPS and Operating Cash Flow for, in certain instances, the full-year 2025 and future periods, as well as profitability growth in 2025, and the underlying drivers of those results, such as the revenue opportunity represented by treatments for Skeletal Conditions, namely VOXZOGO, the expected demand and continued growth of BioMarin's Enzyme Therapies portfolio, including PALYNZIQ, and the expectation regarding the full realization of the benefits of BioMarin's cost transformation program; plans regarding BioMarin's revamped corporate strategy and operating model in 2025 and beyond, including expected growth in the Skeletal Conditions business unit and execution of BioMarin's business development strategy, and its anticipated benefits; the timing of orders for commercial products; BioMarin's ability to meet product demand; the timing of BioMarin's clinical development and commercial prospects, including announcements of data from clinical studies and trials; the clinical development and commercialization of BioMarin's product candidates and commercial products, including (i) expected advancements of pipeline candidates, including BMN 333, BMN 349 and BMN 351, the anticipated initial data read-out for BMN 351 in the second half of 2025, the expected data and data presentation for BMN 333 in the first half of 2026 and plans to initiate a registration-enabling study for BMN 333 in 2026, as well as plans to seek similar agreements with additional global regulators in the coming months; (ii) plans to submit applications to expand PALYNZIQ age eligibility for the treatment of adolescents with phenylketonuria between the ages of 12 and 17 in the U.S. and Europe in the second half of 2025; (iii) expected topline data from the pivotal study in hypochondroplasia in 2026 and launch in 2027; (iv) the expectations regarding global expansion of VOXZOGO for achondroplasia and expected increase in the rate of U.S. expansion in the second half of 2025; (v) plans to advance five new VOXZOGO indications with BioMarin's CANOPY clinical program; and (vi) plans to reach greater number of patients around the world across BioMarin's Enzyme Therapies; the expected benefits and availability of BioMarin's commercial products and product candidates; and potential growth opportunities and trends.
These forward-looking statements are predictions and involve risks and uncertainties such that actual results may differ materially from these statements. These risks and uncertainties include, among others: BioMarin's success in the commercialization of its commercial products; impacts of macroeconomic and other external factors on BioMarin's operations, such as trade wars and potential future pharmaceutical tariffs; results and timing of current and planned preclinical studies and clinical trials and the release of data from those trials; BioMarin's ability to successfully manufacture its commercial products and product candidates; the content and timing of decisions by the U.S. Food and Drug Administration, the European Commission and other regulatory authorities concerning each of the described products and product candidates; the market for each of these products; actual sales of BioMarin's commercial products; and those factors detailed in BioMarin's filings with the Securities and Exchange Commission, including, without limitation, the factors contained under the caption "Risk Factors" in BioMarin's Annual Report on Form 10-K for the fiscal year ended December 31, 2024, as such factors may be updated by any subsequent reports. Stockholders are urged not to place undue reliance on forward-looking statements, which speak only as of the date hereof. BioMarin is under no obligation, and expressly disclaims any obligation to update or alter any forward-looking statement, whether as a result of new information, future events or otherwise.
BioMarin®, BRINEURA®, KUVAN®, NAGLAZYME®, PALYNZIQ®, ROCTAVIAN®, VIMIZIM® and VOXZOGO® are registered trademarks of BioMarin Pharmaceutical Inc., or its affiliates. ALDURAZYME® is a registered trademark of BioMarin/Genzyme LLC. All other brand names and service marks, trademarks and other trade names appearing in this release are the property of their respective owners.
Non-GAAP Information
The results presented in this press release include both GAAP information and Non-GAAP information. Non-GAAP Income is defined by the company as GAAP Net Income excluding amortization of intangible assets, stock-based compensation expense and, in certain periods, certain other specified items, as detailed below when applicable. The company also includes a Non-GAAP adjustment for the estimated tax impact of the reconciling items. Non-GAAP R&D expenses and Non-GAAP SG&A expenses are defined by the company as GAAP R&D expenses and GAAP SG&A expenses, respectively, excluding stock-based compensation expense and, in certain periods, certain other specified items, as detailed below when applicable. Non-GAAP Operating Margin percentage is defined by the company as GAAP Income from Operations, excluding amortization of intangible assets, stock-based compensation expense and, in certain periods, certain other specified items, divided by GAAP Total Revenues. Non-GAAP Diluted EPS is defined by the company as Non-GAAP Income divided by Non-GAAP Weighted-Average Diluted Shares Outstanding. Non-GAAP Weighted-Average Diluted Shares Outstanding is defined by the company as GAAP Weighted-Average Diluted Shares Outstanding, adjusted to include any common shares issuable under the company's equity plans and convertible debt in periods when they are dilutive under Non-GAAP. The company's presentation of percentage changes in total revenues at Constant Currency rates, which is computed using current period local currency sales at the prior period's foreign exchange rates, is also a Non-GAAP financial measure. This measure provides information about growth (or declines) in the company's total revenue as if foreign currency exchange rates had not changed between the prior period and the current period.
BioMarin regularly uses both GAAP and Non-GAAP results and expectations internally to assess its financial operating performance and evaluate key business decisions related to its principal business activities: the discovery, development, manufacture, marketing and sale of innovative biologic therapies. Because Non-GAAP Income, Non-GAAP R&D expenses, Non-GAAP SG&A expenses, Non-GAAP Operating Margin percentage, Non-GAAP Diluted EPS, Non-GAAP Weighted-Average Diluted Shares Outstanding and Constant Currency are important internal measurements for BioMarin, the company believes that providing this information in conjunction with BioMarin's GAAP information enhances investors' and analysts' ability to meaningfully compare the company's results from period to period and to its forward-looking guidance, and to identify operating trends in the company's principal business. BioMarin also uses Non-GAAP Income internally to understand, manage and evaluate its business and to make operating decisions, and compensation of executives is based in part on this measure.
Non-GAAP financial measures are not meant to be considered in isolation or as a substitute for, or superior to comparable GAAP measures and should be read in conjunction with the consolidated financial information prepared in accordance with GAAP. Investors should note that the Non-GAAP information is not prepared under any comprehensive set of accounting rules or principles and does not reflect all of the amounts associated with the company's results of operations as determined in accordance with GAAP. Investors should also note that these Non-GAAP financial measures have no standardized meaning prescribed by GAAP and, therefore, have limits in their usefulness to investors. In addition, from time to time in the future there may be other items that the company may exclude for purposes of its Non-GAAP financial measures; likewise, the company may in the future cease to exclude items that it has historically excluded for purposes of its Non-GAAP financial measures. Because of the non-standardized definitions, the Non-GAAP financial measure as used by BioMarin in this press release and the accompanying tables may be calculated differently from, and therefore may not be directly comparable to, similarly titled measures used by other companies.
The following tables present the reconciliation of GAAP reported to Non-GAAP adjusted financial information:
SOURCE BioMarin Pharmaceutical Inc.
WANT YOUR COMPANY'S NEWS FEATURED ON PRNEWSWIRE.COM?
440k+
Newsrooms &
Influencers
9k+
Digital Media
Outlets
270k+
Journalists
Opted In
GET STARTED
财报临床3期临床结果临床1期寡核苷酸
分析
对领域进行一次全面的分析。
登录
或
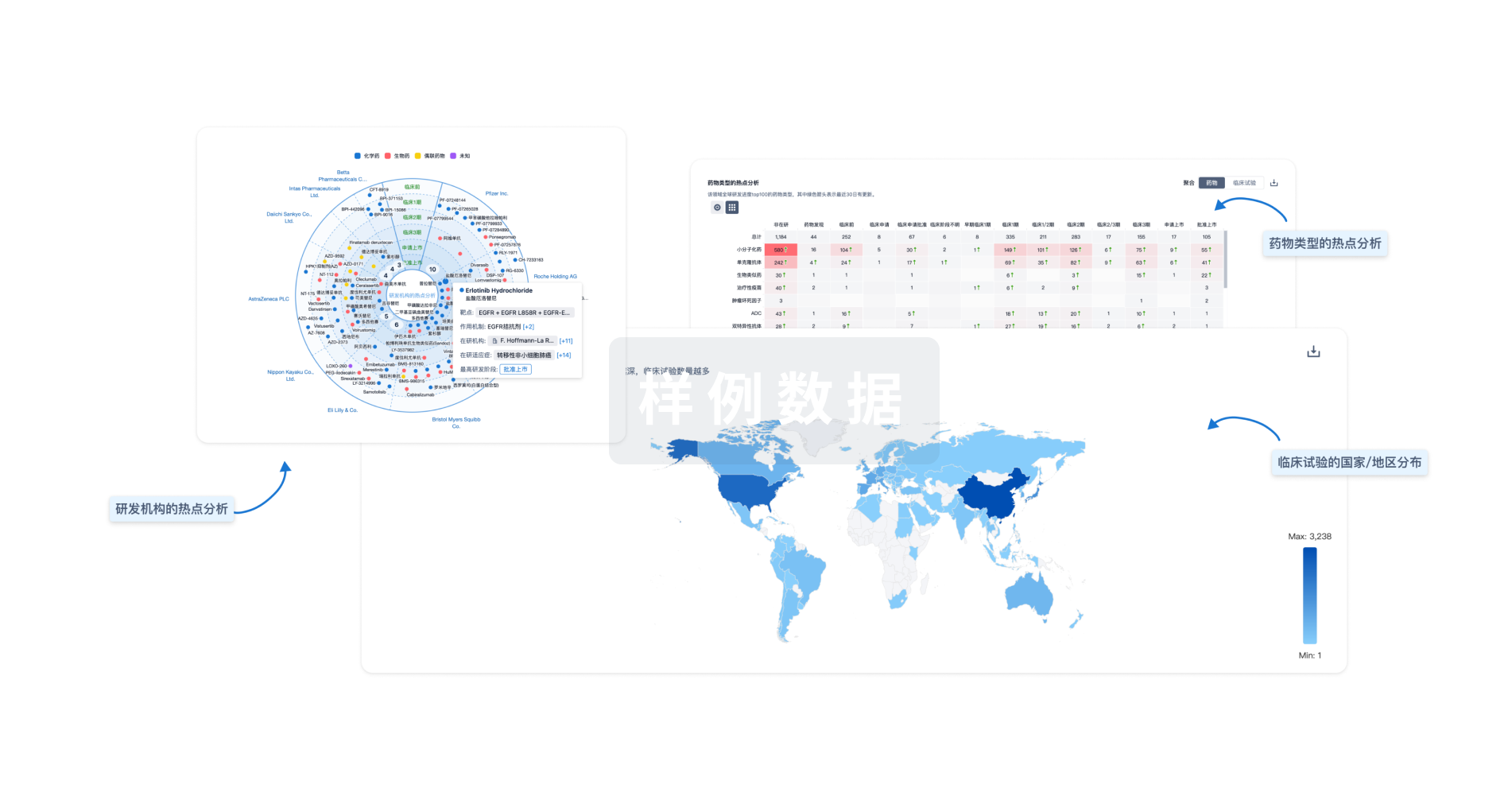
生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用




