预约演示
更新于:2026-03-08

CSL Ltd.
更新于:2026-03-08
概览
标签
感染
其他疾病
呼吸系统疾病
单克隆抗体
预防性疫苗
重组凝血因子
疾病领域得分
一眼洞穿机构专注的疾病领域
暂无数据
技术平台
公司药物应用最多的技术
暂无数据
靶点
公司最常开发的靶点
暂无数据
| 排名前五的药物类型 | 数量 |
|---|---|
| 预防性疫苗 | 21 |
| 小分子化药 | 17 |
| 灭活疫苗 | 8 |
| 非重组凝血因子 | 8 |
| 单克隆抗体 | 8 |
关联
88
项与 CSL Ltd. 相关的药物作用机制 Factor XIIa抑制剂 |
在研机构 |
原研机构 |
在研适应症 |
最高研发阶段批准上市 |
首次获批国家/地区 英国 [+1] |
首次获批日期2025-01-24 |
靶点- |
作用机制 免疫调节剂 |
在研机构 |
原研机构 |
非在研适应症- |
最高研发阶段批准上市 |
首次获批国家/地区 欧盟 [+3] |
首次获批日期2024-04-19 |
作用机制 SARS-CoV-2 S protein抑制剂 |
在研适应症 |
非在研适应症- |
最高研发阶段批准上市 |
首次获批国家/地区 日本 |
首次获批日期2023-11-28 |
433
项与 CSL Ltd. 相关的临床试验NCT07390968
A Phase 2, Multicenter, Double-Blind, Randomized, Controlled Trial of the Safety and Immunogenicity of a Self-Amplifying mRNA COVID-19 Vaccine in Adult Hematopoietic Cell Transplant Recipients
This phase IIb trial compares the effect of LUNAR-COV19 vaccine to Comirnaty vaccine in treating adult patients who have received a hematopoietic cell transplant (HCT). Guidelines recommend repeating severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) vaccination of 3 messenger ribonucleic acid (mRNA) vaccines followed by a fourth booster 3-6 months after treatment. However, vaccination is less effective in HCT patients compared to healthy people due to impaired immune responses. LUNAR-COV19, a self-amplifying mRNA vaccine, may help the body's own immune system recognize the SARS-CoV-2 spike protein and fight the virus by using a special mRNA that copies itself for a stronger response. Vaccines made from mRNA with SARS-CoV-2, such as Comirnaty, may help the body build an effective immune response. This may provide active protection against SARS-CoV-2 infection. LUNAR-COV19 may be safe and tolerable and may generate a better and more durable immune response than the Comirnaty vaccine in adult patients who have received a HCT.
开始日期2026-09-01 |
申办/合作机构 |
NCT07326592
A Phase 4, Multicenter, Double-blind, Study to Investigate the Efficacy, Safety, and Tolerability of 3 Active Doses of Respreeza® / Zemaira® Weekly Intravenous Infusions Administered Over 3 Years as Longterm Maintenance Therapy in Adult Subjects With Emphysema Related to Alpha1 Antitrypsin Deficiency
This is a multicenter, parallel-group, double-blind, randomized phase 4 study designed to identify the optimal dose of CE1226 (2 active doses) to slow disease progression as assessed by reduced rates of annual lung density decline in alpha-1 antitrypsin (AAT) deficient participants over 3 years as compared with the marketed dose 60 milligrams per kilogram (mg/kg).
开始日期2026-04-15 |
申办/合作机构 |
NCT07332091
A Phase 2, Multicenter, Randomized, Placebo-controlled, Double-blind Study of the Efficacy and Safety of Vamifeport in Adult Subjects With HFE-related Hereditary Hemochromatosis (FERROCLEAR Study)
This is a phase 2, multicenter, randomized, placebo-controlled, double-blind, parallel-group, proof-of-concept study to assess vamifeport in adult participants with homeostatic iron regulator gene-related hereditary hemochromatosis (HFE-HH). The primary objective of the study is to assess the effect of vamifeport treatment on magnetic resonance imaging (MRI)-based liver iron concentration (LIC) in adult participants with HFE-HH.
开始日期2026-02-08 |
申办/合作机构 |
100 项与 CSL Ltd. 相关的临床结果
登录后查看更多信息
0 项与 CSL Ltd. 相关的专利(医药)
登录后查看更多信息
593
项与 CSL Ltd. 相关的文献(医药)2026-01-01·Theranostics
Engineered nanoconjugates for simultaneous detection and degradation of stroke-associated microthrombi
Article
作者: Gauberti, Maxime ; Bonnard, Thomas ; Seillier, Célia ; Goux, Didier ; Mangin, Pierre ; Khalin, Igor ; Picot, Audrey ; Vivien, Denis ; de Lizarrondo, Sara Martinez ; Talukdar, Ankita ; Jacqmarcq, Charlène ; Mouriaux, Clarisse ; Schmidt, Peter
Background: Microthrombi obstructing downstream microcirculation in acute ischemic stroke (AIS) are difficult to treat and visualize with current imaging methods. Methods: To address this need, a novel theranostic agent, IO@PDA@tPA, was developed by combining iron oxide microparticles (IO) coated with polydopamine (PDA) and conjugated with recombinant tissue-type plasminogen activator (r-tPA). The amidolytic and fibrinolytic capacities of r-tPA grafted on IO@PDA were assessed using the spectrofluorometric test, the clot lysis assay, and the whole blood halo assay. IO@PDA@tPA was then tested in vivo in a preclinical ischemic stroke model induced by thrombin injection into the middle cerebral artery in both non-diabetic and diabetic mice. Two doses equivalent to 2.5 and 5 mg/kg r-tPA were tested. The presence of microthrombi was monitored via molecular MRI. A series of T 2*-weighted sequences for microthrombi imaging and magnetic resonance angiography (MRA) was performed over 45 min. At 24 h, lesion size, vessel patency, and hemorrhagic transformation were assessed with T2 -weighted imaging, MRA, and T2 * -weighted MRI, respectively. A grip test was performed to assess functional recovery one day before stroke (baseline), and at 24 h and five days after stroke. Additionally, inflammatory processes were evaluated five days post-stroke by flow cytometry in the non-diabetic cohort. Results: This agent exhibited in vitro clot lysis activity. In vivo, administration of IO@PDA@tPA at one-quarter of the standard r-tPA dose enabled both visualization and degradation of microthrombi, as detected by T2 * -weighted MRI. This treatment significantly reduced lesion size and promoted recanalization 24 h after stroke onset. In the hyperglycemic mice cohort, the agent demonstrated efficacy comparable to r-tPA without increasing hemorrhagic risk-a common complication of free r-tPA. Moreover, full functional recovery observed within five days post-stroke. Flow cytometry indicated that IO@PDA@tPA mitigated inflammatory processes. Conclusion: IO@PDA@tPA represents a promising theranostic agent targeting microthrombi in AIS, reducing the required r-tPA dose and limiting associated side effects.
2025-12-31·JOURNAL OF MEDICAL ECONOMICS
Cost-effectiveness and cost-utility analysis of Haemate-P versus other von Willebrand disease treatments in Spain
Article
作者: Panebianco, Marco ; Escolar, Gines ; Yan, Songkai ; McDade, Cheryl L. ; Wilson, Michele R. ; Linden, Stephan ; Barrientos, Laura Vidal ; Mendez, Pablo ; Tomic, Radovan ; Megias-Vericat, Juan E.
OBJECTIVE:
von Willebrand Disease (vWD) is the most common congenital bleeding disorder, with an estimated prevalence in Spain of 0.01%. The aim was to assess the cost-utility of Haemate-P compared with present alternatives in the treatment of vWD in Spain.
METHODS:
A Markov model was developed in Microsoft Excel to estimate the cost-effectiveness of various treatments for vWD over a lifetime horizon. Transition probabilities among health states were based on age and number of bleeding events. Treatment strategies compared included Haemate-P, Fanhdi, and Wilate in long-term prophylaxis (LTP) or on-demand treatment (ODT). Costs and quality-of-life were measured based on patient age, treatment, and number of bleeding events incurred. Both costs and utilities were discounted at 3%. One-way and probabilistic sensitivity analyses were performed.
RESULTS:
When comparing LTP regimens, Haemate-P was less costly and numerically more effective than both Fanhdi (incremental costs = -€1,313,845; incremental quality-adjusted life-years [QALY] = 0.13) and Wilate (incremental costs = -€2,233,940; incremental QALY = 0.29). For ODT, Haemate-P was assumed to have equal effectiveness as Fanhdi and Wilate but reduced the costs by €696,857 and €1,145,780, respectively. Haemate-P prophylaxis was more effective and less costly compared with Haemate-P on-demand in the base case (incremental costs = -€633,317; incremental QALY = 0.90). Results were generally robust to sensitivity analyses.
CONCLUSIONS:
In patients with severe vWD experiencing a high bleed rate, Haemate-P prophylaxis is a less costly and potentially more effective treatment strategy and Haemate-P is cost-saving among on-demand strategies.
2025-12-31·Future Science OA
CAR-T cell therapies: patient access and affordability solutions
Review
作者: Gonçalves, Elisabete
Chimeric Antigen Receptor (CAR)-T cell therapies, as potentially curative treatments, are a group of immunotherapy agents that are changing the paradigm for the treatment of hematologic malignancies. Ongoing research on CAR-T cell therapy is expected to expand the currently approved indications, which, given the high prices of these innovative therapeutic solutions, will increase the pressure on the sustainability of health systems, enhancing the need to establish adjusted financial solutions and promote the implementation of post-marketing monitoring procedures. This study examines the specific challenges in the development of robust clinical evidence to support the value measurement and cost-effectiveness assessment of CAR-T cell therapies and in the selection of adequate financing solutions. Managed Entry Agreements, which create mechanisms in which the risk associated with the uncertainty in long-term outcomes of these therapies is shared between the manufacturer and the payer, have emerged as preferred solutions in several European Union countries. The access barriers to CAR-T cell therapies are described, and recommendations on potential solutions to address affordability concerns using a framework of a life cycle approach to value assessment involving different stakeholders and adapted financing tools are proposed.
1,227
项与 CSL Ltd. 相关的新闻(医药)2026-03-06
2月27日,中国疫苗行业协会血液制品分会第一届第四次全体委员会会议在云南曲靖召开。本次会议由中国疫苗行业协会、中国疫苗行业协会血液制品分会主办,来自全国血液制品生产企业及单采血浆站、血液制品销售企业、相关高校科研院所及血液制品行业上下游设备、试剂、耗材厂商的100余人出席,会议由中国疫苗行业协会血液制品分会副主任委员杨晓锋主持。
中国疫苗行业协会血液制品分会副主任委员杨晓锋主持会议
中国疫苗行业协会血液制品分会主任委员李长清代表主办方致辞。在致辞中,李主任指出,2025 年血液制品行业在挑战中承压前行,在变革中寻求突破,血浆采集创历史新高,超过1.4万吨,全国百吨浆站达20家,血液制品总产量稳步提升,创新驱动取得重要进展,产学研用的协同创新生态正在逐步形成,产品质量标准和生产工艺已获得越来越多国际市场的认可,中国血液制品行业正从“规模扩张”向“高质量发展”坚实迈进。但与此同时,行业面临采浆艰难、产品同质化竞争、高技术壁垒产品研发投入与国际有差距、国家监管政策收紧、医保政策改革与税收政策等挑战。期待行业携手推动自律协同,企业以患者为中心开展创新,顺应国家医保政策,扩大产品适应症,推动专家共识,建立科学定价支付机制,加快国际化建设步伐。
中国疫苗行业协会血液制品分会主任委员李长清致辞
北京博晖创新生物技术集团股份有限公司董事长、总经理沈治卫先生致辞。他表示,博晖生物始终牢记“集人间点滴、筑生命工程”的初心使命,坚守“生命至上、安全为先、质量为本、创新为魂”的核心宗旨。守正创新、笃行实干,始终坚持以敬畏之心对待生产,以严谨之态把控质量,以务实之风经营企业,以协会搭建的平台为契机,进一步深化与行业同仁的全方位、深层次交流合作,共享发展机遇、共破行业难题、共筑行业辉煌!
博晖集团董事长、总经理沈治卫致辞
大会投票选举了增补常务委员,北京博晖创新生物技术集团股份有限公司总经理助理兼血源管理中心总经理何少杰、上海莱士血液制品股份有限公司副总经理宋正敏、南岳生物制药有限公司质量总监阳晖当选。血液制品分会主任委员李长清为新当选的常务委员颁发了聘书。
血液制品分会副秘书长毛晓向与会代表们汇报了2025年度血液制品分会工作报告及2026年度工作计划。报告显示,分会新增调整了部分委员,目前分会共有委员118人,常务委员22人。在全体委员的共同参与下,分会全年高效推进多项重点工作:组织召开2次常务委员会会议,统筹行业重大议题决策;承办第24届中国生物制品大会血液制品分论坛和中国疫苗行业协会(第一届)学术大会暨展览会血液制品分论坛,结合行业关注的热点问题,聚焦“血液制品智能制造与交叉融合发展”和“现行医保政策背景下血液制品的医疗价值”,邀请相关职能部门领导、行业专家、政策研究专家、临床专家现场探讨和交流;开展血液制品生产企业及单采血浆站实验室室间质量评价总结暨检测技术培训会议,提升行业检测标准化水平等工作;组织完成4项团体标准的申报和实施。在行业规范、学术交流、企业合作等方面取得显著成效。2026年分会将继续坚守初心、服务大局,聚焦行业发展与会员需求,强化平台建设、深化务实合作、提升服务效能。以更开放的姿态、更务实的举措凝聚各方力量,推动行业高质量发展,奋力谱写分会工作新篇章。
中国疫苗行业协会血液制品分会副秘书长毛晓作分会年度工作报告
中国医学科学院输血研究所刘彬博士对2025年度全国原料血浆采集情况进行了深入分析。据各企业提交的综合统计数据显示,“十四五”期间,血液制品行业原料血浆采集总量创历史新高,从2021年的9000多吨增长到2025年的 14000多吨,增长近 50%,但同比增速连续两年放缓,2025年增速为5.6%。当前全国325家在运营的单采血浆站主要分布在中西部地区,山东、广西等地站均采浆量表现突出,山东临朐更是突破300吨,成为全国标杆。但献浆员招募仍存在短板,770万在册献浆员在5亿覆盖人口中渗透率仅1.5%,且老龄化趋势明显,年轻献浆员招募成为行业重点任务。刘彬博士指出,要进一步强化献浆员健康与情感关怀,优化献浆流程,推进智能化管理,构建全链条信息化追溯体系,响应国家智慧监管政策要求,建立浆站动态调整机制,提升行业整体效率。
中国医学科学院输血研究所刘彬博士作《2025年度全国血浆采集情况报告》
李长清主任聚焦全球血浆蛋白产品与市场,用数据、视野与专业判断,全景解析产业现状与未来趋势,带来重磅主题分享报告《全球血浆蛋白产品与市场解析》。李主任指出,血浆蛋白是临床刚需、技术密集、高度监管的特殊生物医药,全球市场格局稳定、增长清晰,同时也在技术创新、区域需求变化中不断重塑。从全球市场规模来看:不含重组产品的血液制品市场规模达330亿美元,年复合增速6.9%,皮下注射免疫球蛋白等高端产品成为增长引擎;含重组的单抗治疗药物、基因治疗等产品后,市场规模达450亿美元以上,增速与血浆蛋白市场接近。从企业市场份额来看:CSL、Grifols、武田、Octapharma四大巨头占全球血浆蛋白市场和血液制品市场的76%以上,含重组产品后市场份额有所下降,罗氏等企业份额上升。从市场反馈的数据来看,采浆产能、皮下注射渗透率、需求结构、中国市场定价和准入政策将共同决定全球血液制品市场收入结构和公司份额平衡。
李长清主任作专题报告《全球血浆蛋白产品与市场解析》
中共曲靖市委党校一级调研员马中华书记受邀为大会带来《百年变局的时代坐标》专题演讲。马书记结合国际形势和科技竞争态势,分析了我们当前的经济趋势和未来发展空间。他指出,中国生物医药行业在成长,但与国际巨头仍有差距,应参与重塑国际秩序。随着国内外经济政策和市场因素的影响,未来几年血液制品可能迎来涨价空间。
中共曲靖市委党校一级调研员马中华书记专题演讲
大会合影
疫苗高管变更
2026-03-05
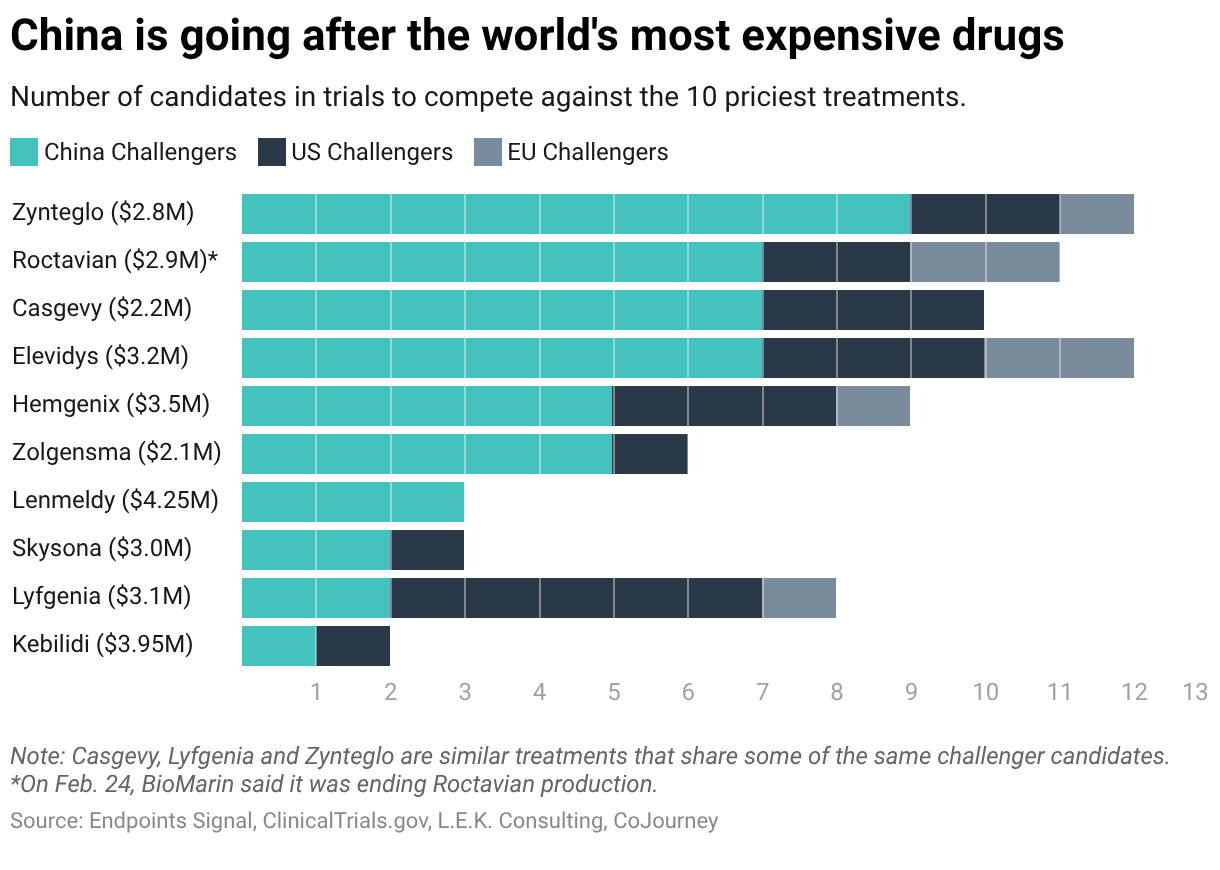
Last April, Chinese regulators approved Belief BioMed’s gene therapy for the bleeding disorder hemophilia B, setting up a challenge to one of the priciest medicines.
The treatment marked the first hemophilia gene therapy in China to be developed and manufactured entirely by a domestic company. Belief BioMed priced it at $350,000, one-tenth the cost of a rival therapy that sells for $3.5 million in the US.
In the year since, it’s become clear that Belief BioMed’s approval is a bellwether: In a global race to develop cheaper and potentially better alternatives to the world’s 10 most expensive medicines, China has pulled ahead.
An
Endpoints News
analysis of clinical trial databases shows that China accounts for 48 of the 77 programs targeting this group of ultra-expensive gene therapies. China’s total is more than double the number in the US and seven times as many as Europe.
Even if only a fraction of these lower-cost alternatives reach the market, it could upend the economics of a gene therapy system built on multimillion-dollar prices.
Shanghai-based Belief BioMed’s 161,000-square-foot facility — the equivalent of three football fields — stands as tangible proof that China’s gene therapy ambitions have gone beyond lab science and into industrial execution.
The company plans to dose its first commercial hemophilia B patient this month, and it’s working to secure approval in the US and additional countries.
“We want to be a global gene therapy company,” said Xiao Xiao, the co-founder and chief science officer of Belief BioMed. “No drug is only for one nation.”
In its expansive pipeline, the company is also conducting clinical trials of two therapies that could compete with top-priced therapies for hemophilia A and the rare disorder Duchenne muscular dystrophy. Belief BioMed is joined by Chinese companies like Shanghai Vitalgen BioPharma and CorrectSequence Therapeutics in chasing many of the most expensive therapies on the market.
For most of the pharmaceutical industry, direct competition is an expected part of the business. That wasn’t necessarily the case with gene therapies.
These medicines, which replace defective genes, require intricate scientific design and manufacturing. A 2024
analysis in the
Journal of Law and the Biosciences
argued that know-how is much tougher to reproduce compared to other parts of drug development.
And it was assumed that gene therapies would cure certain rare conditions with a limited market, discouraging competition. But some first-mover therapies had slow uptake, were
far from cures
or
mainly reached wealthy countries
.
Belief BioMed illustrates how China industrialized gene therapy development, as part of the
country’s wider rise in biotech
.
Trained in China, Xiao’s 35-year career took him from the University of Pittsburgh to the North Carolina company AskBio. He contributed fundamental science behind using AAV vectors to deliver gene therapies. He also navigated scientific challenges and high costs that caused many early US efforts to struggle.
Xiao went back to China more than a decade ago upon seeing Chinese patients unable to afford hemophilia B treatments. While patients in wealthy countries get preventative annual therapy covered by insurance, many in
China only receive treatment
after dangerous bleeding events.
Xiao and his colleagues founded Belief BioMed in 2018 to develop a gene therapy, in the hope that a one-time treatment could transform a chronic condition into a curable disease.
At the time, the Dutch company uniQure’s hemophilia B therapy was in clinical trials, and would be
approved in the US
as Hemgenix in 2022 with a $3.5 million list price, part of a wave of multimillion-dollar gene therapies. (The medicine has since been approved in Europe, Canada, Saudi Arabia, and in some Asia-Pacific countries, though not China.) Xiao sought to develop a less costly — and maybe even better — therapy for China and the larger world.
Belief’s therapy, like Hemgenix, uses an AAV vector to deliver a working copy of the factor IX gene to cells to produce long-term clotting. Xiao’s team engineered a next-generation capsid that’s designed to improve delivery and reduce immune reactions for greater safety and efficacy.
In both early and late-stage studies, a single infusion of Belief’s medicine greatly reduced or eliminated bleeding episodes in the year following treatment, according to findings published
last November in
Nature Medicine
. The clinical results so far appear comparable to Hemgenix, though Belief must dose more commercial patients before making a final comparison.
CSL Behring, which commercializes uniQure’s hemophilia B therapy, said in a statement that it’s confident in the value of Hemgenix given its long-term data. It’s working to expand the therapy to countries where it’s not available.
Belief’s bigger breakthrough may lie less in science and more in its decision to build its own facility, showing how China can independently develop and commercialize gene therapies.
Gene therapy production is notoriously fragile. Scientists grow large batches of specially engineered cells in bioreactors and insert DNA instructions into them. The cells’ own machinery then produces and assembles the viral particles that carry the therapeutic gene.
Belief does everything in-house, including producing the genetic material and vectors, to reduce errors that commonly plague the field. In 2022, for instance, US-based Regenxbio publicly said that
a third-party manufacturing delay
pushed back the start of a study for its Duchenne muscular dystrophy gene therapy.
In addition, Belief sought to rethink gene therapy manufacturing processes that had slowly evolved in the US and Europe. The company designed high-yield cells that churn out larger quantities of therapeutic virus, reworked gene insertion steps to reduce waste, and stripped out costly ingredients embedded in earlier-generation processes.
The streamlined manufacturing process drove down costs, Xiao said. So did China’s system for
quickly testing medicines
in humans. The result, he said, is an economic model that makes it possible to pursue gene therapies for conditions that others have deemed too risky or given up on, such as hemophilia A.
In February, US-based BioMarin said it would withdraw its hemophilia A therapy from the market after
failing to find a buyer
. That’s but one example of Western drugmakers pulling back from the field amid challenging economics.
Other Chinese companies echo Belief’s philosophy to be better and less expensive. YolTech Therapeutics is in clinical trials with a program that could compete with Casgevy, a $2.2 million gene therapy for sickle cell disease.
Rather than removing a patient’s cells and editing them in the lab, as companies in the US have already done, the Shanghai-based biotech
edits cells directly
in the bone marrow with a simple transfusion. Zi Jun Emma Wang, the chief technology officer and co-founder of YolTech, said it’s too early to talk about pricing but there’s potential to be much lower in cost than Casgevy.
“With our cost of goods being an order of magnitude lower than what’s typically the cost for cell therapy, we believe there’s a huge potential,” Wang said.
Helen Chen, head of LEK Consulting’s healthcare practice in Shanghai, recalled visiting Chinese companies 15 years ago that had lists of the world’s top-selling drugs in a bid to potentially compete with them.
“The difference now is that the competition from China is coming out much faster,” Chen said. “Before it was kind of an amusing lab exercise.”
China has the right conditions for gene therapy development, but not necessarily for commercialization, posing business challenges.
In China, it can still be difficult to get payer coverage for these treatments
.
The Chinese government recently expanded insurance
coverage for innovative medicines
. But gene therapies, even when relatively discounted, remain a tough sell. There’s no alternative but to design a lower-cost gene therapy from the beginning.
“We know that in China if you have a $3 million drug, nobody is able to afford it,” said Lijun Wang, the CEO of CoJourney, a company that manufactures gene therapies.
US and European challengers, by contrast, aren’t as bound by cost during development. Their payers balk at high gene therapy costs, but more often relent because the medicines replace a lifetime of chronic care.
After months of reimbursement struggles that delayed patient dosing, Belief recently secured a breakthrough when a city-level program known as Huiminbao agreed to partly cover the costs for its $350,000 therapy. The company partnered with Takeda China on domestic commercialization.
The next test lies abroad. The
FDA is skeptical
of studies from patients in China. But the company has engaged the FDA with its Chinese clinical and manufacturing data, hoping to avoid or minimize expensive US trials. US approval would grant access to the world’s largest pharmaceutical market, and speed approval elsewhere.
Xiao said the therapy’s cost in other markets would be lower than Hemgenix, but added it’s premature to give a concrete price. A discounted gene therapy could be welcome amid
slow uptake for hemophilia B gene therapies
, particularly in low-income countries that struggle to afford them.
“It could have a huge impact,” said hemophilia researcher Glenn Pierce, who has worked with companies like Biogen and Bayer. But, he added, even if it was one-tenth the price, Belief’s therapy could still be too expensive in poorer countries.
Already, China’s growing ability to make low-cost gene therapies is driving worldwide patients to Asia,
as Endpoints previously reported
.
Xiao said that compared to when the company was founded, China is bursting with gene therapy manufacturers. Investors are warming up to the space, aiming to be picks and shovels in a potential gene therapy gold rush.
“We had no choice but to bite the bullet and build out manufacturing. Now it’s everywhere,” Xiao said.
基因疗法上市批准
2026-03-04
·今日头条
会议现场
2月27日,中国疫苗行业协会血液制品分会2026年年会暨第一届第四次全体委员会会议在曲靖召开。本次会议由中国疫苗行业协会、中国疫苗行业协会血液制品分会主办,博晖生物制药(云南)有限公司承办,来自全国的血液制品生产和销售企业、单采血浆站、相关高校科研院所及行业上下游厂商共100余人出席。
李长清致辞
中国疫苗行业协会血液制品分会主任委员李长清代表主办方致辞。他指出,2025年血液制品行业在挑战中承压前行,在变革中寻求突破,血浆采集创历史新高,全国采浆超1.4万吨,百吨浆站达20家,血液制品总产量稳步提升,创新驱动取得重要进展,产学研用的协同创新生态正在逐步形成,产品质量标准和生产工艺已获得越来越多国际市场的认可,中国血液制品行业正从“规模扩张”向“高质量发展”坚实迈进。但与此同时,行业面临采浆艰难、产品同质化竞争、高技术壁垒产品研发投入与国际有差距、国家监管政策收紧、医保政策改革与税收政策等挑战。期待行业携手推动自律协同,企业以患者为中心开展创新,顺应国家医保政策,扩大产品适应证症,推动专家共识,建立科学定价支付机制,加快国际化建设步伐。
沈治卫致辞
北京博晖创新生物技术集团股份有限公司董事长、总经理沈治卫代表承办方致辞。他表示,博晖生物始终牢记“集人间点滴、筑生命工程”的初心使命,坚守“生命至上、安全为先、质量为本、创新为魂”的核心宗旨。守正创新、笃行实干,始终坚持以敬畏之心对待生产,以严谨之态把控质量,以务实之风经营企业,以协会搭建的平台为契机,进一步深化与行业同仁的全方位、深层次交流合作,共享发展机遇、共破行业难题、共筑行业辉煌!
毛晓作报告
血液制品分会副秘书长毛晓向与会代表汇报了2025年度血液制品分会工作报告及2026年度工作计划。报告显示,分会新增调整了部分委员,目前分会共有委员118人,常务委员22人。在全体委员的共同参与下,分会全年高效推进多项重点工作:组织召开2次常务委员会会议,统筹行业重大议题决策;承办第24届中国生物制品大会血液制品分论坛和中国疫苗行业协会(第一届)学术大会暨展览会血液制品分论坛,结合行业关注的热点问题,聚焦“血液制品智能制造与交叉融合发展”和“现行医保政策背景下血液制品的医疗价值”,邀请相关职能部门领导、行业专家、政策研究专家、临床专家现场探讨和交流;开展血液制品生产企业及单采血浆站实验室室间质量评价总结暨检测技术培训会议,提升行业检测标准化水平等工作;组织完成4项团体标准的申报和实施。在行业规范、学术交流、企业合作等方面取得显著成效。2026年分会将继续坚守初心、服务大局,聚焦行业发展与会员需求,强化平台建设、深化务实合作、提升服务效能。以更开放的姿态、更务实的举措凝聚各方力量,推动行业高质量发展,奋力谱写分会工作新篇章。
刘彬作报告
中国医学科学院输血研究所刘彬博士对2025年度全国原料血浆采集情况进行了深入分析。据各企业提交的综合统计数据,“十四五”期间,血液制品行业原料血浆采集总量创历史新高,从2021年的9000多吨增长到2025年的 14000多吨,增长近 50%,但同比增速连续两年放缓,2025年增速为5.6%。当前全国325家在运营的单采血浆站主要分布在中西部地区,山东、广西等地站均采浆量表现突出,山东临朐更是突破300吨,成为全国标杆。但献浆员招募仍存在短板,770万在册献浆员在5亿覆盖人口中渗透率仅1.5%,且老龄化趋势明显,年轻献浆员招募成为行业重点任务。刘彬博士指出,要进一步强化献浆员健康与情感关怀,优化献浆流程,推进智能化管理,构建全链条信息化追溯体系,响应国家智慧监管政策要求,建立浆站动态调整机制,提升行业整体效率。
李长清聚焦全球血浆蛋白产品与市场,全景解析产业现状与未来趋势,并作《全球血浆蛋白产品与市场解析》报告。他指出,血浆蛋白是临床刚需、技术密集、高度监管的特殊生物医药,全球市场格局稳定、增长清晰,同时也在技术创新、区域需求变化中不断重塑。从全球市场规模来看:不含重组产品的血液制品市场规模达330亿美元,年复合增速6.9%,皮下注射免疫球蛋白等高端产品成为增长引擎;含重组的单抗治疗药物、基因治疗等产品,市场规模达450亿美元以上,增速与血浆蛋白市场接近。从企业市场份额来看:CSL、Grifols、武田、Octapharma四大巨头占全球血浆蛋白市场和血液制品市场的76%以上,含重组产品后市场份额有所下降,罗氏等企业份额上升。从市场反馈的数据来看,采浆产能、皮下注射渗透率、需求结构、中国市场定价和准入政策将共同决定全球血液制品市场收入结构和公司份额平衡。
杨晓峰主持
会议投票选举了中国疫苗行业协会血液制品分会增补常务委员,分别为北京博晖创新生物技术集团股份有限公司总经理助理兼血源管理中心总经理何少杰、上海莱士血液制品股份有限公司副总经理宋正敏、南岳生物制药有限公司质量总监阳晖。
中共曲靖市委党校马中华教授受邀作《百年变局的时代坐标》专题演讲。
中国疫苗行业协会血液制品分会副主任委员杨晓锋主持会议。
通讯员:王成通
疫苗高管变更一致性评价
100 项与 CSL Ltd. 相关的药物交易
登录后查看更多信息
100 项与 CSL Ltd. 相关的转化医学
登录后查看更多信息
组织架构
使用我们的机构树数据加速您的研究。
登录
或

管线布局
2026年05月13日管线快照
管线布局中药物为当前组织机构及其子机构作为药物机构进行统计,早期临床1期并入临床1期,临床1/2期并入临床2期,临床2/3期并入临床3期
药物发现
3
7
临床前
临床1期
5
6
临床2期
临床3期
6
1
申请上市
批准上市
59
60
其他
登录后查看更多信息
当前项目
| 药物(靶点) | 适应症 | 全球最高研发状态 |
|---|---|---|
羧基麦芽糖铁 | 缺铁性贫血 更多 | 批准上市 |
人凝血酶原复合物 ( F10 x factor IX x factor VII x thrombin ) | 出血 更多 | 批准上市 |
帕替罗姆 ( Potassium ) | 高钾血症 更多 | 批准上市 |
Human Coagulation Factor Viii / Human Von Willebrand Factor(Csl Behring Llc) ( F10 ) | 血友病A 更多 | 批准上市 |
加达西单抗 ( Factor XIIa ) | 遗传性血管性水肿 更多 | 批准上市 |
登录后查看更多信息
药物交易
使用我们的药物交易数据加速您的研究。
登录
或

转化医学
使用我们的转化医学数据加速您的研究。
登录
或

营收
使用 Synapse 探索超过 36 万个组织的财务状况。
登录
或

科研基金(NIH)
访问超过 200 万项资助和基金信息,以提升您的研究之旅。
登录
或

投资
深入了解从初创企业到成熟企业的最新公司投资动态。
登录
或

融资
发掘融资趋势以验证和推进您的投资机会。
登录
或

生物医药百科问答
全新生物医药AI Agent 覆盖科研全链路,让突破性发现快人一步
立即开始免费试用!
智慧芽新药情报库是智慧芽专为生命科学人士构建的基于AI的创新药情报平台,助您全方位提升您的研发与决策效率。
立即开始数据试用!
智慧芽新药库数据也通过智慧芽数据服务平台,以API或者数据包形式对外开放,助您更加充分利用智慧芽新药情报信息。
生物序列数据库
生物药研发创新
免费使用
化学结构数据库
小分子化药研发创新
免费使用

